Abstract
Cutaneous T-cell lymphoma (CTCL) comprises a spectrum of T-cell lymphomas with primary skin involvement. Mycosis fungoides (MF) and Sezary syndrome (SS) are the common subtypes of CTCL in which patients present with widely diverse profiles of skin involvement and varying extents of extracutaneous disease. Patients with early-stage disease have an excellent prognosis and are managed primarily with skin-directed therapies; however, those with advanced-stage MF or SS often require multiple lines and recurrent courses of systemic therapies. Many options are available when considering systemic agents, and it is often challenging to know how to prioritize therapies to address a patient's objective disease and quality of life issues. Appreciating the disease heterogeneity and understanding the patient's overall disease profile (eg, skin, lymph nodes, blood, large cell transformation) serve as a useful framework in aligning therapies that can optimally treat active sites of disease. Tissue or blood biomarkers can be integrated into our process of prioritizing therapies and personalizing management in MF or SS. Multidisciplinary management and optimizing supportive care are additional key elements for a favorable outcome. Appropriate patients with high-risk disease should be considered for allogeneic hematopoietic stem cell transplant.
Learning Objectives
Understand the clinical heterogeneity of patients with MF and SS
Appreciate the unique quality of life issues associated with cutaneous disease
Identify the clinical, pathological, and other biomarkers that help prioritize therapies and personalize clinical management in MF and SS
Introduction
Mycosis fungoides (MF) and Sezary syndrome (SS) are the most common subtypes of cutaneous T-cell lymphoma (CTCL), and although the skin is the primary site of involvement, the malignant T cells may expand in the lymph node (LN), visceral, and blood compartments.1 In the skin lesions of early-stage MF, significant inflammatory infiltrate of immune stimulated profile is observed. However, as the disease progresses the malignant T cells acquire a Th2 phenotype accompanied by a microenvironment skewed toward a more immune-suppressive Th2 cytokine profile.2,3 A subset of patients with advanced MF or SS undergo a transformation into large cell disease (LCT) that is often associated with more aggressive biological behavior. Patients with MF and SS subgroups are often distinct clinically and biologically but also share similarities, and clinical features can shift from MF to SS and vice versa in the course of a patient's disease.
The clinical management of MF/SS involves therapies developed specifically for patients with CTCL (eg, total skin electron beam therapy [TSEBT], extracorporeal photopheresis, bexarotene, vorinostat, romidepsin) and those originally developed to treat other lymphomas but co-opted for CTCL (eg, brentuximab vedotin [BV], pralatrexate). More recently, new systemic therapies have emerged, built upon rationally selected targets in MF/SS (eg, mogamulizumab [MOGA], anti-KIR3DL2 antibody; Table 1).
Although these newer agents represent a major advancement, they have not altered our fundamental treatment strategy. Only a slim minority of patients will ever be cured of their disease, and most patients will receive numerous lines of therapy during their lives. Each failed treatment takes a physical and psychological toll on patients. We have long known that a one-size-fits-all approach cannot work for this heterogeneous disease. As our therapeutic armamentarium continues to expand, we are now challenged with how to best match patients to the treatments most likely to benefit them. Furthermore, it is unclear how best to sequence or combine therapies, and given the lack of curative therapies, the ones with fewer cumulative toxicities and durable responses are prioritized. In selected high-risk advanced-stage MF or SS, allogeneic hematopoietic stem cell transplant (HSCT) should be considered as a potential curative therapy.
CLINICAL CASE
A 64-year-old male presented with a 2-year history of progressive skin rash that currently affected >80% of the body surface area with generalized erythroderma associated with severe, debilitating itching. Topical and systemic steroids were minimally helpful. Skin biopsies showed an atypical T-cell infiltrate. Blood flow demonstrated an abnormal T-cell population expressing CD3 and CD4 but lacking CD7 and CD26, a phenotype consistent with Sezary cells (SCs), which represented 68% of lymphocytes with an absolute count of 1250/mm3; T-cell receptor next-generation sequencing (NGS) studies supported a clonal T-cell process in the skin and blood. Whole-body positron emission tomography/computed tomography (PET/CT) imaging showed an approximately 2 cm axillary and inguinal lymphadenopathy (LAD) with a maximum standardized uptake value of 3 to 4, consistent with reactive or dermatopathic LNs. A diagnosis of SS was established, presenting with low-burden SCs and clinically reactive LAD and thus clinical stage IVA1. The patient was treated with oral bexarotene with overall global partial response (75% improvement of his skin disease and >50% reduction of his SCs), and supportive care was provided with topical steroids, emollients, and gabapentin for itching. Six months later while continuing maintenance bexarotene, he noted worsening of his skin disease with recurrent erythroderma. A skin biopsy confirmed CTCL without evidence of LCT. His blood Sezary flow now showed >10,000 SCs/mm3. Updated PET/CT findings again supported reactive or dermatopathic LAD with no hepatosplenomegaly.
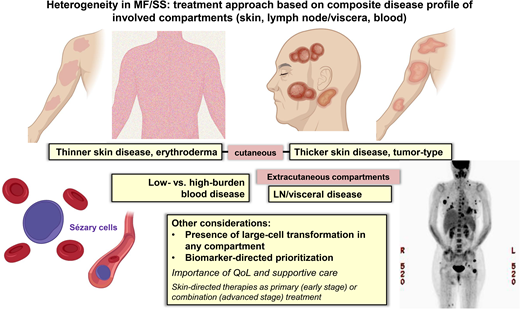
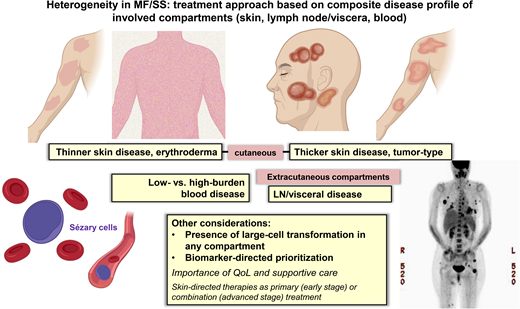
Recognizing disease heterogeneity within the skin and across extracutaneous compartments
MF and SS are unique in their striking heterogeneity of clinical, biological, and molecular features and the need to individualize management for an optimal outcome. This heterogeneity is further complicated by variable clinical behavior across disease compartments (eg, skin, LN, blood). A meaningful clinical outcome of treatment in MF/SS consists of a durable global (composite of all compartments) objective response and improvement of quality of life (QoL) measures,4-7 and achieving these goals requires individualization of therapy. Traditionally, the clinical stage has been a primary consideration in treatment selection; however, additional guidance is often needed given the heterogeneity within stage groupings. The extent/burden of disease or status of other established prognostic factors (eg, LCT) in the skin or other disease compartments further contributes to treatment selection.8
In early-stage MF with patch and/or plaque skin disease, skin-directed treatment is preferred, and improvement of QoL is usually the primary focus of treatment. Primary skin-directed treatments include topical preparations of steroids, mechlorethamine, retinoids, phototherapy (eg, narrowband ultraviolet B), and TSEBT.8 TSEBT is very effective at low doses (eg, 12 Gy) and most appropriate in patients who have widespread thicker or folliculotropic (hair follicle involvement) skin disease.9 If skin- directed treatment options fail to manage a patient's disease, then escalating to systemic agents is appropriate.
Patients with advanced-stage MF and SS have varying clinical presentations ranging from tumor-type skin disease to generalized erythroderma in which the type and extent of their skin and extracutaneous disease (LN and/or blood disease) drive the therapies that best address a patient's overall disease. Many standard treatment options exist for advanced-stage patients, including systemic biological and targeted therapies or traditional cytotoxic chemotherapy.8 The ultimate selection of the primary therapy will be based on the assessment of the skin and the severity of any extracutaneous disease and on aligning therapies that address the most symptomatic and threatening disease.
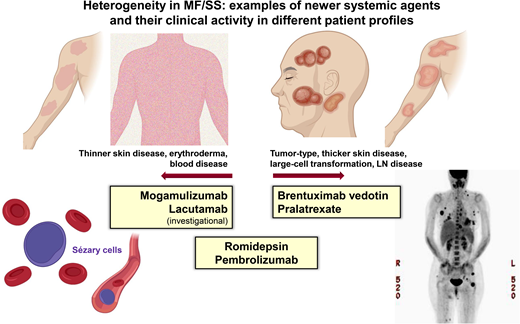
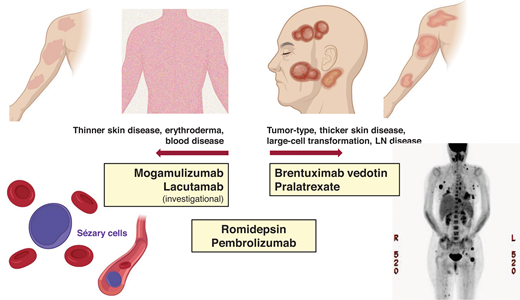
Systemic treatments often exhibit differential efficacy across skin, blood, and nodal compartments (Table 1 and Figure 1). The response interpretation can be further complicated by the varying response criteria and clinical end points used in earlier clinical trials in CTCL; however, more recent trials have utilized the international consensus criteria that have helped compare results across studies.4 There are frequent mixed responses in the skin or across different compartments with disease, and therapies that reduce skin disease may not be effective in clearing the blood or LN disease, and vice versa. For example, in a patient with MF and widespread ulcerating skin tumors (±LCT), we would select treatments with known reliable activity in the skin compartment (eg, TSEBT, BV).9-12 However, in a patient with SS and high-burden Sezary disease, we would opt for therapies that are efficient at reducing the blood disease (eg, MOGA, romidepsin).13,14 Furthermore, some therapies do not have established, meaningful activity in LCT or bulky disease, whereas some therapies have shown activity across all compartments, including LCT disease. Traditional combination chemotherapy regimens in systemic TCLs are often reserved for patients with refractory, advanced disease or when allogeneic HSCT is planned for consolidation.8 Lastly, the potential toxicities and tolerability of chronic therapy must be considered in prioritizing treatments and personalizing management.
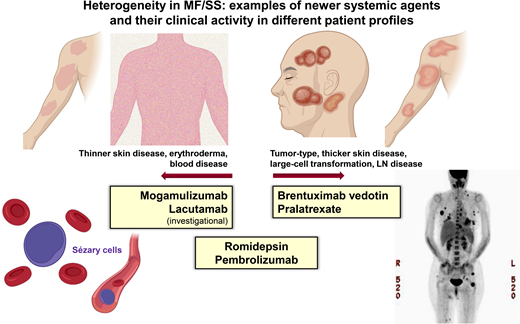
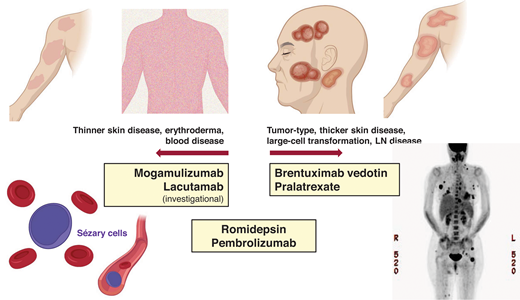
Examples of newer systemic agents and their clinical activity in different patient profiles.
Examples of newer systemic agents and their clinical activity in different patient profiles.
Importance of multidisciplinary care and management of QoL issues: essential role of supportive therapy
Ideally, the management of patients with CTCL involves a multidisciplinary approach. It is helpful to establish collaborative colleagues in hematology/medical oncology, radiation oncology, pathology, and dermatology who are interested in cutaneous lymphoma. To address the extensive skin disease, specialized radiation methods such as TSEBT serve as an important therapeutic alternative; however, access to centers with TSEBT can be an issue. Low-dose TSEBT can be combined with treatments that have great efficacy in the blood and/or LN compartments to improve the overall composite response and response duration.15,16 Patients may have extensive skin wounds or atypical skin infections or develop skin lesions that clinically and/or pathologically mimic CTCL, including an inflammatory skin reaction to lymphoma therapy (eg, MOGA, pembrolizumab). Thus, a close collaboration across specialties is essential in providing appropriate clinical care and optimal outcome in CTCL.
Intolerable itching is a common QoL in CTCL, especially in SS. The effective management of itching also reduces the risk of secondary skin infections as patients often have compromised skin barrier function and are commonly colonized with Staphylococcus aureus.17 Management with an anti-staph supportive regimen has been shown to improve their itching, clear their skin, and lower infection-associated complications.18 The visible nature of CTCL adds a unique negative life impact because it constantly reminds patients of their disease and often prohibits patients from social interaction, intensifying the emotional burden of their struggles.19,20 Thus, in contrast to other indolent lymphomas such as low-grade follicular lymphoma, in which watchful waiting can be acceptable, patients with CTCL are often on constant treatment, and QoL elements may lead to a decision to change therapy without objective disease progression. Optimizing supportive care is essential, and clinical end points such as time to next significant therapy may be useful measures of overall clinical benefit.21
Prioritizing treatment options in SS
In patients with SS, the leukemic subtype of CTCL, clinical management is often stratified by the burden of the blood Sezary disease. Primary systemic therapy for those with a lower Sezary burden (<5,000 SCs/mm3) includes oral bexarotene,22 methotrexate,23 and extracorporeal photopheresis.24 In those with a higher Sezary burden, histone deacetylase inhibitors (eg, romidepsin) or newer therapies, such as MOGA, with more reliable activity in the blood compartment would be appropriate frontline options (Table 1).14 With MOGA, an anti-CCR4 monoclonal antibody in which CCR4 is highly and consistently expressed by SCs, objective blood response was observed in 68% (83/122) of patients with 44% (54/122) achieving complete remission (CR) in the pivotal trial, MAVORIC.13 The median time to response in the blood was 1 month, and the median duration of blood response was 26 months. The skin response rate was 42% with a lower nodal response rate of 17% and a global (composite of all compartments) overall response rate of 28%. However, patients with active LCT were excluded in the MOGA pivotal trial. Given the impressive clinical activity in the blood combined with a favorable tolerability profile, MOGA may be a preferred option in high-burden SS without skin LCT or LN disease. Similarly, the anti-KIR3DL2 antibody (lacutamab) currently under clinical development has been shown to be more effective in reducing blood disease compared to nodal disease (blood response rate [RR] 56% vs nodal RR 11%).25 Conversely, BV, which demonstrated reliable efficacy in the skin (including LCT) and the LN disease,11 does not have established activity in Sezary disease and SS and was excluded in the pivotal trial in CTCL. In SS with extensive LN disease (±LCT), romidepsin may be prioritized over MOGA or other options, as romidepsin has demonstrated consistent clinical activity across compartments (Table 1).
Length of therapy and maintenance strategies
To address the chronic course of MF/SS, newer systemic therapies were developed in which clinical studies allowed patients to continue treatment without a fixed duration until intolerance or disease progression occurred. However, for therapies that have dose-cumulative toxicities such as the peripheral neuropathy associated with BV treatment, we should consider a fixed treatment course (<6-8 cycles) and re-treat with BV later when needed or use reduced doses of BV.10,26.27 MOGA can be continued long-term, but in one-third of patients, it may lead to a rash (median time to rash, 105 days),13,28-30 and the severity of the rash may affect the length of treatment with MOGA. Despite the occurrence of MOGA, patients can be re-treated with MOGA. For chronic therapy in CTCL, treatment options or strategies that minimize immune suppression will be preferred given the high-risk of recurrent skin infections.
After initial disease reduction, a maintenance strategy of a sustainable treatment regimen can be considered. This may involve increasing the treatment interval of intravenous therapies or transitioning to an oral agent.31 Utilizing a less intense maintenance dose regimen of therapies may lower treatment-related toxicities and provide a longer overall clinical benefit. Integrating skin-directed therapy is not only essential in combination with systemic agents but necessary when the systemic disease is cleared, and patients then have primarily skin-limited disease.
CLINICAL CASE (Continued)
Given the higher burden of blood Sezary disease and lack of LCT in the skin, the patient was treated with MOGA and after 2 cycles experienced >90% skin improvement and reduction of itching. His blood Sezary disease cleared promptly without measurable abnormal circulating T cells (B0) by flow cytometry. After 4 months of continued great response with continued blood CR (B0), MOGA was discontinued, and the patient continued topical therapy as needed for limited residual skin disease. The patient's blood Sezary remained clear and his skin near CR off MOGA, but after several months, he experienced disease progression in the skin with rapidly worsening, widespread thick plaques and nodular, tumor-type disease. Skin biopsy showed LCT with 10–40% of neoplastic T cells expressing CD30 by immunohistochemistry. PET/CT now showed more notable LAD, and a core needle sampling showed involvement with T-cell lymphoma (CD30 expression of 30%). Given the significant CD30 expression in the skin and LN compartments, BV was selected as the next therapy.
Management of LCT in MF and SS
LCT in MF or SS is recognized as one of the strongest independent adverse prognostic indicators,32,33 and thus knowledge of LCT is a key factor in treatment selection (Table 1). However, notable inter-rater (pathologist to pathologist) variability is observed in the interpretation of LCT as the criteria is an arbitrary cutoff value (>25% of malignant T cells are large cells) by histopathologic evaluation.34 Accordingly, a subset of patients designated as LCT may have a more indolent course; therefore, appropriate clinical judgment is essential for optimal management. In contrast to systemic lymphomas in which anthracycline-based therapy and autologous transplants are considered in the setting of transformed disease, LCT in MF/SS is managed differently, with greater individualized tailoring for the extent and biology of the transformed disease.
CD30 expression is not a requirement for LCT criteria, but the median CD30 expression is higher in the lesional tissue of patients with LCT compared to those without LCT,35 and thus BV is often a highly effective treatment in MF with LCT.10,12,36 In the phase 3 trial of BV in CD30+ CTCL, the primary end point of an objective response rate lasting >4 months (ORR4) was higher in patients with LCT than without LCT (65% vs 39%).11,12 Pralatrexate and romidepsin, each as a single agent, have also demonstrated clinical activity in MF or SS with LCT,14,37,38 though the data in these single agents are not as robust as in the pivotal trial of BV. MOGA would not be prioritized in patients with LCT given the lack of established data.13 Older agents such as liposomal doxorubicin or gemcitabine have greater toxicity profiles but can be used in MF/SS with LCT in refractory settings or when newer agents are not available.39,40 A recent clinical trial of pembrolizumab in patients with relapsed/refractory MF or SS showed promising clinical activity in patients with LCT and can be considered in relapsed or refractory settings.41
Biomarker-guided selection and integration of therapies in MF/SS-CTCL
Currently, numerous available and investigational therapies in MF/SS-CTCL can target the cell surface molecules of malignant T cells, disrupted cellular pathways, and/or the tumor microenvironment (TME), including the immune milieu (Table 1). Reliable biomarkers that enrich the patient subsets that can benefit with specific therapies would be ideal, and such biomarker information may help select and prioritize therapies among a list of options.
Of the targets on the malignant cell surface in CTCL, CD30 has the most robust data, with a CD30 targeting agent, BV. With the use of more sensitive tools for CD30 detection (eg, multispectral imaging), samples with nondetectable CD30 expression by routine immunohistochemistry (IHC) and light microscopy demonstrated cell-surface CD30 molecules.10 In MF/SS, the inter- and intrapatient (interlesional) CD30 expression levels (by routine IHC) can be highly variable in the skin, and BV has demonstrated significant clinical activity in patients with a spectrum of CD30 expression in 2 independent studies in MF/SS.10,36,42 Patients with >5% CD30max (maximum CD30 level from multiple skin samples) had more reliable overall clinical response compared with those with CD30max <5%; however, meaningful activity was observed across all CD30 levels. Further, in the pivotal study of BV in CD30+ CTCL (ALCANZA), patients with multiple skin biopsies showed a notable variability of CD30 levels, ranging from nondetectable to >50% expression.11,12 Thus, BV is a treatment option even in those with low/negligible levels of CD30 expression, especially in refractory settings.
Therapies such as MOGA can target the malignant cells as well as TME, as the target molecule CCR4 is expressed not only by the malignant T cells but also by the regulatory T cells. CCR4 is a chemokine receptor associated with skin trafficking, but unlike CD30, the molecule is frequently and consistently expressed in the lesional skin (median CCR4, 80%; range of 1%-100% by IHC) and may not be useful as a biomarker in CTCL.13 However, some patients with MF/SS have negligible CCR4 expression, and further studies will be needed to establish the role of MOGA in these patients. Further, assessment for CCR4 expression is not routinely available, so a standardized protocol for CCR4 assessment and a better understanding of its correlation with clinical outcome needs to be established.43 Although both CCR4 and KIR3DL2 are both commonly expressed in skin disease with LCT, neither MOGA nor anti-KIR3DL2 antibody (lacutamab) has established activity data in patients with LCT.25,44
Newer biomarker platforms (eg, actionable NGS panels, TME profiling) may help guide treatment selection and prioritize therapies in selected settings.45,46 The knowledge of Th2 skewing with disease progression has led to the selection of immune therapies that shift the immune profile from Th2 to Th1, including interferons, IL-12, and toll-like receptor agonists. Agents known to deplete regulatory T cells in the TME, such as MOGA or CD25-targeting agents,47 and to activate macrophages (increase the “eat-me” signal), such as agents targeting the CD47-SIRPα axis (eg, SIRPαFc, magrolimab),48,49 may be considered as single agents or part of combination regimens. Patients with genomic data that demonstrate pathogenic alteration in the T-cell immune regulatory axis may benefit from immune therapies that include checkpoint inhibitors.41
In patients with refractory or high-risk disease, using NGS platforms to evaluate for additional prognostic clues or actionable pathogenic genetic variants can help prioritize therapies. For example, JAK inhibitors may be considered in patients with relapsed/refractory disease with a pathogenic mutation of JAK1/3.50 Moreover, we can extrapolate genomic biomarker data from other T-cell lymphomas. For example, CCR4 gain of function mutations have been associated with superior clinical outcomes in adult T-cell leukemia/lymphoma patients treated with MOGA.51 Thus, if such potential pathogenic variants of CCR4 are noted by NGS data, perhaps MOGA can be prioritized over other therapies in high-burden Sezary patients or in refractory disease.
As previously discussed, LCT is a very important clinical and pathologic factor to consider when selecting therapy. An NGS study by Ungewickell et al reported that LCT is associated with a high mutation burden and recurrent PLCG1 alterations.52 Furthermore, recent studies exploring the programmed cell death 1 (PD-1) and programmed cell death 1 ligand 1 (PD-L1) axis show a possible link between PD-L1 structural variants and LCT,45 supporting the hypothesis that patients with LCT may have more TME immune alterations that allow tumor growth. These patients with PD-L1 alteration have experienced clinical benefit with anti-PD-1 antibody such as pembrolizumab.41,53
CLINICAL CASE (continued)
The patient was treated with BV, with great clinical response in the skin and flattening of his thicker, tumor-type skin disease that showed LCT by pathology. PET/CT obtained after 3 cycles of BV showed clinical CR of LN disease. The blood compartment remained clear of measurable disease by flow cytometry. The patient had clinical features consistent with a high-risk prognostic profile, including a history of high blood SC burden, LN disease, and LCT.33 At the initiation of BV, allogeneic HSCT was discussed as a potential curative therapy in this high-risk setting. The patient's sibling was identified as a 10/10 matched donor. The patient was 67 years old at this time point, and he agreed to move forth with allogeneic HSCT using TSEBT and a total lymphoid irradiation/antithymocyte globulin nonmyeloablative regimen.
Allogeneic HSCT as a path to establish durable remission in high-risk MF/SS
Patients with advanced-stage MF and SS have a median survival of <5 years, are often treated with multiple sequential systemic therapies, and eventually become refractory to available agents.33 High-dose chemotherapy followed by autologous HSCT has not shown a durable benefit,54 but allogeneic HSCT that relies on a graft vs lymphoma effect has led to long-term remissions (Table 2).55-61
There is some controversy regarding which subset of patients should move forth for allogeneic transplant in MF/SS and when in their disease course it is appropriate. Allogeneic HSCT should be considered for appropriate patients with stage IIB through IV disease who are refractory to multiple lines of systemic therapy, demonstrate declining duration of clinical benefit, or present with a very high-risk profile. A large international study of prognostic factors in advanced-stage MF and SS (retro-CLIPI) identified 3 risk groups with distinct prognostic outcomes based on the number of key adverse prognostic parameters, including extracutaneous disease and LCT.33 Patients in the high-risk group with a 5-year overall survival (OS) of 28% were clearly appropriate for allogeneic HSCT. A significant proportion of patients in the intermediate-risk (5-year OS, 44%) group may be considered for allogeneic HSCT. The 5-year OS with allogeneic HSCT in the published studies ranged from 32% to 56%,55-62 likely supporting an improved survival benefit provided that most patients who received allogeneic HSCT included those with intermediate- to high-risk retro-CLIPI profiles. The optimal timing for allogeneic HSCT is when the disease is well controlled (CR or near CR of all disease compartments) and before the disease has progressed to a state in which the chance of response or survival with allogeneic HSCT is low. The skin compartment is a frequent site of posttransplant disease relapse, and thus TSEBT is often an important and necessary component of the preparatory strategy in MF/SS. The median age of patients with advanced MF or SS at diagnosis is 60 to 65 years, so an allogeneic regimen with fewer transplant-related toxicities has also been explored.61 For each patient, a decision to transplant requires careful consideration of the long-term benefits and significant risks, including the potential for graft-versus-host disease (GVHD) and disease relapse after transplant.
In patients with aggressive MF or SS, there remains an unmet need for additional safe and effective therapies that can bridge patients to allogeneic HSCT. And especially for those who are not eligible for allogeneic HSCT or have no optimal donors, new and novel immune therapies and approaches are needed. As these innovative therapies are made available, it is essential to integrate them to manage patients successfully and to continue exploring biomarkers that can help us guide our selection or prioritization of treatment options.
Conclusions and future directions
The clinical, pathological, and biological features in MF and SS are highly heterogeneous, with significant inter- and intra-patient variability that requires a personalized approach to management. Available and investigative therapies often have differential activity across disease compartments, and thus in addition to clinical stage and the presence of LCT, compartmental activity is an important element that guides treatment selection. In appropriate high-risk settings, patients should be considered for allogeneic HSCT. The unique QoL issues bring unique challenges in managing patients with CTCL.
Ultimately, in this highly heterogeneous disease group we hope to establish biomarkers and strategies that enrich (better align subsets) for durable clinical response and address resistance/escape mechanisms while minimizing toxicities. More personalized and tolerable immunotherapies may be a pathway to achieve a more durable or sustained clinical response. Advances in biomarker-guided investigations in CTCL will enable us to apply evidence-based approaches in prioritizing therapies and help develop combination strategies that may lead to the clinical synergy of combined agents.
Conflict-of-interest disclosure
Youn H. Kim: Advisory board, Research funding; Innate: Research funding; Corvus: Research funding; Galderma: Advisory board, Research funding; CRISPR therapeutics: Research funding; Secura Bio: Advisory board; Trillium: Research funding.
Off-label drug use
Youn H. Kim: none.