Abstract
Emerging data have enhanced our understanding of cancer-associated thrombosis, a major cause of morbidity and mortality in patients with cancer. This update will focus on recent findings, including the phenomenon of incidental venous thromboembolism (VTE), novel approaches to risk assessment, and the results of randomized clinical trials focusing on prophylaxis of cancer outpatients. Incidental VTE is an important contributor to rates of cancer-associated VTE and, in terms of outcomes, appears to be as consequential for patients as symptomatic VTE. Multiple biomarkers have been studied, with the highest level of evidence for prechemotherapy elevated platelet counts, elevated leukocyte counts, and low hemoglobin. Other candidate biomarkers, including D-dimer and tissue factor, are currently being evaluated. A recently validated risk score for chemotherapy-associated VTE has now been evaluated in more than 10 000 cancer patients in a variety of clinical settings and trials and is ready for clinical use (Level 1 clinical decision rule). Several randomized clinical trials in solid-tumor patients with low-molecular-weight heparins and semuloparin, an ultra-low-molecular-weight heparin, demonstrate clearly that outpatient thromboprophylaxis is feasible, safe, and effective. Selecting the appropriate patients for prophylaxis, however, continues to be a matter of controversy.
Introduction
An explosion of data in the past few years has enhanced our understanding of cancer-associated thrombosis, a major cause of morbidity and mortality in patients with cancer. This increase in research has followed an increase in clinical events, with the most contemporary reports documenting “unacceptably high” event rates.1 Venous manifestations of cancer-associated thrombosis include deep vein thrombosis (DVT) and pulmonary embolism (PE), as well as visceral or splanchnic vein thrombosis, together described as venous thromboembolism (VTE). Arterial events include stroke and myocardial infarction.
Cancer-associated thrombosis affects the lives of patients significantly. VTE results in a requirement for long-term anticoagulation, a 12% annual risk of bleeding complications, and up to 21% annual risk of recurrent events even with anticoagulation,2 delay or discontinuation of chemotherapy,3 consumption of healthcare resources,4 and a potential impact on patient quality of life. Most importantly, thrombotic events are the second leading cause of death in cancer patients (after cancer itself) and are associated with worsened short-term and long-term survival.5–7 This update will focus on recent and emerging data regarding cancer-associated thrombosis, including the phenomenon of incidental VTE, novel approaches to risk assessment, and the results of randomized clinical trials (RCTs) focusing on prophylaxis of outpatient cancer patients.
Incidental VTE
Increasingly, VTE is being diagnosed as an incidental finding on CT scans ordered for other indications, typically staging or restaging of malignancy. These VTE events are referred to as “incidental” or “unsuspected” VTE. The term “asymptomatic” VTE should be discouraged because patients often have unrecognized symptoms; in a retrospective review of 59 cancer patients with incidentally diagnosed PE and matched controls, approximately 75% were symptomatic with higher rates of reported fatigue and shortness of breath than controls.8 The prevalence of incidental PE on routine radiographic studies of the chest performed for cancer staging or monitoring of other disease such as pulmonary nodules is 1.5%-3.4% per scan.9–11 The rate of incidental PE increases to 4%-9% in hospitalized cancer patients receiving routine scans.9,11 Incidental VTE is an important contributor to rates of cancer-associated VTE. For example, in an analysis of pancreatic cancer patients, incidental VTE comprised 33.3% of PEs, 21.4% of DVTs, and 100% of visceral VTEs.12
It is important to understand that incidental VTE appears to be as consequential for patients as symptomatic VTE. In a recent analysis, rates of VTE recurrence, bleeding, and mortality were similar in cancer patients with incidental VTE compared with cancer patients with suspected VTE.13 In a case-control study, cancer patients with incidental VTE had significantly worse survival (hazard ratio [HR] = 1.51; 95% confidence interval [CI], 1.01-2.27; P = 0.048) compared with matched cancer patients without VTE.14 In the pancreatic cancer study discussed above, DVT (HR = 25; 95% CI, 10-63; P < .0001), PE (HR = 8.9; 95% CI, 2.5-31.7; P = .007) and incidental visceral events (HR = 2.6; 95% CI, 1.6-4.2; P = .0001) were all independently associated with mortality, although anticoagulants reduced these risks by 70% (26%-88%; P = .009).12 A study in lung cancer patients also showed that both incidental (HR = 2.4; P = .01 vs patients with no VTE) and suspected VTE (HR = 2.7; P < .002) were associated with worse survival.15 Although randomized data are not available regarding treatment of incidental VTE, based on these cohort studies, my practice is to use therapeutic anticoagulation (preferably extended-duration low-molecular-weight heparin [LMWH]) for all incidental PE, DVT, and acute, symptomatic visceral vein thrombi.
Risk assessment: biomarkers
Multiple biomarkers have been linked to cancer-associated thrombosis (Table 1). The highest level of evidence currently exists for components of the complete blood count: prechemotherapy elevated platelet counts, elevated leukocyte counts, and low hemoglobin levels are all associated with chemotherapy-associated VTE.16,17 Given that the complete blood count is obtained routinely in cancer patients, these can be considered extremely cost-effective biomarkers.
Select biomarkers predictive of cancer-associated thrombosis

*Investigational or not widely available.
D-dimer is another widely studied (and available) biomarker predictive of cancer-associated VTE. In colorectal cancer, patients with elevated D-dimer (defined as > 0.3 mg/L) had a 20% (95% CI, 12%-31%) 1-year incidence of DVT versus 5% (95% CI, 2%-12%) for other patients (adjusted HR = 6.53; 95% CI, 1.58-27.0).18 Elevated D-dimer was also associated with increased risk of VTE (HR = 1.8; 95% CI, 1.0-3.2; P = .048) in the Vienna Cancer and Thrombosis Study (CATS) registry.19 It should be noted, however, that many cancer patients have elevated D-dimer levels and there is no consensus on the cutoff levels predictive of cancer-associated thrombosis.
Several research groups have focused on tissue factor (TF), the physiologic initiator of hemostasis that is widely expressed across multiple malignancies. TF is released into the circulation in the form of microparticles, and levels can be detected in cancer patients. Assays to evaluate TF include immunohistochemical grading of TF expression on tumor cells, measurement of TF antigen using ELISA, TF microparticle procoagulant activity, and impedance-based flow cytometry, but there is no consensus “standard” TF assay. Initial reports suggested a significant association of elevated TF with subsequent VTE.20,21 However, the majority of these data were derived from patients with specific cancers, particularly pancreatic cancer.22 More recently, in a recent large study of cancer patients with a heterogeneous mixture of cancer patients, elevated procoagulant microparticles (although not TF specific) were not found to be predictive of VTE.23 Further, in a prospective analysis of subgroups of the Vienna CATS registry, TF was predictive of VTE in pancreatic but not brain or colorectal cancers.24 TF is still to be considered an investigational biomarker, with potential value in pancreatic and other select cancers. Other candidate biomarkers include soluble P-selectin and thrombin-generation assays, but confirmatory studies are awaited.25,26
Risk assessment: a risk score
Risk assessment tools can incorporate multiple variables to identify patients or subpopulations at risk for events. A recently developed risk score can identify cancer patients at high risk for VTE using a combination of easily available clinical and laboratory variables (Table 2).16 This risk score for chemotherapy-associated VTE was originally derived from a development cohort of 2701 patients and was then validated in an independent cohort of 1365 patients from a prospective registry. Observed rates of VTE in the development and validation cohorts were 0.8% and 0.3% in the low-risk category, 1.8% and 2% in the intermediate-risk category, and 7.1% and 6.7% in the high-risk category, respectively. This model was first externally validated in a prospective population by the Vienna CATS study in 819 cancer patients.27 The 6-month cumulative probabilities of developing VTE in this study population were 1.5% (score of 0), 3.8% (score of 1), 9.4% (score of 2), and 17.7% (score ≥ 3). Several other retrospective and prospective studies have further validated this risk score, although rates vary between studies because of varying patient selection and follow-up periods (Table 3). The risk score was recently found to be the only predictor of VTE in an analysis of 1412 patients enrolled in phase 1 studies.3 The score was evaluated in a prespecified subgroup analysis of the SAVE-ONCO study, a large RCT of thromboprophylaxis in cancer outpatients on chemotherapy, as well as in a post hoc subgroup analysis of the Prophylaxis of Thromboembolism During Chemotherapy Trial (PROTECHT) study.28,29 The score was both prognostic of VTE and predictive of benefit from thromboprophylaxis in both trials. Altogether, the risk score has now been evaluated in more than 10 000 cancer patients in a variety of clinical settings and trials and is therefore validated and ready for clinical use to assess for risk of cancer-associated VTE (level 1 clinical decision rule).30 An expansion of the original risk score with the inclusion of 2 additional biomarkers, D-dimer and soluble P-selectin, has been described by the Vienna group.27 This expanded risk score, although promising, requires further validation in other studies. The P-selectin assay, which is required for the expanded model, is not widely available, which may limit its practical use.
Predictive model for chemotherapy-associated VTE16
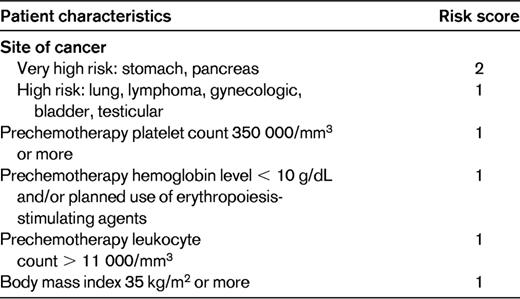
High-risk score, ≥ 3; intermediate-risk score, 1-2; low-risk score, 0.
Rates of VTE in select studies validating a risk score for chemotherapy-associated VTE
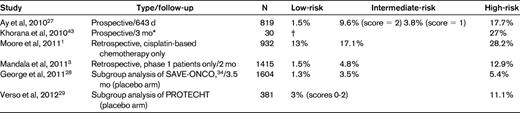
High-risk score, ≥ 3; intermediate-risk score, 1-2; low-risk score, 0.
*Included 4 weekly screening ultrasonographies.
†Enrolled only high-risk patients.
A myeloma-specific risk assessment algorithm with recommendations for prophylaxis has been proposed by the International Myeloma Working Group.31 This risk assessment tool is based on expert consensus and has not been validated prospectively or retrospectively.
Cancer outpatients: to prophylax or not to prophylax
VTE is a largely preventable illness and multiple anticoagulants are available and used in a variety of settings as primary prophylaxis. Thromboprophylaxis is currently recommended for cancer inpatients without contraindications by the American Society of Clinical Oncology (ASCO) and National Comprehensive Cancer Network (NCCN) guidelines. These recommendations are based on extrapolation from large trials conducted in the medically ill population, which included a minority of patients with malignancy. Unfortunately, no cancer-specific studies have been conducted, and this remains a major knowledge gap in the field. Current efforts to improve prophylaxis, supported by regulatory authorities including the United States Surgeon General and Joint Commission, have focused on hospitalized and postsurgical patients. However, cancer treatment paradigms have shifted, with a majority of cancer therapy occurring in the outpatient setting. It would follow that a majority of VTE events also occur in the outpatient setting, and this was demonstrated in a recent analysis of more than 17 000 patients, in which nearly 80% of VTE in cancer occurred in outpatients. Therefore, efforts to reduce the public health burden of cancer-associated VTE need to take the cancer outpatient population into account when developing prophylaxis strategies.
Recent RCTs have focused on outpatient prophylaxis, but have taken varied approaches to risk stratification. One approach has to been to focus on common cancers known to be associated with VTE. The PROTECHT study evaluated daily nadroparin, a LMWH, in patients with locally advanced or metastatic lung, gastrointestinal, pancreatic, breast, ovarian, and head/neck cancers on chemotherapy.32 Overall, 2% of the treatment group and 3.9% of the placebo group developed a thromboembolic event (1-sided 95% CI, 0.303%; P = .02) with a nonsignificant increase in major bleeding. The largest cancer thromboprophylaxis trial was published in early 2012. SAVE-ONCO was a prospective, double-blind, multicenter study of more than 3200 patients with locally advanced or metastatic solid tumors (lung, pancreas, stomach, colorectal, bladder, or ovary) randomized to daily subcutaneous semuloparin (a novel ultra-LMWH) or placebo.33 Patients receiving prophylactic semuloparin had a 64% relative risk reduction of VTE (HR = 0.36; 95% CI, 0.21-0.60]; P < .0001; 1.2% vs 3.4%) compared with placebo with no significant increase in major bleeding. Semuloparin is not currently available for clinical use, although regulatory approval is being pursued in several countries.
A second approach has been to focus on single sites of cancer known to be very high risk for VTE, particularly pancreas and multiple myeloma. Two recent RCTs focused on pancreatic cancer. In the CONKO-004 study, VTE occurred in 5% of patients randomized to enoxaparin (1 mg/kg daily for 3 months, then 40 mg daily) versus 14.5% in the observation arm (P < .01).34 In the FRAGEM study, patients were randomized to full therapeutic dose of dalteparin versus observation.35 All-type VTE during treatment (< 100 days from randomization) was reduced from 23% to 3.4% (P = .002), (risk ratio = 0.145; 95% CI, 0.035-0.612), an 85% risk reduction.
Two large studies have focused on thromboprophylaxis in myeloma. The first studied thromboprophylaxis with either LMWH or low-dose aspirin or low-fixed dose warfarin in 667 newly diagnosed myeloma patients.36 In this substudy of 2 RCTs, patients treated with 1 of 3 specific thalidomide-containing regimens were randomly assigned to receive LMWH (enoxaparin 40 mg/d), aspirin (100 mg/d), or warfarin (1.25 mg/d). VTE occurred in 5% in the LMWH group, 6.4% in the aspirin group, and 8.2% in the warfarin group (P = not significant). Only 3 major bleeding episodes were recorded. The investigators concluded that LMWH, warfarin, and aspirin are likely to be similarly effective prophylactic regimens except in elderly patients, in whom warfarin showed less efficacy than LMWH. In another study of newly diagnosed myeloma treated with lenalidomide, 342 patients in a substudy were randomized to low-dose aspirin (100 mg/d) or enoxaparin 40 mg/d. VTE occurred in 2.3% of the aspirin group and 1.2% in the LMWH group (absolute difference, 1.07%; P = .452). Given the relative efficacy, the investigators suggested that aspirin could be an effective alternative to LMWH as prophylaxis in this setting.
Novel oral anticoagulants have not been rigorously tested in prophylaxis in the outpatient setting. A recent pilot study of apixaban, a factor Xa inhibitor, evaluated tolerance in a randomized phase 2 design.37 Subjects receiving either first- or second-line chemotherapy for advanced or metastatic lung, breast, gastrointestinal, bladder, ovarian or prostate cancers, cancer of unknown origin, myeloma, or selected lymphomas were randomized to 5, 10, or 20 mg/d of apixaban or placebo for 12 weeks. The primary outcome was either major bleeding or clinically relevant nonmajor bleeding. Major bleeding occurred in 2.2% (95% CI, 0.26%-7.5%) of 93 patients receiving apixaban, with no fatal bleeds. Only 3 symptomatic VTEs occurred (all in the placebo arm). Therefore, it appears that apixaban is safe and feasible to use as outpatient prophylaxis. Effectiveness in reducing VTE requires evaluation in the phase 3 setting, and at this point apixaban cannot be recommended for clinical use in this setting.
These studies demonstrate that outpatient thromboprophylaxis is feasible, safe, and effective. Which patients should be selected for prophylaxis, however, continues to be a matter of controversy. The relatively low event rates seen in the PROTECHT and SAVE-ONCO trials are used as arguments against broad recommendations of prophylaxis for unselected cancer patients, a point of view this author agrees with. Again, the process of risk stratification becomes key. In this context, the risk score discussed earlier is not only prognostic of VTE, but also predictive of benefit from prophylaxis. When the risk score was applied to the SAVE-ONCO population, rates in the placebo arm were higher and risk reduction was therefore greater in high-risk patients (5.4% in the placebo arm vs 1.4% in the semuloparin arm, for score ≥ 3 [HR = 0.27] compared with 1.3% vs 1%, respectively, for score = 0 [HR = 0.71]).29 When the risk score was applied to the PROTECHT population in a post hoc analysis, nearly 12% of the population was in the high-risk subgroup.29 Rates of VTE in the high-risk subgroup were reduced from 11.1% (5 of 45) in the placebo arm to 4.5% in the nadroparin arm (relative risk = 0.38; 95% CI, 0.09-1.53; number need to treat, 15). High-risk patients defined by this score are currently the subject of a National Institutes of Health–sponsored prophylaxis study using dalteparin. Current guidelines have not taken into account these more recent studies and outpatient prophylaxis is recommended by ASCO and NCCN only for high-risk myeloma patients receiving thalidomide- or lenalidomide-based combination regimens.
Treatment of cancer-associated VTE: where are the data?
Extended-duration LMWH for at least 6 months is currently the standard of care for the treatment of acute DVT and PE in active cancer patients. The most robust evidence to support this comes from the Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer (CLOT) trial. This study randomized 676 cancer patients with VTE to receive initial dalteparin followed by 6 months of either dalteparin or warfarin with a target international normalized ratio of 2.5.38 Fifteen percent of patients treated with warfarin developed recurrent VTE, compared with 7.9% of patients treated with dalteparin (HR = 0.48; 95% CI, 0.30-0.77). This is an absolute risk reduction of 7.8% or a number needed to treat of 12 to prevent one recurrent VTE. Other smaller studies with tinzaparin and enoxaparin and a Cochrane systematic review all support the use of LMWH as the preferred treatment for cancer-associated VTE.39 The optimal duration of anticoagulation in cancer patients with VTE is not known, but extended anticoagulation beyond the standard 6 months should be considered, especially for those with active cancer and/or those receiving anticancer treatments. Management of recurrent VTE on anticoagulation is a difficult clinical problem with few published data. Patients on warfarin (despite the recommendation for LMWH) should be switched to LMWH therapy. For patients already on LMWH, dose escalation by 20%-25% may be beneficial.40 Inferior vena caval filters should be used conservatively and temporarily when true contraindications to anticoagulation exist.
The safety and efficacy of novel anticoagulants in the therapeutic setting is the subject of ongoing and recent trials.41,42 Unfortunately, completed studies have enrolled a very small number of cancer patients (approximately 5% of study populations) and have used warfarin, rather than LMWH, as the control arm. No cancer-specific treatment studies have been conducted. At the time of this writing, the use of novel oral anticoagulants for the treatment of cancer-associated VTE is not supported by published studies. Evidence-based changes in current treatment algorithms are eagerly awaited by clinicians and patients alike.
Acknowledgments
The author is supported by grants from the National Cancer Institute (K23 CA120587), the National Heart, Lung and Blood Institute of the National Institutes of Health (1R01HL095109-01), and the V Foundation.
Disclosures
Conflict-of-interest disclosure: The author has received research funding from Sanofi-Aventis and Leo Pharma and has consulted for and received honoraria from Sanofi-Aventis. Off-label drug use: None disclosed.
Correspondence
Alok A. Khorana, MD, 601 Elmwood Ave, Box 704, Rochester, NY 14642; Phone: 585-273-4150; Fax: 585-273-1042; e-mail: alok_khorana@urmc.rochester.edu.
