Abstract
Mrs. A. is a 73-year-old woman who has developed increasing fatigue and lower back pain over the past year. The pain limits her exercise tolerance such that she can now walk only 1 block. She is a retired schoolteacher who does volunteer efforts in her community but has limited her activities due to fatigue. Karnofsky performance status is 70%. She has a history of chronic hypertension treated with a diuretic, adult-onset diabetes mellitus treated with metformin, and hypothyroidism treated with levothyroxine. Initial evaluation reveals anemia, renal dysfunction, an elevated total protein, and an L2 compression fracture on lumbosacral radiographs. Results of initial and subsequent evaluation are shown below, and she is referred to a hematologist for further evaluation, which revealed the following: calcium 9.0 mg/dL, creatinine 3.2 mg/dL with estimated creatinine clearance using the Modification of Diet in Renal Disease equation of 15 mL/min, hemoglobin 9.6 g/dL, total protein 11 g/dL, albumin 3.2 g/dL, immunoglobulin A (IgA) λ M protein 6.8 g/dL, total IgA 7.2 g/dL, IgG 0.4g/dL, IgM 0.03 g/dL, free κ <0.01 mg/L, free λ 1000 mg/L, serum free light chain ratio <0.01, β-2–microglobulin 4.2, viscosity 3.0, lactate dehydrogenase 200 U/L, urine protein electrophoresis: 125 mg/dL with 30% M protein, and urine immunofixation: λ light chain. Skeletal bone survey showed lytic lesions in femurs and humeri and diffusely in ribs bilaterally as well as compression fractures at T4, T6, and L2. Bone marrow biopsy revealed λ-restricted plasma cells comprising 50% of the bone marrow core. Fluorescence in situ hybridization testing on marrow showed that del 17p was present in 80% of the plasma cells. Mrs. A. is informed of the diagnosis of multiple myeloma and the need for therapy. She requests consultation with 2 of the leading world experts. However, she wants to be treated near her home and does not want treatment on a clinical trial.
Learning Objectives
Appreciate that performance status and comorbidities are not adequate to encompass aging-associated vulnerabilities that impact treatment tolerance and prognosis in older adults with multiple myeloma
Apply measures of frailty to treatment considerations in older adults with multiple myeloma
Consider the intersection of treatment options and geriatric concerns in older adults with multiple myeloma from diagnosis through relapse
Introduction
In approaching initial treatment recommendations for an older patient with newly diagnosed multiple myeloma, 2 tasks should simultaneously be undertaken: staging the malignancy and “staging the aging.” Staging the myeloma will inform selection of the optimal treatment from a disease-focused perspective, assuming typical tolerance of the regimen. Staging the aging will provide insight into the patient’s physiologic aging and vulnerability to toxicity of therapy. With respect to the former, disease-focused prognostic markers can aid in categorizing the myeloma biology and inform treatment approaches. In this case, the patient has an elevated β-2–microglobulin, low albumin, normal lactate dehydrogenase, and high-risk chromosomal abnormalities, yielding Revised International Stating System stage II.1
In parallel with the malignancy staging, we must consider the patient’s physiologic age. Given the aging population and concomitant increase in the number of older adults with cancer, clinicians increasingly recognize the need to categorize the varied health status of older adults and incorporate the geriatric medicine principles into the care of older adults with cancer.2 Numerous studies have shown that Karnofsky or Eastern Cooperative Oncology Group (ECOG) performance status does not fully capture the level of functional limitation in an older adult with cancer and that comorbidities are independent from performance status.3,4 Similarly, frailty cannot be categorically equated with comorbidity or functional dependence, which are interrelated but distinct concepts.5 Recently, the American Society of Clinical Oncology issued guidelines recommending, at minimum, evaluation of function, comorbidity, falls, depression, cognition, and nutrition in older adults with cancer.6
The relevance of these guidelines in the care of older adults with myeloma is supported by the prevalence of geriatric impairments in this population. In a cohort of 869 older adults with myeloma, most of whom had an ECOG performance status of 0 or 1, 14% were dependent in 2 or more activities of daily living, and 18% were dependent in 3 or more instrumental activities of daily living.7 In a cohort of 40 patients who underwent comprehensive geriatric assessment, 62.5% were dependent on 1 or more instrumental activities of daily living. Other geriatric syndromes are extremely common as well. In a cohort of 801 patients with myeloma, common comorbidities included renal impairment (68%), cardiac impairment (45%), and pulmonary impairment (32%). In a cohort of 24 older men with myeloma, 1/2 screened positive for nutritional impairment, 1/2 were frail using the Rockwood clinical frailty scale, 1/4 were impaired in performing the Timed Up and Go Test (an objective measure of physical performance), and 29% screened positive for risk of depression.8 Falls were reported by about 30% of older adults with myeloma in a 400-patient cohort, more than in noncancer controls.9 Geriatric impairments are very common in older adults with myeloma, and their presence may influence an individual’s risk of adverse outcomes.
The International Myeloma Working Group (IMWG) has developed a model of frailty based on geriatric factors associated with adverse outcomes in older adults with myeloma (Table 1). The model was developed in a cohort of 869 older patients with myeloma enrolled in clinical trials. The model was built using factors associated with overall survival, including patient age, comorbidities, and functional status (activities of daily living/instrumental activities of daily living), to categorize patients as fit, intermediate fit, or frail.7 The resultant frailty status was shown to be associated with grade ≥3 nonhematologic adverse events, drug discontinuation, and lower overall survival. The model has since been externally validated in a cohort of 125 patients, with frail patients having a hazard ratio (HR) for mortality of 6.06 (95% confidence interval, 1.35-27.25) after adjusting for International Staging System Stage, cytogenetics, and therapy.10 Of note, when the IMWG frailty model was applied in a younger cohort (69% under age 65), frailty was associated with nonhematologic toxicity but not treatment discontinuation or overall survival.11 In our case presentation, we are unable to ascertain the patient’s IMWG frailty status with the data provided. A Karnofsky performance status of 70% is defined as “[c]ares for self; unable to carry on normal activity or do active work.” This suggests that the patient is independent in activities of daily living (eg, bathing, toileting, and transferring), but we do not know her level of independence in instrumental activities of daily living (eg, meal preparation, medication management, and finances).
Comparison of select risk prediction models relevant to older adults with multiple myeloma
| Factors associated with increased risk . | IMWG7 . | Revised Myeloma Comorbidity Index13 . | Geriatric Assessment in Hematology Scale16,17 . | Cancer and Aging Research Group Toxicity Tool*72,73 . | CRASH score†74 . | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameter . | Points . | Parameter . | Points . | Parameter . | Points . | Parameter . | Points . | Parameter . | Points . | |
| Age | Age 76-80 | 1 | 60-69 | 1 | — | ≥72 | 2 | — | ||
| Age >80 | 2 | ≥70 | 2 | — | — | |||||
| Performance/functional status | Any ADL dependence | 1 | KPS 80-90 | 2 | Gait speed ≤0.8 m/s | 1 | ≥1 falls in past 6 mo | 3 | ECOG 1-2 | 1 |
| Any IADL dependence | 1 | KPS < 70% | 3 | Any ADL dependence | 1 | Dependent in taking medications | 1 | ECOG 3-4 | 2 | |
| Limited in walking 1 block | 2 | |||||||||
| Comorbidities | Charlson Comorbidity Index ≥2 | 1 | Renal disease: eGFR < 60 | 1 | Diabetes, BMI > 25 kg/m2 | 1 | Creatinine clearance <34 mL/min | 3 | — | |
| Moderate/severe pulmonary disease | 1 | Cancer, lung disease, heart failure, or smoking‡ | ||||||||
| Medications/polypharmacy | — | — | ≥5 medications | 1 | — | |||||
| Nutrition | — | — | ≤8 on MNA-SF | 1 | — | MNA < 28 | 2 | |||
| Cognition | — | — | ≥3 errors on SPMSQ | 1 | — | MMS any errors | 2 | |||
| Psychosocial | — | — | Felt depressed 3-7 d of past week | 1 | Decreased social activities due to physical or emotional health | 1 | ||||
| Other | — | Moderate/severe frailty phenotype | 1 | Self-reported health fair or poor | 1 | Anemia | 3 | |||
| Hearing fair or worse | 2 | |||||||||
| Cytogenetics | — | Unfavorable | 1 | — | — | |||||
| Total score | Fit | 0 | Fit | 0-3 | Range | 0-8 | Range | 0-19 | Range | 0-8 |
| Intermediate fit | 1 | Intermediate | 4-6 | |||||||
| Frail | 2 | Frail | 7-9 | |||||||
| Factors associated with increased risk . | IMWG7 . | Revised Myeloma Comorbidity Index13 . | Geriatric Assessment in Hematology Scale16,17 . | Cancer and Aging Research Group Toxicity Tool*72,73 . | CRASH score†74 . | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameter . | Points . | Parameter . | Points . | Parameter . | Points . | Parameter . | Points . | Parameter . | Points . | |
| Age | Age 76-80 | 1 | 60-69 | 1 | — | ≥72 | 2 | — | ||
| Age >80 | 2 | ≥70 | 2 | — | — | |||||
| Performance/functional status | Any ADL dependence | 1 | KPS 80-90 | 2 | Gait speed ≤0.8 m/s | 1 | ≥1 falls in past 6 mo | 3 | ECOG 1-2 | 1 |
| Any IADL dependence | 1 | KPS < 70% | 3 | Any ADL dependence | 1 | Dependent in taking medications | 1 | ECOG 3-4 | 2 | |
| Limited in walking 1 block | 2 | |||||||||
| Comorbidities | Charlson Comorbidity Index ≥2 | 1 | Renal disease: eGFR < 60 | 1 | Diabetes, BMI > 25 kg/m2 | 1 | Creatinine clearance <34 mL/min | 3 | — | |
| Moderate/severe pulmonary disease | 1 | Cancer, lung disease, heart failure, or smoking‡ | ||||||||
| Medications/polypharmacy | — | — | ≥5 medications | 1 | — | |||||
| Nutrition | — | — | ≤8 on MNA-SF | 1 | — | MNA < 28 | 2 | |||
| Cognition | — | — | ≥3 errors on SPMSQ | 1 | — | MMS any errors | 2 | |||
| Psychosocial | — | — | Felt depressed 3-7 d of past week | 1 | Decreased social activities due to physical or emotional health | 1 | ||||
| Other | — | Moderate/severe frailty phenotype | 1 | Self-reported health fair or poor | 1 | Anemia | 3 | |||
| Hearing fair or worse | 2 | |||||||||
| Cytogenetics | — | Unfavorable | 1 | — | — | |||||
| Total score | Fit | 0 | Fit | 0-3 | Range | 0-8 | Range | 0-19 | Range | 0-8 |
| Intermediate fit | 1 | Intermediate | 4-6 | |||||||
| Frail | 2 | Frail | 7-9 | |||||||
ADL, activity of daily living; BMI, body mass index; CRASH, Chemotherapy Risk Assessment for High Age Patients; eGFR, estimated glomerular filtration rate; IADL, instrumental activity of daily living; KPS, Karnofsky Performance Status; MMS, Mini Mental Status Exam; MNA, Mini Nutritional Assessment; MNA-SF, Mini Nutritional Assessment—Short Form; SPMSQ, Short Portable Mental Status Questionnaire.
Developed and validated in solid tumor malignancies; original model includes points for gastrointestinal or genitourinary malignancies and chemotherapy type.
Developed and validated in primarily solid tumor malignancies; original model includes points for chemotherapy type. Variables listed are in the model for nonhematologic toxicity of chemotherapy.
Another frailty model, the Revised Myeloma Comorbidity Index (R-MCI), predicts survival in older adults with myeloma. The R-MCI incorporates specific comorbidities (ie, renal or pulmonary disease), Karnofsky performance status, age, frailty as defined by Fried et al,12 and cytogenetics (Table 1). This model was developed in 552 patients with a median age of 62 and validated in 249 patients with a median age of 63. Using this model, patients categorized as frail had a 9-fold greater risk of death compared with those categorized as fit (HR, 9.57; 95% confidence interval, 6.52-14.03).13 When the R-MCI and IMWG were directly compared in a cohort of 125 patients with myeloma, patients categorized as frail using the IMWG model had an HR for mortality of 6.06, whereas patients categorized as frail by the R-MCI had an adjusted HR for mortality of 8.34 (95% confidence interval, 1.69-41.17).10 In our case, although we do not have data on the patient’s frailty phenotype12 (which requires data on grip strength, weight loss, gait speed, self-reported exhaustion, and low physical activity), she would already be categorized as frail with the R-MCI based on her renal impairment, age, performance status, and cytogenetics, and she would be expected to have a greater risk for mortality.
Additional models incorporating components of geriatric assessment have been developed and validated in myeloma and other cancer populations. A recently published model applied the Accumulation of Deficits approach to operationalizing frailty in older adults with myeloma and showed that frailty was prognostic, with HR for mortality of 1.63 (95% confidence interval, 1.26-2.11).14,15 The Geriatric Assessment in Hematology scale16,17 incorporates domains not assessed in the IMWG or R-MCI frailty models, including gait speed, nutrition, cognition, and depression, which as mentioned above, are prevalent in older adults with myeloma. It was developed in 164 patients with multiple myeloma, chronic lymphocytic leukemia, myelodysplastic syndrome, and acute myeloid leukemia; it has criterion validity, although data on its utility in predicting chemotherapy toxicity and survival are awaited. The Cancer and Aging Research Group Risk Prediction Tool and the Chemotherapy Risk Assessment for High Age Patients Score have been widely adopted in the geriatric oncology literature as tools to predict toxicity of chemotherapy, although their utility in hematologic malignancies is unknown.6
There is no single optimal regimen for all older patients with newly diagnosed multiple myeloma. The suggested induction regimen for this patient is influenced by both disease-related factors and her current functional status. Her high-risk cytogenetic abnormality with 17p deletion warrants consideration of a 3-drug bortezomib-based combination regimen. The IMWG consensus on the treatment of multiple myeloma with high-risk cytogenetics and the European Myeloma Network consensus on practice in older adults with myeloma both concluded that bortezomib-based regimens may partly overcome the adverse prognostic effect of 17p deletion.18,19 In the SWOG S0777 trial of lenalidomide, bortezimib, and dexamethasone (RVd) vs lenalidomide and dexamethasone (Rd), the median progression-free survival was 38 months in the high-risk patients treated with RVd compared with 16 months in the high-risk patients treated with Rd.20 In a recent review of frontline therapies for older adults with high-risk cytogenetics, Avet-Loiseau and Facon21 note that, among patients with high-risk cytogenetics, progression-free and overall survival tended to be longer with triplet regimens. Table 2 highlights regimens that have been evaluated in as frontline therapy in older adults with multiple myeloma, including response rates, progression-free survival, overall survival, and toxicities.
Range of reported outcomes from trials for patients with newly diagnosed multiple myeloma who are transplant ineligible
| Regimen . | Overall response rate, % . | Complete response rate, % . | Median PFS, mo . | Median OS, mo . | 3-y OS in high-risk subset, % . | Early deaths/death due to toxicity, % . | Treatment discontinuation due to adverse events, % . | Grade ≥3 fatigue, % . | Grade ≥3 neuropathy, % . |
|---|---|---|---|---|---|---|---|---|---|
| Proteosome inhibitor based | |||||||||
| VD/VP24,25 | 64-73 | 3-8 | 14.0-14.7 | 49.8 | NR | NR | 29 | 11 | 22 |
| VMP24,25,35,75-78 | 70-86 | 4-30 | 17.1-31 | 74%-87% 3-y OS | 56.1 | 3-6 | 2.3-34 | 2-8 | 7-20 |
| VCD-lite/VCP25,79 | 64-67 | 2-29 | 15.2-24.2 | 29.7 | NR | 7.1-7.9 | 14 | 6.1 | 6.1 |
| Immunomodulatory agent based | |||||||||
| Rd36,41,80-82 | 70-81 | 3-22 | 8.9-25.3 | 30.5-62.3 | NR | 4.6 | 7-19 | 2-11 | 0-2 |
| MPR80,83 | 68 | 3-11 | 14-24 | 62% 3-y OS | NR | 0.7-2.3 | 4-18 | 2-3 | 0-3 |
| MPR+R maintenance83-85 | 70.4-84 | 11.2-16 | 18.7-31 | 69%-70% 3-y OS | NR | 2 | 16-41 | 5 | 0-2 |
| CPR80 | 74 | 0.5 | 20 | 68% 4-y OS | NR | 3.6 | 15 | 2 | 3 |
| Proteosome inhibitor + immunomodulatory agent | |||||||||
| RVD-lite32 | 86 | 44 | 35.1 | NR | NR | NR | 4 | 16 | 2 |
| VMPT-VT78,86 | 89 | 38 | 35.3 | 61% 5-y OS | NR | 4 | 23 | 6 | 16.8 |
| VTD/VTP24,76,77 | 80-81 | 4-28 | 15.4-34 | 43-51.5 | 55 | 5 | 17-38 | 12 | 9-27 |
| Proteosome inhibitor + monoclonal antibody | |||||||||
| VMP-dara35 | 90.9 | 42.6 | NR | NR | NR | 3.2 | 4.9 | NR | 1.4 |
| Regimen . | Overall response rate, % . | Complete response rate, % . | Median PFS, mo . | Median OS, mo . | 3-y OS in high-risk subset, % . | Early deaths/death due to toxicity, % . | Treatment discontinuation due to adverse events, % . | Grade ≥3 fatigue, % . | Grade ≥3 neuropathy, % . |
|---|---|---|---|---|---|---|---|---|---|
| Proteosome inhibitor based | |||||||||
| VD/VP24,25 | 64-73 | 3-8 | 14.0-14.7 | 49.8 | NR | NR | 29 | 11 | 22 |
| VMP24,25,35,75-78 | 70-86 | 4-30 | 17.1-31 | 74%-87% 3-y OS | 56.1 | 3-6 | 2.3-34 | 2-8 | 7-20 |
| VCD-lite/VCP25,79 | 64-67 | 2-29 | 15.2-24.2 | 29.7 | NR | 7.1-7.9 | 14 | 6.1 | 6.1 |
| Immunomodulatory agent based | |||||||||
| Rd36,41,80-82 | 70-81 | 3-22 | 8.9-25.3 | 30.5-62.3 | NR | 4.6 | 7-19 | 2-11 | 0-2 |
| MPR80,83 | 68 | 3-11 | 14-24 | 62% 3-y OS | NR | 0.7-2.3 | 4-18 | 2-3 | 0-3 |
| MPR+R maintenance83-85 | 70.4-84 | 11.2-16 | 18.7-31 | 69%-70% 3-y OS | NR | 2 | 16-41 | 5 | 0-2 |
| CPR80 | 74 | 0.5 | 20 | 68% 4-y OS | NR | 3.6 | 15 | 2 | 3 |
| Proteosome inhibitor + immunomodulatory agent | |||||||||
| RVD-lite32 | 86 | 44 | 35.1 | NR | NR | NR | 4 | 16 | 2 |
| VMPT-VT78,86 | 89 | 38 | 35.3 | 61% 5-y OS | NR | 4 | 23 | 6 | 16.8 |
| VTD/VTP24,76,77 | 80-81 | 4-28 | 15.4-34 | 43-51.5 | 55 | 5 | 17-38 | 12 | 9-27 |
| Proteosome inhibitor + monoclonal antibody | |||||||||
| VMP-dara35 | 90.9 | 42.6 | NR | NR | NR | 3.2 | 4.9 | NR | 1.4 |
CPR, cyclophosphamide, prednisone, and lenalidomide; MPR, melphalan, prednisone, and lenalidomide; MPR+R, melphalan, prednisone, and lenalidomide with lenalidomide maintenance; NR, not reported; OS, overall survival; PFS, progression-free survival. VCD-lite, weekly bortezomib, cyclophosphamide, and dexamethasone; VCD, bortezomib, cyclophosphamide, and dexamethasone; VD, bortezomib and dexamethasone; VMP, bortezomib, melphalan, and prednisone; VMP-dara, bortezomib, melphalan, prednisone, and daratumumab; VMPT-VT, bortezomib, melphalan, prednisone, and thalidomide with bortezomib and thalidomide maintenance; VP, bortezomib and prednisone; VTD, bortezomib, thalidomide, and dexamethasone; VTP, bortezomib, thalidomide, and prednisone.
Although a triplet would be optimal, our patient’s current functional limitations warrant some caution with a triplet regimen at standard dosing. We know that patients enrolled in clinical trials tend to have better performance status, fewer comorbidities, and better survival than those ineligible for clinical trials, and therefore, the data from clinical trials may not directly apply to every patient in clinic.22,23 Indeed, in studies that include patients who are more vulnerable than those typically enrolled in trials of transplant-ineligible patients, survival with 3-drug regimens was similar to that of 2-drug regimens due to increased rates of toxicity.24,25
Given that the patient meets criteria for frailty based on the R-MCI, she may be at greater risk for toxicities of therapy. We know that early discontinuation of therapy due to toxicity is associated with shorter survival.26 Although the approach has not been validated in multiple myeloma, the “start low—go slow” principle used in geriatrics has been applied in treatment trials in other malignancies. In this approach, empiric initial dose modifications are followed by dose escalation if the older patient tolerates the initial dosing well.27 Empiric dose modifications based on vulnerabilities in older adults with myeloma have been proposed and are a reasonable approach (Table 3),19,28-31 although they await prospective evaluation of their impact on toxicity and efficacy. In this case, I would proceed initially with weekly subcutaneous bortezomib and dexamethasone alone, with the anticipation that there would be improvement in her renal function and possibly, her functional status as her pain improves, thereby allowing us to escalate to a triplet regimen.
Suggested dose modifications for older adults with myeloma
| Drug . | Dose level 0 . | Dose level − 1* . | Dose level − 2* . |
|---|---|---|---|
| Dexamethasone | 40 mg days 1, 8, 15, 22 every 4 wk | 20 mg days 1, 8, 15, 22 every 4 wk | 10 mg days 1, 8, 15, 22 every 4 wk |
| Or 20 mg on day of and day after bortezomib87 | Or 10 mg on day of and day after bortezomib | ||
| Prednisone | 2 mg/kg days 1-4 of 4- to 6-wk cycle | 1 mg/kg days 1-4 of 4- to 6-wk cycle | 0.3-0.5 mg/kg days 1-4 of 4- to 6-wk cycle |
| Or 60 mg/m2 days 1-4 of 6-wk cycle | Or 30 mg/m2 days 1-4 of 6-wk cycle | Or 10-15 mg/m2 days 1-4 of 4- to 6-wk cycle | |
| Bortezomib | 1.3 mg/m2 days 1, 4, 8, 11 every 3 wk | 1.3 mg/m2 days 1, 8, 15, 22 every 5 wk | 1.0 mg/m2 days 1, 8, 15, 22 every 5 wk |
| Ixazomib | 4 mg days 1, 8, 15 every 4 wk | 3 mg days 1, 8, 15 every 4 wk | 2.3 mg days 1, 8, 15 every 4 wk |
| Carfilzomib | 20 mg/m2 days 1, 2, 8, 9, 15, 16 in cycle 1; 27 mg/m2 in cycle 2+ every 4 wk | 20 mg/m2 days 1, 2, 8, 9, 15, 16 in cycle 1; 27 mg/m2 days 1, 8, 15 in cycle 2+ every 4 wk | 20 mg/m2 days 1, 8, 15 every 4 wk |
| Lenalidomide | 25 mg days 1-21 every 4 wk | 15 mg days 1-21 every 4 wk | 10 mg days 1-21 every 4 wk |
| Pomalidomide | 4 mg days 1-21 every 4 wk | 3 mg days 1-21 every 4 wk | 2 mg days 1-21 every 4 wk |
| Thalidomide | 100-200 mg daily | 50-100 mg daily | 50 mg every other day; 50 mg daily |
| Melphalan | 0.25 mg/kg days 1-4 every 4-6 wk | 0.18 mg/kg days 1-4 every 4-6 wk | 0.13 mg/kg days 1-4 every 4-6 wk |
| Cyclophosphamide | 300 mg/m2 days 1, 8, 15 (±22) every 4 wk | 150 mg/m2 days 1, 8, 15 every 4 wk | 75 mg/m2 days 1, 8, 15 every 4 wk |
| Daratumumab | 16 mg/kg weekly for cycles 1-2 (4-wk cycles), then every other week for cycles 3-6, then every 4 wk | No age- or frailty-related dose modifications | No age- or frailty-related dose modifications |
| Elotuzumab | 10 mg/kg weekly for cycles 1-2 (4-wk cycles), then every other week | No age- or frailty-related dose modifications | No age- or frailty-related dose modifications |
| Drug . | Dose level 0 . | Dose level − 1* . | Dose level − 2* . |
|---|---|---|---|
| Dexamethasone | 40 mg days 1, 8, 15, 22 every 4 wk | 20 mg days 1, 8, 15, 22 every 4 wk | 10 mg days 1, 8, 15, 22 every 4 wk |
| Or 20 mg on day of and day after bortezomib87 | Or 10 mg on day of and day after bortezomib | ||
| Prednisone | 2 mg/kg days 1-4 of 4- to 6-wk cycle | 1 mg/kg days 1-4 of 4- to 6-wk cycle | 0.3-0.5 mg/kg days 1-4 of 4- to 6-wk cycle |
| Or 60 mg/m2 days 1-4 of 6-wk cycle | Or 30 mg/m2 days 1-4 of 6-wk cycle | Or 10-15 mg/m2 days 1-4 of 4- to 6-wk cycle | |
| Bortezomib | 1.3 mg/m2 days 1, 4, 8, 11 every 3 wk | 1.3 mg/m2 days 1, 8, 15, 22 every 5 wk | 1.0 mg/m2 days 1, 8, 15, 22 every 5 wk |
| Ixazomib | 4 mg days 1, 8, 15 every 4 wk | 3 mg days 1, 8, 15 every 4 wk | 2.3 mg days 1, 8, 15 every 4 wk |
| Carfilzomib | 20 mg/m2 days 1, 2, 8, 9, 15, 16 in cycle 1; 27 mg/m2 in cycle 2+ every 4 wk | 20 mg/m2 days 1, 2, 8, 9, 15, 16 in cycle 1; 27 mg/m2 days 1, 8, 15 in cycle 2+ every 4 wk | 20 mg/m2 days 1, 8, 15 every 4 wk |
| Lenalidomide | 25 mg days 1-21 every 4 wk | 15 mg days 1-21 every 4 wk | 10 mg days 1-21 every 4 wk |
| Pomalidomide | 4 mg days 1-21 every 4 wk | 3 mg days 1-21 every 4 wk | 2 mg days 1-21 every 4 wk |
| Thalidomide | 100-200 mg daily | 50-100 mg daily | 50 mg every other day; 50 mg daily |
| Melphalan | 0.25 mg/kg days 1-4 every 4-6 wk | 0.18 mg/kg days 1-4 every 4-6 wk | 0.13 mg/kg days 1-4 every 4-6 wk |
| Cyclophosphamide | 300 mg/m2 days 1, 8, 15 (±22) every 4 wk | 150 mg/m2 days 1, 8, 15 every 4 wk | 75 mg/m2 days 1, 8, 15 every 4 wk |
| Daratumumab | 16 mg/kg weekly for cycles 1-2 (4-wk cycles), then every other week for cycles 3-6, then every 4 wk | No age- or frailty-related dose modifications | No age- or frailty-related dose modifications |
| Elotuzumab | 10 mg/kg weekly for cycles 1-2 (4-wk cycles), then every other week | No age- or frailty-related dose modifications | No age- or frailty-related dose modifications |
Because of her high-risk cytogenetics, a 3-drug regimen would be preferred, potentially after a 2-drug “prephase” as described above. The combination of RVd is superior to Rd alone with respect to response rate, progression-free survival, and overall survival.20 However, the rate of grade 3 neurologic toxicity using twice weekly intravenously administered bortezomib was 33%. Because of the high rate of toxicity, a modification of this regimen, termed “RVD-lite,” was designed specifically for older adults and tested in a phase 2 study. In this study, bortezomib was administered subcutaneously and weekly for 4 weeks, with 1 week off, for 9 cycles followed by 6 cycles of consolidation.32 The overall response rate was 86%, with a median progression-free survival of 35 months. Yet, importantly, this efficacy did not come at the cost of toxicity: the rate of discontinuation due to toxicity was only 4%, and the rate of grade 3 neuropathy was only 2%. As mentioned above, early discontinuation of therapy due to toxicities is associated with poorer survival, whereas a cumulative bortezomib exposure of >39 mg/m2 (which would be 7.5 cycles of RVD-lite) is associated with superior survival33 ; therefore, initial therapeutic selection to optimize the chances of continuing therapy is ideal. Although the combination of lenalidomide and bortezomib is not specifically approved for initial therapy, its use is supported by the National Comprehensive Cancer Network guidelines34 and the IMWG consensus on the treatment of individuals with high-risk cytogenetics.18
It is a testament to the rapid advances in our field that another new regimen (bortezomib, melphalan, prednisone [VMP], and daratumumab) was approved by the Food and Drug Administration in May 2018 for older adults with newly diagnosed multiple myeloma who are transplant ineligible.35 In our patient's case, she would not have been a candidate for the ALCYONE trial due to her renal impairment at presentation, and therefore, the role for the daratumumab-VMP regimen in this patient is unclear.
Had the patient in the case presented with different features, other considerations may have informed treatment options. For example, in a patient with standard-risk cytogenetics, the RVD-lite regimen could be considered, but there may be more room for personalization of therapy. For example, if the patient expresses a strong preference for an all orally administered regimen and her renal function allowed, Rd alone, as per the FIRST trial, would be reasonable.36 In a patient who had not experienced the functional decline seen in this patient, who met criteria as outlined above for being “fit,” induction therapy followed by consolidation with high-dose therapy and autologous stem cell transplantation could be considered. Numerous studies have shown similar outcomes between younger patients and selected fit older patients who undergo high-dose therapy and autologous stem cell transplant as well as improved survival among older patients who undergo high-dose therapy and autologous stem cell transplant compared with older adults who do not undergo this treatment.37-40
It should also be noted that steroids, particularly traditional high doses of dexamethasone (ie, cumulative doses ≥480 mg per 4-week cycle), may be particularly toxic in older adults.41,42 Dose adjustments as outlined in Table 3, with lower doses of dexamethasone or the substitution of prednisone for dexamethasone, may be appropriate.41,43
In addition to selecting the initial antimyeloma regimen, we must also attend to her supportive care. The plain radiographs, which showed compression fractures in this case, should be followed with magnetic resonance imaging of the whole spine. If there is any soft tissue component concerning for impending neurologic compromise, radiation therapy may be considered to those focal areas. Although radiation and systemic therapy are generally not coadministered, phase 1 data support the safety of concomitant bortezomib and radiation therapy.44 Concomitant treatment with radiation therapy for symptomatic focal lesions and systemic therapy for systemic disease (renal insufficiency in this case) may be required in some patients. The patient may be a candidate for vertebroplasty for her symptomatic L2 lesion and will require use of a bone-modifying agent. Given her significant renal impairment, her therapeutic options include pamidronate or denosumab. Denosumab is noninferior to zoledronic acid for the prevention of skeletal-related events; however, cost is substantially higher for denosumab. Given its ease of subcutaneous administration, denosumab is an alternative to pamidronate in myeloma patients with renal insufficiency. The IMWG recommends against the use of pamidronate or zoledronic acid in patients with a creatinine clearance <30 mL/min,45 whereas the American Society of Clinical Oncology advises slowing the rate of infusion of pamidronate to 4 to 6 hours for patients with creatinine >3 mg/dL or creatinine clearance <30 mL/min.46 Current guidelines generally recommend bone-modifying therapy for 2 years.45,46
Mrs. A. opts for RVD-lite (oral lenalidomide, subcutaneous bortezomib, and dexamethasone)
After 6 monthly cycles, reevaluation reveals M protein by serum protein electrophoresis: none; immunoglobulin A (IgA) quantification: 0.1 g/dL; β-2–microglobulin 1.4; creatinine 1.2 mg/dL; free κ 15 mg/L; free λ 25 mg/L; serum free light chain ratio 0.60 (normal); and UPEP: only trace λ.
This follow-up data show improvement in the patient’s renal function, although it is important to note that, despite her normal serum creatinine, her glomerular filtration rate using either the Chronic Kidney Disease Epidemiology Collaboration (CKD-Epi) or the Modification of Diet in Renal Disease (MDRD) equation is still <60 mL/min per 1.73 m2. In addition, her monoclonal paraprotein is negative, but given her positive urine immunofixation, she is in very good partial response. As in younger patients, depth of response in older patients correlates with progression-free and overall survival.47 Regarding duration of therapy, in the phase 2 study of RVD-lite, the initial treatment plan was for 9 35-day cycles. As long as she is tolerating the regimen without significant neuropathy, RVD-lite may be continued to increase her depth of response. However, the cumulative sensory neuropathy from bortezomib may interfere with daily functions and contribute to an increased risk of falls. Continued conversation about the patient's goals of care, preferences, and values will ensure that we do not induce toxicity that hastens functional decline in our pursuit of greater control of the myeloma.
Mrs. A. has an excellent response and opts for continued therapy for 6 more cycles
Serum M protein is not detectable, IgA is 0.5 g/dL, and serum immunofixation is negative. However, she does have grade 2 peripheral neuropathy.
The patient has attained a complete response. Achieving minimal residual disease (MRD) negativity is associated with better outcomes, overcoming the negative prognostic impact of high-risk cytogenetics.48,49 However, despite its prognostic import, it is not known how MRD assessment should influence treatment recommendations. Several lines of evidence support continuous therapy regardless of the depth of response. In the FIRST trial, continuous therapy with lenalidomide was associated with prolonged progression-free survival compared with fixed duration therapy, although it should be noted that overall survival was similar with fixed duration lenalidomide or continuous lenalidomide.36 In a pooled analysis of 3 phase 3 trials, over 1300 patients were randomized to therapy that was either of fixed duration or continuous. Continuous therapy was associated with prolongation of progression-free survival and overall survival, with an HR of 0.69 in the overall survival analysis.50 Thus, continuous treatment should be recommended as long as significant toxicity does not develop. Given that the patient now has grade 2 peripheral neuropathy, it would be appropriate to discontinue bortezomib and continue with lenalidomide alone.
Mrs. A. is treated with maintenance lenalidomide at 15 mg by mouth daily for 21 of 28 days
She remains in remission for 2.5 years but then, develops new lytic bone lesions with M protein 0.5 g/dL.
The patient has a number of therapeutic options at this point. With multiple new agents and different drug classes, there is tremendous flexibility to tailor a regimen, taking into account her prior treatment, residual toxicities of prior therapy, and her current state of health. The agents that she has not yet been exposed to include carfilzomib, ixazomib, thalidomide, pomalidomide, elotuzumab, and daratumumab as well as the conventional alkylating agents melphalan and cyclophosphamide. Given that her myeloma is symptomatic with new bony lesions and refractory to lenalidomide, ixazomib and elotuzumab are less attractive options, because they are approved in combination with lenalidomide.51,52 If her prior peripheral neuropathy has resolved, her disease may still be sensitive to bortezomib, and the combination of bortezomib, daratumumab, and dexamethasone could be considered53 as could the combination of pomalidomide, bortezomib, and dexamethasone.54 However, about 1/3 of patients with grade ≥2 neuropathy will not have improvement in neuropathy after discontinuing therapy.55 If she were exposed to further bortezomib and her neuropathy worsened, as a grade 3 neuropathy, this toxicity would mean that the neuropathy is now interfering with her ability to independently complete her activities of daily living. Patient preferences become an important consideration: 59% of older adults with cancer prioritize maintaining their independence over length of life.56 An attractive therapeutic option with less neurotoxicity would be the combination of pomalidomide, daratumumab, and dexamethasone, which was recently approved in the United States, with an overall response rate of 60% and a median progression-free survival of 8.8 months.57 Given that our patient is receiving this regimen in her second line of therapy, her likelihood of response and duration of benefit may be even greater than the typical patient in that trial given that patients who received the combination in second or third line tended to have a higher response rate than those receiving it later in their course.
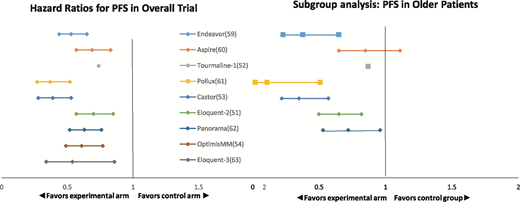
Therapeutic trials in patients with relapsed/refractory myeloma typically do not have age-based eligibility restrictions, and the outcomes of the older patients enrolled in the pivotal trials are typically similar to those of the younger patients enrolled (Figure 1).51-54,58-63 However, the proportion of older adults enrolled tends to be small; the trials tend to have exclusion criteria based on performance status, renal function, and cardiac and pulmonary comorbidities that de facto exclude older patients.58 The small proportion of older adults who are enrolled may not be representative of the older population with relapsed/refractory myeloma in general. Extrapolating from the pivotal trials and applying the results (both disease control and toxicities) to older adults who would not have been eligible for trials may overestimate their benefit and underestimate toxicity.22,23
Comparison of HRs for progression-free survival in overall study population vs older subgroup in recent randomized trials in relapsed/refractory multiple myeloma. Endeavor trial: carfilzomib-dexamethasone vs bortezomib-dexamethasone (older subgroup ≥75, N = 143); Aspire trial: carfilzomib-lenalidomide-dexamethasone vs l lenalidomide-dexamethasone (older subgroup ≥65, N = 393); Tourmaline-1 trial: ixazomib-lenalidomide-dexamethasone (older subgroup >75, N = 108); Pollux trial: daratumumab-bortezomib-dexamethasone vs bortezomib-dexamethasone (older subgroup ≥75, N = 64); Castor trial: daratumumab-lenalidomide-dexamethasone vs lenalidomide-dexamethasone (older subgroup ≥65, N = 241); Eloquent-2 trial: elotuzumab-lenalidomide-dexamethasone vs lenalidomide-dexamethasone alone (older subgroup ≥65, N = 370); Panorama trial: panobinostat-bortezomib-dexamethasone vs bortezomib-dexamethasone (older subgroup ≥65, N = 323); OptimisMM trial: pomalidomide-bortezomib-dexamethasone vs bortezomib-dexamethasone (no subgroup analysis published); and Eloquent-3 trial: elotuzumab-pomalidomide-dexamethasone vs pomalidomide-dexamethasone alone (no subgroup analysis published).
Comparison of HRs for progression-free survival in overall study population vs older subgroup in recent randomized trials in relapsed/refractory multiple myeloma. Endeavor trial: carfilzomib-dexamethasone vs bortezomib-dexamethasone (older subgroup ≥75, N = 143); Aspire trial: carfilzomib-lenalidomide-dexamethasone vs l lenalidomide-dexamethasone (older subgroup ≥65, N = 393); Tourmaline-1 trial: ixazomib-lenalidomide-dexamethasone (older subgroup >75, N = 108); Pollux trial: daratumumab-bortezomib-dexamethasone vs bortezomib-dexamethasone (older subgroup ≥75, N = 64); Castor trial: daratumumab-lenalidomide-dexamethasone vs lenalidomide-dexamethasone (older subgroup ≥65, N = 241); Eloquent-2 trial: elotuzumab-lenalidomide-dexamethasone vs lenalidomide-dexamethasone alone (older subgroup ≥65, N = 370); Panorama trial: panobinostat-bortezomib-dexamethasone vs bortezomib-dexamethasone (older subgroup ≥65, N = 323); OptimisMM trial: pomalidomide-bortezomib-dexamethasone vs bortezomib-dexamethasone (no subgroup analysis published); and Eloquent-3 trial: elotuzumab-pomalidomide-dexamethasone vs pomalidomide-dexamethasone alone (no subgroup analysis published).
She was treated with pomalidomide, dexamethasone, and daratumumab and achieved partial response
Relapse is observed with new lower back pain and L4 compression fracture.
At the time of myeloma progression, restaging the malignancy should be accompanied by “restaging the aging.” Although the frailty models described at the start of the case were developed and validated in older adults with newly diagnosed multiple myeloma, the same principles likely apply. In our case, about 5 years have intervened since the initial presentation. Our patient is now 78 years old. In addition to residual toxicities of prior therapy and the impact of the disease itself on her overall health, other aging-associated conditions may have developed. Her preexisting hypertension may have progressed to diastolic congestive heart failure, which would make carfilzomib problematic given its association with cardiac toxicities.64 Similarly, her underlying neuropathy may put her at greater risk for worsening peripheral neuropathy related to thalidomide.65 If she has developed cognitive impairment, this may be a barrier to adherence to an all orally administered regimen, such as ixazomib and dexamethasone with or without cyclophosphamide.66-68 Her social support network may now be more limited; in a survey of older caregivers of patients with cancer, over 2/3 were themselves experiencing poor health or serious health conditions, which may present a barrier to frequent visits for parenteral therapy if she is dependent on a caregiver for transportation.69 Formal assessment of the patient's comorbidities, functional status, medications, history of falls, cognition, psychosocial status, and social support will be essential in considering subsequent therapeutic options.6
Clinical trials are an important option at every therapeutic juncture. Unfortunately, older adults are systematically underenrolled in clinical trials.70 This is less commonly due to specific age restrictions, but rather, it is de facto exclusion related to comorbidities or excluded medications required for comorbid conditions. For example, our patient's creatinine clearance of <60 mL/min is a common exclusion criteria for clinical trials, even when the study drug is not renally cleared. Overly restrictive eligibility criteria limit the generalizability of results; recognition of this has led to calls to broaden inclusion criteria and provide clear rationale for exclusions.71
In summary, the approach to the care of an older adult with myeloma should include incorporation of the principles of geriatrics interwoven with disease-focused considerations. Models of frailty, incorporating just a few additional items not gathered in routine oncology assessment, can aid is stratifying the patient’s risk of toxicity and mortality and inform shared decision making.
Correspondence
Tanya M. Wildes, Division of Medical Oncology, Washington University School of Medicine, 660 South Euclid Ave, Campus Box 8056, St. Louis, MO 63110; e-mail: twildes@wustl.edu.
References
Competing Interests
Conflict-of-interest disclosure: T.M.W. has received research funding from Janseen.
Author notes
Off-label drug use: None disclosed.