Abstract
Matched unrelated donors (URD) are the most frequent source of stem cells for allogeneic hematopoietic cell transplantation (HCT) to date, with HCT performed mainly under conventional immunosuppression by methotrexate and cyclosporine. In this setting, every single allelic donor–recipient mismatch for HLA-A, -B, -C, -DRB1 (8/8), but not for HLA-DQB1, -DPB1, has a significant negative effect on overall survival (OS). When several 8/8 HLA-matched URD are available, donor age is the most important factor impacting OS. Moving forward from the traditional way of counting the number of donor–recipient HLA allele mismatches to biology-driven algorithms for functional matching has led to the unraveling of an association between permissive, low-risk HLA-DPB1 mismatches and improved outcome after URD HCT for malignant disease but not for nonmalignant disease. Functional HLA matching might prove to have increasing importance for URD selection in the era of new immunosuppressive regimens that have the potential to substantially reshuffle the role of HLA mismatches in URD HCT.
Learning Objectives
Appreciate that HLA-A, -B, -C, -DRB1 donor–recipient mismatches reduce the probability of overall survival in unrelated HCT under conventional immune prophylaxis
Learn that when several 8/8 HLA-matched URD are available, donor age is the most important factor influencing survival
Understand the functional matching can identify permissive, low-risk HLA-DPB1 disparities associated with improved survival in the presence of reduced relapse and GvHD after HCT for malignant disease but not for nonmalignant disease
Bear in mind that novel immunosuppressive regimens with posttransplant cyclophosphamide might reshuffle the role of HLA mismatches in URD HCT, emphasizing the need for new biology-driven approaches to functional matching
Introduction
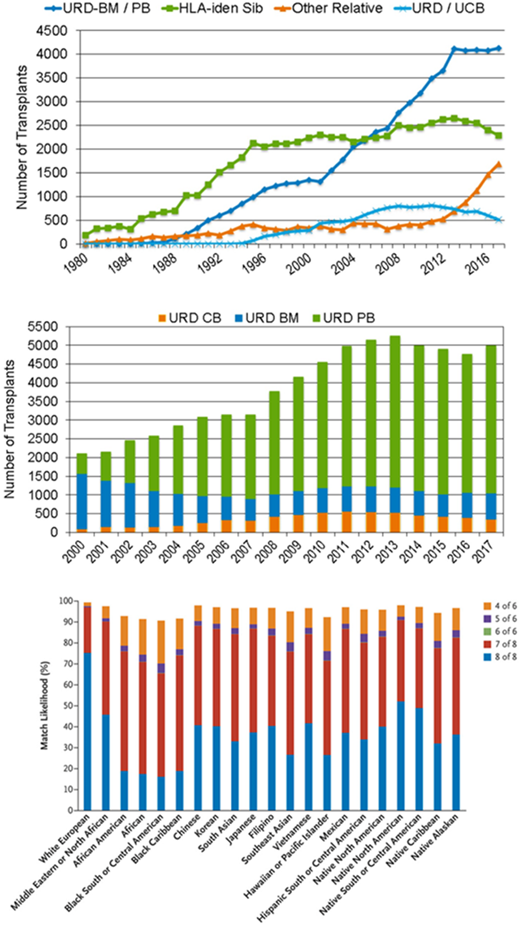
Hematopoietic cell transplantation (HCT) from unrelated donors (URD) has come a long way since the first such transplant was performed for acute leukemia back in 1979.1 Reflecting both the decreasing number of siblings in families from the Western world and the increasing safety of HCT due to improved immune suppression regimens as well as refined tissue-typing techniques, URD have become the preferred source of hematopoietic stem cells (HSC) for well over a decade.2 The significant rise in HCT from haploidentical donors over the last 5 years has been at the expense of HLA-identical sibling and umbilical cord blood donors, but not of URD, whose numbers remain stable (Figure 1A). The rise in URD HCT was paralleled by an increasing use of peripheral blood stem cells (PBSC) over bone marrow (BM) in the last decade (Figure 1B). In the same time frame, the number of available adult volunteers enrolled in the worldwide registries has dramatically increased, partly due to modern techniques of high-resolution HLA tissue typing by next-generation sequencing, reaching an all-time record of more than 35 million donors in 2019 (World Marrow Donor Association [WMDA]; https://statistics.wmda.info/). Hence, many patients, particularly those of white European descent, have a high likelihood to identify one or several well-matched URD3 (Figure 1C). Clinicians are therefore frequently spoiled for choice in routine donor selection, and evidence-based URD–recipient matching criteria represent a clinical need. The National Marrow Donor Program (NMDP) is providing guidelines for donor selection that are updated on a regular basis.4 These take into account both HLA and non-HLA factors, including recent advancements in the understanding of the biology of beneficial and adverse immune effects in URD HCT, which are paving the way toward new strategies of functional matching. The present educational review highlights these issues, initiating with a short paragraph on the HLA nomenclature and typing techniques, knowledge of which is necessary to understand the relevant messages on URD selection.
Donor type, HSC source, and URD availability for allogeneic HCT. (A) Donor type. Shown are the numbers of HCTs from URD, HLA-identical siblings, other relatives (ie, mainly haploidentical family), and umbilical cord blood performed in the United States between 1980 and 2017.2 (B) HSC source. Shown are the numbers of URD HCTs from BM, PBSC, and umbilical cord blood performed in the United States between 2000 and 2017.2 (C) URD availability. Shown is the probability of finding a URD at different levels of HLA matching for adult patients (>20 years of age) from different ethnic groups.3
Donor type, HSC source, and URD availability for allogeneic HCT. (A) Donor type. Shown are the numbers of HCTs from URD, HLA-identical siblings, other relatives (ie, mainly haploidentical family), and umbilical cord blood performed in the United States between 1980 and 2017.2 (B) HSC source. Shown are the numbers of URD HCTs from BM, PBSC, and umbilical cord blood performed in the United States between 2000 and 2017.2 (C) URD availability. Shown is the probability of finding a URD at different levels of HLA matching for adult patients (>20 years of age) from different ethnic groups.3
HLA typing terminology and techniques
The history of HCT is inextricably bound to the history of HLA, and not surprisingly, the discovery of both fields was honored by the Nobel Prize, awarded to Jean Deausset in 1980 and to Donnall Thomas in 1990.5 The observation that histocompatibility matching between recipient and donor is required for the engraftment of allogeneic HSC and the prevention of lethal graft-versus-host disease (GVHD) was paralleled by the increasing appreciation of the polymorphism of HLAs on white blood cells in the 1960s. HLA was soon to be recognized as the most polymorphic gene system in eukaryotes, and to date 17 191 HLA-A, -B, -C class I and 6716 HLA-DRB1, -DQB1, -DPB1 class II alleles have been described (IPD-IMGT/HLA database, release 3.37.0, 2019-07-10).6
HLA molecules are cell surface αβ-heterodimers of 2 immunoglobulin chains, of which only 1 carries allelic variability. For HLA class I, the polymorphic α-chain (heavy chain) is encoded by the A, B, C loci in the major histocompatibility complex (MHC) on the short arm of chromosome 6, whereas the nonpolymorphic β-chain (light chain) is encoded on the long arm of chromosome 15 (ie, outside of the MHC).7 In contrast, the α- and β-chains of HLA class II have similar molecular weight and are both encoded in the MHC; however, the HLA-DR α-chain is not polymorphic, in contrast to the highly polymorphic HLA-DR β-chain. Therefore, HLA typing for HCT donor selection generally is focused on the polymorphic α-chains of HLA class I A, B, C and β-chains of HLA class II DRB, DQB1, DPB1.
HLA typing was initially performed using serological antibodies, which had led to their discovery as an antigen system on white blood cells.8 On the basis of antibody reactivity, a limited number of approximately 100 serological types were identified, which are used in the serological typing terminology. With the advent of molecular typing techniques in the 1990s, a parallel molecular terminology was introduced (details can be found in the nomenclature reports at http://hla.alleles.org).9 Molecular nomenclature is characterized by an asterisk (*) after the HLA locus designation, missing in the serology nomenclature, and up to 4 so-called fields divided by colons (:). These fields designate the allele group, generally corresponding to the serological epitopes (first field), a specific HLA protein with a unique amino acid sequence (second field), a synonymous nucleotide substitution within the coding region (third field), and differences in the noncoding region (fourth field).
The term “HLA typing at low resolution” refers to the first field and is equivalent to serological resolution, achievable both by serological typing and by molecular standard techniques. The term “HLA typing at high resolution” generally refers to the second field and can be achieved by conventional molecular typing techniques (sequence-specific priming or hybridization, Sanger sequencing), as well as by high-throughput targeted next-generation sequencing. The latter has led to a dramatic improvement in the level of typing resolution for donors enrolled in the registries, particularly in the Western world, which in turn contributes to shortening URD search times.7 Recently, the advent of so-called third-generation sequencing of long DNA stretches has allowed high-throughput typing of the entire exon–intron region of HLA genes, resulting in “HLA typing at ultra-high resolution” with unambiguous typing of all 4 fields.10 Most available evidence on the role of HLA in URD HCT, however, is based on second-field high-resolution typing, and the term “allele matching” therefore is used to refer to this level of resolution in the present review.
Clinical case
A 60-year-old white man in good overall clinical condition is scheduled for allogeneic HCT to treat high-risk acute myeloid leukemia in first complete remission (CR1). Owing to the lack of a suitable HLA-matched sibling, a URD search is initiated. The patient is typed at high resolution for HLA-A, -B, -C, -DRB1, -DQB1, and -DPB1. Among 4 donors with second-field 6 loci typing already available, 3 are 8/8 and one is 7/8 matched to the patient (Table 1). The latter is 28 years of age, and the 8/8 donors are 40 to 45 years old. All 8/8 donors are matched also for HLA-DQB1 (10/10), and one of them is also HLA-DPB1 identical (12/12), whereas the other 2 carry an HLA-DPB1 allele mismatch in the graft-versus-host (GVH) direction. Of these, one is permissive according to the T-cell epitope 3-group model (TCE3) and low risk according to the expression single-nucleotide polymorphism proxy (SNP proxy), whereas the other one is TCE3 nonpermissive and not classifiable by the SNP proxy. A decision is made to proceed with the 10/10, HLA-DPB1 TCE3-permissive, SNP proxy low-risk URD. Ten weeks after initiation of the URD search, the transplant is performed using granulocyte colony-stimulating factor–stimulated PBSC, with the patient still in CR1. The conditioning regimen is myeloablative, and standard immune prophylaxis with short-course methotrexate and cyclosporine is used. The posttransplant course is without complications, except for grade 1 cutaneous acute GVHD on day +28, which resolves within a few days under treatment with topical corticosteroids. Cytologic and molecular analysis of the BM aspirate at discharge reveals sustained complete remission and full donor chimerism. Two years after HCT, the patient is alive and well in continued complete remission without signs of chronic GVHD.
HLA matching status of the patient in the clinical case and potential unrelated donors
| Subject . | Age (y) . | HLA-A* . | HLA-B* . | HLA-C* . | HLA-DRB1* . | HLA-DQB1* . | HLA-DPB1* . | TCE3† . | SNP proxy‡ . | HLA allele matching . |
|---|---|---|---|---|---|---|---|---|---|---|
| Patient | 60 | 03:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 04:01, 04:02 | 3/3 | A/A | |
| Donor 1 | 42 | 03:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 04:01, 04:02 | 3/3, NA | A/A, NA | 8/8 (12/12) |
| Donor 2 | 44 | 03:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 02:01, 04:02 | 3/3, P | A/A, LR | 8/8 (11/12) |
| Donor 3 | 41 | 03:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 02:01, 03:01 | 3/2, NP | A/G, NA | 8/8 (10/12) |
| Donor 4 | 28 | 02:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 14:01, 09:01 | 2/1, NP | G/G, NA | 7/8 (9/12) |
| Subject . | Age (y) . | HLA-A* . | HLA-B* . | HLA-C* . | HLA-DRB1* . | HLA-DQB1* . | HLA-DPB1* . | TCE3† . | SNP proxy‡ . | HLA allele matching . |
|---|---|---|---|---|---|---|---|---|---|---|
| Patient | 60 | 03:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 04:01, 04:02 | 3/3 | A/A | |
| Donor 1 | 42 | 03:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 04:01, 04:02 | 3/3, NA | A/A, NA | 8/8 (12/12) |
| Donor 2 | 44 | 03:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 02:01, 04:02 | 3/3, P | A/A, LR | 8/8 (11/12) |
| Donor 3 | 41 | 03:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 02:01, 03:01 | 3/2, NP | A/G, NA | 8/8 (10/12) |
| Donor 4 | 28 | 02:01, 24:02 | 07:02, 38:01 | 07:02, 12:03 | 01:01, 13:01 | 05:01, 06:03 | 14:01, 09:01 | 2/1, NP | G/G, NA | 7/8 (9/12) |
Mismatched alleles in the donor are indicated in bold.
LR, low risk because donor 2 and the recipient differ only for a single HLA-DPB1 allele and the mismatched alleles both carry the low-expressing rs9277534-A variant; NA, not applicable because donor and recipient are identical for both HLA-DPB1 alleles (donor 1) or mismatched for both HLA-DPB1 alleles (donors 3 and 4); NP, nonpermissive because one (donor 3) or both (donor 4) of the mismatched HLA-DPB1 alleles in the donor (in bold) belong to a different TCE group from the patient’s; P, permissive because the mismatched HLA-DPB1 allele in the donor (in bold) belongs to the same TCE group as the patient’s; SNP, single-nucleotide polymorphism; TCE3, T-cell epitope 3-group model.
Functional HLA-DPB1 matching by TCE3.12 The numbers indicate to which of the 3 TCE groups (1, 2, or 3) the first (before the slash) and the second (after the slash) HLA-DPB1 allele in each individual belongs. Classification by TCE3 can be performed via a tool freely available online at https://www.ebi.ac.uk/ipd/imgt/hla/dpb.html.
Functional HLA-DPB1 matching by the SNP proxy.13 The letters A and G refer to the nucleotide carried at the rs9277534 SNP by the first (before the slash) and the second (after the slash) HLA-DPB1 allele in each individual.
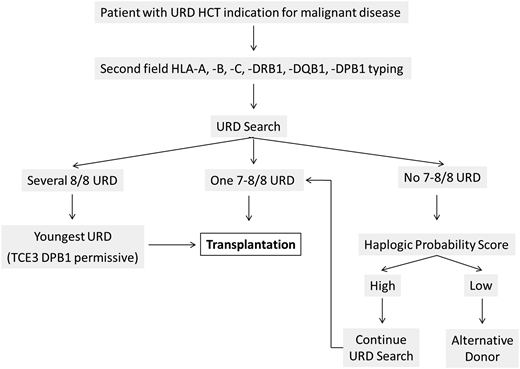
This case illustrates a standard URD selection workflow for patients with several 8/8 URD (Figure 2). At the time of medical indication for an unrelated HCT, high-resolution typing of the patient is immediately performed for all 6 HLA loci. Several well-matched URD can readily be identified for this patient of white European descent.3 The search focuses on donors with high-resolution 6 locus HLA typing already available to avoid the need for time-consuming complementary typing of the donors. The 8/8 URD are preferred over the 7/8 donor despite the younger age of the latter, because the effect of donor age is secondary to HLA-A, -B, -C, -DRB1 matching according to NMDP guidelines.4 Because there is no marked age difference between the 8/8 donors, the next focus is functional matching for HLA-DPB1. Given that the transplantation indication is high-risk acute myeloid leukemia, a donor with an HLA-DPB1 mismatch in the GVH direction is preferred over the 12/12 donor because HLA-DPB1 GVH disparity has been shown to reduce the risk of relapse.11 To keep the risk of severe GVHD limited, the donor with functional HLA-DPB1 mismatch TCE3 permissive12 and low risk according to the SNP proxy13 is selected using the relevant online DPB T-Cell Epitope tool (https://www.ebi.ac.uk/ipd/imgt/hla/dpb.html) for TCE3 selection. The timely execution of the search in conjunction with the availability of several well-matched URD has allowed the clinicians to perform the transplantation in the short period of only 10 weeks from search initiation to actual HSC infusion. The early disease status (CR1) of the patient has not progressed during that time. The limited extent of acute GVHD experienced by the patient is likely reflective of limited T-cell alloreactivity, possibly helpful to eradicate residual leukemia cells if present after conditioning. All these factors favored the long-term positive outcome for this patient.
Flowchart for the selection of URD. A patient with malignant disease indication for URD HCT should be typed at high resolution (ie, the second field for the 6 HLA loci (-A, -B, -C, -DRB1, -DQB1, -DPB1) and a donor search initiated immediately. If only one 8/8 or 7/8 donor is available, this donor can be selected. In case of several 8/8 URD, the youngest donor should be selected,4 possibly with an HLA-DPB1 mismatch in the GVH direction permissive according to TCE3 and low risk according to the SNP proxy algorithms.11 If no 8/8 or 7/8 donor is retrieved in the search, the probability of identifying such a donor should be assessed by the HapLogic probability score.23 In case of high probability, the donor search should continue if the disease status of the patient does not impose extreme urgency, such as by requesting additional HLA typing on partially typed potential candidate donors. In case of low probability or extreme clinical urgency, alternative donors should be considered. For patients with nonmalignant disease, permissive HLA-DPB1 TCE3 mismatches hold no graft-versus-leukemia advantage, and the best 8/8 donor is the youngest with allele level HLA-DPB1 matching (12/12; see text).
Flowchart for the selection of URD. A patient with malignant disease indication for URD HCT should be typed at high resolution (ie, the second field for the 6 HLA loci (-A, -B, -C, -DRB1, -DQB1, -DPB1) and a donor search initiated immediately. If only one 8/8 or 7/8 donor is available, this donor can be selected. In case of several 8/8 URD, the youngest donor should be selected,4 possibly with an HLA-DPB1 mismatch in the GVH direction permissive according to TCE3 and low risk according to the SNP proxy algorithms.11 If no 8/8 or 7/8 donor is retrieved in the search, the probability of identifying such a donor should be assessed by the HapLogic probability score.23 In case of high probability, the donor search should continue if the disease status of the patient does not impose extreme urgency, such as by requesting additional HLA typing on partially typed potential candidate donors. In case of low probability or extreme clinical urgency, alternative donors should be considered. For patients with nonmalignant disease, permissive HLA-DPB1 TCE3 mismatches hold no graft-versus-leukemia advantage, and the best 8/8 donor is the youngest with allele level HLA-DPB1 matching (12/12; see text).
HLA allele matching in unrelated HCT
For unrelated HCT, HLA typing of at least HLA-A, B, C, DRB1 should be performed at high resolution (ie, at minimum the second field). This is in contrast to HCT from related donors (HLA-identical or -haploidentical), in whom low-resolution (first field equivalent to serological resolution) typing can be sufficient if HLA genotypes can be determined by family studies.8
Initially, allogeneic HCT was performed exclusively from genotypically HLA-identical siblings, who are in most cases fully matched for 12/12 HLA class I and II alleles.1 With the advent of calcineurin inhibitors for the prevention of GVHD, allogeneic HCT from URD became increasingly feasible in the 1980s under the assumption that clinical risks were directly proportional to the number of mismatches at each of the 6 HLA loci.14 Large retrospective studies to prove this concept became feasible only in the beginning of the 2000s, with increasing numbers of unrelated HCT performed all over the world and outcome data being made available through international collaboration, streamlined in organizations such as the Center for International Blood and Marrow Transplant Research (CIBMTR) and the International Histocompatibility Working Group. This led to the first seminal observation that both antigen (first field) and allele level (second field) recipient–donor matching for HLA-A, -B, -C, -DRB1, but not for -DQB1 and -DPB1, is significantly associated with overall survival (OS), disease-free survival, and nonrelapse mortality (NRM)15 (Table 2). This association, made in the context of BM as the main graft source, was later confirmed also for PBSC16 (Table 2), as well as in a large body of subsequent studies over the last 15 years. In a recent meta-analysis, the significant negative impact on OS of a single mismatch compared with a full match at 5 HLA loci was confirmed in the pooled analysis of 5364 URD HCT from 7 different studies.17 Locus specificity of this association was consistently shown for HLA class I -A, -B, -C mismatches, whereas, interestingly, it was found in some but not all studies for HLA class II -DRB1. In particular, HLA-DRB1 mismatching was not associated with OS in Japanese patients, suggesting a possible impact of ethnicity.18 A recent study reported an association with worse clinical outcome also for ultra-high-resolution mismatches; however, these data warrant validation in independent studies.10
Multivariate survival associations of single HLA-A, -B, -C, -DRB1 mismatches in unrelated donor hematopoietic cell transplantation
| Outcome . | BM* . | PBSC† . | ||
|---|---|---|---|---|
| 7/8 (n = 985)‡ . | P value . | 7/8 (n = 803)‡ . | P value . | |
| OS | 1.25 (1.13-1.38) | <.001 | 1.26 (1.11-1.43) | <.001 |
| DFS | 1.23 (1.12-1.36) | <.001 | 1.17 (1.03-1.32) | .012 |
| NRM | 1.40 (1.25-1.56) | <.001 | 1.38 (1.15-1.66) | <.001 |
| Outcome . | BM* . | PBSC† . | ||
|---|---|---|---|---|
| 7/8 (n = 985)‡ . | P value . | 7/8 (n = 803)‡ . | P value . | |
| OS | 1.25 (1.13-1.38) | <.001 | 1.26 (1.11-1.43) | <.001 |
| DFS | 1.23 (1.12-1.36) | <.001 | 1.17 (1.03-1.32) | .012 |
| NRM | 1.40 (1.25-1.56) | <.001 | 1.38 (1.15-1.66) | <.001 |
Bold type indicates statistical significance.
BM, bone marrow; DFS, disease-free survival; NRM, nonrelapse mortality; OS, overall survival; PBSC, peripheral blood stem cells.
Data are from Lee et al.15 Baseline is the 8/8 transplants (n = 1840) against the 7/8 transplants. Ninety-two percent of hematopoietic cell transplantations were performed with BM. Models were adjusted for disease, disease stage, Karnofsky performance status, donor–patient cytomegalovirus serology, patient race and age, use of T-cell depletion, use of total body irradiation, graft source, donor age, patient–donor sex match, and year of transplantation.
Data are from Fürst et al.16 Baseline is the 8/8 transplants (n = 1511) against the 7/8 transplants. Eighty-seven percent of hematopoietic cell transplantations were performed with PBSC. Models were adjusted for disease stage, donor–patient cytomegalovirus serology, patient age, use of T-cell depletion, conditioning regimen intensity, patient–donor sex match, year of transplantation, time to transplantation, and donor origin (national vs international).
Shown are the relative risk values followed by the 95% confidence intervals in parentheses.
Role of GVHD prophylaxis
The aforementioned associations between donor–recipient HLA allele mismatches and survival endpoints (Table 2) were observed in transplants performed under conventional GVHD prophylaxis using cyclosporine and methotrexate. In this setting, the current NMDP guidelines indicate an 8/8 HLA-A, -B, -C, DRB1 matched URD as the best possible choice,4 and the donor selection in the present clinical case was based on this recommendation. It should be noted that clinical practice of posttransplant immune suppression has seen important changes over the last decade. First, centers in Europe, but not in the United States, are increasingly using in vivo T-cell depletion by antithymocyte globulin in URD HCT. However, this does not seem to significantly impact the relevance of HLA allele matching for outcome.19 Second, and importantly, the introduction of posttransplant cyclophosphamide (PtCy) has allowed the successful transplantation of T-cell–replete HSC across an entire HLA haplotype donor–recipient disparity.20 This seems to be related to the selective PtCy-mediated elimination of alloreactive T cells activated early after transplantation, together with the expansion of regulatory T cells. These clinical and experimental observations raise the possibility that PtCy might represent a game changer for the role of HLA mismatches also in URD HCT.21 Clinical exploration of PtCy in this setting is only at its beginnings, and further study aimed at unraveling the biological mechanisms and clinical effects mediated by this agent in HCT from different donor sources is clearly warranted.
Donor search time matters
The negative association between recipient–donor HLA allele mismatches and posttransplant survival was observed in multivariate analysis, independently of other patient-, donor-, and transplantation-related variables15-17 (Table 2). Nevertheless, the impact of HLA disparity was markedly greater in patients with early disease status than in those with more advanced disease status at transplantation.15,22 This shows that the timing of the donor search plays an important role in the clinical benefit of optimal matching. In the present clinical case, it was possible to initiate the transplantation procedure within only 10 weeks of the donor search start, creating favorable conditions for a positive outcome. This was certainly facilitated by the ready availability of second-field 6 loci HLA typing for 4 potentially suitable donors for this patient in the worldwide registries, obviating the need for time-intensive additional testing of only partially typed candidate donors. Unfortunately, according to the WMDA Global Trend Report 2017 (www.wmda.info), only 26.1% of the more than 35 million enrolled donors are second-field high-resolution typed for all 6 HLA loci. Additional efforts to improve the quality of donor HLA typing in the worldwide registries are clearly warranted. Moreover, clinicians should attempt to estimate the probability for their patient to identify a well-matched donor at the start of the search. In the present clinical case, the patient was of white European descent and did not present uncommon HLA haplotypes, indicating that his probability of finding an 8/8 URD was at least 75%.3 For patients from other ethnic groups, and those with uncommon HLA haplotypes, the probabilities may, in contrast, be as low as 16% and are in any case highly dependent on the specific HLA haplotype combination present in each individual patient. The NMDP has made available an online system for URD match likelihood prediction, which has recently been further refined using a Search Prognosis categorization, to facilitate personalized decisions on the prompt pursuit of alternative donors if needed.23 This tool should facilitate the decision whether to pursue a URD search or immediately opt for an alternative donor transplantation when no 7/8 or 8/8 matched URD can be identified in the initial search (Figure 2). This problem should be carefully considered also in light of the underlying disease. A faster turn to alternative donors is justified for patients with high-risk disease than for patients with lower-risk disease or even nonmalignant transplantation indication.
Donor age and other non-HLA factors
Many patients, in particular those of white European descent with common HLA haplotypes, have a choice between several 8/8 URD. In a recent analysis of 202 consecutive 10/10 HCT procedures at a single center in Germany, the search had identified more than one 10/10 URD for 189 (93.5%) of the patients (K.F., unpublished data). This is in line with observations from the NMDP showing that in >3400 patients of European, Hispanic, African American, and Asian descent who received an 8/8 URD HCT, 83% to 94% had more than one 8/8 donor to choose from (Martin Maiers, unpublished data). The important issue of how to prioritize different non-HLA factors for donor selection in these cases has been addressed in an elegant recent study from the CIBMTR aimed at constructing a hierarchical algorithm of donor characteristics associated with OS.24 The characteristics under consideration were donor age, sex, cytomegalovirus (CMV) status, blood group, race/ethnicity, parity, and HLA-DQB1 and DPB1 matching status. Interestingly, in 2 independent cohorts of 8/8 URD HCT, each comprising well over 4000 cases, donor age was the only factor consistently validated as being associated with OS in a linear fashion. These findings are in line with a previous study demonstrating significantly lower OS for each decade increase in donor age.25 On this basis, the current NMDP guidelines recommend the selection of the youngest among several 8/8 URD4 (Figure 2). Following these recommendations, the use of URD <32 years of age increased from 50% to >70% in the last decade in the United States (Michelle Kuxhausen and Stephen R. Spellman, unpublished data). According to the WMDA Global Trend Report 2017 (www.wmda.info), 38.9% of enrolled donors are <35 years old, emphasizing the need for accrual of donors among the younger population.
Regarding other non-HLA donor factors, no specific recommendations are given by the NMDP guidelines, owing to conflicting results of different studies.4 An important parameter in this context is CMV, both donor-recipient CMV serology and posttransplant CMV reactivation, the latter having been associated with increased NRM but also with reduced relapse risk for myeloid malignancies.26,27 A survival advantage for CMV-seronegative patients who received transplants from CMV-seronegative donors has been convincingly shown.28 Nevertheless, neither in the donor selection algorithm mentioned above24 nor in an independent German study with a similar scope19 could CMV serological status be graded with respect to other non-HLA factors as influencing HCT outcome. Similarly, a reduction of relapse risk by posttransplant CMV reactivation has been observed in some studies but not others,27 and it might be dependent on the absence of in vivo T-cell depletion. The mechanisms underlying this potentially relevant observation remain unclear and could be related to innate (memory natural killer [NK] cells, γδ T cells) as well as adaptive (heterologous immunity mediated by alloreactive αβ T cells) immunity and/or to direct antileukemia effects. Further study is clearly warranted to more definitively establish the role of CMV in allogeneic HCT.
Among non-HLA immunogenetic factors, the role of killer immunoglobulin-like receptors (KIRs) responsible for NK cell alloreactivity is a subject of intense research and debate. Like HLA, KIR genes are highly polymorphic, but their variation regards not only nucleotide variability but also the number of different KIR genes, with haplotypes coding for predominantly activating or inhibitory receptors.29 HLA class I antigens carrying specific epitopes serve as KIR ligands on NK cells, which are thought to undergo a process of education similar to T cells by the presence or absence of an individual’s self-HLA KIR ligand.30 Based on all this, different models for an impact of KIR in URD HCT have been developed, including missing self, missing ligand, receptor ligand, and KIR haplotype status,31 as well as more subtle associations such as the copresence of specific activating or inhibitory KIRs with specific HLA class I allotypes. To facilitate the consideration of KIR as a parameter in the donor search, KIR genotyping is being included by some laboratories in the initial profiling of donors at enrollment.32 Nevertheless, the role of KIR in URD HCT remains controversial and is possibly dependent on other competing factors such as in vivo T-cell depletion.31 More research is warranted in this important field before recommendations can be issued on how to consider KIRs in URD selection. The same holds true for other immunogenetic parameters, including nonclassical HLA-E or MHC class I chain–related genes, for which a clear consensus on their respective roles in URD HCT has yet to be reached.
Unrelated HCT for the cure of malignant blood disorders: Jekyll and Hyde
The power of allogeneic HCT to cure malignant hematologic blood disorders is dependent on the control of residual leukemia by donor immune cells infused with the graft or reconstituting after transplantation—the graft-versus-leukemia (GVL) effect. GVL is in large part mediated by alloreactive donor T cells recognizing mismatched minor or major histocompatibility antigens. Most of these antigens, however, are expressed not only by leukemia but also by the patient’s healthy tissues, and the same alloreactive donor T cells mediating GVL also cause GVHD, one of the major complications of allogeneic HCT.1 The interdependency of the 2 phenomena has been known since the early days of transplantation, when it was shown that some degree of GVHD is needed to protect patients from disease recurrence, with exceedingly high relapse rates in HCT from monozygotic twins without any major or minor histocompatibility antigen mismatches.1 The ultimate clinical success of HCT is therefore dependent on the ability to shift the balance from “Hyde” (GVHD) to “Jekyll” (GVL). A large variety of parameters can be exploited to this effect, including pharmacological approaches (ie, use of immunosuppressive agents favoring regulatory T cells), graft manipulation (ie, selective depletion of alloreactive T-cell effectors) or posttransplantation adoptive cellular therapy (ie, infusion of specific T-cell subsets such as regulatory T cells or naïve T-cell–depleted infusions).
Despite the GVL effect, relapse remains the major cause of death in patients surviving more than 100 days after allogeneic HCT. The frequency of relapse-related death is slightly higher after transplantation from an HLA-identical sibling than after URD transplantation (57% vs 48%), arguing in favor of a reduction of relapse by greater genetic disparity. In contrast, no significant differences in relapse rates after HLA-identical sibling and URD or haploidentical HCT were observed in different retrospective studies.33 This might be influenced by differences in disease characteristics, conditioning regimens, and GVHD prophylaxis and treatment between the cohorts. In URD HCT, the only 2 loci for which allele mismatches were shown to be associated with reduced relapse were HLA-C and -DPB1.33 In both cases, relapse protection was counterbalanced by increased GVHD, again reflecting the “Jekyll” and “Hyde” nature of allogeneic HCT and abrogating any survival benefit. For HLA-DPB1, this problem can at least partially be overcome by functional matching.
From HLA allele counting to functional matching
Traditionally, the risks associated with donor–recipient HLA disparity have been assumed to be directly proportional to the number of mismatches at a given locus, attributing the same potential immunogenicity to every polymorphic HLA variant.14 Numerous pieces of evidence exist to suggest that this view is too simplistic, however. First, mismatches for some (HLA-A, -B, -C, DRB1) but not other (HLA-DQB1, -DPB1) HLA loci negatively impact survival, demonstrating locus-specific functional differences.18 Second, the finding that mismatches involving specific polymorphic amino acids in HLA class I have a greater impact on transplantation outcome than others34 hints at differential immunogenicity that might be dictated by the repertoire of peptides presented in the HLA binding groove, which in turn is critically dependent on specific amino acids lining the groove.35 Third, it has been shown that HLA mismatches involving alleles with low expression levels are associated with lower risks of adverse events after HCT.36 Based on all this, 2 conceptually different but functionally similar models for permissive, low-risk HLA mismatches in URD HCT have been developed, building on structural similarity and expression levels of the mismatched HLA in the recipient, respectively. Both models have so far been tested only for HLA-DPB1 disparities present in >80% of 10/10 URD-recipient pairs,11 but they might potentially apply also to other HLA loci.
According to the first model, termed “TCE3,” HLA-DPB1 alleles can be divided into 3 different TCE groups on the basis of their structural polymorphism. HLA-DPB1 mismatches within the same TCE group elicit low levels of T-cell alloreactivity and are permissive (ie, associated with low clinical risks), whereas HLA-DPB1 mismatches across different TCE groups elicit high levels of T-cell alloreactivity and are nonpermissive (ie, associated with higher clinical risks).11 Functional TCE3 matching can be performed using an online tool freely available at https://www.ebi.ac.uk/ipd/imgt/hla/dpb.html, with recommended use of version 2.0 that was updated to the most recent evidence.35 Permissive TCE3 mismatches were shown to be associated with reduced relapse risk compared with HLA-DPB1 allele matches, but with lower severe GVHD and NRM than nonpermissive mismatches, resulting in improved OS12 (Table 3). These associations were confirmed in different independent studies.22 Based on this, the TCE3 algorithm has been incorporated into URD search tools from the NMDP and the German Registry, and its use is recommended by the current NMDP guidelines to select between several 8/8 URD4 (Figure 2). Over the years, some variations of the TCE3 model have been proposed, including the consideration of 4 groups (TCE4)37 and the calculation of a linear “delta functional distance” score between patient and donor based on the relative functional weight of their HLA-DPB1 amino acid polymorphism.11,36 However, these models have yet to be independently validated, and currently the TCE3 model is the recommended state of the art.
Multivariate models for associations between HLA-DPB1 matching status according to allele or functional matching in unrelated donor hematopoietic cell transplantation
| Outcome . | HLA-DPB1 . | |||||
|---|---|---|---|---|---|---|
| Allele* . | Functional TCE3* . | Functional SNP proxy† . | ||||
| Match (n = 1216)‡ . | P value . | NP (n = 1654)‡ . | P value . | High (n = 481)‡ . | P value . | |
| OS | 0.96 (0.87-1.06) | .40 | 1.15 (1.05-1.25) | .002 | 1.13 (0.95-1.35) | .16 |
| aGVHD grades 3-4 | 0.84 (0.69-1.03) | .09 | 1.31 (1.11-1.54) | .002 | 1.50 (1.12-2.01) | .007 |
| Relapse | 1.34 (1.17-1.54) | <.0001 | 0.89 (0.77-1.02) | .10 | 0.89 (0.68-1.17) | .40 |
| Outcome . | HLA-DPB1 . | |||||
|---|---|---|---|---|---|---|
| Allele* . | Functional TCE3* . | Functional SNP proxy† . | ||||
| Match (n = 1216)‡ . | P value . | NP (n = 1654)‡ . | P value . | High (n = 481)‡ . | P value . | |
| OS | 0.96 (0.87-1.06) | .40 | 1.15 (1.05-1.25) | .002 | 1.13 (0.95-1.35) | .16 |
| aGVHD grades 3-4 | 0.84 (0.69-1.03) | .09 | 1.31 (1.11-1.54) | .002 | 1.50 (1.12-2.01) | .007 |
| Relapse | 1.34 (1.17-1.54) | <.0001 | 0.89 (0.77-1.02) | .10 | 0.89 (0.68-1.17) | .40 |
Bold type indicates statistical significance.
aGVHD, acute graft-versus-host disease; NP, nonpermissively HLA-DPB1–mismatched transplantations; OS, overall survival; SNP, single-nucleotide polymorphism; TCE3, T-cell epitope 3-group model.
Data are from Fleischhauer et al.12 Baseline is the 8/8 TCE3 permissively HLA-DPB1–mismatched transplantations (n = 2539) against the HLA-DPB1 allele–matched (Match) transplantations or against the TCE3 nonpermissively HLA-DPB1–mismatched transplantations. Models were adjusted for disease severity, patient age, patient/donor sex and cytomegalovirus status, hematopoietic stem cell source, use of T-cell depletion, year of transplantation, conditioning regimen, and donor registry (Japanese Registry vs others).
Data are from Petersdorf et al.13 Baseline is the 8/8 transplants with a single HLA-DPB1 mismatch in the graft-versus-host direction, where the mismatched allele in the donor carried the SNP proxy A and the mismatched allele in the recipient also carried the SNP proxy A (n = 413), against the same type of pairing but where the mismatched allele in the recipient carried the SNP proxy G. Models were adjusted for disease severity, patient age, patient/donor sex and cytomegalovirus status, hematopoietic stem cell source, use of T-cell depletion, year of transplantation, and conditioning regimen.
Shown are the relative risk values followed by the 95% confidence intervals in parentheses.
The second model of low-risk HLA-DPB1 mismatches is based on expression levels predicted by the biallelic G/A rs9277534 SNP in the 3′ untranslated region,13 with high and low expression associated with the G and A variants, respectively. This SNP proxy for expression is in tight linkage disequilibrium with specific HLA-DPB1 alleles and can therefore be inferred from the HLA-DPB1 type. Interestingly, there is a good though not exclusive association between the SNP proxy variants and the aforementioned TCE groups,38 resulting in a significant though not complete overlap between high- and low-risk predictions by the TCE and SNP proxy models. Of note, the latter has been validated for transplantation with unidirectional HLA-DPB1 mismatches in the GVH direction, resulting in roughly 50% of unclassifiable pairs.37, 38 Transplantation from 8/8, low-risk SNP proxy URD was associated with significantly lower acute GVHD than high-risk SNP proxy URD in 2 independent studies,13,39 whereas no significant associations were observed with relapse and OS. Selecting an 8/8 URD with an HLA-DPB1 mismatch that is TCE3 permissive and low risk by the SNP proxy algorithms is likely to improve OS and limit acute GVHD (Figure 2).
Unrelated HCT for the cure of nonmalignant disease
Allogeneic HCT is also a powerful tool to provide a definitive cure for nonmalignant disorders, including genetic immunodeficiency syndromes or hemoglobinopathies, which represented the indication for 7.9% of such transplantations performed in Europe in 2017.40 The considerations for URD selection in this setting are different from those for the patients with malignant disease. First, the GVL effect is obviously not needed, and HLA mismatching is predominantly associated with toxic GVHD, abolishing the aforementioned rationales for functional matching and making the selection of a 12/12 matched URD the first priority. Second, graft rejection is a major concern in these patients who have a fully functional immune system and are frequently sensitized against allogeneic HLA by multiple blood transfusions. This should be considered, and donor-specific antibodies for any HLA mismatch in the HVG direction, even at HLA-DPB1, should be avoided.
Conclusions
URD HCT remains the most widely used platform of allogeneic HCT, with the largest body of evidence and the longest clinical follow-up. The NMDP guidelines for URD selection that are the basis of the present review should be regarded as not being cut in stone, because the role of histocompatibility is likely to undergo significant changes in the era of new immunosuppressive regimens. In this scenario, further development of biology-driven functional matching algorithms appears to be a promising approach to tackle URD selection in years to come.
Correspondence
Katharina Fleischhauer, Institute for Experimental Cellular Therapy, University Hospital Essen, Hufelandstrasse 55, 45122 Essen, Germany; e-mail: katharina.fleischhauer@uk-essen.de.
References
Competing Interests
Conflict-of-interest disclosure: K.F. declares no competing financial interests.
Author notes
Off-label drug use: None disclosed.