Abstract
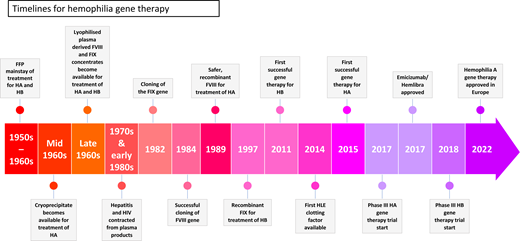
The cloning of the factor VIII (FVIII) and factor IX (FIX) genes in the 1980s has led to a succession of clinical advances starting with the advent of molecular diagnostic for hemophilia, followed by the development of recombinant clotting factor replacement therapy. Now gene therapy beckons on the back of decades of research that has brought us to the final stages of the approval of 2 products in Europe and United States, thus heralding a new era in the treatment of the hemophilias. Valoctocogene roxaparvovec, the first gene therapy for treatment of hemophilia A, has been granted conditional marketing authorization in Europe. Another approach (etranacogene dezaparvovec, AMT-061) for hemophilia B is also under review by regulators. There are several other gene therapy approaches in earlier stages of development. These approaches entail a one-off infusion of a genetically modified adeno-associated virus (AAV) engineered to deliver either the FVIII or FIX gene to the liver, leading to the continuous endogenous synthesis and secretion of the missing coagulation factor into the circulation by the hepatocytes, thus preventing or reducing bleeding episodes. Ongoing observations show sustained clinical benefit of gene therapy for >5 years following a single administration of an AAV vector without long-lasting or late toxicities. An asymptomatic, self-limiting, immune-mediated rise in alanine aminotransferase is commonly observed within the first 12 months after gene transfer that has the potential to eliminate the transduced hepatocytes in the absence of treatment with immunosuppressive agents such as corticosteroids. The current state of this exciting and rapidly evolving field, as well as the challenges that need to be overcome for the widespread adaptation of this new treatment paradigm, is the subject of this review.
Learning Objectives
Introduce basic terms and gene therapy concepts
Provide the science behind hemophilia gene therapy
Present outcomes from ongoing and completed gene therapy trials in hemophilia
Indicate limitations of gene therapy
Introduction
Hemophilia A and B are congenital bleeding disorders caused by a dysfunction or deficiency of coagulation factor (F) VIII or IX, respectively. The incidence of hemophilia A is ~1 in 5000 and that of hemophilia B is 1 in 25 000 live male births. Despite the genetic and biochemical differences, these disorders are clinically indistinguishable from each other, with the symptoms varying depending on the residual plasma coagulation factor levels. Patients who produce less than 1% (<1 IU/dL) FVIII or FIX activity have a severe phenotype that is characterized by recurrent spontaneous musculoskeletal, soft tissue, and other life-threatening bleeds such as intracranial hemorrhage, as well as excessive bleeding during and following surgery or trauma.1 Repeated episodes of intra-articular bleeding cause severe, progressive, destructive arthropathy with deformity leading to complete loss of joint function and attendant disability. Patients with moderate hemophilia have residual clotting factor levels of 1% to 5% of normal. Spontaneous bleeding episodes are infrequent in these individuals but they are still at risk from trauma-induced bleeds. In patients with mild hemophilia, bleeding is usually restricted to traumatic events, but even these individuals have an increased risk of death from intracranial bleeding when compared to those without hemophilia.2
The current standard of care for patients severely affected by hemophilia in countries with developed economies consists of regularly administered prophylaxis with factor concentrates intended to maintain a factor level above 1% of normal FVIII or FIX activity. Regular prophylaxis started in early childhood can largely prevent arthropathy.3 When continued throughout life, prophylaxis leads to near normalization of life expectancy.4 The relatively short half-life of FVIII and FIX in the circulation necessitates frequent intravenous administration of factor concentrates (at least 2-3 times a week), which is demanding and expensive, while leading to a “sawtooth” pattern of factor levels in plasma that is high immediately after infusion, falling rapidly to near baseline, which is when breakthrough bleeding can occur. Patients with hemophilia, therefore, have to carefully plan periods of increased physical activities, such as sport, which people living without hemophilia can hardly imagine.
New modified synthetic formulations of IX fused to proteins with long half-life such as albumin or Fcγ have greatly improved the stability profile.5 These extended half-life products are particularly effective in hemophilia B, allowing reduction of injection frequency to once weekly or even once biweekly while maintaining higher trough levels (FIX >5%). Efanesoctocog alfa, a FVIII-Fc fusion linked to the FVIII-binding D’D3 domains of von Willebrand factor, using XTEN linkers, has an increased half-life of at least 38 hours, raising the prospects of extended protection in patients with hemophilia A with a weekly dosage.6 Nonclotting factor products capable of securing hemostasis in patients with a bleeding diathesis are also making a significant impact. Emicizumab, an approved bispecific FVIII mimetic antibody, has significantly improved the treatment of hemophilia A. One arm of this bispecific binds to factor IXa and the other to factor X, facilitating the conversion of factor X into its active form and leading to restoration of hemostasis to a degree comparable to factor VIII level of about 15% of normal in patients with hemophilia A with or without inhibitors.7 Emicizumab has attractive pharmacokinetic attributes that allow less frequent (weekly to monthly) subcutaneous administration. Unlike FVIII, emicizumab is active in plasma all the time and is associated with microangiopathy and thrombosis, when used in combination with activated prothrombin complex concentrates.8 Approaches to rebalancing the clotting cascade by targeting tissue factor pathway inhibitor and antithrombin are in late-stage clinical trials.9
These novel therapies are beginning to change the clinical management of the hemophilias in countries with developed economies by decreasing infusion frequency, thus improving compliance with prophylaxis, offering alternatives to inhibitor patients, and easing route of administration. To date, none of these advances have affected the standard of care for 80% of patients with hemophilia, who live in parts of the world with economies in transition or development.10 These patients have little or no access to factor concentrates and have a reduced life expectancy, with very few surviving beyond their teenage years.
Rationale for gene therapy for hemophilia
Even set against this scenario of widening therapeutic choice for the hemophilias, gene therapy offers a strikingly attractive option of a cure through endogenous production of FVIII or FIX following gene transfer of a functional copy of the respective gene. The hemophilias have always been considered good candidates for gene therapy because their clinical manifestations are due to lack of a single protein that circulates in minute amounts in the bloodstream. Years of clinical experience and natural history studies show that a small increase in circulating levels of the deficient clotting factor to 5% of normal significantly modifies the bleeding diathesis. Thus, the therapeutic goal for gene therapy of hemophilia is modest in comparison to most monogenetic disorders. FVIII and FIX levels in plasma correlate well with risk of bleeding, and the levels of these proteins in circulation can be readily assessed with methods established in most routine coagulation laboratories across the world. Furthermore, tight regulation of transgene expression is not necessary since a wide range of FIX or FVIII is expected to be beneficial and nontoxic. Animal models such as FVIII- and FIX-knockout mice11-13 and dogs with hemophilia A or B14,15 have facilitated extensive preclinical evaluation of gene therapy strategies. The efficiency of therapy can be assessed easily by just measuring plasma levels of FVIII or FIX as these correlate well with the risk of bleeding.
Gene delivery vectors
The most efficient way to introduce therapeutic genes into target somatic cells, a process referred to as transduction, is to use modified, naturally occurring viruses as vectors, since they are highly evolved to transfer their own DNA in the central process of their life cycles. Adeno-associated viral (AAV) vectors are currently the vectors of choice for gene therapy of a range of monogenetic disorders, including the hemophilias. Three AAV-based biologics, including Luxturna (voretigene neparvovec-rzyl; for Leber congenital amaurosis), Zolgensma (onasemnogene abeparvovec-xioi; spinal muscular atrophy), and Glybera (alipogene tiparvovec; familial lipoprotein lipase deficiency), have received marketing approval with several other products under regulatory review. AAV vectors are derived from a small, single-stranded, DNA-based wild-type parvovirus that is thought to be nonpathogenic in humans, weakly immunogenic, and replication deficient, requiring a helper virus (usually adenovirus or herpesvirus) for replication and productive infection. It, therefore, has the best safety profile among vectors of viral origin. Additionally, recombinant AAV vectors are entirely devoid of wild-type viral coding sequences, thus reducing the potential for invoking cell- mediated immune response to foreign viral proteins. Although most of the early gene therapy studies focused on AAV2, a common human isolate that was the first to be characterized, there are now over 100 naturally occurring AAV serotypes with differing tropism and immune-biological properties available for gene therapy approaches. These vectors are effective at targeting rapidly dividing as well as postmitotic tissues such as the liver. The AAV transgene is relatively easy to manipulate, enabling the addition of stronger tissue-specific promoters and codon optimization of the transgene complementary DNA (cDNA) to improve levels of expression. Codon optimization in this setting refers to an approach to improve gene expression and increase translation efficiency of the AAV transgene by using codons that are used naturally by genes (eg, albumin) that are expressed at high levels in the liver. Similarly, AAV capsid engineering is bringing forward novel synthetic capsids capable of selective tissue tropism, higher potency, and larger packaging capacity. One limitation of AAV vectors is their small packaging size (~5.0 kb, including inverted terminal repeats) compared with other viral vectors. However, several strategies have been investigated to enable delivery of a large therapeutic gene, including the use of a shortened version of the gene that encodes a truncated but functional protein.
Hemophilia B gene therapy trials with AAV vectors
Early AAV-mediated gene therapy trials focused on hemophilia B in part because the FIX cDNA is relatively small (1.5 kb), and its expression pathway is significantly less complex than that of FVIII. The first study to show circulating therapeutic levels of FIX following AAV gene transfer was pioneered by Katherine High's group at Children's Hospital of Philadelphia (CHOP) (NCT00515710). This study entailed the administration of AAV2 vectors into the hepatic artery, following selective catheterization, to mediate efficient gene transfer of the liver of patients with severe hemophilia B. In 1 out of a total of 7 participants in this study, plasma FIX levels increased to ~10% of normal levels before unexpectedly declining to baseline values. This participant was treated at the highest dose level (2e12 vg/kg), and the decline in transgenic protein coincided with an asymptomatic, transient, 10-fold rise in liver transaminases, which returned to baseline values over the subsequent weeks, as the FIX levels declined. Decline in FIX expression associated with liver toxicity was not observed in animal models, including nonhuman primates exposed to 10-fold higher doses, and was thought to be due to a capsid-specific, cytotoxic T-cell response directed against the transduced hepatocytes.16
The first report of sustained FIX expression at therapeutic levels in patients with severe hemophilia B came from our group in a trial sponsored by St. Jude Children's Research Hospital and University College, London (UCL NCT00979238) at the start of the past decade. The key aspect that contributed to the success of this study was the use of vector pseudotyped with AAV serotype 8 capsid. This offered 2 advantages over AAV2 vectors: (1) remarkable tropism of AAV8 for liver, leading to efficient transduction of hepatocytes following administration of the vector in the peripheral circulation, thus facilitating a simple noninvasive route of vector administration that dispensed with the need for invasive procedures for catheterization of the hepatic artery in patients with a bleeding diathesis17,18 and (2) lower seroprevalence of AAV8 in humans (~25% compared with over 70% for AAV2).19 Anti-AAV antibodies have the potential to block gene transfer, and so the lower seroprevalence rate of AAV8 in humans afforded us the luxury of excluding a smaller proportion of patients with neutralizing anti-AAV8 antibodies from the clinical trial. Another differentiating aspect of this study was the use of a 2.3-kb mini-FIX expression cassette that contained a codon- optimized FIX cDNA under the control of a liver-specific promoter engineered to be packaged as a self-complementary AAV dimer in a single virion to mediate higher levels of FIX expression.
Ten subjects sequentially received a single intravenous infusion of self-complementary AAV2/8-LP1-hFIXco at a dose of 2e11 vg/kg, 6e11 vg/kg, or 2e12 vg/kg over a 2-year period between 2010 and 2012. Stable, long-term FIX expression at 1% to 8% of normal FIX activity was established in all 10 subjects. Asymptomatic, transient elevation of alanine aminotransferase (ALT) accompanied by a fall in steady-state factor IX was observed in 4 of 6 subjects recruited to the 2e12-vg/kg dose level between 6 and 10 weeks after gene transfer. Bilirubin, alkaline phosphatase, and γ-glutamyl-transferase levels remained in the normal range. Each of the 4 patients with raised liver enzymes received a short tapering course of prednisolone, leading to normalization of ALT and aspartate aminotransferase levels with preservation of FIX transgene expression in the range of 2% to 5% of normal FIX activity. Cessation of corticosteroids was not associated with a subsequent rise in ALT levels. Notably, 2 subjects treated at the high dose level had no evidence of transaminitis and did not require treatment with steroids. They both achieved stable factor IX expression of between 5% and 8% normal FIX activity. We did not observe any correlation between a specific human leukocyte antigen type and risk of transaminitis following AAV gene transfer. Transgenic FIX activity levels have remained stable in all 10 subjects over a period of follow-up that extends 10 years and is associated with a significant reduction in the annual FIX concentrate usage and frequency of spontaneous bleeding.20 Importantly, the quality of life of these individuals has improved dramatically as they are now able to undertake activities that previously provoked bleeds without suffering from bleeding episodes. No late toxicity was observed and neutralizing antibodies to FIX were not detected in any patient. Ongoing monitoring of the liver does not show any evidence of long-lasting damage.20 Therefore, the reactive use of corticosteroids upon detecting a rise in ALT, potentially suppressing an immune response–directed AAV against transduced hepatocytes, helped to preserve transgenic FIX levels long term.
The gene therapy studies that followed (Table 1) differed in their selection of AAV capsid, configuration of the vector genome, design of the expression cassette, and method of vector manufacture (mammalian system vs insect cell–baculovirus method). In general, higher vector doses were required for therapeutic transgene expression when the vector preparations were made using the insect cell–baculovirus method. For instance, AAV5 serotype pseudotyped vectors (AMT-060; UniQure Therapeutics) made using the insect cell–baculovirus method but containing the same FIX gene cassette as that used in the St Jude–UCL trial resulted in mean FIX activity levels of 6.9% despite using a log higher vector dose of 2e13 vg/kg.21 Raised ALT levels were observed in 3 of 10 patients recruited to AMT-060, requiring treatment with corticosteroids. In 3 other phase 1/2 clinical trials (DTX101 [NCT02618915], AAV8-hFIX19 [NCT01620801], and BAX 335 [NCT01687608]), transaminitis led to loss of transgenic FIX despite administration of prednisolone at an AAV dose of 1e12 vg/kg or higher.22,23 This suggests that corticosteroids may not work in all circumstances.
Recent hemophilia B gene therapy trial using AAV
| Sponsor . | Transgene . | No. of CpG motifs in transgene . | Serotype . | Genome format . | Method of vector delivery . | Dose range (vg/kg) . | Mean stable FIX activity levels . | No. of patients with transaminitis . | Current status . |
|---|---|---|---|---|---|---|---|---|---|
| Avigen/CHOP | Wild-type FIX | 19 | AAV2 | ssAAV | IM | 2e11 to 1.8e12 | Transient at a maximum level of 1.6% | 0 | Closed |
| Avigen/CHOP Coagulin-B | Wild-type FIX | 19 | AAV2 | ssAAV | Intrahepatic artery | 2e11 to 2e12 | Transient with a maximum of 12% | 1/2 at highest dose | Closed |
| St Jude/UCL | Codon-optimized FIX | 0 | AAV8 | scAAV | Systemic | 2e11 to 2e12 | 5.1% | 4/6 patient at highest dose | Closed |
| Takeda (Baxalta; BAX 335) | Codon-optimized FIX + Padua mutation | 99 | AAV8 | scAAV | Systemic | 2e11 to 3e12 | Transient except in 1 patient who had expression of ~20% at last report | 2/6 patients treated at or above 1e12 received steroids in response to ALT elevations and 1 patient received prophylactic steroids. | Closed |
| Spark Therapeutics (SPK-9001, fidanacogene elaparvovec) | Codon-optimized FIX containing the Padua mutation | 0 | AAV- Spark100 | ssAAV | Systemic | 5e11 | 19.8% at 5 years | 3/15 patients | Transitioned to phase 3 with Pfizer |
| uniQure (AMT-060) | Codon-optimized FIX | 0 | AAV5 | ssAAV | Systemic | 5e12-2e13 | 6.9% 5.2% at 5e12-vg/kg dose and 7.2% at 2e13-vg/kg dose at 5 years | 2/5 at highest dose | A new program, AMT-061, that contains the FIX Padua in development with CSL Behring |
| CSL Behring (AMT-061, etranacogene dezaparvovec) | Codon-optimized FIX containing the Padua mutation | ?0 | AAV5 | ?ssAAV | Systemic | 2e13 | 36.9% | 9/54 | Under regulatory review |
| Dimension Therapeutics (DTX101) | Codon-optimized FIX | 96 | AAVrh10 | ssAAV | Systemic | 1.6e12-5e12 | 6.7% | 3/3 at highest dose | Closed |
| Freeline Therapeutics (FLT-180a, verbrinacogene setparvovec) | Codon-optimized FIX containing the Padua mutation | 5 | AAV-S3 synthetic capsid | ssAAV | Systemic | 3.84e11-1.28e12 | 30%-279% | 8/10 | Clinical trials ongoing |
| Belief BioMed (BBM-H901) | Codon-optimized FIX containing the Padua mutation | 0 | AAV843 synthetic capsid | scAAV | Systemic | 5e12 | 36.9% | 2/10 | Clinical trials ongoing |
| Sangamo Bioscience (SB-FIX) | Codon-optimized FIX | Not known | AAV6/zinc finger–mediated targeted integration into the albumin locus in hepatocytes | ssAAV | Systemic | Unknown | Unknown | Closed |
| Sponsor . | Transgene . | No. of CpG motifs in transgene . | Serotype . | Genome format . | Method of vector delivery . | Dose range (vg/kg) . | Mean stable FIX activity levels . | No. of patients with transaminitis . | Current status . |
|---|---|---|---|---|---|---|---|---|---|
| Avigen/CHOP | Wild-type FIX | 19 | AAV2 | ssAAV | IM | 2e11 to 1.8e12 | Transient at a maximum level of 1.6% | 0 | Closed |
| Avigen/CHOP Coagulin-B | Wild-type FIX | 19 | AAV2 | ssAAV | Intrahepatic artery | 2e11 to 2e12 | Transient with a maximum of 12% | 1/2 at highest dose | Closed |
| St Jude/UCL | Codon-optimized FIX | 0 | AAV8 | scAAV | Systemic | 2e11 to 2e12 | 5.1% | 4/6 patient at highest dose | Closed |
| Takeda (Baxalta; BAX 335) | Codon-optimized FIX + Padua mutation | 99 | AAV8 | scAAV | Systemic | 2e11 to 3e12 | Transient except in 1 patient who had expression of ~20% at last report | 2/6 patients treated at or above 1e12 received steroids in response to ALT elevations and 1 patient received prophylactic steroids. | Closed |
| Spark Therapeutics (SPK-9001, fidanacogene elaparvovec) | Codon-optimized FIX containing the Padua mutation | 0 | AAV- Spark100 | ssAAV | Systemic | 5e11 | 19.8% at 5 years | 3/15 patients | Transitioned to phase 3 with Pfizer |
| uniQure (AMT-060) | Codon-optimized FIX | 0 | AAV5 | ssAAV | Systemic | 5e12-2e13 | 6.9% 5.2% at 5e12-vg/kg dose and 7.2% at 2e13-vg/kg dose at 5 years | 2/5 at highest dose | A new program, AMT-061, that contains the FIX Padua in development with CSL Behring |
| CSL Behring (AMT-061, etranacogene dezaparvovec) | Codon-optimized FIX containing the Padua mutation | ?0 | AAV5 | ?ssAAV | Systemic | 2e13 | 36.9% | 9/54 | Under regulatory review |
| Dimension Therapeutics (DTX101) | Codon-optimized FIX | 96 | AAVrh10 | ssAAV | Systemic | 1.6e12-5e12 | 6.7% | 3/3 at highest dose | Closed |
| Freeline Therapeutics (FLT-180a, verbrinacogene setparvovec) | Codon-optimized FIX containing the Padua mutation | 5 | AAV-S3 synthetic capsid | ssAAV | Systemic | 3.84e11-1.28e12 | 30%-279% | 8/10 | Clinical trials ongoing |
| Belief BioMed (BBM-H901) | Codon-optimized FIX containing the Padua mutation | 0 | AAV843 synthetic capsid | scAAV | Systemic | 5e12 | 36.9% | 2/10 | Clinical trials ongoing |
| Sangamo Bioscience (SB-FIX) | Codon-optimized FIX | Not known | AAV6/zinc finger–mediated targeted integration into the albumin locus in hepatocytes | ssAAV | Systemic | Unknown | Unknown | Closed |
IM, intramuscular; scAAV, self-complementary AAV; ssAAV, single stranded AAV.
Since the risk of transaminitis appeared to be vector dose dependent, occurring at a high frequency, at doses >1e12 vg/kg, a strategy to reduce or avoid this complication would be to improve vector performance so that low doses of AAV vectors could be used to mediate plasma FIX activity levels at >5% of normal. With this in mind, the next generation of hemophilia B trials used a FIX cDNA containing a naturally occurring gain-of-function (Padua) mutation in humans characterized by leucine (R338L) instead of arginine at position 338 in the catalytic domain. This mutation enhances FIX activity (FIX:C) by 5- to 8-fold for a given amount of FIX antigen. Therefore, a small increase in plasma FIX antigen levels would lead to a substantial increase in plasma FIX clotting activity. There were concerns that the use of a variant transgene would increase the risk of FIX inhibitor development, but preclinical studies in a canine model of hemophilia B showed that FIX-R338L expression after gene transfer did not result in inhibitor formation and, furthermore, was able to induce tolerance.24
The first clinical study to evaluate the FIX-R338L transgene in patients with hemophilia B (BAX 335 [NCT01687608]) showed that peak FIX activity at 30% to 58% could be achieved at the high-dose level (3e12 vg/kg), but unfortunately, expression declined to basal levels in all but 1 patient, who continues to express at the 20% level. The FIX cDNA used in this and the previously mentioned DTX1010 (NCT02618915) study had a higher level of unmethylated cytidine-phosphate-guanosine (CpG) motifs than in other studies. Both studies were associated with loss of transgenic FIX despite administration of prednisolone. This led to the hypothesis that an excess of unmethylated CpG motifs, which are common in bacterial but not mammalian DNA, triggered a Toll-like receptor 9 response, leading to loss of transduced hepatocytes with an associated transaminitis that is not responsive to corticosteroids.25
UniQure also incorporated the Padua gain-of-function mutation into the FIX sequences of AMT-060 to create AMT-061 (NCT03489291), which showed enhancement of FIX activity level in 3 patients with severe hemophilia B of ~8-fold higher than levels previously reported with AMT-060 at the same dose.26 This was followed by an open-label phase 3 study (HOPE-B, NCT03569891) in 54 patients with moderate to severe hemophilia B in the United States and in Europe. At 1.5 years after a 2e13-vg/kg dose of AMT-061, mean plasma FIX activity levels were 36.9% of normal, resulting in a 64% reduction in bleeding rates and a 97% drop in annual FIX usage. Notably, these benefits were seen regardless of preexisting antibodies against AAV5.27
In another study (SPK-9001, fidanacogene elaparvovec, NCT02484092), sustained mean FIX activity of 22.9% ± 9.9% was observed in the 15 subjects following a single administration of vector at a dose of 5e11 vg/kg. A reduction in annualized bleeding rate (ABR) to 0.4 ± 1.1 postinfusion compared to 8.9 ± 14.0 prior to gene therapy.28 SPK-9001 consists of bioengineered capsid pseudotyped AAV vector containing the FIX-R338L transgene with a lower number of CpG.29 Transaminitis occurred in 3 subjects who responded well to steroids. All patients were able to stop FIX prophylaxis with a 98% reduction in ABR. There was no evidence of inhibitor formation to FIX-Padua. A follow-on report described stable FIX activity levels in this cohort of patients at 5 years of ~19.8%.30 Pfizer has taken on further development of this technology in an ongoing phase 3 trial (NCT03861273).
Stable FIX activity levels of 36.9% at 1 year were observed in 10 patients with hemophilia B following systemic administration of 5e12 vg/kg BBM-H901 in an investigator-initiated phase 1 trial in China (NCT04135300).31 BBM-H901 is a dimeric form of a codon-optimized FIX expression cassette containing the Padua mutation under the control of a liver-specific promoter, encapsidated with an engineered, liver-tropic AAV capsid. Preconditioning before vector administration included 1 week of prednisolone at a dose of 1 mg/kg followed by an 8-week taper. Two (20%) participants had elevation of ALT and aspartate aminotransferase accompanied by a decrease of FIX activity.
Freeline Therapeutics coupled a synthetic capsid (AAVS3), adapted for more efficient transduction of human hepatocytes with a modified FIX expression cassette containing the R338L mutation (FLT180a), and in the B-AMAZE trial, patients received 1 of 4 doses of this vector (3.84e11, 6.4e11, 8.32e11, or 1.28e12 vg/kg) together with a prophylactic immune management regimen in order to manage the risk of transaminitis in the knowledge that higher gene transfer may translate into a greater risk of transaminitis. All patients had dose-dependent increases in FIX levels following treatment with FLT180a, with sustained FIX activity in 9 of 10 patients at a median follow-up of ~2 years. Five patients had FIX levels in the normal range (51%-78%), three patients had levels ranging from 23% to 43%, and 1 high-dose patient (1.28e12 vg/kg) was at 260%. An arteriovenous fistula thrombosis occurred in this patient. A 75% reduction in ABR was observed despite a 96% reduction in annualized FIX consumption. Treatment was generally well tolerated, with transient transaminitis being the most common FLT180a-related adverse event.32
Thus, with advances in gene transfer technology, it is has become possible to achieve plasma FIX activity levels in the mild range on a consistent basis and with some AAV vector formulation and even normal and supraphysiological FIX activity levels, a feat thought to be impossible almost a decade ago.
AAV vectors and gene therapy for hemophilia A
The limited packaging capacity of AAV vectors (4.7 kb) and the poor expression profile of FVIII have hindered the use of these vectors for gene therapy of hemophilia A. However, we developed an AAV-based gene transfer approach that addresses both the size constraints and inefficient FVIII expression. The FVIII B-domain, which is not required for cofactor activity, was removed to reduce the size of the FVIII expression cassette. Human factor VIII expression was improved 10-fold by reorganization of the wild-type cDNA of human FVIII according to the codon usage of highly expressed human genes.33 BioMarin, which licensed this construct, commenced the first clinical trial for hemophilia A using AAV5 pseudotyped vectors (Table 2) made using the baculovirus–insect cell manufacturing method. This vector (AAV5-hFVIII-SQ, BMN 270) was initially tested in 9 men with severe hemophilia A over a dose range of 6e12 to 6e13 vg/kg in the context of a rapid vector phase 1/2 dose escalation trial (NCT02576795, BMN 270-201, valoctocogene roxaparvovec).34,35 Factor VIII expression was <3 IU/dL in the low- and intermediate-dose cohorts. Of the 7 patients treated at the 6e13-vg/kg dose level, mean FVIII activity was 64 IU/dL (Table 2) using a chromogenic assay at 1 year. The levels declined to 36 IU/dL and 33 IU/dL in years 2 and 3, respectively, after gene transfer. This trend has continued, and at 5 years, the mean FVIII activity was 11.6 IU/dL (unpublished report).
Hemophilia A gene therapy trial using AAV
| AAV vector . | Study . | Outcomes . | Current status . |
|---|---|---|---|
| BMN 270-201: A Phase 1/2, Dose-Escalation, Safety, Tolerability, and Efficacy Study of Valoctocogene Roxaparvovec, an Adenovirus-Associated Virus Vector-Mediated Gene Transfer of Human Factor VIII in Patients With Severe Hemophilia A (NCT02576795) | FVIII expression <3% in the low- and intermediate-dose cohorts. 6e13-vg/kg dose cohort (n = 7), mean FVIII activity at 1, 2, and 3 years of 64 IU/dL, 36 IU/dL, and 33 IU/dL (Chromogenic). 4e13-vg/kg dose cohort (n = 6): FVIII activity at years 1 and 2 = 21 IU/dL and 15 IU/dL, respectively (Chromogenic). >90% reduction in ABR and FVIII concentrate infusion in both groups. | Closed to recruitment. LTFU continues. EU conditional approval granted. | |
| BMN 270-203: A Phase 1/2 Safety, Tolerability, and Efficacy Study of Valoctocogene Roxaparvovec, an Adeno-Associated Virus Vector-Mediated Gene Transfer of Human Factor VIII in Hemophilia A Patients with Residual FVIII Levels ≤1IU/dL and preexisting Antibodies Against AAV5 (NCT03520712) | Enrolling subjects; no data reported. | ||
| BioMarin (BMN 270; AAV5-hFVIII-SQ valoctocogene roxaparvovec) | BMN 270-205: Safety, Tolerability, and Efficacy Study of Valoctocogene Roxaparvovec in Hemophilia A With Active or Prior Inhibitors (NCT04684940) | Enrolling subjects; no data reported. | |
| BMN 270-301: A Phase 3 Open-Label, Single-Arm Study to Evaluate the Efficacy and Safety of BMN 270, an Adeno-Associated Virus Vector-Mediated Gene Transfer of Human Factor VIII in Hemophilia A Patients with Residual FVIII Levels ≤1IU/dL (NCT03370913) | FVIII activity levels (N = 132) at ~1 year = 41.9 IU/dL (Chromogenic). 98.6% decline in ABR and 83.8% reduction in FVIII concentrate usage. Transaminitis in 85.8% of the participants. | Target recruitment completed. LTFU continues. Under regulatory review. | |
| BMN 270-302: Phase 3 Study to Evaluate Efficacy/Safety of Valoctocogene Roxaparvovec an AAV Vector-Mediated Gene Transfer of hFVIII at a Dose of 4x1013 vg/kg in Hemophilia A Patients With Residual FVIII Levels ≤1IU/dL Receiving Prophylactic FVIII Infusions (GENEr8-2) (NCT03392974). | Phase 3: closed to recruitment due to lack of interest in the low-vector dose. LTFU continues. | ||
| BMN 270-303: A Phase 3b, Single Arm, Open-Label Study to Evaluate the Efficacy and Safety of BMN 270, an Adeno-Associated Virus Vector-Mediated Gene Transfer of Human Factor VIII, With Prophylactic Corticosteroids in Hemophilia A Patients (GENEr8-3) (NCT04323098) | Enrolling subjects; no data reported. | ||
| UCL/AAV8 FVIII V3 peptide instead of B-domain | Gene Therapy for Haemophilia A (GO-8, NCT03001830) | 4 doses under evaluation: 6e11, 2e12, 4e12, or 6e12 vg/kg. Longest follow-up in 2e12-vg/kg dose cohort shows stable expression of FVIII out to 3 years at 15 IU/dL. | Open |
| Spark/Roche (SPK-8011) AAV-LK03-FVIII-SQ | SPK-8011-101: Gene Transfer, Dose-Finding Safety, Tolerability, and Efficacy Study of SPK-8011 (a Recombinant Adeno-Associated Viral Vector With Human Factor VIII Gene) in Individuals With Hemophilia A (NCT03003533) Together With Long-Term Follow-up Study (NCT03432520) | 4 cohorts (N = 18) over 5e11 to 2e12 vg/kg; mean FVIII expression ~12.9% ± 6.9% (1-stage clotting assay); 2 participants lost all expression because of transaminitis refractory to immunosuppression; 91% reduction in ABR; FVIII concentrate usage down by 96.4% | Closed to recruitment. LTFU continues. |
| Spark/Roche (SPK-8016) AAV-FVIII-SQ using a novel capsid | SPK-8016-101: Dose-Finding Study of SPK-8016 Gene Therapy in Patients With Hemophilia A to Support Evaluation in Individuals With FVIII Inhibitors (NCT03734588) | Recruitment closed. No data reported. | |
| Pfizer/Sangamo Bioscience (SB-525; giroctocogene fitelparvovec) AAV6 FVIII-SQ | SB-525-1603: A Phase 1/2, Open-Label, Adaptive, Dose-Ranging Study to Assess the Safety and Tolerability of SB-525 (PF-07055480) (Recombinant AAV2/6 Human Factor 8 Gene Therapy) in Adult Subjects With Severe Hemophilia A (Alta Study) (NCT03061201) | 4 cohorts (9e11, 2e12, 1e13, and 3e13 vg/kg; N = 11). Mean FVIII activity: ~43% and 25.7% at years 1 and 2, respectively @ 3e13-vg/kg dose level (N = 5); chromogenic assay. Transaminitis was detected in 5 of the 11 participants. | Closed to recruitment. LTFU continues. |
| C0371004: Phase III. An Open-Label, Non-investigational Product, Lead-in Study to Evaluate at Least 6 Months of Prospective Efficacy and Safety Data of Factor Replacement Therapy in the Usual Care Setting of Moderately Severe to Severe Adult Hemophilia B Subjects (FIX:C ≤2%) Who Are Negative for Nab to AAV Vector-Spark100 and Moderately Severe to Severe Hemophilia A Adult Subjects (FVIII:C ≤1%) Who Are Negative for Nab to AAV Vector Sb-525 Capsid (AAV6), Prior to the Respective Therapeutic Ph 3 Gene Therapy Studies (NAB Protocol) (NCT03587116) | Recruiting 250 subjects in this 6-month lead-in study to support the phase 3 intervention study. | ||
| C3731003: Phase 3, Open-Label, Single-Arm Study to Evaluate the Efficacy and Safety of PF-07055480 (Recombinant AAV2/6 Human Factor VIII Gene Therapy) in Adult Male Participants With Moderately Severe to Severe Hemophilia A (FVIII:C ≤1%) (AFFINE) (NCT04370054) | Recruitment target = 60 patients. Phase 3 restarted following temporary regulatory hold for high FVIII levels (>150%) in some participants. | ||
| Bayer/Ultragenyx Therapeutics BAY 2599023 (DTX-201) AAVhu37 capsid pseudotyped FVIII-SQ | A Phase 1/2 Open-Label Safety and Dose-Finding Study of BAY2599023 (DTX201), an Adeno-Associated Virus (AAV) hu37-Mediated Gene Transfer of B-Domain Deleted Human Factor VIII, in Adults With Severe Hemophilia A (NCT03588299) | 9 participants enrolled sequentially into 1 of 3 dose cohorts (0.5e13, 1e13, and 2e13 vg/kg). FVIII expression levels for up to >23 months. Transaminitis observed in 5/9 participants. | |
| Takeda (TAK 754) AAV8-FVIII-SQ | A Study of BAX 888 in Male Adults With Severe Hemophilia A (NCT03370172) | 2e12- and 6e12-vg/kg dose cohorts assessed. Study suspended following transaminitis-mediated loss of FVIII expression in all participants. | |
| AAV vector . | Study . | Outcomes . | Current status . |
|---|---|---|---|
| BMN 270-201: A Phase 1/2, Dose-Escalation, Safety, Tolerability, and Efficacy Study of Valoctocogene Roxaparvovec, an Adenovirus-Associated Virus Vector-Mediated Gene Transfer of Human Factor VIII in Patients With Severe Hemophilia A (NCT02576795) | FVIII expression <3% in the low- and intermediate-dose cohorts. 6e13-vg/kg dose cohort (n = 7), mean FVIII activity at 1, 2, and 3 years of 64 IU/dL, 36 IU/dL, and 33 IU/dL (Chromogenic). 4e13-vg/kg dose cohort (n = 6): FVIII activity at years 1 and 2 = 21 IU/dL and 15 IU/dL, respectively (Chromogenic). >90% reduction in ABR and FVIII concentrate infusion in both groups. | Closed to recruitment. LTFU continues. EU conditional approval granted. | |
| BMN 270-203: A Phase 1/2 Safety, Tolerability, and Efficacy Study of Valoctocogene Roxaparvovec, an Adeno-Associated Virus Vector-Mediated Gene Transfer of Human Factor VIII in Hemophilia A Patients with Residual FVIII Levels ≤1IU/dL and preexisting Antibodies Against AAV5 (NCT03520712) | Enrolling subjects; no data reported. | ||
| BioMarin (BMN 270; AAV5-hFVIII-SQ valoctocogene roxaparvovec) | BMN 270-205: Safety, Tolerability, and Efficacy Study of Valoctocogene Roxaparvovec in Hemophilia A With Active or Prior Inhibitors (NCT04684940) | Enrolling subjects; no data reported. | |
| BMN 270-301: A Phase 3 Open-Label, Single-Arm Study to Evaluate the Efficacy and Safety of BMN 270, an Adeno-Associated Virus Vector-Mediated Gene Transfer of Human Factor VIII in Hemophilia A Patients with Residual FVIII Levels ≤1IU/dL (NCT03370913) | FVIII activity levels (N = 132) at ~1 year = 41.9 IU/dL (Chromogenic). 98.6% decline in ABR and 83.8% reduction in FVIII concentrate usage. Transaminitis in 85.8% of the participants. | Target recruitment completed. LTFU continues. Under regulatory review. | |
| BMN 270-302: Phase 3 Study to Evaluate Efficacy/Safety of Valoctocogene Roxaparvovec an AAV Vector-Mediated Gene Transfer of hFVIII at a Dose of 4x1013 vg/kg in Hemophilia A Patients With Residual FVIII Levels ≤1IU/dL Receiving Prophylactic FVIII Infusions (GENEr8-2) (NCT03392974). | Phase 3: closed to recruitment due to lack of interest in the low-vector dose. LTFU continues. | ||
| BMN 270-303: A Phase 3b, Single Arm, Open-Label Study to Evaluate the Efficacy and Safety of BMN 270, an Adeno-Associated Virus Vector-Mediated Gene Transfer of Human Factor VIII, With Prophylactic Corticosteroids in Hemophilia A Patients (GENEr8-3) (NCT04323098) | Enrolling subjects; no data reported. | ||
| UCL/AAV8 FVIII V3 peptide instead of B-domain | Gene Therapy for Haemophilia A (GO-8, NCT03001830) | 4 doses under evaluation: 6e11, 2e12, 4e12, or 6e12 vg/kg. Longest follow-up in 2e12-vg/kg dose cohort shows stable expression of FVIII out to 3 years at 15 IU/dL. | Open |
| Spark/Roche (SPK-8011) AAV-LK03-FVIII-SQ | SPK-8011-101: Gene Transfer, Dose-Finding Safety, Tolerability, and Efficacy Study of SPK-8011 (a Recombinant Adeno-Associated Viral Vector With Human Factor VIII Gene) in Individuals With Hemophilia A (NCT03003533) Together With Long-Term Follow-up Study (NCT03432520) | 4 cohorts (N = 18) over 5e11 to 2e12 vg/kg; mean FVIII expression ~12.9% ± 6.9% (1-stage clotting assay); 2 participants lost all expression because of transaminitis refractory to immunosuppression; 91% reduction in ABR; FVIII concentrate usage down by 96.4% | Closed to recruitment. LTFU continues. |
| Spark/Roche (SPK-8016) AAV-FVIII-SQ using a novel capsid | SPK-8016-101: Dose-Finding Study of SPK-8016 Gene Therapy in Patients With Hemophilia A to Support Evaluation in Individuals With FVIII Inhibitors (NCT03734588) | Recruitment closed. No data reported. | |
| Pfizer/Sangamo Bioscience (SB-525; giroctocogene fitelparvovec) AAV6 FVIII-SQ | SB-525-1603: A Phase 1/2, Open-Label, Adaptive, Dose-Ranging Study to Assess the Safety and Tolerability of SB-525 (PF-07055480) (Recombinant AAV2/6 Human Factor 8 Gene Therapy) in Adult Subjects With Severe Hemophilia A (Alta Study) (NCT03061201) | 4 cohorts (9e11, 2e12, 1e13, and 3e13 vg/kg; N = 11). Mean FVIII activity: ~43% and 25.7% at years 1 and 2, respectively @ 3e13-vg/kg dose level (N = 5); chromogenic assay. Transaminitis was detected in 5 of the 11 participants. | Closed to recruitment. LTFU continues. |
| C0371004: Phase III. An Open-Label, Non-investigational Product, Lead-in Study to Evaluate at Least 6 Months of Prospective Efficacy and Safety Data of Factor Replacement Therapy in the Usual Care Setting of Moderately Severe to Severe Adult Hemophilia B Subjects (FIX:C ≤2%) Who Are Negative for Nab to AAV Vector-Spark100 and Moderately Severe to Severe Hemophilia A Adult Subjects (FVIII:C ≤1%) Who Are Negative for Nab to AAV Vector Sb-525 Capsid (AAV6), Prior to the Respective Therapeutic Ph 3 Gene Therapy Studies (NAB Protocol) (NCT03587116) | Recruiting 250 subjects in this 6-month lead-in study to support the phase 3 intervention study. | ||
| C3731003: Phase 3, Open-Label, Single-Arm Study to Evaluate the Efficacy and Safety of PF-07055480 (Recombinant AAV2/6 Human Factor VIII Gene Therapy) in Adult Male Participants With Moderately Severe to Severe Hemophilia A (FVIII:C ≤1%) (AFFINE) (NCT04370054) | Recruitment target = 60 patients. Phase 3 restarted following temporary regulatory hold for high FVIII levels (>150%) in some participants. | ||
| Bayer/Ultragenyx Therapeutics BAY 2599023 (DTX-201) AAVhu37 capsid pseudotyped FVIII-SQ | A Phase 1/2 Open-Label Safety and Dose-Finding Study of BAY2599023 (DTX201), an Adeno-Associated Virus (AAV) hu37-Mediated Gene Transfer of B-Domain Deleted Human Factor VIII, in Adults With Severe Hemophilia A (NCT03588299) | 9 participants enrolled sequentially into 1 of 3 dose cohorts (0.5e13, 1e13, and 2e13 vg/kg). FVIII expression levels for up to >23 months. Transaminitis observed in 5/9 participants. | |
| Takeda (TAK 754) AAV8-FVIII-SQ | A Study of BAX 888 in Male Adults With Severe Hemophilia A (NCT03370172) | 2e12- and 6e12-vg/kg dose cohorts assessed. Study suspended following transaminitis-mediated loss of FVIII expression in all participants. | |
ABR, annualized bleed rates; EU, European Union; LTFU, long-term follow-up.
An additional 6 patients were treated at a lower dose level of 4e13 vg/kg, leading to mean FVIII activity at the end of years 1 and 2 of 21 IU/dL and 15 IU/dL, respectively.34,35 By 4 years, the mean FVIII activity levels had dropped to 5.6 IU/dL (unpublished report). Across the total 13 patients in the 4e13- and 6e13-vg/kg dose levels, a >90% reduction in ABR and FVIII concentrate infusion was observed when compared to pregene therapy values that appears to be maintained out to 4 and 5 years in the 4e13- and 6e13-vg/kg cohorts, respectively.
In the largest gene therapy trial in patients with hemophilia, BioMarin assessed BMN 270 in open-label, single-arm, multicenter, phase 3 trial (NCT03370913, GENEr8-1) in 134 patients with severe hemophilia A at a dose of 6e13 vg/kg. The safety and efficacy results have recently been published36 and show mean FVIII activity levels between 49 and 52 weeks of 41.9 IU/dL in the 132 human immunodeficiency virus–negative participants in that trial. This was associated with a 98.6% decline in ABR and an 83.8% reduction in FVIII concentrate usage. Transaminitis was observed in 85.8% of the participants and managed with transient immunosuppression. None of the participants developed inhibitors to FVIII, malignancy, or thromboembolic events. The phase 3 data appear to be consistent with the initial observations in phase 1/2.
In the ongoing, UCL-sponsored, phase 1/2 clinical trial (GO-8; NCT03001830), we are evaluating an identical construct to that used in the BioMarin study except for the inclusion of a 17–amino acid peptide comprising 6 N-linked glycosylation motifs (V3) from the human FVIII B-domain (AAV-HLP-hFVIII-V3) that are highly conserved through evolution.37 In murine studies, AAV-HLP-hFVIII-V3 mediated expression of FVIII at 3-fold higher levels when compared to AAV-HLP-hFVIII-SQ, encoding the conventional B-domain–deleted FVIII. The safety and efficacy of AAV8 serotype pseudotyped HLP-hFVIII-V3, manufactured in mammalian HEK 293T cells, have been assessed in 9 adult men with severe hemophilia A who received 1 of 4 doses of this vector (6e11, 2e12, 4e12, or 6e12 vg/kg) together with a prophylactic immune management regimen. With a follow-up extending to 4 years in some patients, FVIII expression levels appear to be more stable than in the BioMarin trials. In the GO8 2e12-vg/kg cohort, FVIII activity levels have remained stable over a period of 3 years at ~15% of normal using a chromogenic assay. No participant has developed a FVIII inhibitor.38
Spark Therapeutics dosed 18 patients with SPK-8011 (NCT03003533), an AAV vector containing a codon-optimized human factor VIII gene under the control of a liver-specific promoter pseudotyped with a bioengineered capsid, LK03 capsid, manufactured using the HEK-293 process.39 These patients were treated in 4 dose cohorts over a dose range of 5e11 to 2e12 vg/kg. Sixteen of the 18 participants had sustained FVIII expression at a median follow-up of 33.4 months (range, 3.7-47.6), with 2 losing all expression because of transaminitis that was refractory to immunosuppression. In the patients with sustained FVIII expression, there does not appear to be a clear relationship between vector dose and plasma FVIII levels, with an overall mean FVIII activity level of 12.9% ± 6.9% using a 1-stage clotting assay that shows ~1.5 times higher FVIII activity when compared to chromogenic assay methods. There was no apparent decrease in FVIII activity over a 2-year follow-up period. A 91% reduction in annualized bleeding rate was observed in patients with stable expression, with a reduction in annual FVIII concentrate usage of 96.4%. This program is currently in phase 3 evaluation.
Pfizer/Sangamo Therapeutics (SB-525, NCT04370054, giroctocogene fitelparvovec) announced results of their ongoing phase 1/2 trial in 11 patients with severe hemophilia A treated in 4 dose cohorts (9e11, 2e12, 1e13, and 3e13 vg/kg) with reactive corticosteroid administration for transaminitis. The codon- optimized BDD-FVIII transgene is packaged in AAV serotype 6 capsid and manufactured using the baculovirus–insect cell manufacturing method. At the last update (ASH 202140 ), mean FVIII activity level, as measured by a chromogenic assay in the 3e13-vg/kg dose cohort (N = 5) at 2 years, was 25.7%, a drop from a mean of ~43% at 1 year after gene transfer. Transaminitis was detected in 5 of the 11 participants enrolled in the study, including 3 participants in the 3e13-vg/kg cohort. A phase 3 evaluation of SB-525 (NCT04370054) has been restarted recently following a temporary regulatory hold for protocol amendment following the observance of FVIII levels greater than 150% in some participants in the trial.
Takeda recently suspended screening and further enrollment in the TAK-754, an AAV8 gene therapy study (NCT03370172) for hemophilia A in which subjects had been enrolled into the 2e12- and 6e12-vg/kg dose cohorts. All subjects lost FVIII expression following transaminitis.41
BAY 2599023 phase 1/2 study uses an AAVhu37 capsid to deliver a B-domain–deleted FVIII codon-optimized cDNA, under the control of a liver-specific promoter/enhancer element. Thus far, 9 participants have been enrolled sequentially into 1 of 3 dose cohorts (0.5e13, 1e13, and 2e13 vg/kg).42 Transaminitis was observed in 5 of 9 participants requiring a short course of corticosteroids. FVIII expression levels appear to be in the therapeutic range over a period of >23 months, enabling patients in cohorts 2 and 3 to come off prophylaxis with FVIII concentrates. Enrollment into the fourth cohort (4e13 vg/kg) is under way.
These differing AAV gene therapy trials offer substantial choices to the hemophilia community and provide an opportunity for patients with preexisting antibodies to 1 serotype to be treated by alternative serotypes. In contrast to hemophilia B, higher vector doses appear to be required for hemophilia A. Additionally, expression of FVIII appears to be declining over time following AAV gene transfer in contrast to hemophilia B and may reflect the fact that an oversized transgene is used in most hemophilia A studies, which may influence durability. This decline in transgene expression in the absence of transaminitis appears to be most pronounced in patients with the highest level of FVIII expression. While the reason for this late decline in FVIII activity remains unclear, potential explanations include (1) loss of the episomally retained, oversized FVIII AAV transgene from the transduced hepatocytes and (2) silencing of the FVIII transgene.
Obstacles to wider use of AAV vector technology
Safety considerations
Thus far, the risk of liver toxicity, accompanied by loss or reduction of transgene expression, appears to be the most worrying toxicity associated with liver-targeted delivery of AAV, as described before. Transaminitis appears to occur with all AAV capsids regardless of the AAV genome configuration, transgene promoter, or method of manufacture. It is a self-limiting phenomenon that may be controlled with corticosteroids alone or in combination with other immune-suppressive agents in some patients. The precise pathophysiologic basis for transaminitis remains unclear, in part because it has not been possible to recapitulate this toxicity in animal models.24,43 Clinical data suggest that the number of CpG motifs in the transgene may play a role as discussed previously. It remains unclear, in that instance, why the transaminitis is limited to the initial period after gene transfer as there have been no reports of late recurrence of transaminitis beyond a 2-year period. Of note, in this respect, is the recent report of a link between wild-type AAV2 infection and an outbreak of acute hepatitis in children who were coinfected with adenovirus or, less commonly, human herpesvirus 6. It is unclear if AAV2 is pathogenic in these children or just a useful persistent biomarker of recent adenoviral infection.44
As expected, all subjects in these trials develop long-lasting AAV capsid-specific humoral immunity. While the rise in anti-AAV IgG does not have direct clinical consequences, its persistence at high titers precludes subsequent successful gene transfer with a vector of the same serotype, in the event that transgene expression should fall below therapeutic levels.
The risk of insertional mutagenesis following AAV-mediated gene transfer has been judged to be low because proviral DNA is maintained predominantly in an episomal form. This is consistent with the fact that wild-type AAV infection in humans, although common, is not associated with oncogenesis. However, deep sequencing studies show that integration of the AAV genome can occur in the liver.45,46 Indeed, a recent publication has found wild-type AAV2 genome fragments integrated in the proximity of known proto-oncogenes in a small percentage of human hepatocellular carcinoma (HCC) specimens.47 However, the pathogenic role of AAV2 in this setting is not certain. In December 2020, HCC was reported in a patient enrolled in the HOPE-B gene therapy trial for hemophilia B. The patient had multiple risk factors for HCC, including a history of hepatitis B and C, evidence of nonalcoholic fatty liver disease, smoking history, family history of cancer, and advanced age. An investigation including whole- genome sequencing of the tumor and adjacent tissue showed that etranacogene dezaparvovec was unlikely to have caused the HCC in this subject. An increased incidence of HCC has, however, been reported in the mucopolysaccharidoses type VII mouse model following perinatal gene transfer of AAV potentially through integration and disruption of an imprinted region rich in microRNAs and small nucleolar RNAs on mouse chromosome 12.48 Biomarin reported tumors on necropsy of mice at 52 weeks after administration of 2e14 vg/kg of AAV5–phenylalanine hydroxylase gene therapy in a tumor-prone mouse model that harbored 2 germline mutations: one to eliminate the phenylalanine hydroxylase gene and a second to render the mice immunodeficient. Further investigations are ongoing. Other studies in mice have failed to recapitulate this finding, and collectively, the available data in mice as well as larger animal models suggest that AAV has a relatively low risk of tumorigenesis.49 Nevertheless, safety consideration remains paramount and will require careful long-term monitoring of patients, likely beyond the 5 years of follow-up mandated by the US Food and Drug Administration.
Scale-up of vector production
Continued progression toward flexible, scalable production and purification methodologies is ongoing to support the commercialization of AAV biotherapeutics. The most widely used method for the generation of AAV entails the transient transfection of HEK-293 cells with plasmids encoding the necessary vector, helper, and packaging genes. This method is cumbersome, but progress has been made on improving the productivity to supply phase 3/market authorization hemophilia gene therapy trials.50 Another method being used by several biopharma companies because of its scalability is one based on baculovirus grown in SF9 insect cells.51 However, infectivity of AAV made using the baculovirus system is low due in part to lower levels of VP1 incorporation into the AAV capsid. Attention is currently focused on the downstream purification process so that the purity of clinical-grade AAV preparation can be improved.
Preexisting neutralizing anti-AAV antibody
Between 20% and 70% of patients have preexisting neutralizing anti-AAV antibodies (NAbs) to specific AAV serotypes, which can block efficient gene transfer. These patients are currently excluded from gene therapy trials, but NAbs represent a major limitation to the broad applicability of gene therapy to the hemophilia population. One strategy to overcome NAbs that works well in animal models is to switch AAV serotype17 but may not be applicable in humans due to cross-reactivity of NAbs. Alternatively, NAbs could be overcome by using immunosuppression, plasmapheresis, or by simply increasing vector dose or adding empty capsids.52
Affordability of AAV gene therapy
It is likely that gene therapy will command a high price, at least initially, in order to recoup the development cost. However, successful gene therapy offers the advantage of continuous endogenous expression of clotting factor, which will eliminate breakthrough bleeding and microhemorrhages, thereby reducing comorbidities and the need for frequent medical interventions while improving quality of life. Thus, gene therapy has the potential to yield significant savings for the health care system and society in general but may still prove to be unaffordable for patients living in developing or emerging economies.
Alternative gene therapy approaches that show promise
The AAV genome is maintained in an episomal format, raising the potential for loss of transgene expression with division of the transduced cell. This has engendered interest in targeting approaches.53 One such approach, “GeneRide,” entails the use of AAV vectors to deliver a promoterless FIX gene flanked by “guide DNA arm” that is several hundred base pairs long that matches a specific locus such as the albumin locus. Once in the target cell nucleus, the AAV transgene is integrated at a specific site that matches with the “guide DNA arms” within the patient's genome using the host cell's homologous recombination machinery.54 This approach has been successful in mice. Early studies in humans have been complicated by reports of thrombotic microangiopathy. Zinc-finger nucleases and clustered regularly interspaced short palindromic repeat (CRISPR)/CRISPR associated protein 9 (CRISPR/Cas9) have the potential to mediate targeted integration of the FVIII or FIX gene in a “safe harbor” using nonhomologous end joining.55 Recently, Sangamo Therapeutics, together with Georgetown University, announced the treatment of the first patient with hemophilia B (SB-FIX, NCT02695160) with a gene-editing approach in which the targeted integration of the FIX cDNA into the intron of the host cell albumin gene is promoted by zinc-finger nucleases following systemic administration of 3 separate AAV vectors. A potential concern with this approach is the effect of persistent expression of the endonuclease following gene transfer.
Integrating vectors, based on lentivirus, to propagate integration of the FIX or FVIII transgene into target cells is also under evaluation in hematopoietic stem cells or blood outgrowth endothelial cells following ex vivo manipulations. The development of a new generation of lentiviral vectors designed for efficient delivery of the transgene to the liver following systemic delivery of vectors carrying FVIII and FIX genes shows great promise and supports the further evaluation of this approach in the clinic.56
Conclusion
The availability of convincing evidence of long-term expression of transgenic FVIII and FIX at therapeutic levels resulting in amelioration of the bleeding diathesis following AAV-mediated gene transfer is an important step toward development of curative gene therapy. The first gene therapy for hemophilia has been approved by regulators in the United Kingdom. Market approval of a number of other gene therapy approaches for the hemophilias are on the horizon, all with the potential to change the treatment paradigm for patients with severe hemophilia. Several obstacles still remain, but the field is progressing at a rapid pace to resolve many of the limitations of gene therapy, thus facilitating broad availability for this disruptive therapeutic. Success in the hemophilia spaces will facilitate the development of gene therapy for other monogenetic disorders, particularly those with limited or nonexistent treatment options.
Conflict-of-interest disclosure
Amit C. Nathwani: no competing financial interests to declare.
Off-label drug use
Amit C. Nathwani: nothing to disclose.