Abstract
Patient- and leukemia-specific factors assessed at diagnosis classify patients with acute myeloid leukemia (AML) in risk categories that are prognostic for outcome. The induction phase with intensive chemotherapy in fit patients aims to reach a complete remission (CR) of less than 5% blasts in bone marrow by morphology. To deepen and sustain the response, induction is followed by consolidation treatment. This postremission treatment of patients with AML is graduated in intensity based on this favorable, intermediate, or adverse risk group classification as defined in the European Leukemia Network (ELN) 2022 recommendations. The increment of evidence that measurable residual disease (MRD) after induction can be superimposed on risk group at diagnosis is instrumental in tailoring further treatment accordingly. Several techniques are applied to detect MRD such as multiparameter flow cytometry (MFC), quantitative (digital) polymerase chain reaction (PCR), and next-generation sequencing. The clinical implementation of MRD and the technique used differ among institutes, leading to the accumulation of a wide range of data, and therefore harmonization is warranted. Currently, evidence for MRD guidance is limited to the time point after induction using MFC or quantitative PCR for NPM1 and core binding factor abnormalities in intermediate-risk patients. The role of MRD in targeted or nonintensive therapies needs to be clarified, although some data show improved survival in patients achieving CR-MRD negativity. Potential application of MRD for selection of conditioning before stem cell transplantation, monitoring after consolidation, and use as an intermediate end point in clinical trials need further evaluation.
Learning Objectives
Recognize that measurable residual disease (MRD) assessment is recommended as prognostic factor in intensively treated patients as measured after induction chemotherapy
Understand that molecular MRD monitoring at the end of treatment and during follow-up is recommended for selected patients
Appreciate that clinical relevance of MRD at other time points and different therapies (targeted/nonintensive) needs further evaluation
Realize that standardization of MRD measurements is essential for in vitro diagnostic regulations and use of MRD as an intermediate end point
CLINICAL CASE
A female patient of 65 years of age was referred to our hospital. Bone marrow (BM) aspiration showed a high proportion of myeloblasts by morphology and multiparameter flow cytometry (MFC), which led to the diagnosis of acute myeloid leukemia (AML). The disease was classified as de novo AML, not otherwise specified.1 The patient was stratified as European Leukemia Network (ELN) 2022 intermediate risk2 based on normal karyotype, an FLT3/ITD mutation (at low allelic ratio, although ELN 2022 does not require allelic ratio), and a DNMT3A mutation. At diagnosis, the leukemia was characterized by MFC and revealed a leukemia-associated phenotype (LAIP) with the combination of cluster of differentiation markers (CD) of CD34+CD13+CD7+.
The patient's age, medical history, and overall health indicated her eligibility for intensive 7 + 3 chemotherapy (cytarabine continuously for 7 days, along with short infusions of an anthracycline on each of the first 3 days). She was enrolled in a randomized trial comparing standard 7 + 3 chemotherapy with midostaurin to 7 + 3 chemotherapy including a different (more targeted) tyrosine kinase inhibitor. After the first induction cycle, the patient reached morphologic complete remission (CR; <5% blasts in BM by microscopy). MFC–measurable residual disease (MRD) showed 0.15% LAIP cells of the total white blood cells. Considering the applied MFC-MRD assay, this was deemed MRD positive (≥0.1%). Retrospective analysis using next-generation sequencing (NGS)–MRD confirmed MRD positivity for FLT3/ITD at 0.36%. A donor search was initiated, and she received the second chemotherapy cycle. After this second cycle, MFC-MRD showed MRD negativity of 0.04% (MRD negative), while the post hoc analysis identified the patient MRD positive for FLT3/ITD with a variant allele frequency (VAF) of 0.09%. The patient received myeloablative conditioning and peripheral blood (PB) stem cells from a matched unrelated donor for allogeneic stem cell transplantation (alloSCT), and the patient achieved CRMRD negative by both MRD techniques and resumed the FLT3 inhibitor after blood count recovery.
Disease monitoring was performed at 1 and 3 months, at which time MFC and NGS identified MRD relapse by MRD conversion from negative to positive. In the BM, the MFC-MRD was 0.2% of a new LAIP (CD34+CD13+CD56+), while the original LAIP was absent, and FLT3/ITD was detectable with a VAF of 1.6%. Another BM after 2 weeks confirmed MRD relapse (MFC 0.3%, NGS 2.6%). This coincided with a mixed donor/patient T-cell chimerism detected in PB.3 The immunosuppression was decreased and donor lymphocyte infusions were started.4,5 This resulted in a decrease in the LAIP cells, and MRD was cleared entirely after 3 months. After 2 years, the MRD monitoring was stopped, and the patient was followed up by clinical visits and standard laboratory evaluation.
The ongoing trial the patient was enrolled in is designed to evaluate MRD as a secondary end point for surrogacy of the clinical end-point event-free survival (EFS). In case the primary end-point EFS is improved by the new tyrosine kinase inhibitor and MRD is indeed significantly lower after a defined period of treatment, it is ideally suited to establish surrogacy of MRD for faster and more efficient evaluation of new treatment strategies.
In this review, we discuss the pros and cons of different MRD technologies, MRD results, and their clinical implications based on the available data.
Introduction
AML is a heterogeneous disease in both biology and clinical outcome. Main reasons for poor outcome are refractory disease (no CR) or relapse. To tailor therapy, estimating the risk of poor response to treatment is now imperative. Therefore, after the initial assessment of AML, the diagnostic workup needs to be more detailed to determine the prognostic risk group. This is mainly based on cytogenetic and molecular aberrations, leading to 3 ELN risk groups (favorable, intermediate, and adverse).2 First-line treatment of patients with AML aims at reaching CR by eradicating the majority of leukemia cells. This commonly consists of 1 or 2 cycles of intensive induction chemotherapy. For FLT3/ITD-mutated patients, addition of the multitargeted kinase inhibitor midostaurin significantly increased EFS and overall survival (OS)6 and is now standard of care. The induction phase is then followed by consolidation treatment to extend the response, and the intensity is commonly selected based on risk group. First-line treatment options with increasing intensity are (1) up to 3 cycles of chemotherapy (commonly intermediate- dose AraC), (2) myeloablative chemotherapy rescued by autologous stem cell transplantation, and (3) allogenic stem cell transplantation (alloSCT). Increasing treatment intensity will improve antileukemia efficacy but comes with adverse side effects and increased nonrelapse mortality (Figure 1). According to ELN, alloSCT is the preferred consolidation treatment for patients with an estimated relapse risk exceeding 35% to 40%.2,7 It needs to be emphasized that intensity of treatment is not solely based on laboratory testing but on patient-related factors as well, such as older age or comorbidities.
Considerations for postremission clinical decision-making. For patients with relatively low (<40%) risk of relapse, postremission consolidation may be deintensified to non-alloSCT (another cycle of chemotherapy or autologous stem cell transplantation). Several studies indicate this for favorable-risk patients and MRD-negative intermediate-risk patients.19-21 AlloSCT has a stronger antileukemia effect but comes with more side effects and is therefore recommended for MRD-positive intermediate-risk patients and adverse risk patients, and some studies also showed benefit for FLT3/ITD-positive patients in first remission.28,29 By MRD monitoring, early return of disease can be detected, which for non-alloSCT can be salvaged by alloSCT,22 and for alloSCT, an increase in MRD levels can be an indication for further stimulating the antileukemia effect by donor lymphocyte infusions and a decrease of immunosuppression.4,5 Created by BioRender.
Considerations for postremission clinical decision-making. For patients with relatively low (<40%) risk of relapse, postremission consolidation may be deintensified to non-alloSCT (another cycle of chemotherapy or autologous stem cell transplantation). Several studies indicate this for favorable-risk patients and MRD-negative intermediate-risk patients.19-21 AlloSCT has a stronger antileukemia effect but comes with more side effects and is therefore recommended for MRD-positive intermediate-risk patients and adverse risk patients, and some studies also showed benefit for FLT3/ITD-positive patients in first remission.28,29 By MRD monitoring, early return of disease can be detected, which for non-alloSCT can be salvaged by alloSCT,22 and for alloSCT, an increase in MRD levels can be an indication for further stimulating the antileukemia effect by donor lymphocyte infusions and a decrease of immunosuppression.4,5 Created by BioRender.
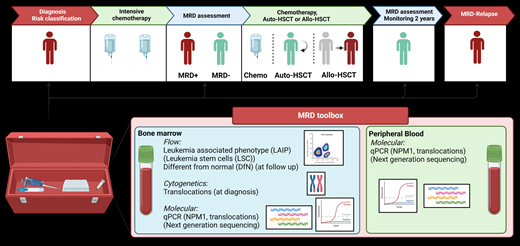
The ELN-AML guideline2 recommends refinement of morphologic response assessment by more sensitive MRD assessment, and the ELN-MRD guidelines8 provide the tools for standardized MRD evaluation (Table 1). Flow cytometry is commonly used for AML diagnosis, and hence the technique is widely available. MFC-MRD testing still needs expertise and strict criteria due to the rather extensive and subjective gating strategies. In addition, MFC may not be sensitive enough (10−3-10−4) to detect minimal numbers of residual cells. Molecular techniques such as quantitative polymerase chain reaction (PCR) or digital PCR are more sensitive (10−6) but only suitable for patients with specific molecular aberrations (NPM1 mutation or core binding factor [CBF] translocations). This may be resolved when several mutations can be followed simultaneously using NGS.9 FLT3/ITD has been a difficult target to monitor MRD due to the variable length of the insertions and the duplicity of sequences.10 However, NGS techniques have been applied11 and the getITD analysis algorithm is increasingly used for MRD monitoring.12
Different techniques for measuring residual disease
| Technique . | Methods . | Material . | Cutoff . | Advantage . | Disadvantage . |
|---|---|---|---|---|---|
| Multiparameter flow cytometry | |||||
| LAIP13 | Identify and follow a specific LAIP | BM | 0.1%* | LAIPs found in 90% of patients | Misses clonal evolution |
| Different from normal17 | Identify leukemia cells in the empty spaces of normal BM flow plots | BM | Any MRD | No need for a diagnostic sample | Needs ample experience on regenerating normal BM |
| LAIP-based different from normal48 | Identify a LAIP at diagnosis but measure the whole panel to detect emerging cell populations | BM | 0.1% | Usable for almost all patients | Still needs experience on background LAIP levels |
| Targeted molecular9 | |||||
| qPCR | NPM1 | BM/PB | 2% copies per ABL copies | Highly sensitive, rapid, standardizable | Can be low-level present with no clinical consequences |
| qPCR | Translocations† | BM/PB | 2% copies per ABL copies or 3-4 log reduction | Highly sensitive, rapid, standardizable | BM cannot be as measured frequently as PB |
| Next-generation sequencing | |||||
| NGS24,25 | NPM1 | BM/PB | 0.01%-0.1% | Can be well standardized, reproducible, sensitive, flexible | Not yet standardized, not comparable to qPCR yet, expensive, time consuming |
| NGS15,23,26 | FLT3/ITD | BM/PB | 0.01% | Can be well standardized, reproducible, sensitive, flexible | Expensive and clinical application needs to be validated |
| Technique . | Methods . | Material . | Cutoff . | Advantage . | Disadvantage . |
|---|---|---|---|---|---|
| Multiparameter flow cytometry | |||||
| LAIP13 | Identify and follow a specific LAIP | BM | 0.1%* | LAIPs found in 90% of patients | Misses clonal evolution |
| Different from normal17 | Identify leukemia cells in the empty spaces of normal BM flow plots | BM | Any MRD | No need for a diagnostic sample | Needs ample experience on regenerating normal BM |
| LAIP-based different from normal48 | Identify a LAIP at diagnosis but measure the whole panel to detect emerging cell populations | BM | 0.1% | Usable for almost all patients | Still needs experience on background LAIP levels |
| Targeted molecular9 | |||||
| qPCR | NPM1 | BM/PB | 2% copies per ABL copies | Highly sensitive, rapid, standardizable | Can be low-level present with no clinical consequences |
| qPCR | Translocations† | BM/PB | 2% copies per ABL copies or 3-4 log reduction | Highly sensitive, rapid, standardizable | BM cannot be as measured frequently as PB |
| Next-generation sequencing | |||||
| NGS24,25 | NPM1 | BM/PB | 0.01%-0.1% | Can be well standardized, reproducible, sensitive, flexible | Not yet standardized, not comparable to qPCR yet, expensive, time consuming |
| NGS15,23,26 | FLT3/ITD | BM/PB | 0.01% | Can be well standardized, reproducible, sensitive, flexible | Expensive and clinical application needs to be validated |
qPCR, quantitative polymerase chain reaction.
LAIP cells/CD45-expressing cells.
RUNX1-RUNX1T1, CBFB-MYH11, PML-RARA, KMT2A-MLLT3, DEK-NUP214, BCR-ABL.
MRD at diagnosis
For accurate risk classification,2 the leukemic cells are molecularly characterized by cytogenetic analysis and DNA sequencing. When BM is available (or, if not, PB), MFC-MRD can be performed to characterize the patient-specific LAIP at diagnosis, thereby allowing to follow the LAIP in the MRD setting. A LAIP is defined as CD marker expression combinations present on >10% of blasts that are not found in BM from healthy individuals. A LAIP can be identified in about 90% of patients.13 Measurement of MFC-MRD is recommended when patients do not harbor NPM1 mutations or CBF translocations. Otherwise, patients should be monitored by quantitative PCR or digital PCR for NPM1 mutations or CBF translocations.8 Based on the recent data on FLT3/ITD monitoring by NGS, we expect a rapid uptake of this approach to refine or replace MFC-MRD monitoring as in our patient.14,15 Furthermore, mutations such as DNMT3A, TET2, and ASXL1 (DTA) mutations may reflect clonal hematopoiesis of indeterminate potential and can be often present with a relatively high VAF in nonmalignant clones and therefore cannot be used for MRD assessment.14,16
MRD after the first cycle of chemotherapy
For accurate detection of MFC-MRD after chemotherapy, BM material is required and patients should be in morphologic CR. For the MFC-MRD measurement, the LAIP approach would require only measuring the flow cytometry panel tube that contains the LAIP found at diagnosis. However, during therapy, small cell populations that were undetectable at diagnosis may be selected and proliferate, causing an immune phenotypic shift that the LAIP approach can miss. Another MFC-MRD approach can detect these emerging cell populations and is referred to as different-from-normal, which can also be used when no diagnostic sample is available and is essential to recognize clonal evolution.17 ELN recommendations advocate the combined use by measuring all antibody panels at follow-up to assess MRD as LAIP-based different-from-normal,8 allowing to monitor the dominant LAIP cell population and the possible emergence of a new LAIP.
Often, the search for a donor for alloSCT starts at diagnosis, while some argue the time point after the first cycle of chemotherapy would suffice, since very few patients who are MRD negative become MRD positive after the second cycle and the effort of searching a donor could be spared for nonresponding and MRD-positive patients.18 For adverse risk patients, it is debated whether patients with MRD-positive CR should receive a second cycle of chemo or immediately undergo alloSCT.19
MRD after the second cycle of chemotherapy (postremission therapy)
MFC-MRD with a cutoff of 0.1% after induction therapy is now established as important prognostic factor for outcome.8 The clinical implementation of MRD to guide further postremission strategies varies widely among treatment centers. Recently, several studies indicated that alloSCT could safely be omitted19-21 for many intermediate-risk patients, and those who relapsed could be salvaged effectively with alloSCT.22 The data of Zhang and collaegues19 suggest that alloSCT in favorable- and intermediate-risk patients may only benefit MRD-positive patients. Published data of the 3 trials were highly comparable, as shown in Figure 2. To gather further evidence that MRD-negative intermediate-risk patients can be safely treated with non-alloSCT treatment, a randomized trial would be required showing noninferiority of the non-alloSCT treatment approach compared to alloSCT.
Prognostic effect of consolidation treatment per ELN risk group and MRD status. In favorable- and intermediate-risk patients, non-alloSCT treatment does not negatively affect survival compared to alloSCT in MRD-negative patients. In adverse risk patients, the alloSCT is beneficial independent of MRD. Combined data (means and standard error of the mean) calculated from Zhang et al,19 Venditti et al (intermediate risk-patients),20 and Tettero et al.22
Prognostic effect of consolidation treatment per ELN risk group and MRD status. In favorable- and intermediate-risk patients, non-alloSCT treatment does not negatively affect survival compared to alloSCT in MRD-negative patients. In adverse risk patients, the alloSCT is beneficial independent of MRD. Combined data (means and standard error of the mean) calculated from Zhang et al,19 Venditti et al (intermediate risk-patients),20 and Tettero et al.22
Although some studies recently showed its potential prognostic relevance,15,23 FLT3/ITD MRD is not as well clinically validated as NPM1.24,25 Current data show that FLT3/ITD is a reliable MRD marker before alloSCT at a ≥0.01% VAF cutoff (Table 1).15,23,26 This also applied to patients treated with an FLT3 inhibitor, as shown in the Quantum first trial, which confirmed the 0.01% cutoff as an ideal cutpoint.27 Residual FLT3/ITD MRD should be considered an indication for alloSCT, while it is unclear how the unfavorable prognostic effect of MRD before alloSCT may be mitigated.
Should every FLT3/ITD-positive patient undergo alloSCT? While this is common practice in the United States,28,29 there are data that question this approach. A post hoc analysis of the Ratify trial identified 318 patients with available data comparing allogeneic hematopoietic cell transplantation and non–allogeneic hematopoietic cell transplantation treatment approaches.30 Patients were grouped according to ELN 2017 risk groups based on NPM1 mutation status and FLT3/ITD allelic ratio. Patients with favorable risk (NPM1 mutated and FLT3/ITD low allelic ratio) had long-term survival at 75% when treated with midostaurin independent of alloSCT. Although the survival of the intermediate-risk group was worse with approximately 50% long-term survival, there was no OS difference with or without alloSCT in the midostaurin group. Only the ELN adverse risk group had a significant benefit from alloSCT, especially in the midostaurin-treated group. The ELN 2022 classification has abandoned the FLT3/ITD allelic ratio and groups all FLT3/ITD-positive patients in the intermediate-risk group. The recent studies evaluating FLT3/ITD NGS-MRD show a long-term survival for MRD-negative patients of 50% to 75%.15,23,26 It is thus conceivable that the mutation profile at diagnosis (eg, NPM1 mutated, FLT3/ITD low allelic ratio), the FLT3/ITD MRD status after 2 cycles of chemotherapy, or a combination of both can identify FLT3/ITD-mutated patients who will not benefit from alloSCT. Recently, the results of the BMT-CTN 1506 (MORPHO) study were presented, which evaluated giltertinib maintenance after alloSCT in FLT3/ITD-mutated patients.31 Gilteritinib treatment was associated with an improved relapse-free survival (hazard ratio, 0.515; 95% CI, 0.316-0.838) for the 50.5% of patients with detectable MRD pre- or post-alloSCT, compared to those without detectable MRD. The effect of gilteritinib on OS in patients with detectable MRD has not been reported yet. More research is needed to assess the clinical application of MRD in this subgroup of patients.
MRD monitoring after consolidation completion
The current evidence for accurate MRD monitoring at follow-up is only strong enough for NPM1 and CBF molecular aberrations.8 To further establish the role of MFC-MRD for disease monitoring, this is often still measured every 3 months for the first 2 years after consolidation or alloSCT, or when there are clinical indications of disease recurrence. Collecting these data in clinical trials is warranted as MFC-MRD monitoring after consolidation is not yet common clinical practice but may be beneficial for early intervention options and early relapse detection (Table 2). There are several considerations for using MRD for monitoring. First, the type of patient material matters. For MFC-MRD, PB is not well investigated, and MFC-MRD studies suggested that MRD in PB has higher specificity but 10-fold less sensitivity than BM.32,33 Molecular MRD can be very sensitive, so for this technique, PB is considered suitable. Second, the threshold for MFC-MRD positivity at follow-up and subsequent potential clinical intervention still needs to be established. The cutoff of 0.1% may clearly indicate a high risk for relapse, but lower levels may also already indicate recurrent disease. It is also suggested that kinetics, measured as a certain level of increase in MRD, point to disease recurrence. For molecular techniques, a 1-log increase is considered a useful parameter,8 but for MFC-MRD, sufficient data are lacking. Third, the duration of monitoring needs to be clarified. Previous reports suggest that most relapses occur within 2 years. PB-MRD can be analyzed more frequently (every 4-6 weeks) than BM (every 3 months). Because some relapses occur very fast when a small clone has gained a growth advantage, MRD monitoring would preferably be done in PB. Our patient case showed early positivity after alloSCT, which has been described in 7.5% of patients monitored by MFC-MRD shortly after alloSCT.34
Clinical studies using MRD in adult AML
| Trial/study group . | Patients . | MRD technique . | Key finding . |
|---|---|---|---|
| MRD guidance after induction in complete remission in intermediate-risk patients | |||
| GIMEMA 1310, risk-adapted, MRD-directed therapy for young adults with newly diagnosed AML20 | N = 429 ≤60 years | MFC | Similar 2-year survival in MRD-negative intermediate-risk patients receiving autoSCT compared to MRD-positive patients receiving alloSCT. |
| HOVON-SAKK 132: Addition of lenalidomide to intensive treatment in younger and middle-aged adults with newly diagnosed AML21 | N = 780 AML/high-risk MDS ≤65 years | MFC and NPM1 | No difference in outcome between MRD-negative and MRD-positive intermediate-risk patients, while MRD-negative patients were considered eligible for non-alloSCT treatment. |
| ChiCTR-TRC-10001202 and 10001209 NCT03021330: Prognostic effect and clinical application of early MRD by flow cytometry on de novo AML19 | N = 769 <60 years | MFC | Overall survival of MRD-negative patients in favorable and intermediate-risk groups was comparable between transplant and nontransplant patients. |
| Post-remission measurable residual disease directs treatment choice and improves outcomes for patients with intermediate-risk acute myeloid leukemia in CR149 | N = 235 Intermediate risk 14-60 years | MFC | Retrospective analysis of real-world data postremission MRD directs treatment choice and improves outcomes for patients with intermediate-risk AML in CR1. |
| Current retrospective studies showing relevance ofFLT3/ITD as MRD marker | |||
| DNA sequencing to detect residual disease in adults with AML prior to hematopoietic cell transplant15 | N = 1075 AML in CR1 before transplant ≥18 years | NGS | Persistence of FLT3-ITD or NPM1 variants in blood at an allele fraction of 0.01% or higher was associated with increased relapse and worse survival compared with those without variants detected. |
| Prognostic value of FLT3-ITD residual disease in AML23 | N = 161 FLT3-ITD+ AML in CR1 after induction ≥18 years | NGS | FLT3-ITD MRD is prognostic and better identifies patients at risk for relapse compared to MFC or NGS-based NMP1 MRD alone. |
| Pretransplant FLT3-ITD MRD assessed by high-sensitivity PCR-NGS determines posttransplant clinical outcome26 | N = 104 Pretransplant AML 17-68 years | NGS | Pre-HCT detection of FLT3-ITD MRD is related to poor prognosis and can be an indication for future MRD-directed therapeutic strategies. |
| Allogenic stem cell transplantation conditioning/donor selection | |||
| Impact of conditioning intensity of allogeneic transplantation for acute myeloid leukemia with genomic evidence of residual disease (CIBMTR)50 | N = 190 ≥18 years in first CR | NGS: FLT3, NPM1, IDH1, IDH2, and/or KIT variants | MAC rather than RIC in patients with AML with genomic evidence of MRD before alloHCT can result in improved survival. |
| Impact of pre-transplant induction and consolidation cycles on AML allogeneic transplant outcomes: a CIBMTR analysis in 3113 AML patients51 | N = 3113 ≥18 years | MFC, cytogenetics and molecular | Detectable MRD at the time of MAC alloHCT did not impact outcomes while detectable MRD preceding RIC alloHCT was associated with an increased risk of relapse. |
| Measurable residual disease, conditioning regimen intensity, and age predict outcome of allogeneic hematopoietic cell transplantation for acute myeloid leukemia in first remission52 | N = 2292 (EBMT) ≥18 years | MFC and molecular | Patients aged <50 y with AML CR1 MRD-positive status should preferentially be offered MAC alloHCT. Prospective studies are needed to address whether patients who are AML CR1 MRD negative may be spared the toxicity of MAC regimens. |
| Haploidentical allograft is superior to matched sibling donor allograft in eradicating pre-transplantation MRD of AML patients as determined by MFC: a retrospective and prospective analysis53 | N = 339 ≤60 years | MFC | For MRD-positive patients, haploSCT was associated with lower incidence of relapse and better survival, suggesting a stronger antileukemia effect compared to matched sibling donor transplantation. |
| Monitoring during maintenance | |||
| Prospective phase II (NCT00801489): Common kinase mutations do not impact optimal molecular responses in CBF AML treated with fludarabine, cytarabine, and G-CSF based regimens54 | N = 174 ≥18 years | Optimal PCR response FLT3, RAS, and KIT | Attainment of PCR <0.01% during/after consolidation improved RFS and was more important than achieving early optimal PCR response (post C1 PCR <0.1%). |
| Observational cohort: MRD status and FLT3 inhibitor therapy in patients with FLT3/ITD mutated AML following allogeneic hematopoietic cell transplantation55 | N = 34 ≥18 years | NGS: FLT3/ITD | Prognostic significance of NGS-based MRD monitoring for FLT3/ITD and the ability of post-alloSCT maintenance to prevent relapse and death. |
| Measurable residual disease-guided treatment with azacitidine to prevent haematological relapse in patients with myelodysplastic syndrome and acute myeloid leukaemia (RELAZA2): an open-label, multicentre, phase 2 trial56 | N = 53 ≥18 years CR-MRD+ | qPCR: NPM1, DEK-NUP214, RUNX1-RUNX1T1, CBFb-MYH11 | MRD-guided treatment with azacitidine prevented or substantially delayed hematologic relapse with an acceptable safety. |
| Monitoring after treatment | |||
| Posttransplant MRD and T-cell chimerism status predict outcomes in patients allografted with AML/MDS (FIGARO)3 | N = 187 ≥18 years Peri-alloSCT | MFC and T-cell chimerism | Post-alloSCT MRD is an important predictor of outcome and is most informative when combined with T-cell chimerism. |
| Bone marrow CD34+ molecular chimerism as an early predictor of relapse after alloSCT in patients with AML (PROMISE)57 | N = 168 <75 years After alloSCT | Molecular WT1 and CD34+ chimerism | Molecular chimerism and WT1 after alloSCT (first and third months) are useful MRD markers. When considered together at third month, CD34+ molecular chimerism could represent an earlier predictor of relapse compared to WT1. |
| MRD in nonintensively treated patients | |||
| Undetectable measurable residual disease is associated with improved outcomes in AML irrespective of treatment intensity. Retrospective analysis of real world data58 | N = 635 (250 nonintensively treated) ≥18 years | MFC: excluding APL and CBF AML | Achievement of MRD negativity should be the key objective of AML therapy in both high- and low-intensity treatment regimens. |
| Measurable residual disease response and prognosis in treatment-naïve acute myeloid leukemia with venetoclax and azacitidine (VIALE-A)46 | N = 190 (164 in CRc for MRD assessment) ≥18 years | MFC | Patients who achieved CRc and MRD negativity at any time point during treatment with venetoclax and azacitidine had longer duration of response, EFS, and OS than responding MRD-positive patients |
| Ibrutinib added to 10-day decitabine for older patients with AML and higher risk MDS (HOVON-SAKK 135)59 | N = 144 Unfit AML/high-risk MDS >60 years | MFC | After 3 cycles of treatment, 28 (49%) of 57 patients were MRD negative. In this limited number of cases, MRD revealed no apparent impact on outcome. |
| MRD characterization for targeted therapy | |||
| ALLG AMLM26 phase 1B/2 study investigating novel therapies to target early relapse and clonal evolution as pre-emptive therapy in AML (INTERCEPT): a multi-arm, precision-based, recursive, platform trial60 | Patients with MRD-relapse ≥18 years | MFC and molecular | Ongoing study: The primary end point is MRD response (≥1 log10 reduction in molecular MRD or flow MRD <0.1%) within 100 days of the first dose of study drug. |
| Intermediate end point | |||
| Early assessment of clofarabine effectiveness based on measurable residual disease, including AML stem cells (HOVON-SAKK 102)38 | N = 291 ≤65 years | MFC including leukemia stem cells and NPM1 | Lower levels of MRD were found in clofarabine-treated patients than in patients treated without clofarabine in the intermediate-I risk group. |
| Umbrella trial in myeloid malignancies: the MyeloMATCH National Clinical Trials Network Precision Medicine Initiative61 | Older adults, MDS and young adults | MFC and NGS | Ongoing study: Assignment of treatment to patients based on biomarkers related to the disease such as mutations and MRD. |
| Trial/study group . | Patients . | MRD technique . | Key finding . |
|---|---|---|---|
| MRD guidance after induction in complete remission in intermediate-risk patients | |||
| GIMEMA 1310, risk-adapted, MRD-directed therapy for young adults with newly diagnosed AML20 | N = 429 ≤60 years | MFC | Similar 2-year survival in MRD-negative intermediate-risk patients receiving autoSCT compared to MRD-positive patients receiving alloSCT. |
| HOVON-SAKK 132: Addition of lenalidomide to intensive treatment in younger and middle-aged adults with newly diagnosed AML21 | N = 780 AML/high-risk MDS ≤65 years | MFC and NPM1 | No difference in outcome between MRD-negative and MRD-positive intermediate-risk patients, while MRD-negative patients were considered eligible for non-alloSCT treatment. |
| ChiCTR-TRC-10001202 and 10001209 NCT03021330: Prognostic effect and clinical application of early MRD by flow cytometry on de novo AML19 | N = 769 <60 years | MFC | Overall survival of MRD-negative patients in favorable and intermediate-risk groups was comparable between transplant and nontransplant patients. |
| Post-remission measurable residual disease directs treatment choice and improves outcomes for patients with intermediate-risk acute myeloid leukemia in CR149 | N = 235 Intermediate risk 14-60 years | MFC | Retrospective analysis of real-world data postremission MRD directs treatment choice and improves outcomes for patients with intermediate-risk AML in CR1. |
| Current retrospective studies showing relevance ofFLT3/ITD as MRD marker | |||
| DNA sequencing to detect residual disease in adults with AML prior to hematopoietic cell transplant15 | N = 1075 AML in CR1 before transplant ≥18 years | NGS | Persistence of FLT3-ITD or NPM1 variants in blood at an allele fraction of 0.01% or higher was associated with increased relapse and worse survival compared with those without variants detected. |
| Prognostic value of FLT3-ITD residual disease in AML23 | N = 161 FLT3-ITD+ AML in CR1 after induction ≥18 years | NGS | FLT3-ITD MRD is prognostic and better identifies patients at risk for relapse compared to MFC or NGS-based NMP1 MRD alone. |
| Pretransplant FLT3-ITD MRD assessed by high-sensitivity PCR-NGS determines posttransplant clinical outcome26 | N = 104 Pretransplant AML 17-68 years | NGS | Pre-HCT detection of FLT3-ITD MRD is related to poor prognosis and can be an indication for future MRD-directed therapeutic strategies. |
| Allogenic stem cell transplantation conditioning/donor selection | |||
| Impact of conditioning intensity of allogeneic transplantation for acute myeloid leukemia with genomic evidence of residual disease (CIBMTR)50 | N = 190 ≥18 years in first CR | NGS: FLT3, NPM1, IDH1, IDH2, and/or KIT variants | MAC rather than RIC in patients with AML with genomic evidence of MRD before alloHCT can result in improved survival. |
| Impact of pre-transplant induction and consolidation cycles on AML allogeneic transplant outcomes: a CIBMTR analysis in 3113 AML patients51 | N = 3113 ≥18 years | MFC, cytogenetics and molecular | Detectable MRD at the time of MAC alloHCT did not impact outcomes while detectable MRD preceding RIC alloHCT was associated with an increased risk of relapse. |
| Measurable residual disease, conditioning regimen intensity, and age predict outcome of allogeneic hematopoietic cell transplantation for acute myeloid leukemia in first remission52 | N = 2292 (EBMT) ≥18 years | MFC and molecular | Patients aged <50 y with AML CR1 MRD-positive status should preferentially be offered MAC alloHCT. Prospective studies are needed to address whether patients who are AML CR1 MRD negative may be spared the toxicity of MAC regimens. |
| Haploidentical allograft is superior to matched sibling donor allograft in eradicating pre-transplantation MRD of AML patients as determined by MFC: a retrospective and prospective analysis53 | N = 339 ≤60 years | MFC | For MRD-positive patients, haploSCT was associated with lower incidence of relapse and better survival, suggesting a stronger antileukemia effect compared to matched sibling donor transplantation. |
| Monitoring during maintenance | |||
| Prospective phase II (NCT00801489): Common kinase mutations do not impact optimal molecular responses in CBF AML treated with fludarabine, cytarabine, and G-CSF based regimens54 | N = 174 ≥18 years | Optimal PCR response FLT3, RAS, and KIT | Attainment of PCR <0.01% during/after consolidation improved RFS and was more important than achieving early optimal PCR response (post C1 PCR <0.1%). |
| Observational cohort: MRD status and FLT3 inhibitor therapy in patients with FLT3/ITD mutated AML following allogeneic hematopoietic cell transplantation55 | N = 34 ≥18 years | NGS: FLT3/ITD | Prognostic significance of NGS-based MRD monitoring for FLT3/ITD and the ability of post-alloSCT maintenance to prevent relapse and death. |
| Measurable residual disease-guided treatment with azacitidine to prevent haematological relapse in patients with myelodysplastic syndrome and acute myeloid leukaemia (RELAZA2): an open-label, multicentre, phase 2 trial56 | N = 53 ≥18 years CR-MRD+ | qPCR: NPM1, DEK-NUP214, RUNX1-RUNX1T1, CBFb-MYH11 | MRD-guided treatment with azacitidine prevented or substantially delayed hematologic relapse with an acceptable safety. |
| Monitoring after treatment | |||
| Posttransplant MRD and T-cell chimerism status predict outcomes in patients allografted with AML/MDS (FIGARO)3 | N = 187 ≥18 years Peri-alloSCT | MFC and T-cell chimerism | Post-alloSCT MRD is an important predictor of outcome and is most informative when combined with T-cell chimerism. |
| Bone marrow CD34+ molecular chimerism as an early predictor of relapse after alloSCT in patients with AML (PROMISE)57 | N = 168 <75 years After alloSCT | Molecular WT1 and CD34+ chimerism | Molecular chimerism and WT1 after alloSCT (first and third months) are useful MRD markers. When considered together at third month, CD34+ molecular chimerism could represent an earlier predictor of relapse compared to WT1. |
| MRD in nonintensively treated patients | |||
| Undetectable measurable residual disease is associated with improved outcomes in AML irrespective of treatment intensity. Retrospective analysis of real world data58 | N = 635 (250 nonintensively treated) ≥18 years | MFC: excluding APL and CBF AML | Achievement of MRD negativity should be the key objective of AML therapy in both high- and low-intensity treatment regimens. |
| Measurable residual disease response and prognosis in treatment-naïve acute myeloid leukemia with venetoclax and azacitidine (VIALE-A)46 | N = 190 (164 in CRc for MRD assessment) ≥18 years | MFC | Patients who achieved CRc and MRD negativity at any time point during treatment with venetoclax and azacitidine had longer duration of response, EFS, and OS than responding MRD-positive patients |
| Ibrutinib added to 10-day decitabine for older patients with AML and higher risk MDS (HOVON-SAKK 135)59 | N = 144 Unfit AML/high-risk MDS >60 years | MFC | After 3 cycles of treatment, 28 (49%) of 57 patients were MRD negative. In this limited number of cases, MRD revealed no apparent impact on outcome. |
| MRD characterization for targeted therapy | |||
| ALLG AMLM26 phase 1B/2 study investigating novel therapies to target early relapse and clonal evolution as pre-emptive therapy in AML (INTERCEPT): a multi-arm, precision-based, recursive, platform trial60 | Patients with MRD-relapse ≥18 years | MFC and molecular | Ongoing study: The primary end point is MRD response (≥1 log10 reduction in molecular MRD or flow MRD <0.1%) within 100 days of the first dose of study drug. |
| Intermediate end point | |||
| Early assessment of clofarabine effectiveness based on measurable residual disease, including AML stem cells (HOVON-SAKK 102)38 | N = 291 ≤65 years | MFC including leukemia stem cells and NPM1 | Lower levels of MRD were found in clofarabine-treated patients than in patients treated without clofarabine in the intermediate-I risk group. |
| Umbrella trial in myeloid malignancies: the MyeloMATCH National Clinical Trials Network Precision Medicine Initiative61 | Older adults, MDS and young adults | MFC and NGS | Ongoing study: Assignment of treatment to patients based on biomarkers related to the disease such as mutations and MRD. |
alloHCT, allogeneic hematopoietic cell transplantation; APL, acute promyelocytic leukemia; autoSCT, autologous stem cell transplantation; CRc, complete remission rate; EBMT, European Society for Blood and Marrow Transplantation; haploSCT, haploidentical stem cell transplantation; HCT, hematopoietic cell transplantation; MAC, myeloablative conditioning; MDS, myelodysplastic syndrome; RFS, relapse free survival; RIC, reduced intensity conditioning.
MRD relapse
MRD relapse is defined by the ELN recommendations independent of the technique that is used: “MRD relapse is defined as either (1) conversion of MRD negativity to MRD positivity independent of the MRD technique or (2) increase of MRD ≥1 log10 between any 2 positive samples measured in the same tissue (PB or BM) in patients with low-level MRD. Conversion from negative to positive MRD in PB or BM should be confirmed within 4 weeks, in a second consecutive sample, preferably with a BM sample.”8 After MRD relapse, alloSCT can be beneficial for curation.22 The most suitable technique, best time points, duration of MRD monitoring after alloSCT, and for which subsets of patients are still under investigation. An early indication that NGS-MRD is also prognostic after relapse was recently published for patients treated with fludarabine, high dose cytarabine (Ara-C), idarubicin and granulocyte-colony stimulating factor (G-CSF) and venetoclax, where MRD-negative patients had an excellent survival.35
Intermediate end point
Since many new drugs are currently available, MRD as intermediate end point would be worthwhile to fasten the process of drug approval.36 The association between MRD and prognostic value has been very well established for intensively treated patients.37 However, limited positive trials have included the measurement of MRD to show that the treatment effects are associated with a corresponding decline in MRD or the number of MRD-negative patients.38 Thus, for the regulatory agencies to accept MRD as intermediate end point, additional data are required.39
Discussion
MRD monitoring in AML is quickly evolving from risk stratification to clinical decision-making to guide therapy, monitor the disease, detect MRD relapse early, and improve the drug development process by using MRD as an intermediate end point for clinical trials. For any MRD-including trial design, it is instrumental to have the exact information on when and what clinical decisions are made on MRD. Some clinical aspects may change the treatment decision, and the prognostic value of MRD is lost when treatment is guided based on MRD.21 In addition, it still needs to be assessed whether quality of life improves with omitting or delaying alloSCT, since these data are currently lacking.
With the new technologies and insights that are currently under investigation, we may improve patient management in the future. For MFC-MRD, one of the advancements would be detecting the leukemia stem cell (LSC) load, measured as a percentage of CD34+/CD38⁻/LSCmarker+ cells per CD45-expressing cells.40 Several studies have shown that a high LSC load was associated with a relatively poor outcome at diagnosis and after induction.41-44 Although promising, more evidence is needed to incorporate LSC load into treatment decisions.
The NGS-based MRD detection is promising but requires standardization. The clinical utility of NGS has been reported for FLT3/ITD and NPM1 mutations. To move this field further, consensus should be accomplished in various aspects such as selection of the most relevant molecular markers, sequencing approaches, sampling tissue (BM or PB), cutoffs, and timing of sampling.
In conclusion, MRD is an important prognostic factor to inform consolidation treatment after CR in intermediate-risk patients. Based on recent MRD data from clinical trials and real-world clinical practice, more evidence is being gathered for the broader use of MRD for the management of AML (Table 2). Still, clearly more data are necessary to determine how MRD (cutoffs, kinetics, patient material, risk group, etc) can best be implemented in clinical practice. The relevance of MRD using current MRD techniques also needs additional studies when novel (targeted) or nonintensive treatments are used.45-47 Improved standardization of MRD measurements should enable the combination of data from different trials and is essential in the light of the in vitro diagnostics regulation. When harmonized MRD data become available, additional MRD applications may be implemented in newly updated guidelines in the coming years.
Acknowledgments
The authors thank Prof. G. J. Ossenkoppele for critically reading the manuscript.
Conflict-of-interest disclosure
Jacqueline Cloos serves an advisory role for Novartis; has received research grants for her institution from Novartis, Merus, Takeda, Genentech, and BD Biosciences; and has received a royalty/license from Navigate and BD Biosciences.
Lok Lam Ngai: no competing financial interests to declare.
Michael Heuser serves an advisory role for Abbvie, BMS, Glycostem, Servier, PinotBio, Amgen, Pfizer, and LabDelbert; has received honoraria from Certara, Jazz Pharmaceuticals, Janssen, Novartis, Pfizer, and Sobi; and has received research funding to his institution from Abbive, Agios, Astellas, BergenBio, BMS, Glycostem, Jazz Pharmaceuticals, Karyopharm, Loxo Oncology, and PinotBio.
Off-label drug use
Jacqueline Cloos: nothing to disclose.
Lok Lam Ngai: nothing to disclose.
Michael Heuser: Use of venetoclax in relapsed/refractory AML patients and in combination with FLAG-IDA is neither approved by FDA nor EMA. Giltertinib maintenance after alloSCT in FLT3/ ITD-mutated patients is neither approved by FDA nor EMA.