Abstract
Because of the breakdown of malaria control programs, the constant emergence of drug resistant parasites, and, possibly, climatic changes malaria poses a major problem for the developing countries. In addition, because of the speed of international travel it is being seen with increasing frequency as an imported disease in non-tropical countries. This update explores recent information about the pathophysiology of the disease, its protean hematological manifestations, and how carrier frequencies for the common hemoglobin disorders have been maintained by relative resistance to the malarial parasite.
In Section I, Dr. Louis Miller and colleagues consider recent information about the pathophysiology of malarial infection, including new information about interactions between the malarial parasite and vascular endothelium.
In Section II, Dr. David Roberts discusses what is known about the complex interactions between red cell production and destruction that characterize the anemia of malaria, one of the commonest causes of anemia in tropical countries.
In Section III, Dr. David Weatherall reviews recent studies on how the high gene frequencies of the thalassemias and hemoglobin variants have been maintained by heterozygote advantage against malaria and how malaria has shaped the genetic structure of human populations.
I. The Pathogenic Basis of Malaria**This section originally appeared in Nature [2002;Feb 7;415(6872):673-9] and has been reprinted with permission of the publisher.
This section originally appeared in Nature [2002;Feb 7;415(6872):673-9] and has been reprinted with permission of the publisher.
Louis H. Miller, MD,** Laboratory of Parasitic Diseases, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD 20892
KEMRI-Wellcome Trust Collaboration Programme, Center for Geographic Medicine Research Coast, Post Office Box 230, Kilifi, Kenya kmarsh@kilifi.mimcom.net
Malaria Research and Training Center, Bamako, Mali okd@mrtcbko.malinet.ml
Acknowledgments: We thank John Barnwell (CDC, Atlanta, GA) for sharing data before publication.
Malaria is today a disease of poverty and underdeveloped countries. In Africa, mortality remains high because there is limited access to treatment in the villages. We should follow in Pasteur’s footsteps by using basic research to develop better tools for the control and cure of malaria. Insight into the complexity of malaria pathogenesis is vital for understanding the disease and will provide a major step towards controlling malaria. Those of us who work on pathogenesis must widen our approach and think in terms of new tools such as vaccines to reduce disease. The inability of many countries to fund expensive campaigns and anti-malarial treatment requires that our new tools be highly effective and affordable.
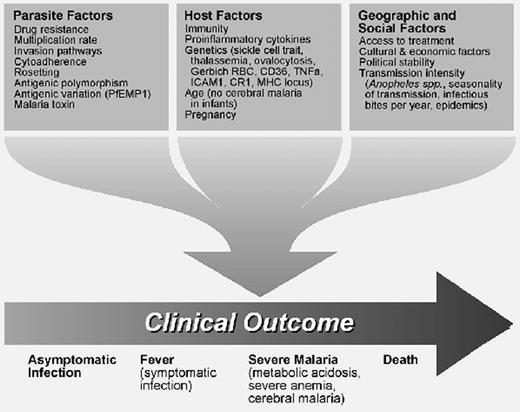
Millions of children die from malaria in Africa every year.1 But the clinical outcome of an infection in a child depends on many factors (Figure 1 ). These factors, often ill-defined, determine the outcome in each child. The top priority must be disease prevention because of the inability of the mothers to access or afford optimal treatment, and the ever-evolving drug resistance. Prevention may be effected through vector control such as insecticide-treated bednets or through the development of antimalarial vaccines.
Malaria: The Disease
Over the last 10 years, there have been several key shifts in our understanding of what constitutes severe malaria, and these shifts define the issues in pathogenesis that need to be explored to better treat sick children. First has been the increasing recognition that severe malaria is a multisystem disorder, even when the most dramatic manifestations may appear to involve a single organ such as the brain. In particular, metabolic acidosis, often profound, has been recognized as a major pathophysiologic feature that cuts across the classical clinical syndromes of cerebral malaria and severe malarial anemia.2 It is the single most important determinant of survival and directly leads to a common, but previously poorly recognized, syndrome of respiratory distress.3 In most cases, this is predominantly (but not exclusively) a lactic acidosis.4 Children with severe malaria have multiple causes of lactic acidosis, from increased production by parasites, through direct stimulation by cytokines, to decreased clearance by the liver; however, most important by far is likely to be the combined effects of several factors in reducing oxygen delivery to tissues.5 A key feature of the biology of Plasmodium falciparum is the ability of infected red blood cells (RBC) to adhere to the linings of small blood vessels. Such sequestered parasites provide considerable obstruction to tissue perfusion. In addition, it is becoming clear that in severe malaria there may be marked reductions in the deformability of uninfected RBC.6,7 The pathogenesis of this abnormality is not clear, but the extremely strong correlation with acidosis suggests that it may play a major role in compromising tissue blood flow. Patients with malaria are often dehydrated and relatively hypovolemic,8 potentially exacerbating microvascular obstruction by reducing perfusion pressure. RBC destruction is also an inevitable part of malaria, and anemia further compromises oxygen delivery.
The second and related shift in thinking about severe disease is the realization that there is no simple one-to-one correlation between clinical syndromes of severe disease and pathogenic processes. Thus, severe anemia may arise from multiple poorly understood processes including acute hemolysis of uninfected RBC and dyserythropoiesis, as well as through the interaction of malaria infection with other parasite infections and with nutritional deficiencies.9 It is increasingly evident that for many desperately sick children, a simple one pathogenone disease model is not adequate, as bacteremia due to common pathogens may be present with acute malaria and may be a factor in mortality.10,11 Even the rigorously defined syndrome of cerebral malaria actually comprises children who have arrived at the point of coma through a variety of routes. In many of these children, coma seems to be a response to overwhelming metabolic stress rather than a primary problem in the brain. Such children are often profoundly acidotic and may regain consciousness remarkably quickly following appropriate resuscitation,12 suggesting that cerebral malaria in this instance cannot be a consequence of the classical histologic picture.
Similarly, it has recently been recognized that a significant proportion of children in coma are, in fact, experiencing covert status epilepticus,13 which responds rapidly to appropriate anticonvulsant therapy. The pathogenesis of this condition is unknown, but again the speed of resolution argues against classical views of pathogenesis. The picture that emerges is of multiple processes leading to a common picture. These distinctions are of much more than academic value: they have direct implications for therapy, but they also identify the research questions for improving therapy of sick children.
Severe malaria is complex and probably cannot be accurately represented by any single schema; however, our current understanding of the way several key pathogenic processes combine to lead to severe disease invokes several basic processes: rapid expansion of infected RBC mass, destruction of both infected and uninfected RBC, microvascular obstruction, and inflammatory processes that combine to lead to reduced tissue perfusion. This in turn may lead to downstream processes at a cellular level that further exacerbate the situation. These general processes, which affect many tissue beds, may also be focused on specific organs in some situations, for instance the brain in cerebral malaria or the placenta during malaria in pregnancy. This could reflect both host-specific factors (for example, an increased likelihood to express particular receptors on cerebral endothelium) and parasite-specific factors (for example, the expression of molecules on the infected RBC surface that are particularly suited for binding to certain receptors). In this article we review the major advances in pathogenesis with the hope that they will lead to new tools to prevent disease before children are so sick that they need hospitalization.
Although the disease must ultimately be understood in humans, much of our knowledge of pathogenesis depends on studies in nonhuman species and in vitro culture of P. falciparum. Invasion of hepatocytes and RBC studied in rodent malarias P. berghei and P. yoelii and the rhesus malaria P. knowlesi, respectively, provided insight about these processes. Inflammatory cytokines are often studied in rodent malarias. In addition, these species are important for screening of drugs and vaccines, including human malarias in New World primates.
The Plasmodium Life Cycle and Pathogenesis
P. falciparum and, to a much lesser extent, P. vivax14 are the main causes of disease and death from malaria (Figure 2, see Color Figures, 512). Mosquitoes inject parasites (sporozoites) into the subcutaneous tissue and less frequently directly into the bloodstream; from there, sporozoites travel to the liver. Recent evidence indicates that sporozoites pass through several hepatocytes before invasion is followed by parasite development.15 The co-receptor on sporozoites for invasion involves, in part, the thrombospondin domains on the circumsporozoite protein and on thrombospondin-related adhesive protein (TRAP). These domains bind specifically to heparin sulfate proteoglycans on hepatocytes in the region in apposition to sinusoidal endothelium and Kuppfer cells.16 Within the hepatocyte, each sporozoite develops into tens of thousands of merozoites, each able to invade a RBC on release from the liver. Disease begins only once the asexual parasite multiplies within RBC. This is the only gateway to disease. P. falciparum and P. vivax within RBC develop over 48 hours, producing around 20 merozoites in a mature parasite, each able to invade other RBC. A small proportion of asexual parasites converts to gametocytes that are critical for the transmission of the infection to others through female anopheline mosquitoes but cause no disease. Here the strategy of P. vivax differs from that of P. falciparum. P. vivax develops into gametocytes soon after release of merozoites from the liver; P. falciparum gametocytes develop much later. Early treatment of clinical malaria attacks by anti-bloodstage chemotherapy for P. falciparum also kills the developing gametocytes; P. vivax transmits before the symptomatic stage of the disease.
Invasion of RBCs
The sequence of invasion is probably similar for all Plasmodium spp. The parasite must engage receptors17 on RBC for binding and undergo apical reorientation,18 junction formation,19 and signaling. The parasite then induces a vacuole derived from the RBC plasma membrane and enters the vacuole by a moving junction. Three organelles on the invasive (apical) end of parasites (rhoptries, micronemes, and dense granules) define the phylum Apicomplexa. Receptors for invasion of RBC by merozoites and for invasion of liver by sporozoites are found in micronemes,20 on the cell surface, and in rhoptries. The distribution of these receptors within an organelle may protect the parasite from antibody-mediated neutralization, as the release after contact with the RBC may limit their exposure to antibody.
A critical question in parasite biology remains the identification of the signaling pathways to release organellar contents on contact with a host RBC (Figure 2, 512). Malaria parasites have intracellular signaling pathways through phosphoinositide, cyclic AMP (cAMP), and calcium-dependent pathways. What remains completely unknown is which merozoite surface molecules recognize the RBC surface and signal for the invasion process. These events include release of critical molecules from apical organelles and the initiation of the actin-myosin moving junction that brings the parasite within the vacuole that forms in the RBC. The TRAP protein interacts with skeletal proteins in malaria sporozoites and in Toxoplasma gondii,21 but the equivalent molecule for merozoites has yet to be identified.
Both P. falciparum and P. vivax can cause severe anemia, but only P. falciparum causes the multiple complications of cerebral malaria, hypoglycemia, metabolic acidosis, and respiratory distress. Certain differences in the biology of the two parasites partially explain the differences in patterns of disease. First, P. falciparum can invade a large percentage of the RBC, whereas P. vivax is limited to reticulocytes. Similar differences are found between virulent and avirulent P. yoelii. Both invade reticulocytes preferentially, but once the reticulocytes are consumed, virulent P. yoelii can invade all RBC, leading to higher parasitemia and death. A recent study of severe compared to uncomplicated falciparum malaria suggested a similar pattern, with the virulent P. falciparum invading all RBC and the avirulent parasites invading only a subpopulation.22
A second difference is the surprising redundancy of invasion pathways in P. falciparum, lacking in P. vivax. P. vivax invades only Duffy blood group-positive RBC23 and is largely limited to reticulocytes. In West Africa, where the Duffy blood group is missing on RBC, P. vivax essentially disappeared. The Duffy negative blood group has also occurred independently in Papua New Guinea,24 an area of high endemicity of P. vivax. The limitations in invasion of P. vivax have led to the discovery of two families of parasite receptors: i) the parasite molecule that binds to the Duffy blood group system and Duffy binding-like (DBL) homologous proteins of P. falciparum and P. knowlesi,25 and ii) the parasite reticulocyte-binding proteins of P. vivax26 and reticulocyte binding-like (RBL) homologous proteins of P. falciparum27 and P. yoelii.28 The various members of the DBL and RBL families may recognize different RBC receptors than the Duffy blood group or the receptor on reticulocytes. The receptor grouping into DBL and RBL refers to the family of homologous parasite proteins, not the specificity on the RBC for binding.
There is a large family of RBL genes in P. yoelii. Each of the merozoites within a single infected RBC can express a different member of the RBL family.29 If each has a different RBC-binding specificity, then the parasite has a greater chance for survival. Thus, although the full details for the DBL and RBL families are unknown, they clearly determine much of the flexibility for invasion by the various Plasmodium spp. This flexibility, in turn, determines the maximum parasitemia and disease caused by the various parasites.
P. falciparum can use its multiple redundant alternative pathways to invade at equal or reduced efficiency RBC lacking a particular receptor such as sialic acid.30,31 Three sialoglycoprotein-dependent pathways involving RBC and parasite co-receptors have been identified: i) glycophorin A and the parasite DBL protein, EBA-175,32 ii) glycophorin C/D and the DBL parasite protein, BAEBL,33 and iii) a trypsin-resistant pathway involving a P. falciparum RBL protein.27 A fourth may involve sialic acid on glycophorin B.27
Despite markedly reduced invasion of glycophorin A-negative RBC, only glycophorin B mutations occur in Africa. Gerbich RBC fail to express glycophorin D and express an altered glycophorin C and have reduced binding to the parasite molecule, BAEBL. Gerbich RBC are rare in most parts of the world except in the falciparum-endemic regions of Papua New Guinea, where the allele frequency approaches 50%.34 Such redundancy and alternative pathways are a major advantage to the survival of P. falciparum in response to changes in host genetics. The parasite, however, may become less virulent as it adapts to survival in these deficient RBC.
Studying the DBL and RBL families has begun to yield a molecular understanding of the diverse invasion pathways for P. falciparum and other Plasmodium spp. Although other parasite proteins on the merozoite surface and in apical organelles have been proposed as receptors,35–,37 there is no direct evidence so far. Because invasion is such a complex series of events from RBC binding, to apical reorientation, to entry, it seems likely that multiple proteins are involved for efficient invasion. For example, evidence has been presented that invasion requires cleavage of a RBC surface protein by a parasite serine protease.38 This parasite enzyme has yet to be identified. Thus, the molecular and cellular events surrounding each step in invasion still remain to be elucidated. Understanding these pathways will give insight into parasite virulence and will facilitate rational vaccine design against merozoite invasion.
Binding of RBC to Vascular Endothelium and Placenta
An important difference between P. falciparum and other human malarias is the way that P. falciparum modifies the surface of the RBC for adherence of both asexual parasites and gametocytes to the endothelium and asexual parasites within placenta. As a result, only ring forms of P. falciparum are found within the circulating blood (for review see refs. 39–,41). The surface of P. falciparum trophozoite- and schizont-infected RBC is covered with knob-like excrescences that are the contact points with host cells.42 The adherence protects the parasite from destruction, as non-adherent mature parasitized RBC are rapidly cleared within the spleen.43
Trying to decipher the highly complex and pathogenic adhesion process emphasizes how much we have learned and how little we understand. To answer whether and how sequestration can lead to pathogenesis, we should first look at how the parasite sequesters. The P. falciparum adhesion process is comparable to adhesion of leukocytes, where most parasites first tether, then roll, before becoming firmly adherent.44,45 Most host receptors are involved with tethering and rolling but are unable to support firm adhesion under flow on their own.44,46 Binding to these host receptors is important, as it significantly increases adhesion that may allow the parasite to efficiently bind to endothelium of various organs.47 Only two receptors, CD36 and chondroitin sulfate A (CSA), provide stable stationary adherence.44,46
Parasites sequester in various organs including heart, lung, brain, liver, kidney, subcutaneous tissues, and placenta. The various endothelial cells in these organs and syncytiotrophoblasts in placenta express different and variable amounts of host receptors. To successfully adhere to these cells, the parasite can bind to a large number of receptors39 (Figure 3, see Color Figures, 512). The adhesion phenotype is not homogenous, and different parasites can bind to variable numbers and combinations of host receptors.48,49 This variability is believed to affect the tissue distribution and pathogenesis of parasites.
Amazingly, a single parasite protein, the P. falciparum erythrocyte membrane protein 1 (PfEMP1) expressed at the infected erythrocyte surface, mediates its binding to the various receptors.39–,41 PfEMP1 is encoded by the large and diverse var gene family that is involved in clonal antigenic variation and plays a central role in P. falciparum pathogenesis.50–,52 The multiple adhesion domains located at the extracellular region of PfEMP1 can simultaneously recognize several host receptors. These domains contain variable numbers of different (5 types) DBL domains, so named for the homology to the DBL domains involved in RBC invasion and 1-2 cysteine-rich interdomain regions (CIDR).52,53 The binding domains for several host receptors were recently mapped to various DBL and CIDR domains.39,54 The diversity within this gene family is extensive, and numerous var genes appear in the parasite population. Although each parasite expresses a single var gene55 (of 50 in its genome), this can change at a rate of up to 2% per generation.56
In most cases, the binding to host endothelium does not lead to pathogenesis, as most infections result in malaria that is devoid of complications.57 What leads to the transition from uncomplicated to a serious infection such as cerebral malaria is unclear at present. An intriguing possibility is that expression of particular binding properties will lead to distinct patterns of sequestration and to pathogenic consequences. One example is the sequestration of infected RBC within the placenta that causes premature delivery, low birth weight, and increased mortality in the newborn and anemia in the mother. Parasitized RBC isolated from placentas have a unique adhesion property different from parasites collected from non-pregnant individuals.48,58 These parasites bound to CSA and failed to adhere to CD36, the critical host receptor for sequestration in microvasculature. The apparent dichotomy in adhesion to these receptors was selected to allow the parasite to sequester not to endothelium but in placenta, perhaps a site of reduced immunity. Indeed, CSA-binding parasites express PfEMP1 with a DBLγ domain that binds CSA and a non-CD36-binding CIDR1.59,60 In contrast, CD36-adherent parasites express a PfEMP1 with a CD36-binding CIDR1.60
Sequestration of parasites in the brain may be related to cerebral malaria and may involve the ICAM-1 receptor.41 Although infected RBC bind to brain endothelium at autopsy, it is unknown whether this represents a different distribution of adhesion from uncomplicated malaria. An increase in ICAM-1 expression in brain endothelium may explain differences in parasite adhesion in cerebral malaria.61,62 The role of sequestration in other severe complications of malaria remains unclear. Pathogenic connections between adhesion and host receptors are supported by both a nonsense in the gene of the adhesion receptor CD36 that is associated with protection from severe malaria, and the link between complement receptor 1 and ABO blood group antigens and rosetting (the binding of uninfected RBCs).40,63,64 Several investigators have suggested that simultaneous binding to multiple receptors might be associated with more severe cases of malaria,65 but specific data are lacking. Some properties, such as rosetting40 and clumping,66 appear at higher frequencies in cases of severe disease, but these associations have not been found in all studies and their effect on pathogenesis remains obscure. One possibility is that competition (for adhesion) between parasites drives some of them to develop new adhesion properties and sequester in less desirable locations that lead to pathogenesis.
Although dissecting various individual interactions is a good experimental approach, the outcome of an infection and progression into pathology depends on the specific and dynamic combination of the host and the parasite properties. Clinical disease also changes with age, immunity, and transmission rates.57 Immunity to malaria plays a major role in controlling disease and pathogenesis. The properties of PfEMP1 as an adhesion protein (to avoid parasite destruction within the spleen) cannot be separated from its involvement in immune evasion by clonal antigenic variation that can lead to chronicity of the infection. Even after many exposures, humans are not refractory to malaria parasites but develop clinical immunity that prevents symptomatic disease. This type of immunity limits disease and, although the individual may harbor low numbers of parasites, they do not develop into a symptomatic infection.57 The role of anti-PfEMP1 antibodies in protection from pathogenic infections is highlighted again in placental malaria. Exposure to parasites that sequester in the placenta during pregnancy induces strain-transcending immunity that blocks adhesion of infected erythrocytes to CSA and may protect the mother and fetus from placental malaria in subsequent pregnancies.67
During the development of clinical immunity, particularly during early childhood, strain-specific antibodies to PfEMP1 play a significant role by preventing infection with previously encounter isolates.68,69 This protection can be significant during and after infections with virulent isolates. Bull et al69,70 showed evidence for the existence of rare and prevalent isolates and that parasites causing severe disease tend to express a subset of variant surface antigens (PfEMP1). Moreover, these isolates were expressed preferentially in children who were less able to recognize (by antibody-mediated agglutination) a large number of isolates. Children exposed once or twice to non-cerebral severe malaria acquire immunity that protects them from this form of the disease.71 Hence, exposure to pathogenic forms of P. falciparum can protect against these parasites, leading to selection of possibly less virulent parasites in subsequent infections. Despite its variation, regions of PfEMP1 are restricted by function (e.g., binding to CD36 or CSA), and these regions may be potential vaccine targets.
How adhesion progresses to pathology is a major question that remains unresolved. Several mechanisms that might cause damage to host endothelium and organs have been proposed, including obstruction of blood flow and systemic or local production and deposition of proinflammatory cytokines (see below). Parasite adhesion can also affect the endothelium by inducing or blocking signal transduction mediated by host receptors such as CD36. The recent advances in adhesion research will hopefully provide leads for the mechanism of adhesion related pathogenesis.
The Proinflammatory Immune Response and Pathogenesis
Antibodies and the proinflammatory response protect against the asexual blood stages of some rodent malarias and probably also human malaria. The protection mediated by the proinflammatory response may relate to the cytokines TNF-α and IFN-γ and the release of mediators such as nitric oxide (NO). Clark proposed that mediators, especially NO, are also central to disease.72 It is perfectly logical that these are involved in bone marrow suppression and cerebral malaria, but the data are lacking to prove this role. Furthermore, no model exists for the study of cerebral malaria. One hypothesis suggests that TNF-α induces endothelial cells from brain to express ICAM-1,73 as vessels in the brain have increased expression of ICAM-1 in cerebral malaria.62 Although NO has been proposed as the cause of cerebral malaria, NO is at higher levels systemically in uncomplicated malaria than in cerebral malaria.74 Coma could be caused by local increases in NO in the brain and not increased levels in blood, but this has not been measured. Indeed, total nitrate plus nitrite levels in the CSF of children with cerebral malaria are low, and it has been suggested that this may exacerbate n-methyl-D-aspartate-mediated neurotoxicity due to excitotoxins such as quinolinic acid.75
Data suggesting that a toxin of malarial origin drives the proinflammatory response are interesting,76,77 but the physiologic significance is unproven at present. The evidence that a particular molecule is involved in induction of the proinflammatory response was developed by an assay for the release of TNF-α by macrophages in vitro. Isolation of subcellular components from the parasite and the in vitro assay led to the identification of the GPI anchor from parasite proteins MSP1 and MSP2 as an inducer of proinflammatory cytokines. Antibodies to the GPI anchor were associated with lack of disease in adults,78 but proof that this is causally related is lacking.
Modifications in the immune response to malaria that may not be malaria specific have been identified. Infection with P. falciparum causes apoptosis of mononuclear cells in infected humans.79 In an animal model, infection with a rodent malaria to which they had previously been exposed led to apoptosis of T cells immune to malaria and not those immune to ova, a malaria-unrelated antigen.80 The cells that were eliminated were proinflammatory T cells, producing IFN-γ and IL-2, but not IL-4. It is unclear whether this is specific to malaria or a more general phenomenon.
Study of genetic differences between populations may inform our understanding of the immune system. Fulanis, an ethnic group in Burkina Faso, have higher antibody to many malarial antigens and less disease than two other ethnic groups in the same village who are bitten by equal numbers of infected mosquitoes.81 The molecular basis is unknown, but the innate immune system may be interacting with the adaptive system to increase antibody titers. The importance of these differences also comes from the study of insecticide-impregnated bednets.82 The use of insecticide-treated bednets has reduced the infection in Fulanis, but not among other ethnic groups in the same area. Possibly, the higher antibody titers in Fulanis are sufficient to take advantage of the reduction in infectious bites as a result of insecticide–treated bednets.
Perspectives
The clinical outcome of an infection in a child in Africa depends on multiple factors (Figure 1). In our attempt to understand disease, we often take a reductionist view to study individual components of the parasite and human in an attempt to identify factors that have a major impact on disease outcome. Such factors can be targets for intervention through development of new tools such as vaccines. Success in the development and implementation of these new tools will depend on a connection with scientists from endemic countries of Africa who have a better understanding of local customs and are experienced in communicating with the poorest people in villages of Africa.
II. Clinical and Hematological Features of Malaria
Climent Casals-Pascual, MB, MSc, and David J. Roberts,*
Blood Research Laboratory, National Blood Service - Oxford Centre, John Radcliffe Hospital, Headington, Oxford, OX3 9DU, United Kingdom
Acknowledgments: The work of Dr. Roberts is supported by the National Blood Service and the Howard Hughes Medical Institute. The work of Dr. Casals-Pascual is supported by the University of Oxford. We thank Professors Marsh, Snow, Warrell, and White and Drs. Newton, English, Crawley, Krishna, and colleagues in Oxford for very many helpful discussions.
Malaria is a major public health problem in tropical areas, and it is estimated that malaria is responsible for 1 to 3 million deaths and 300-500 million infections annually. The vast majority of morbidity and mortality from malaria is caused by infection with P. falciparum, although P. vivax, P. ovale, and P. malariae also are responsible for human infections. This review will focus on the principal clinical and hematological features of falciparum malaria and also highlight some of the particular problems faced by those managing patients and potential carriers of malaria in nonendemic areas.
Clinical Features of Malaria
The signs and symptoms of malaria infection in humans are caused by the asexual blood stage of the parasite. The ratio of numbers of deaths to infections from malaria suggests that infection with blood stage parasites may result in a wide range of outcomes and pathologies. Indeed, the spectrum of severity ranges from asymptomatic infection to rapidly progressive, fatal illness. The clinical presentation of malaria infection is particularly influenced by host age, by immune status with respect to malaria and pregnancy, and by the species, genotype, and perhaps the geographical origin of the parasite. The characteristics of falciparum malaria have been most extensively studied in African children, and it is most appropriate to begin by describing the features of malaria in this group.
Malaria in African Children
Symptoms may appear on average 12 days (but occasionally 6 months or more) after inoculation of sporozoites into the bloodstream. An infection may be asymptomatic in those with acquired or innate immunity to malaria; others with no or partial immunity may suffer from a severe acute illness.
Uncomplicated or mild malaria
Prodromal symptoms of malaria include headache (which is often severe and dominates the presenting complaint), myalgia, and coughing that precede the typical sequence of shaking chills, fever, and sweating associated with a paroxysm of fever. The erythrocytic cycle in falciparum malaria is usually synchronized so, in the initial stages of infection, fever occurs on days 1 and 3 (and thus is a tertian fever). In advanced infections the pattern of fever becomes less regular, even continuous. Nausea, vomiting, diarrhea, and abdominal pain may accompany fever. In an uncomplicated infection, signs are few, with the notable absence of lymphadenopathy or rash, but include splenomegaly and mild jaundice. If the course of treatment is incomplete or if the parasites are resistant to the treatment given, then parasites may recrudesce and once more cause a patent infection. Follow-up of treated cases is therefore essential.
Severe malaria
In some children, malaria causes more serious disorders. They may present with prostration or inability to take oral fluids or, in younger children, inability to suckle. As disease progresses they may exhibit a number of syndrome(s) of severe disease including coma, respiratory distress, anemia, and hypoglycemia and may also have a high rate of bacteremia.1 On admission, children usually give a history of only a few days’ illness.
Exclusion of intercurrent diseases may be difficult. The parasitemia is an unreliable guide to the disease severity, and severe disease may occur in the face of undetectable parasitemia where the parasites are sequestered and their growth cycle is tightly synchronized. Thus, clinical assessment must always seek to exclude other illnesses, in particular acute respiratory infection, bacterial meningitis, encephalitis, Reye’s syndrome and septicemia which may mimic respiratory distress, coma, and/or multisystem disease due to malaria.
Cerebral malaria. The cardinal signs of ‘cerebral malaria’ are reduced consciousness and coma. A core of patients have signs consistent with a diffuse metabolic encephalopathy, although patients who present with reduced consciousness may have other distinct pathophysiological disturbances including seizures, metabolic acidosis, and hypoglycemia. The pathology of ‘cerebral malaria’ is linked with the sequestration of parasitized erythrocytes in postcapillary venules of the cerebral circulation, although the functional disturbance(s) that lead to an encephalopathy are poorly understood.2,3
The depth of coma can be measured at the bedside using a simplification of the Glasgow coma scale devised initially in Blantyre, Malawi.4 As unconsciousness deepens, the patient fails to localize pain and may demonstrate abnormal posturing (decorticate rigidity, decerebrate rigidity, and opisthotonus), pupillary changes, absent corneal reflexes, and abnormal respiratory patterns including hypoventilation and periodic breathing.5 Retinal hemorrhages are common.
Seizures are a prominent feature of cerebral malaria. Children may suffer simple febrile seizures, have prolonged or multiple seizures, or have features suggesting a focus of epileptic activity, including localized motor seizures. Status epilepticus and prolonged post-ictal periods are not uncommon. Interestingly, simultaneous EEGs and video recordings of comatose clinical patients have shown that generalized seizures may occur with minimal physical signs, such as twitching of finger(s), conjugate or nystagmoid deviation of the eyes, or hypoventilation with excessive salivation.6 Such patients may recover consciousness after anticonvulsant therapy. Similarly, children presenting with metabolic acidosis and/or hypoglycemia may recover after appropriate treatment.
The progression of signs in cerebral malaria is variable, but sometimes a cephalo-caudal progression of signs is seen including isolated cranial nerve palsies and brain stem signs culminating in respiratory arrest. This sequence of events and direct measurement of cerebrospinal fluid (CSF) pressure in children presenting in a coma has suggested that, unlike nonimmune adults with cerebral malaria, at least some children with cerebral malaria may have intracranial hypertension.7 This has led to re-evaluation of the risk of early lumbar puncture in excluding meningitis in parasitemic, comatose children, and now some clinicians suggest delaying lumbar puncture and commencing empirical antibiotic treatment, to cover bacterial meningitis, with concurrent antimalarial treatment.8
Children who recover from cerebral malaria usually do so within 48 hours, although a significant minority suffers from neurological sequelae. Up to 15% of children may have hemiplegia, ataxia, dysphasia, hearing difficulty, visual problems including cortical blindness, or epilepsy. Over half of these children make a full recovery, but there is a residue of serious disability caused by cerebral malaria, including abnormal cognitive function and behavior.8,9
Anemia. The blood stage of falciparum malaria may cause life-threatening anemia, and hemoglobin of less than 5 g/dL is considered to represent severe disease. Anemia may become worse after treatment begins, particularly if the parasitemia is high. The anemia is typically normocytic and normochromic, with a notable absence of reticulocytes, although microcytosis and hypochromia may be present due to the very high frequency of alpha and beta thalassemia traits and/or iron deficiency in many endemic areas.10 However, difficulties in assessing, not only the absolute, but also the ‘functional’ iron stores complicate the diagnosis and treatment of iron deficiency in the context of acute malaria infection. In such circumstances the only diagnostic test of iron deficiency may be the response to iron supplementation. The indication(s) for and duration of treatment have not, however, been established. Although chronic hemolysis may increase folate requirements, a frank deficiency is uncommon in children presenting with acute malaria, at least in East Africa.
The pathophysiology of severe anemia is a complex but relatively neglected area of study. Certainly, malaria gives ample reasons for both increased destruction and reduced production of red cells. Red blood cells are destroyed as parasites complete their growth cycle, although some parasites may be removed from erythrocytes as immature ring forms by phagocytic cells.11 Infected erythrocytes may also be phagocytosed by macrophages following opsonization by immunoglobulins and/or complement components. Other effector cells and mechanisms are less well defined but may include antibody-dependent cytotoxicity and natural killer (NK) cells.
The survival of uninfected erythrocytes is also reduced. The activity and the number of macrophages are increased in malarial infection. Moreover, the signals for recognition of uninfected erythrocytes for removal by macrophages are enhanced. Uninfected erythrocytes bind increased amounts of immunoglobulin and/or complement as detected in the direct antiglobulin test (DAT or Coomb’s Test).12,13 The specificity of the immunoglobulins on the surface of the red cells has remained controversial. These antibodies do not have a particular specificity but are more likely to represent immune complexes absorbed onto the surface of red blood cells by complement receptors including CR1 (CD35).14
No discussion of the pathology of malarial anemia is complete without consideration of ‘blackwater fever.’ The sudden appearance of hemoglobin in the urine indicating severe intravascular hemolysis leading to hemoglobinemia and hemoglobinuria received particular attention in early studies of anemia in expatriates living in endemic areas. There was an association between blackwater fever and the irregular use of quinine for chemoprophylaxis. This drug can act as a hapten and stimulate production of a drug-dependent complement-fixing antibody. Recent studies of sudden intravascular hemolysis have shown it is rare in Africa, but more common in Southeast Asia and Papua New Guinea, where it is associated with G6PD deficiency and treatment with a variety of drugs including quinine, mefloquine, and artesunate.15
Reticulocytopenia has been observed in numerous clinical studies of malarial anemia. The histopathological study of the bone marrow of children with malarial anemia shows erythroid hyperplasia, with dyserythropoiesis, cytoplasmic and nuclear bridging, and irregular nuclear outline.16 The functional abnormality has not been defined, but an increased proportion of the erythroid progenitors are found in the G2 phase compared with normal controls.
The prime candidates for the host factors mediating dyserythropoiesis have been growth factors and cytokines. Serum erythropoietin (Epo) was appropriately raised in a study of African children with malarial anemia. However, other studies in adults from Thailand and Sudan have concluded that the Epo concentration, although raised, was inappropriate for the degree of anemia.
The concentrations of tumor necrosis factor α (TNF-α) and interferon (IFN)-γ have been correlated with the severity of the disease,17 and high levels of TNF-α have been shown to suppress erythropoiesis. These cytokines may also contribute to reduced production of Epo and to increased erythrophagocytosis.
The possibility has been raised that high levels of the Th2-type cytokine interleukin-10 (IL-10) might prevent the development of severe malarial anemia. Low levels of IL-10 have been described in African children with severe malarial anemia.18 However, the mechanism of protection from anemia by IL-10 is unclear.
The hypothesis that parasite products directly stimulate the production of inflammatory cytokines, including TNF-α, has been widely promoted. The glycosylphosphatidylinositol (GPI) anchor of malarial membrane proteins may cause cellular dysfunction, but a role for this toxin in dyserythropoiesis remains to be established.19 Other toxic products may exist. During its blood stage, the malaria parasite proteolyses host hemoglobin, releasing heme as a by-product. β-Hematin forms as a crystalline cyclic dimer of oxidized heme and is complexed with protein and lipid products as malarial pigment or hemozoin. The function of monocytes and macrophages is severely inhibited after ingestion of hemozoin. Here, the biologically active moieties may be lipoperoxides such as 4-hydroxynonenal (4-HNE) and 15(R, S) hydroxyeicosatetraenoic acid (HETE) produced by oxidation of membrane lipids (reviewed in 20). Their effect on other cellular functions, such as erythropoiesis, has not been established.
Anemia in falciparum malaria is clearly multifactorial and there is a strong argument that erythrocyte destruction and ineffective erythropoiesis play equal parts in the etiology of malarial anemia.
Respiratory distress and metabolic acidosis. Respiratory distress is common in severe disease and has been shown to be an independent predictor of poor outcome.21 It is defined by tachypnea, by deep, gasping breathing, and by use of the secondary muscles of respiration and usually represents metabolic acidosis, although acute respiratory infection must be carefully excluded.22,23 Acidosis is largely due to excessive lactic acid although other anions may contribute to the anion gap. Salicylate toxicity can also cause a metabolic acidosis when inappropriate home treatment with aspirin occurs. Some children presenting with respiratory distress are dehydrated and may be resuscitated with saline. However, the majority of children presenting with respiratory distress are severely anemic, have a metabolic acidosis secondary to reduced oxygen-carrying capacity, and respond to rapid transfusion of fresh blood (reviewed in 24). However, in pregnant women with malaria and nonimmune adults with malaria, transfusion must be given with careful hemodynamic monitoring (see below).
A minority of children with respiratory distress does not respond to appropriate resuscitation. They probably represent a heterogeneous clinical group and may have renal failure, systemic bacterial infection, or a more profound syndrome of systemic disturbance due to malaria parasites.
Hypoglycemia. Hypoglycemia is most common in children and in pregnant women with severe disease.4,25,26 Hypoglycemia may be evident at presentation or may occur during treatment and sometimes in the face of glucose infusions. In children, insulin levels are appropriate and hypoglycemia appears more likely to follow impaired hepatic gluconeogenesis and increased consumption of glucose in peripheral tissues and by parasites.27 In adults, hypoglycemia has been associated with hyperinsulinemia, and it has been postulated that malaria-derived products and/or quinine directly or indirectly stimulate the beta cells of the pancreatic islets.25 Whatever the pathogenesis of the condition, it is clear that all severely ill patients with malaria should be carefully monitored and aggressively treated for hypoglycemia.
Outcome of malaria infection
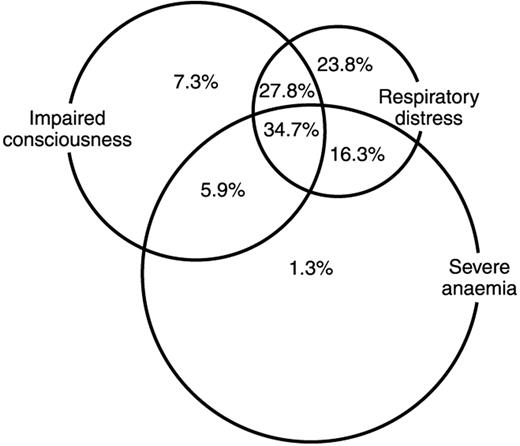
The prognosis of severe falciparum malaria is poor, with a case fatality rate of 15-20% in African children. Mortality is higher when multiple syndromes of severe disease are present (Figure 4 ). A number of clinical and laboratory parameters have been associated with poor outcomes, including deep coma, respiratory distress, hypoglycemia (blood or plasma glucose < 2.2 mM), metabolic acidosis (lactate > 5 mM), raised CSF lactate, renal failure (creatinine > 265 μmol), hyperparasitemia, appearance of pigmented parasitized erythrocytes, and leucocytosis.4,21,22,28 Severe malaria in nonimmune adults also has a high case-specific mortality, and here the rate is probably dependent on the availability of critical care facilities to support multiorgan failure (Table 1 ).
Syndromes of Severe Malaria at Different Ages
One unexplained but consistent feature of the epidemiology of clinical malaria is the age distribution of syndromes of severe disease. Children born in endemic areas are protected from severe malaria in the first 6 months of life by the passive transfer of maternal immunoglobulins and by expression of fetal hemoglobin. Beyond infancy, the presentation of disease changes from predominantly severe anemia in children aged between 1 and 3 in areas of high transmission to predominantly cerebral malaria in older children in areas of lower transmission.29 In this light, the picture of cerebral malaria with multisystem disease in nonimmune adults represents an extreme end of the spectrum in the relationship between age, transmission, and syndrome of severe disease. The reasons for this pattern are unknown but probably include age-specific responses to malaria and also cross-reactive responses activated during infection in older children.
Malaria in Nonimmune Adults
Many travellers (and returning residents with significant levels of clinical immunity) present with what is effectively mild malaria as seen in African children. However, in nonimmunes, severe disease can progress very rapidly and cause life-threatening illness. In this group, multiorgan failure is more prominent than in children with malaria, and some features of the syndromes of malaria differ from those described above for African children (Table 1).30
Cerebral malaria
Coma is a very prominent feature of severe illness in nonimmunes, although neurological sequelae are recorded less frequently. However, epilepsy and psychiatric disturbances have been found at increased frequency in a large series of Vietnam veterans who suffered from cerebral malaria.31
Respiratory failure
Deteriorating respiratory function with widespread pulmonary edema may develop during the disease and carries a very poor prognosis. Some cases of pulmonary edema may be secondary to fluid overload and/or rapid correction of dehydration. However, other patients have adult respiratory distress syndrome (ARDS) with a normal pulmonary wedge pressure, suggesting that the primary abnormality is endothelial damage and excessive permeability. The primary causes of endothelial damage are unknown.32
Renal failure
Renal impairment is common in severe malaria in nonimmune adults, and the microscopic pathology is acute tubular necrosis: glomerulonephritis is rare.33 In some cases acute tubular necrosis may be precipitated by intravascular hemolysis. The pathogenesis in other cases is unclear. Patients may require short-term dialysis for acidosis, fluid overload, hyperkalemia, or rapidly rising creatinine.
Bleeding disorders
Disordered coagulation and clinical evidence of bleeding are not infrequent in adults, and patients may present with bleeding at injection sites, gums, or epistaxis.
Other complications
Hemoglobinuria secondary to intravascular hemolysis and jaundice are more common in adults than in children. As in children, concurrent bacteremia is common.
Malaria in Pregnancy
During pregnancy, women are both more susceptible to malaria infection and also more likely to develop hypoglycemia and pulmonary edema. In pregnant women, malaria infection, often without fever, may nevertheless cause anemia and placental dysfunction. This effect is greatest in primigravidae and has been attributed to the adhesion of parasitized erythrocytes to chrondrotin sulfate A and hyaluronic acid in the placenta. Fetal growth is impaired and babies born to women with placental malaria are on average 100 g lighter than controls born to women without malaria. The subsequent contribution of malaria to infant mortality is substantial.34
Features of P. vivax, P. ovale, and P. malariae Infection
In P. vivax and P. ovale malaria high parasitemias are rare, as invasion of erythrocytes is limited to reticulocytes. The parasites do not appear to sequester in the peripheral circulation or cause organ-specific syndromes. Mortality is limited to occasional deaths from splenic rupture or from intercurrent illnesses in already weakened individuals. Nevertheless, P. vivax malaria has been clearly associated with anemia during pregnancy and with low birth weight in children of affected mothers. In these cases, cytokines or other inflammatory mediators appear to cause placental dysfunction.35P. malariae infection is also rarely fatal but is distinguished by the persistence of blood-stage parasites for up to 40 years. It can, however, cause a progressive and fatal nephrotic syndrome.
White Cell, Platelet, and Coagulation Changes in Acute Malaria Infection
Changes in leucocyte and platelet counts are present in malaria. In addition, there are significant effects on leucocyte function.
White cells
Malaria is accompanied by a modest leucocytosis, although leukopenia may also occur. Occasionally, leukemoid reactions have been observed. Leukocytosis has been associated with severe disease.4 A high neutrophil count may also suggest intercurrent bacterial infection. Monocytosis and increased numbers of circulating lymphocytes are also seen in acute infection, although the significance of these changes is not established.36 However, malarial pigment is often seen in neutrophils and in monocytes and has been associated with severe disease and unfavorable outcome.37
There is a significant dysfunction of myeloid cells in malaria. The adhesive phenotypes expressed by falciparum-infected erythrocytes were previously thought simply to permit sequestration of parasites in the peripheral circulation. Recent work has illuminated how malaria-infected erythrocytes may modulate the function of macrophages and myeloid dendritic cells through the adhesion of malaria-infected cells to CD36 and/or CD51 on host cells (for review, see Urban and Roberts20). Furthermore, the function of monocytes and macrophages may be inhibited by the action of hemozoin from digested hemoglobin.38 These observations suggest that the inhibition of phagocytosis and of other inflammatory responses, mediated by adhesion of infected erythrocytes to myeloid cells and by ingestion of hemozoin, may influence the outcome of infection and facilitate survival of both parasite and host.
Platelets
Thrombocytopenia is almost invariable in malaria and so may be helpful as a sensitive but nonspecific marker of active infection. However, severe thrombocytopenia (< 50 × 109 l1) is rare. Increased removal of platelets may follow absorption of immune complexes, but there is no evidence for platelet-specific alloantibodies. By analogy with erythropoiesis, there may be a defect in thrombopoiesis but this has not been established.
Thrombocytopenia is not associated with disease severity, although, somewhat paradoxically, platelets have been shown to contribute to disease pathology in animal and in human malaria.39 Moreover, in human infections, platelets may form ‘clumps’ with infected erythrocytes.40 Therefore, one explanation of this paradox could be that low levels of platelets may not only be a marker of parasite burden but may also be protective from severe disease.
Coagulation
Abnormalities of laboratory tests of hemostasis, suggesting activation of the coagulation cascades, occur in acute infection, particularly in adults. Patients may present with bleeding at injection sites or mucous membranes. However, histological evidence of intravascular fibrin deposition is notably absent in those dying from severe malaria.2 However, Factor XIII, normally responsible for cross-linking fibrin, is inactivated during malaria infection, and these data may explain low levels of fibrin deposition in the face of increased procoagulant activity.41
Chronic Malaria Infection and the Transmission of Malaria by Transfusion
One of the outstanding problems for those managing the blood supply in nonmalaria endemic countries is the prevention of the transmission of malaria by blood transfusion. In nonimmunes, falciparum malaria would almost invariably develop within 3 months of return from a malaria-endemic area. Furthermore, the infection is symptomatic at very low levels of parasitemia. Nonimmune potential blood donors could be safely excluded from transmitting P. falciparum if they remain asymptomatic 6 months after return from the tropics, although the exact time limits for exclusion of donors who have returned from the tropics are widely debated.
However, malaria is readily transmissible by blood transfusion from semi-immune donors harboring an asymptomatic infection several years after return from a malaria-endemic area.42,43 These donors can in principle be identified by the presence of high titer antibodies against blood-stage antigens.42 Previously, blood donors who were semi-immune to malaria and carrying asymptomatic infection were identified by an ELISA test to detect anti-blood-stage malaria antibodies, using an extract of blood-stage parasites grown in Aotus monkeys. However, the sensitivity of the test, latterly supplied commercially using a stock of antigen derived from the original preparation, declined until it was insufficiently reliable for use in the National Blood Service in the UK.
Commercial tests for the detection of antimalarial antibodies using recombinant malarial antigens are under evaluation. The antigens used in these kits are vaccine candidates and are proteins expressed on the surface of the invasive blood stages of the malaria parasites. However, these same proteins are targets of protective immune responses and are therefore antigenically diverse and elicit highly variable natural antibody responses. It remains to be seen if kits based on antigenically diverse antigens will detect antimalarial antibodies from prospective blood donors with sufficient sensitivity.
Diagnosis
In spite of the distinct clinical syndromes of severe disease, malaria is often misdiagnosed outside endemic areas as the initial signs of disease are nonspecific. Examination of traditional Giemsa-stained thick or thin films will in most cases confirm the diagnosis. The distinctive features of the different types of malaria are described in standard texts.44 Examination of thick films for malaria parasites is a skill honed by regular practice and probably requires specific training for those working in nonendemic areas. Occasionally, severe disease from malaria can be present without detectable parasites in the peripheral circulation, and empirical therapy should be commenced if the patient is seriously ill and there is clinical suspicion of malaria infection.
The traditional methods of diagnosis have been challenged but not superseded by the application of modern methods. Microscopy with fluorescent stains (QBC), polymerase chain reaction assays, and some automated blood cell analyzers offer new approaches, but these are not in wide use.45 More recently, dipstick tests for the detection of parasite antigens HRP2 and pLDH have been developed (Parasight-F [Becton Dickinson, Cockeysville, MD, USA], ICT [ICT Diagnostics, Sydney, Australia], and OptiMAL [Flow Inc., Portland, OR, USA]). These methods may be a useful adjunct in a busy lab, but the tests lose sensitivity at low parasitemias (< 10,000/μL) and have not been licensed for use by the Food and Drug Administration.46
Treatment
Malaria requires urgent effective chemotherapy to prevent progression of disease; such chemotherapy may be the most crucial public health intervention to reduce global mortality from malaria. In severely ill patients, good nursing care is vital. Monitoring and treatment of fits and hypoglycemia are essential and antipyretics should be given.47
Chemotherapy
The drug treatment of malaria must take account of the expected pattern of drug resistance in the area where the infection was contracted, the severity of clinical disease, and the species of parasite. The spread of drug-resistant parasites and the optimal use of affordable, effective drugs are of continual concern, and these topics have been reviewed recently.48,49
Transfusion
Blood transfusion is in principle a straightforward solution to the treatment of severe malarial anemia, although controversy exists over the trigger for transfusion and the rate of administration of blood. The standard regimes of cautious and slow delivery of blood have been challenged by the demonstration that rapid initial flow rates may correct lactic acidosis. However, in nonimmunes and pregnant women, blood transfusion must be accompanied by careful hemodynamic monitoring to avoid precipitating or exacerbating pulmonary edema.
No formal controlled trials for the transfusion of patients with malaria have been performed. Whatever clinical guidelines emerge, blood transfusion in the heartland of malaria endemic areas is beset by many practical and theoretical problems, including the absence of well-characterized donor panels and the residual risk of HIV transmission in the serological window of infectivity without detectable antibodies (estimated at 1 in 2000). At a practical level, positive indirect antiglobulin tests in acute infection may make the exclusion of alloantibodies difficult. Depending on the clinical urgency and transfusion history, the least serologically incompatible blood may have to be given.
Exchange transfusion
One therapeutic option available in North America and in Europe for the urgent treatment of nonimmune patients with severe disease would be exchange blood transfusion. This procedure removes nonsequestered, infected erythrocytes and possibly circulating ‘toxins.’ In the absence of evidence from trials for the use of exchange transfusion in malaria, some have suggested that this treatment could be given for hyperparasitemia (> 20%) in severely ill nonimmune patients.8,50
Conclusion
The clinical features of malaria are diverse in severity and syndrome and include coma, severe anemia, and respiratory distress. In nonimmunes, severe disease may include renal and/or respiratory failure. Emerging themes in clinical management include the management of seizures and of respiratory distress. No reliable nonmicroscopic methods are available for the detection of low parasitemia or of those potential blood donors who may be harboring asymptomatic infection. The optimal chemotherapy regimes for endemic areas and for nonimmune patients are under continuous review as drug-resistant parasites emerge, but there is no specific adjunctive treatment for established disease. However, our understanding of both the parasitological and clinical aspects of pathophysiology is fragmentary, and a detailed description of the pertinent disease processes may lead to novel approaches to treat or to prevent malaria.
III. Genetic Variability in Host Response to Malaria
David J. Weatherall, MD*
Weatherall Institute of Molecular Medicine, University of Oxford, John Radcliffe Hospital, Headington, Oxford, OX3 9DS
Acknowledgments. The author’s work was supported by the Medical Research Council and The Wellcome Trust. We thank Liz Rose for typing this manuscript.
This review is based in part on a recent overview of the genetics of susceptibility to infectious disease: Weatherall DJ, Clegg JB. Genetic variability in response to infection: malaria and after. Genes Immun. In press.
The notion that variations in host response to infection might have a genetic basis is not new.1 At the 8th International Congress of Genetics in Stockhölm in 1948, Neel and Valentine, in order to explain the remarkably high frequencies of thalassemia in some of the immigrant populations in the United States, calculated a mutation rate for the disease of 1:2500. Haldane felt that this was unlikely and that these remarkable gene frequencies must be the result of heterozygote selection. “The corpuscules of anaemic heterozygotes are smaller than normal, and more resistant to hypertonic solutions. It is at least conceivable that they are also more resistant to attacks by the sporozoa which cause malaria, a disease prevalent in Italy, Sicily and Greece, where the gene is frequent.”2 Although it has been suggested that the concept of genetic resistance to infection was already established by the late 1940s,3 a recent reassessment of this question leaves little doubt of the originality and importance of what became known as the malaria hypothesis.4
Of more than 100 species of malarial parasite (Plasmodium), there are only four that have man as their natural vertebrate host; P. falciparum, P. malariae, P. vivax, and P. ovale. Because malaria has in the past and continues to be one of the major killers of mankind, information about individual genetic susceptibility is of broad biological interest. However, with the advent of potential vaccines against malarial infection, and the problems of testing their efficacy in the field, it now becomes of considerable practical importance to be able to determine the frequency and degree of natural protection. Over recent years, a considerable amount of progress has been made toward this end.
Inherited Disorders of Hemoglobin
Collectively, the inherited disorders of hemoglobin are the most common monogenic diseases in man. They comprise the structural hemoglobin variants and the thalassemias, inherited defects in the synthesis of the α or β chains of human adult hemoglobin. Although hundreds of structural hemoglobin variants have been identified,5 only 3—Hb S, Hb C, and Hb E—reach polymorphic frequencies.6,7 The gene for Hb S is distributed widely throughout sub-Saharan Africa, the Middle East, and parts of the Indian subcontinent, where carrier frequencies range from 5-40% or more of the population. Hb C is restricted to parts of West and North Africa. Hb E is found in the eastern half of the Indian subcontinent and throughout Southeast Asia, where, in some areas, carrier rates may exceed 60% of the population. The thalassemias have a high incidence in a broad band extending from the Mediterranean basin and parts of Africa, throughout the Middle East, the Indian subcontinent, Southeast Asia, Melanesia, and into the Pacific Islands.4,7 The carrier frequencies for β thalassemia in these areas range from 1-20%, though rarely greater, while those for the milder forms of α thalassemia are much higher, ranging from 10-20% in parts of sub-Saharan Africa, through 40% or more in some Middle Eastern and Indian populations, to as high as 80% in northern Papua New Guinea and isolated groups in Northeast India.
Analysis of these conditions at the molecular level has provided invaluable information about their heterogeneity and population genetics. Studies of globin gene haplotypes—that is, the patterns of restriction fragment-length polymorphisms in the α or β globin gene clusters associated with these conditions8–,10—has provided important information about their evolution.11 They suggest that the sickle cell mutation may have occurred at least twice, once in Africa and once in either the Middle East or India. Similar data have been interpreted as pointing to multiple origins for the Hb S gene in Africa and the Hb E gene in Asia. This seems unlikely, however, and a more plausible explanation for much of the haplotype diversity observed in association with these variants is that it reflects redistribution on different backgrounds by gene conversion and recombination.11 Over 200 different mutations have been found to underlie β thalassemia; each high-frequency population has its own particular mutations.4,12 The genetics of α thalassemia is more complex, particularly since the α globin genes are duplicated.4 There are two major forms of α thalassemia: αo thalassemia, in which both linked α globin genes are deleted, and α+ thalassemia, in which one of the pairs of linked genes is deleted. The homozygous states for these conditions are represented as --/-- and -α/-α. Both these conditions are extremely heterogeneous at the molecular level, and many different size deletions have been found to cause both α+ and αo thalassemia. As in the case of β thalassemia, the high-frequency regions for a thalassemia have different sets of mutations.4,12
The Structural Hemoglobin Variants and Malaria
The extensive evidence which indicated that the sickle cell trait offers protection against P. falciparum malaria has been reviewed previously.11,13 More recent studies in West Africa suggest that the greatest impact of Hb S seems to be to protect against either death or severe disease—that is, profound anemia or cerebral malaria—while having less effect on infection per se.14 The mechanism for its protective effects has also been reviewed recently and probably reflects both impaired entry into, and growth of parasites in, red cells.1,4 Recent studies in West Africa suggest that the relatively high frequencies of Hb C have also been maintained by resistance to P. falciparum malaria.15 In this case, there is evidence for both heterozygote and homozygote resistance, and the authors suggest that, unlike the sickle cell mutation, this may be an example of transient polymorphism, based largely on the perceived lack of clinical disability or hematological changes of Hb C homozygotes. However, if this were the case, it would be difficult to understand why the frequency of Hb C is not higher in African populations. Since it is not absolutely clear whether homozygotes for this variant are completely unaffected by the condition, further work will be required to substantiate this interesting suggestion. To date, there is no formal evidence for the protective effect of Hb E against malaria, although its population distribution and phenotypic properties of a mild form of β thalassemia suggest that this is very likely to be the case.4
Thalassemia and Malaria
Curiously, it has taken much longer to provide any solid backing for Haldane’s original hypothesis that thalassemia carriers are protected against malaria. This long and frustrating story has been reviewed recently.4 While early studies in Sardinia suggested that there was a relationship between the distribution of thalassemia and malaria in the past, this correlation was not observed in other populations. Furthermore, until the molecular era, it was almost impossible to distinguish between selection, drift, and migration of founder effects as the basis for the population distribution of the thalassemias. However, once it became apparent that each high-frequency area has its own particular thalassemia mutations, it seemed more likely that they had arisen independently and then expanded due to local selective pressures. More recent studies have provided strong evidence that this is the case, at least for the α thalassemias.
The frequency of α+ thalassemia in the Southwest Pacific follows a clinal distribution from northwest to southeast, with the highest frequencies on the north coast of New Guinea and the lowest in New Caledonia.16 These frequencies show a strong correlation with malaria endemicity, as recorded in pre-eradication surveys. On the other hand, there is no geographical correlation of malarial endemicity with other polymorphic markers in this region. The possibility that α thalassemia had been introduced from the mainland populations of Southeast Asia, and that its frequency had been diluted as they moved south across the island populations, was excluded when it was found that the molecular forms of α thalassemia in Melanesia and Papua New Guinea are different to those of the mainland and are set in different α globin gene haplotypes.16 Although these findings provided strong circumstantial evidence that the high frequency of α thalassemia in the Southwest Pacific is the result of malarial selection, it was also found that the disease occurs with gene frequencies varying from 1% to 15% elsewhere in this region, from Fiji in the west to Tahiti and beyond in the east, and in the Micronesian atolls. This was worrying because malaria has never been recorded in these island populations. However, further studies showed that in Polynesia almost 100% of α+ thalassemia can be accounted for by a single mutation that had been previously defined in Vanuatu. Furthermore, this mutation was on the common Vanuatuan α globin gene haplotype. These observations provided strong evidence that the occurrence of the α thalassemia gene in these nonmalarious areas was the result of population migration.17
These population studies suggesting a protective effect of α thalassemia against P. falciparum malaria have been augmented more recently by a prospective case-control study of nearly 250 children with severe malaria admitted to Madang Hospital on the north coast of Papua New Guinea, a region where there is a very high rate of malaria transmission. Compared with normal children, the risk of contracting severe malaria, as defined by the strictest World Health Organization guidelines, was 0.4 for α+ thalassemia homozygotes, and 0.66 for α+ heterozygotes. These studies provide direct evidence for a very strong protective effect of α+ thalassemia against malaria, in both the heterozygous and the homozygous state.18
Molecular analyses of the β globin genes in thalassemic and nonthalassemic individuals in different populations have provided some, albeit indirect, evidence that β thalassemia has also arisen from selection.4 As already mentioned, every population has a different set of β thalassemia mutations. The β globin gene haplotype distribution is divided into 2 regions, the 3′ and 5′ subhaplotypes, which are separated by a recombination hotspot.8,19 However, it turns out that particular β thalassemia mutations are closely associated with specific β globin gene haplotypes, most strongly with the 3′ subhaplotype, which contains the β globin gene, but also with substantial linkage to the 5′ subhaplotype, despite the fact that the haplotypes are separated by the hotspot.9,11 These observations suggest that a recent cause is responsible for the expansion of the β thalassemia mutations; migration has not had sufficient time to disperse them, unlike the normal β globin gene background haplotypes, nor has recombination yet disrupted these linkages.4,11,20 An excellent example of this relationship is seen in Vanuatu, where the sequences of a 3-kilobase (3-kb) region around the β gene from 60 normal chromosomes showed 17 different alleles, involving 19 polymorphic sites. In contrast, 12 β thalassemia chromosomes carrying the common mutation in that region were totally monomorphic over the same region.21 It seems clear, therefore, that the β thalassemia genes throughout malarial regions of the world have been amplified to a high frequency so recently that none of the other forces—migration, recombination, drift, and so on—have had sufficient time or opportunity to bring them into genetic equilibrium with their haplotype backgrounds.
But although these recent studies have provided very strong evidence that the thalassemias have reached their current frequencies by heterozygote selection against malaria, less progress has been made toward an understanding of the likely mechanisms involved. The extensive literature reporting in vitro studies of invasion and development of P. falciparum in thalassemic red cells has been reviewed recently.4 In short, although some abnormalities have been found in the more complex hemoglobinopathies, in the milder forms—that is, those that would have had to come under selection to maintain high gene frequencies—no abnormalities of invasion or growth have been reported. Although several attempts have been made to monitor parasite growth over a number of cycles, these studies have given inconsistent results. More consistent findings have been obtained in analyses of the binding of malaria hyperimmune serum to the surface of P. falciparum–infected thalassemic red cells, in which it has been found consistently that infected cells bind significantly more antibody per unit area than control cells.22 While there is good evidence that the rate of decline of Hb F production in heterozygous β thalassemic infants is retarded,4 and some limited evidence from short-term culture experiments that Hb F retards the growth and development of malarial parasites in vitro,23 this form of protection would be applicable to only β thalassemia.
A completely different mechanism for the possible protection against malaria afforded by α thalassemia was suggested by studies of a large cohort of children, with and without thalassemia, on an island with holoendemic malaria in Vanuatu. It was found that the incidence of uncomplicated malaria and the prevalence of splenomegaly, an index of malaria infection, were significantly higher in very young children with α thalassemia than in normal children. Moreover, the effect was most marked in the youngest children and with the nonlethal parasite, P. vivax.24 It was suggested that the early susceptibility to P. vivax, which may reflect the more rapid turnover of red cells in α thalassemic infants,25 may be acting as a natural vaccine by inducing cross-species protection against P. falciparum.
There are other hints that protection by thalassemia may have at least some degree of immunological involvement. As mentioned earlier, the surface antigen expression in P. falciparum–infected α thalassemic red cells is almost twice that of normal cells, a phenomenon that may lead to better presentation of parasite antigens to the immune system. Furthermore, rosette formation, which has been associated with cerebral malaria, appears to be reduced in thalassemic red cells.26 But perhaps the most important piece of indirect evidence comes from the case control study described earlier, which revealed that α thalassemia not only protects against severe malaria but almost equally against hospitalization from other infectious diseases.18
In short, although there are now extensive data in support of Haldane’s hypothesis that the high frequencies of thalassemia have been maintained by heterozygote, or, as it now appears, for some forms of mild homozygote advantage against malaria, the mechanisms involved are far more complex than those that he proposed. It is now clear that it is not simply the properties of the smaller, under-hemoglobinized red cells that are responsible for the protective effect. Rather, it appears to reflect a much more complex series of events, at least some of which may turn out to have an immunological basis.
Other Red Cell Polymorphisms and Protection Against Malaria
It has been believed for a long time that the high prevalence of individuals in Africa who do not carry the Duffy blood group antigen reflects the protective effect of this genotype against infection with P. vivax. This variant disrupts the Duffy antigen/chemokine receptor (DARC) promoter and alters a GATA-1 binding site, which inhibits DARC expression on red cells and therefore prevents DARC-mediated entry of P. vivax.27,28 This is a milder form of malaria, at least at the present time, and unless it was more severe in the past there may be another explanation for the high prevalence of those who do not carry the Duffy antigen in African populations. A variety of other milder associations between blood group antigens and susceptibility to malaria have been reported.29
There is very strong evidence that glucose-6-phosphate dehydrogenase (G6PD) deficiency, an X-linked disorder that affects millions of individuals in tropical countries, is also protective against P. falciparum malaria. As with thalassemias, several hundred different mutations are responsible for this condition, and their pattern varies between different populations.30 Both hemizygous males and heterozygous females have been found to be protected against severe malaria in both East and West Africa,31 and distribution studies, in Vanuatu, for example,32 have shown a strong correlation with malaria. As is the case for the thalassemias, the mechanisms of protection are still not clear. Work in this field has been reviewed recently; the most likely protective mechanisms appear to be impaired parasite growth or more efficient phagocytosis of parasitized red cells at an early stage of maturation.1
Another remarkable example of a malaria-related balanced polymorphism involves the mutation in band 3 of the red cell membrane that causes the Melanesian form of ovalocytosis, a condition that is extremely common throughout Melanesia and that appears to be lethal in homozygotes.33 This is a particularly interesting polymorphism because heterozygotes appear to be fully susceptible to malarial infection and yet are offered almost complete protection against the development of cerebral malaria.34,35 This observation suggests that the defect in the red cell membrane also alters the interactions between the parasitized cell and the vascular endothelium. The nature of this interaction remains to be characterized.
Human Leukocyte Antigen (HLA) Genes
The human major histocompatibility complex is the most polymorphic region of the human genome that has been analyzed in detail to date. There is increasing evidence that selective pressure by infectious diseases has contributed to this remarkable degree of variability. Structural analyses have suggested that these polymorphisms predominantly affect sites that are critical for peptide epitope binding.1
In the case of malaria, there is strong evidence for associations between both HLA Class I and II alleles and susceptibility to P. falciparum malaria. Thus, at least in parts of Africa, the Class I B53 allele and the Class II DRB1*1352 provide considerable resistance against the severe manifestations of malaria—that is, profound anemia and cerebral malaria.14 Further studies have identified a peptide from the parasite liver-stage antigen-1 (LAS-1) that is an epitope for specific CD8+ cytotoxic T lymphocytes (CTL) that lyse target cells expressing this antigen.36 These observations suggest that parasite-specific CTL are present after natural infection and that this may be at least one mechanism for the HLA-B53 association. So far, data of this type are lacking for other populations.
Other Polymorphisms
After the early successes in identifying polymorphic systems that modify host responses to malaria, a number of other unrelated polygenes that have a similar effect were found. TNF-α, a cytokine that is secreted by white blood cells and has widespread effects on immune activation, has been analyzed in a number of studies. Several different polymorphisms in the promoter regions of the gene for TNF-α have been identified and have been associated with particularly severe forms of malaria.37 One of these, TNF-α-308, may cause increased expression of TNF-α.38 Another influences transcription-factor binding and is associated with an increased risk of cerebral malaria.39
Another particularly fruitful area of recent research in this field is the investigation of polymorphisms of molecules involved in the adhesion of parasitized red cells to the vascular endothelium. The observation that CD36, an important molecule of this kind, is common in Africa led to investigations into its role in malaria. CD36 deficiency has been associated with both susceptibility and resistance to severe forms of malaria.40,41
Recently, a variant of the intracellular adhesion molecule 1 (ICAM1), ICAMKilifi, has been found more commonly in Kenyan children with severe malaria, although it is not associated with more severe disease in West Africans.42,43 Thus, although these relationships need a great deal more work, it seems likely that genetic variation of both effectors of the immune system and adhesion molecules may play an important role in variable response to P. falciparum malaria.
Further evidence that there may be hitherto unidentified immune mechanisms responsible for variations in individual response to P. falciparum malaria has come from extensive population studies in West Africa. Analyses of sympatric ethnic groups with very similar exposures to malaria have shown remarkable differences in infection rates, malaria morbidity, and the prevalence and levels of antibodies to a variety of P. falciparum antigens.44 Extensive investigation of these populations showed no differences in the use of malaria-protective measures or any other sociocultural or environmental factors that might have modified these responses.
Murine Models of Malaria
The growing evidence that at least part of the genetically determined variation in response to severe malaria in humans has an immune basis suggests that more loci that mediate responses of this kind remain to be discovered. So far, total-genome searches for putative loci of this kind have not been reported in the case of human malaria. However, there is growing evidence that analyses of murine malaria using this approach may be of considerable value for further clarifying the genes involved in humans.
Among the murine malaria models, P. chabaudi offers a valuable experimental model of the human form of malaria. Affected animals have analogous blood-stage antigens, the organism invades reticulocytes and mature erythrocytes, infection is associated with suppression of B- and T-cell responses, and the parasite is sequested in the liver and spleen. Recently, there has been considerable success in starting to identify some of the loci involved in variable susceptibility to the consequences of P. chabaudi-induced malaria in different strains of mice. These studies have involved the identification of resistant strains by cross-breeding and then using total genome searches to identify the loci involved in reduced susceptibility.45– 47
Using this approach, a number of chromosomal regions in the mouse genome have been pinpointed as likely to contain a resistance loci for murine malaria. For example, the identification of the Char4 region on mouse chromosome 3, which contains a number of possible candidate genes for malaria resistance, can now be used to search for possible associations of the corresponding human syntenic chromosomal region with susceptibility and/or severity of disease in endemic areas of malaria.47 This new approach of combining murine and human genetics offers an extremely promising way of further exploring genetic variability in response to malarial infections.
There has also been considerable interest in utilizing transgenic mice containing different human globin genes as models for exploring the mechanisms of varying susceptibility to malaria in carriers for particular hemoglobin disorders. So far, although some of these models have reproduced findings which have been observed in human beings, they have failed to point to any of the mechanisms involved in protection against P. falciparum infection by different hemoglobin variants. This field has been reviewed recently.48
Evolutionary Implications
As we have seen, a recurrent pattern is emerging for the distribution and molecular pathology of the human malaria-related polymorphisms. Despite the remarkable degree of protection offered against heterozygotes by the sickle cell gene, and its frequent appearance in Africa, the Middle East, and India, it has never been found farther east than India. Similarly, although Hb E reaches extremely high frequencies throughout Southeast Asia, it is not seen farther west than the eastern parts of the Indian subcontinent. Recent studies of the β globin gene haplotypes associated with the βS Senegal mutation have suggested that it is recent (45-70 generations) in origin.49 Both the α and β thalassemias occur throughout the tropical climes, with the exception of Central and South America, but in each of the high-frequency populations there are different sets of mutations; their relationships to haplotypes of the β globin gene suggest that the expansion of the β thalassemia mutations must have occurred fairly recently. Indeed, all these observations are in keeping with the notion that whatever has been responsible for achieving the extraordinarily high gene frequencies for these conditions has been of fairly recent origin.
Recent studies of haplotype diversity and linkage disequilibrium at the human G6PD locus provide further evidence of the recent origin of alleles that confer malarial resistance.50 In an analysis of 2 G6PD haplotypes, it was found that 2 common variants appeared to have evolved independently between 3000 and 11,000 years ago. These observations on the fairly recent, in evolutionary terms, appearance of genetic polymorphisms that confer resistance to malaria are in keeping with estimates of the spread of P. falciparum derived from studies of polymorphic systems of the parasite. For example, a recent analysis of 25 intron sequences of P. falciparum, involving both general metabolic and housekeeping genes, showed very few nucleotide polymorphisms and suggested that the parasite originated in something like its present form between 9000 and 20,000 years ago.51 These figures are in general agreement with other studies of polymorphic genes of P. falciparum.52 This timescale is in keeping with the notion that it was the development of agriculture somewhere between 5000 and 10,000 years ago that provided the conditions necessary for the effective spread of malaria.
Summary
The picture that is emerging, therefore, is that during the relatively short exposure to severe forms of malaria by human populations, a wide range of different genetic polymorphisms have been utilized to modify individual response to this lethal disease. This has had a profound effect on the genetic constitution of these populations. Not only, as evidenced by the hemoglobin disorders and G6PD, has it left in its wake some extremely common monogenic diseases, but it may well have changed the capacity of these populations to respond to other infections. Clearly, we have identified only the tip of the iceberg of the remarkable genetic variation that exposure to malaria has left in its wake.
The clinical outcome of a malarial infection in an Africa child depends on many parasite, host, geographic and social factors. These converge in the child to result in a range of outcomes, from an asymptomatic infection to severe disease and death.
The clinical outcome of a malarial infection in an Africa child depends on many parasite, host, geographic and social factors. These converge in the child to result in a range of outcomes, from an asymptomatic infection to severe disease and death.
The spectrum of clinical malaria in a malaria endemic area from a series of 1800 children admitted to hospital in Kilifi, Kenya.
The areas of the circles in this Venn diagram are roughly proportional to the number of children in each group. The case fatality rate is given in brackets.
Adapted from Marsh et al. N Engl J Med. 1995;332:1399.21
The spectrum of clinical malaria in a malaria endemic area from a series of 1800 children admitted to hospital in Kilifi, Kenya.
The areas of the circles in this Venn diagram are roughly proportional to the number of children in each group. The case fatality rate is given in brackets.
Adapted from Marsh et al. N Engl J Med. 1995;332:1399.21