Abstract
In Section I, Dr. Stephen O’Brien reviews the latest data on the clinical use of imatinib (STI571, Gleevec, Glivec) in CML. His review focuses on the use of imatinib in newly diagnosed chronic phase patients and summarizes cytogenetic and molecular response data, as well as use of the agent at high doses and in combination with other drugs. A brief summary of the prospective international Phase III studies that are currently ongoing is also provided, and the issues of resistance and definition of suboptimal therapeutic response are also covered. Finally, therapeutic decision-making and treatment strategy are considered.
In Section II, Dr. Ayalew Tefferi considers the latest developments in the biology and therapy of myeloid metaplasia/myelofibrosis. Dr. Tefferi covers what is currently understood of the biology of the disease and reviews established therapies for the condition as well as novel agents that are being used in clinical trials. The development of optimal management strategies for the disease is considered.
In Section III, Dr. Peter Valent reviews the classification of mast cell proliferative disorders and covers the clinical and pathological presentation of this group of neoplasms. He reviews the state-of-the-art regarding the molecular biology of mastocytosis along with diagnostic criteria and novel treatment concepts.
I. Optimizing Therapy in Chronic Myeloid Leukemia
Stephen G. O’Brien, MD, PhD*
Univeristy of Newcastle, Royal Victoria Infirmary, Department of Hematology, Newcastle Upon Tyne NNE1 4LP, UK
The year 2004 has seen a number of significant developments in chronic myeloid leukemia (CML). This section summarizes the discoveries and observations of the last year that are relevant to optimizing therapy for CML patients. A number of groups are attempting to formulate a consensus on current practice and in particular there is a widescale initiative under the auspices of the European Leukemia Network (www.leukemia-net.org) that should lead to widespread optimization of the care of patients with CML.
Should All Newly Diagnosed Patients Be Treated with Imatinib?
This question is frequently asked and opinions still vary. However, there is an inexorable shift toward a trial period of imatinib therapy for most if not all newly diagnosed patients because of the efficacy of imatinib and its minimal morbidity and mortality compared to allografting. As described below, it is now possible to formulate evidence-based criteria that allow the definition of “suboptimal response” to the drug and it is patients with such suboptimal response who might reasonably be offered an allograft. Such an approach can be fairly easily justified for a newly diagnosed 40 year old for whom only a matched unrelated donor is available; however, the decision is much more difficult for a 20 year old with a sibling donor. Although the ongoing response rates with imatinib monotherapy continue to be impressive1 (see below), less than 10% of patients appear to be “disease free” based on polymerase chain reaction (PCR) analysis,2 and there are small numbers of concerning reports of patients, occasionally children,3 developing blast crisis from complete cytogenetic response. Allogeneic transplant should therefore be considered for younger patients especially if a sibling donor is available.
An update (3.5 years’ follow-up on all patients) of the IRIS (0106) study1 will be presented at the American Society of Hematology (ASH) 2004 meeting and the maturing data from this study continue to be the most valuable source of information on the longer-term use of imatinib in newly diagnosed patients. In the original report, after a median follow-up of 19 months, imatinib showed significantly superior tolerability and hematologic and cytogenetic responses and also reduced time to progression when compared with interferon (IFN)-alpha combined with cytosine arabinoside (Ara-C).1 In 553 patients major and complete cytogenetic responses at 18 months were 87.1% and 76.2%, respectively, in the imatinib arm and 34.7% and 14.5% in the IFN and Ara-C arm. The rate of freedom from progression was 96.7% in the imatinib-treated group and significantly less at 91.5% in the combination treatment group. As well as being clinically superior, a comprehensive quality-of-life study has also demonstrated the superiority of imatinib over IFN.4 A review of the 2.5-year follow-up data at the 2003 ASH meeting confirmed that there had been a slight increase in imatinib response rates with further follow-up: 90% major cytogenetic response rate and 82% complete cytogenetic response rate. It is also clear that diagnostic Sokal and/or Hasford scores have an impact on response rates, as shown in Figure 1A (see Color Figures, page 515), and on progression-free survival (Figure 1B; see Color Figures, page 515). After 2.5 years of follow-up, the overall survival in the study was 95% (Figure 2; see Color Figures, page 515). Whether these excellent results will be maintained requires longer follow-up, but so far imatinib resistance does not seem to be a major problem in patients treated in chronic phase. Some groups have reported improved survival in single cohorts of patients,5 but only longer observation will allow a robust analysis of the impact of imatinib on the long-term survival of CML patients.
How Should Patients on Drug Therapy Be Monitored?
There are essentially four laboratory tests that can be used to monitor drug therapy: (1) peripheral blood counts; (2) cytogenetic analysis; (3) real-time quantitative PCR (RQ-PCR) for BCR-ABL mRNA; and (4) assessment of ABL kinase domain mutations.6–,9 The aim of ongoing assessment is to identify those patients who are not responding optimally to standard-dose imatinib monotherapy so that an alternative treatment strategy can be considered. As described below, there is now good evidence to suggest that achievement of complete cytogenetic response should be the major therapeutic target and it has also been demonstrated that the achievement of a 3-log reduction in BCR-ABL/ABL ratio confers superior progression-free survival,2 suggesting that a good “‘molecular” response is also important. ABL kinase domain mutations that cause most cases of imatinib resistance are well-defined and easily detectable, and provide a powerful tool for predicting which patients may develop drug resistance.10 Realistically most hematologists do not have the facilities and resources to perform all of these studies on patients at every visit. So a practical approach to molecular monitoring is necessary.
An excellent recent study by the Australian group has suggested an appropriate approach to “molecular”monitoring of patients on imatinib therapy.11 In 214 imatinib-treated patients with chronic-phase disease Branford et al determined whether there was a difference in the incidence of mutations between the patients with a greater than 2-fold rise in BCR-ABL and those with stable or decreasing levels. Of 56 patients, 34 (61%) with a greater than 2-fold rise had detectable mutations. Only 1 of 158 patients (0.6%) with stable BCR-ABL levels had a detectable mutation. This approach (selecting for mutation analysis only those patients with a 2-fold increase in BCR-ABL levels) therefore detected 34 of 35 (97%) patients who were found to harbor a mutation. This PCR-based selection of patients for mutation analysis is a very useful practical method that is time, labor and cost-effective and paves the way for a more rational, and indeed very powerful, molecular approach to identifying patients in whom imatinib might not be the best treatment in the longer term. Furthermore, recent technological advances in the analysis of heteroduplex DNA by denaturing high-performance liquid chromatography (dHPLC–“Wave” technology) have made the routine detection of mutations more straightforward.12– 14
One could make a case for molecular monitoring alone: no more marrows, forget morphology, dispense with laborious cytogenetic testing. However, until PCR and mutation analysis are more broadly and reliably quality assured, such a move may be a little premature and some would argue strongly that conventional cytogenetics should not be dropped at this stage.15
How Should a Suboptimal Response to Imatinib Be Defined?
This is becoming perhaps the key question in imatinib therapy at present as it is likely to become the major determinant of which patients proceed to allografting, a much riskier therapy than taking imatinib but one with the potential to cure. An analysis of data from the IRIS0106 study presented at the ASH 2003 meeting has proven very useful in attempting to predict, based on early cytogenetic response, which patients are likely to achieve a complete cytogenetic response (CCR) over time. These data are illustrated in Figures 3 and 4 (see Color Figures, page 515–516) and indicate that (1) the achievement of CCR is important in avoiding disease progression; (2) achievement of CCR is beneficial no matter how long it takes to get there; (3) a patient is unlikely to achieve CCR if they have not achieved any significant cytogenetic response by 6 months; and (4) a patient is unlikely to achieve CCR if they have not achieved a major cytogenetic response by 12 months. Other studies have also shown that failure to achieve a significant cytogenetic response on imatinib therapy is an adverse prognostic feature.16 There is also an increasing body of evidence that the development of cytopenias while on imatinib therapy is associated with a significantly reduced chance of obtaining a complete cytogenetic response17 but this is somewhat difficult to quantify.
Based on these data, and incorporating what we know from the RQ-PCR and mutation data described above, one can formulate a set of criteria to define sub-optimal response; these are shown in Table 1 . These criteria would suggest that during the first year from diagnosis, as well as routine blood counts and chemistry analyses at clinic visits, patients should ideally have 20 ml blood taken (in 4 EDTA tubes) for PCR at 3-month intervals and have their bone marrow cytogenetic response analyzed at 6 month intervals.
How Should Patients with Suboptimal Response Be Managed?
Having established that we now have reasonably reliable methods for identifying patients who may not do well on imatinib, the obvious question is how such patients should be treated. The key issue for suboptimal responders is whether, if a suitable donor is available, the patient should be transplanted and this possibility should be actively explored. Some, but certainly not all, patients with suboptimal response have mutations in the ABL kinase domain,7 and various strategies have been suggested to bypass the effects of such mutations.18,19 None are particularly satisfactory. Increasing the dose of imatinib seems, in some patients, to overcome resistance or improve suboptimal response, but in most cases this response is short lived.20 The best hope for the future is to switch to, or add in, one or more of the new drugs described below.
Can Response to Therapy Be Predicted at Diagnosis?
As noted above, the Sokal (and Euro/Hasford) scores seem to still be useful in selecting good and poor prognosis patients (Figure 2; see Color Figures, page 515) although these scores have not been specifically formulated for imatinib-treated patients. Other laboratory approaches such as assessment of derivative 9q deletion status,21 and assessment of telomere shortening22 appear useful. Gene expression profiling as applied to CML is still in its infancy but the technology seems to show promise23–,27 in identifying patients who respond well (or don’t respond well) to imatinib and changes in gene expression profiles during therapy may also be informative.28
Can Imatinib Be Safely Stopped in Patients Who Have Achieved a Good Response?
This is another rather imponderable question to which there is no good answer at present. There are anecdotal reports of patients who have achieved and maintained a complete cytogenetic response and the outcome has varied.29 In contrast there are reports of patients developing blast crisis shortly after stopping imatinib,30 so it remains uncertain how safe it is to stop the drug. Investigators are beginning to consider for the first time whether it might be reasonable with very close monitoring to stop therapy in patients who have maintained, for example, a complete molecular response for 2 years, but at the moment there are very few data to inform this particular debate.
What Are the Questions Being Asked in Current Large-Scale Clinical Trials?
A number of investigator-led Phase III prospective randomized trials are currently underway that seek to address similar questions. Most of these Phase III trials are evaluating the role of higher doses of imatinib31 or imatinib in combination with interferon or Ara-C32 and have both survival and molecular endpoints (www.spirit-cml.org). Higher doses of imatinib (800 mg daily) have been evaluated, particularly by the MD Anderson Cancer Center group, in a nonrandomized context, and very encouraging data have emerged. However, the utility of higher doses of imatinib needs to be evaluated in a Phase III setting, and it will be some years before robust answers emerge from these current studies.
New Therapeutic Developments
There are a number of new therapeutic agents being evaluated in CML (Table 2 ) and during the rush to find the “son of Glivec” there have inevitably been some disappointing leads. However, some agents just coming into clinical trials look promising. AMN107 appears to be a useful compound that in vitro can bypass some of the mechanisms of imatinib resistance: initial trials are currently being planned. BMS354825 is another compound that is causing much interest in the CML community. A combined abl and src tyrosine kinase inhibitor, “825,” has only recently gone into Phase I clinical studies. Promising initial results have been obtained with an encouraging toxicity profile. The agent can bypass in vitro the effects of ABL kinase domain mutations that cause imatinib resistance but, for reasons that are not clear as yet, 825 is not effective against the T315I mutation.33,34
With promising agents in development, it seems likely that in the future drug therapy will incorporate combinations of oral medication, perhaps also with IFN and/or Ara-C. Whether these drugs should be combined sequentially or simultaneously will no doubt be explored, but care must be taken to ensure that we make real progress rather than falling into a confusion of uninformative trials. True global collaboration on such projects will be essential in order to expedite improvements in patient care. The situation is becoming very similar to the development of human immunodeficiency virus (HIV) therapy, where developing multiple drug combinations, assessing viral load (BCR-ABL RQ-PCR) and viral mutation (ABL kinase domain mutation), demonstrating improvements in survival, and coping with strong patient lobbying are all challenges that face researchers. I suspect that we have much to learn from the experience of our colleagues who work in the HIV field, particularly in terms of trial design and analysis.
Imatinib has been a remarkably successful therapy for CML, and many have thought that this achievement could not be repeated. However, with the advent of further promising agents, what had previously been considered unlikely or impossible—the cure of CML by drug therapy alone—may well be within our grasp. In the meantime, the intelligent combination of drug therapy with the selected application of modern allografting technologies offers the best chance of cure for most patients.
II. Myelofibrosis: Update on Pathogenesis and Treatment
Ayalew Tefferi, MD*
Mayo Clinic, 200 First Street, SW, Rochester MN 55905
Myelofibrosis with myeloid metaplasia (MMM) is a clonal stem cell disease that is characterized by intense bone marrow stromal reaction including collagen fibrosis, osteosclerosis, and angiogenesis.1 The clinical phenotype includes progressive anemia, massive splenomegaly, hepatosplenic as well as non-hepatosplenic extramedullary hematopoiesis (EMH), and a leukoerythroblastic blood smear. Average life expectancy is estimated at 5–7 years but may approach 15 years in young patients with good prognostic factors. However, quality of life is often compromised by frequent red blood cell (RBC) transfusions as well as a markedly enlarged spleen that is usually accompanied by both mechanical discomfort and profound constitutional symptoms. Cause of death in MMM includes leukemic transformation that occurs in 10%–20% of patients in the first 10 years. MMM can present de novo (agnogenic myeloid metaplasia) or develop in the setting of either polycythemia vera (post-polycythemic myeloid metaplasia) or essential thrombocythemia (post-thrombocythemic myeloid metaplasia) at a rate of 10%–20% after 15–20 years of follow-up.
Pathogenesis
The primary clonal process in myelofibrosis
It is generally agreed that MMM is a clonal disorder arising from an early stem cell, thereby affecting both the myeloid and lymphoid lineages.2 However, the nature of the disease-causing genetic mutation remains elusive. Recurrent cytogenetic abnormalities that are seen in approximately 50% of chemotherapy-naïve patients with MMM include del(20q11q13), del(13q12q22), trisomy 8, trisomy 9, t(1;7), del(12p11p13), monosomy or long arm deletions involving chromosome 7, and trisomy 1q.3 However, the individual lesions occur in only the minority of patients, and none are specific to MMM. Furthermore, the application of molecular cytogenetic studies by fluorescence in situ hybridization (FISH) did not disclose additional, karyotypically occult, structural lesions.4
Other genetic studies of pathogenesis have included mutation screening for type III receptor tyrosine kinases (c-kit, c-fms, flt-3)5 and genome-wide scanning for loss-of-heterozygosity (LOH).6 The results from such studies have been largely unrevealing despite the identification of frequent (20%–43%) LOH sites involving chromosomes 1p, 1q, 2p, 3q, and 3p.6 On the other hand, gene expression studies have suggested a more consistent alteration of gene function including both down-regulation (e.g., RARβ2 located at 3p24, BCL1 located at 11q13, cdc2 located at 10q21) and up-regulation (e.g., HMGA2 located at 12q13-q15, FKBP51 located at 6p21, GATA2 located at 3q21, JUNB located at 19p13.2) in MMM-derived CD34 cells.6–,9 Other studies have suggested that the JAK/STAT pathway is involved and STAT5 is constitutively activated in both CD34 cells and megakaryocytes from patients with MMM.10 Whether or not the above set of observations will ultimately elucidate the underlying molecular mechanism in MMM remains unknown.
The secondary bone marrow stromal reaction in myelofibrosis
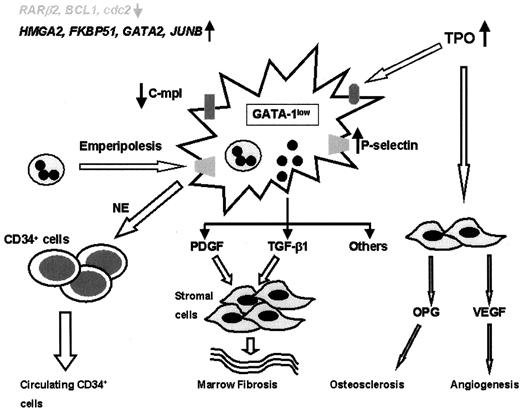
MMM is characterized by polyclonal fibroblast proliferation and alterations in both cellular and extracellular levels of various fibrogenic and angiogenic cytokines. It is currently hypothesized that this bone marrow stromal aberration is reactive and mediated by cytokines derived from clonal megakaryocytes and monocytes (Figure 5 ).1 However, the basis for megakaryocyte/monocyte proliferation and the specific cytokines responsible for the stromal reaction remain unknown.
In mice, either prolonged exposure to excess thrombopoietin (TPO)11 or intrinsic underexpression of GATA-1 (a transcription factor that is important in the maturation of both megakaryocytes and erythrocytes)12 results in megakaryocyte proliferation and an MMM phenotype (megakaryocytic hyperplasia, myelofibrosis, anemia, extramedullary hematopoiesis). In human MMM, however, there is no evidence of either over-expression of TPO13 or underexpression of GATA-1.14 Furthermore, neither TPO nor its receptor (Mpl) is essential for endogenous megakaryocyte growth in human MMM.15 However, MMM megakaryocytes from both the experimental and human disease models display decreased surface expression of Mpl,16 and this might result in decreased clearance of TPO with subsequent contribution to cell proliferation and stimulation of other cytokine production.
In TPO-induced murine MMM, both myelofibrosis (via stem cell–derived transforming growth factor [TGF]-β1) and osteosclerosis (via stroma-derived osteoprotegerin) have been directly linked to specific cytokines.17,18 Similarly TGF-β1, platelet derived growth factor (PDGF), basic fibroblast growth factor (bFGF), vascular endothelial growth factor (VEGF), and tissue inhibitors of matrix metalloproteinases have all been implicated in the bone marrow fibrosis and angiogenesis seen in human MMM.19 Tissue abundance of these cytokines might result from pathologic interaction of megakaryocytes and neutrophils (emperipolesis) that is induced by increased expression of P-selectin by the former.20 Similarly, neutrophil-derived elastase and matrix metalloproteinase might contribute to the abnormal peripheral blood egress of myeloid progenitors in MMM.21
Treatment
Conventional drug therapy in MMM provides palliation in terms of both anemia and splenomegaly. Similarly, both splenectomy and involved-field irradiation engender a defined palliative role in the treatment of MMM. Investigational therapy in MMM includes both experimental drug therapy and allogeneic hematopoietic stem cell transplantation (AHSCT).
Conventional treatment in myelofibrosis
Androgen preparations (e.g., oral fluoxymesterone 10 mg 2 times a day), corticosteroids (e.g., oral prednisone 30 mg/day), and erythropoietin (e.g., 40,000 units subcutaneously once a week) are used as first-line therapy for alleviation of anemia in MMM.1 An approximate 30% response rate with a median remission duration of 1 year is expected from the use of one or more of these treatment modalities. Symptomatic splenomegaly is initially treated with hydroxyurea (e.g., starting dose of 500 mg 2 or 3 times a day) and subsequently with splenectomy.22 Splenectomy is indicated in the presence of drug-refractory splenic pain and/or discomfort, high RBC transfusion requirements, and symptomatic portal hypertension.22 The procedure provides symptomatic relief for the majority of patients and a durable erythroid response in 25% of patients. The experience from our institution suggests that post-splenectomy occurrence of leukemic transformation in MMM represents the natural progression of the disease and is not the result of the procedure itself. Involved-field radiation therapy works best for non-hepatosplenic EMH but has limited value in controlling symptomatic enlargement of the spleen and liver.23
Other cytoreductive drugs that might be considered either prior to splenectomy or in those patients who are poor surgical candidates include intravenous cladribine (5 mg/m2/day in a 2-hour infusion for 5 consecutive days to be repeated for 4–6 monthly cycles),24 oral melphalan (2.5 mg 3 times a week),25 oral busulfan (2–6 mg/day with close monitoring of blood counts),26,27 and intravenous daunorubicin (50 mg/m2 weekly for 4–8 weeks) (unpublished personal experience). In contrast, IFN-α therapy has limited value in alleviating anemia associated with MMM.28
Investigational drug therapy in myelofibrosis
Over the last decade, our group at the Mayo Clinic has explored experimental drug therapy intended to interfere with cytokines believed to mediate the bone marrow fibrosis and angiogenesis in MMM. In this regard, drugs that were shown to be clinically ineffective include imatinib mesylate (inhibits PDGF receptor–associated tyrosine kinase activity),29 IFN-α (non-specific myelosuppressive agent),28 anagrelide (interferes with terminal differentiation of megakaryocytes and platelet production),30 pirfenidone (impairs fibroblast proliferation and collagen synthesis),31 and suramin (inhibits TGF-β binding on fibroblasts).32 In contrast, the drugs that showed promise of activity include thalidomide (has anti-angiogenic activity and also inhibits tumor necrosis factor [TNF]-α production)33,34 and etanercept (a soluble TNF-α receptor that produced a 60% response rate in constitutional symptoms).35
The demonstration of florid bone marrow angiogenesis in MMM36 encouraged the development of several small pilot studies that evaluated the therapeutic value of thalidomide either alone33,34,37–,43 or in combination with other drugs44,45 in MMM. Studies that involved 10 or more patients have demonstrated a response rate of 20%–62% in anemia, 25%–80% in thrombocytopenia, and 7%–30% in splenomegaly.34,37,39,40,42,43 Information from these studies indicates that low-dose thalidomide (50 mg/day) is as effective as higher doses (200 mg/day or more) and that the addition of prednisone to the lower-dose schedule improves drug tolerance and may enhance the erythropoietic activity of the drug.37,44 Long-term analysis of thalidomide-based drug therapy suggests a durable response in a quarter of the patients, including some in whom the drug was discontinued.46 Unusual thalidomide drug effects in MMM, all reversible, include extreme leukocytosis and thrombocytosis. Thalidomide responses in MMM have not been shown to affect either the degree of bone marrow fibrosis or angiogenesis.
Allogeneic stem cell transplantation in myelofibrosis
Treatment with AHSCT, either myeloablative47–,49 or reduced-intensity,50,51 is directed at eradicating the mutant MMM clone. However, the particular treatment modality is associated with significant morbidity and mortality, and is not applicable to the majority of patients with MMM.52 The three largest studies of myeloablative AHSCT (both related and matched unrelated) provide results in a combined total of 147 patients.48,49,53 Engraftment was not a problem, with more than 80% of patients achieving neutrophil engraftment by day 30.53 However, transplant-related morbidity and mortality were significant, resulting in a 5-year survival of only 14% for patients above age 44 years in one study53 and a 2-year overall survival of 41% in another study.49 In the most favorable of the three studies, 20 of the 56 patients had died within 3 years of the transplant, and the reported incidence of chronic graft-versus-host disease (CGVHD) was 59% at a median follow-up period of only 2.8 years.48
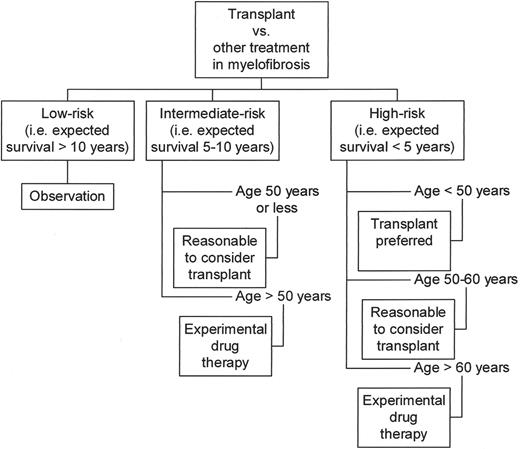
On the positive side of the current experience with myeloablative AHSCT in MMM, younger patients did better in all three studies mentioned above, with projected 5-year survival rates of above 60%. Both clinical and histological remissions were documented in the surviving patients.48,53 Furthermore, transplant outcome was significantly better in good-prognosis patients compared to the outcome in poor-prognosis patients. This latter observation suggests that a window of opportunity may be lost by delaying transplant until the disease progresses. However, it is important to realize that transplant-age patients with good-prognosis MMM can expect a median survival of 15 years.54 This must be weighed against the risk of an upfront mortality rate of at least 20% and a high risk of CGVHD. Taking all these issues into consideration, an evolving guideline for transplant decision making in MMM can be formulated (Figure 6 ).
Reduced-intensity AHSCT has also been attempted in MMM. In a preliminary report from a multicenter study of 20 patients (median age 50 years) with MMM (including 5 who had undergone leukemic transformation), 1-year treatment-related mortality, relapse rate, and survival were 37%, 36%, and 54%, respectively. However, when reduced-intensity AHSCT was performed before transformation, 1-year survival increased to 77%.55 A limited number of other studies, consisting of fewer than 6 study patients each, support the feasibility of reduced-intensity AHSCT in terms of engraftment, attainment of full donor chimerism, and ability to induce both clinical and pathological remissions.50 However, a longer follow-up period as well as a larger sample size are needed to define the durability of full donor chimerism, long-term relapse rate, and quality of life. Such information is critical since the potential beneficiaries of reduced-intensity AHSCT are the subset of patients with good prognosis disease.
Concluding Remarks
Myelofibrosis with myeloid metaplasia is characterized by both shortened survival and poor quality of life, both of which seek therapeutic solutions. In appropriately selected patients, AHSCT promises the possibility of durable disease remission. In this regard, long-term analysis of transplant studies with adequate sample size is key in determining timing of transplant as well as the choice between myeloablative and reduced-intensity conditioning regimens. Experimental drug therapy offers an attractive alternative for patients in need of therapy for palliation of symptoms. The positive experience with the combination of low-dose thalidomide and prednisone therapy in MMM has encouraged our institution to evaluate the value of adding either etanercept (with the rational of optimizing anti-TNF-α activity) or cyclophosphamide (based on in vitro observation of drug synergism) to the combination in ongoing clinical trials. Thalidomide-associated drug side effects might be effectively addressed in the future by the development of thalidomide analogues that are more potent and yet less toxic than the parent drug. One of these agents, CC-5013, has shown significant activity in the treatment of both multiple myeloma and myelodysplastic syndrome (MDS), and it is currently being tested in MMM.
III. Mast Cell Proliferative Disorders: Diagnosis, Classification, and Therapy
Peter Valent, MD,* and Dean D. Metcalfe, MD
Medical University of Vienna, Department of Internal Medicine I, Division of Hematology and Hemostaseology, Währinger Gürtel 18–20, Vienna A-1090, Austria
Mast cell proliferative disorders are characterized by the abnormal growth and accumulation of mast cells (MCs) in one or more organs. Clinical symptoms occur from the overwhelming and sometimes devastating infiltration of tissues by neoplastic cells and/or the release of chemical mediators from MCs. Cutaneous mastocytosis (CM) is considered a benign disease that is confined to the skin and often regresses spontaneously. By contrast, systemic mastocytosis (SM) appears as a clonal persistent disease of myelomastocytic progenitors. The somatic c-kit mutation D816V is detectable within hematopoietic cells in the majority of SM patients. The clinical course in SM is variable. Many patients remain in an indolent stage for decades. In other patients, however, aggressive SM (ASM), an associated clonal hematological non-MC-lineage disease (AHNMD), or mast cell leukemia (MCL), develops. This article provides an overview of mast cell proliferative disorders with special reference to diagnostic criteria and novel treatment concepts.
Introduction
Mast cells are metachromatic cells that, when mature, reside in vascularized tissues in most organs. In contrast to other hematopoietic cells, MCs are extremely long-lived cells with an estimated life span of months to years. MCs are derived from pluripotent and MC-committed hematopoietic progenitors that are detectable in the bone marrow as well as in the peripheral blood.1,2 Circulating MC progenitors have the capability to transmigrate from the blood into tissues where differentiation and maturation occurs.1,2 Cytokines and other factors are involved in the regulation of growth and differentiation of MCs. The most important cytokine is stem cell factor (SCF), which induces development of MCs from uncommitted progenitors.1,2 The effects of SCF on MC-progenitors and mature MCs are mediated through KIT, a tyrosine kinase receptor encoded by the c-kit proto-oncogene.1,2 SCF and KIT are considered essential for the development and differentiation of MCs. Supporting this concept, defects in either the c-kit or scf gene in mice are associated with MC-deficiency. By contrast, “gain-of-function mutations” in c-kit are associated with enhanced survival (and under certain circumstances with autonomous growth) of MCs and their progenitors.3 Such “transforming” mutations, particularly D816V, are often detected in patients with SM.3– 5
The common pathogenetic hallmark of mastocytosis, shared by all disease variants, is the focal accumulation (clustering) of MCs in various tissues and organs.6 Depending on the disease variant, MCs and their progenitors may also show enhanced survival and an increased proliferative capacity. In contrast to CM, monoclonality of SM is well established. Notably, the c-kit point mutation D816V is detectable in the majority (> 80%) of all adult patients with SM.3–,5 The D816V mutation of c-kit is considered to represent an important “hit” contributing to disease development and possibly to the abnormal clustering of neoplastic MCs. However, c-kit D816V alone is unable to act as a fully transforming oncogene.7 Rather, in ASM and MCL, in which the c-kit mutation D816V is also detected and MCs, in addition to clustering, show increased proliferative capacity,8 additional gene defects are likely to be responsible for uncontrolled MC growth.
In this regard it is noteworthy that apart from c-kit D816V, other c-kit mutations (rarely occurring) have been reported in SM as well as chromosomal defects and gene polymorphisms3,9– 11 (Table 3 ). However, the factors determining the course of SM (indolent versus aggressive/leukemic) remain at present unknown. Similarly, little is known about factors contributing to abnormal clustering of MCs in mastocytosis. One hypothesis is that aberrant expression of cell-cell adhesion molecules on MCs, such as CD2, contributes to abnormal clustering. Whether the c-kit mutation D816V or other defects are responsible for abnormal expression of adhesion molecules on MCs in patients with SM remains unknown.
Diagnostic Criteria and WHO Classification
Mastocytosis can be separated into CM and SM. Localized mast cell tumors (mastocytomas and mast cell sarcoma) are very rare. In contrast to CM, SM is a persistent disease with established monoclonality, marked by c-kit D816V, in most cases. Remarkably, in several patients with SM, the c-kit mutation D816V is found not only in MCs, but also in non-MC-lineage hematopoietic cells.12 Based on such data, SM is now considered a myeloproliferative disorder. This concept is consistent with the observation that MCs derive from myelopoietic progenitors and with the relatively high incidence of AHNMDs, including secondary acute myeloid leukemias (AMLs), that occur in these patients. The World Health Organization (WHO) consensus classification of mastocytosis is based on specific criteria that help in the delineation between SM and CM, between SM and myelomastocytic disorders, and between SM and a reactive increase in MCs.13,14 Respective criteria have been termed SM-criteria and are divided into major SM-criteria and minor SM-criteria.13,14 If at least 1 major and 1 minor criterion or at least 3 minor criteria are fulfilled, the diagnosis of SM can be established13,14 (Table 4 ).
The major SM-criterion is the presence of compact dense multifocal MC infiltrates within a bone marrow biopsy section.6,13,14 The most suitable marker for MC detection in such biopsies is tryptase. Compact MC infiltrates can also be detected in extra-medullary organs in SM.6 However, the primary organ to be examined in suspected SM is the bone marrow.
Minor SM criteria relate to the morphology of MCs (spindle shaped, atypical MCs) (Table 5 ), their phenotype (CD2, CD25), elevated serum tryptase (> 20 ng/mL), and demonstration of codon 816 mutations of c-kit.13–,16 The application of such criteria is considered crucial in the diagnostic work-up, since MCs may also increase and even form focal infiltrates in reactive MC hyperplasia or in myelomastocytic disorders. For example, advanced myeloid neoplasms may exhibit an increase in diffusely spread MCs in the bone marrow without histological, cytological or biochemical evidence of SM. If the percentage of MCs in these patients exceeds 10% of all nucleated cells in the bone marrow smear or the differential blood count, it is appropriate to establish the diagnosis of myelomastocytic leukemia.13– 15
Categories of Mastocytosis
Cutaneous mastocytosis
The delineation of subcategories of cutaneous mastocytosis (CM) is based on inspection and biopsy of the dermis. Based on these aspects, three major variants have been defined: maculopapular CM (urticaria pigmentosa), diffuse CM, and solitary mastocytoma of skin. Most patients are children. By definition, the disease is confined to the skin. The serum total tryptase level in CM is usually < 20 ng/mL.13 In many children, skin lesions regress during puberty. Progression of pediatric CM to SM is unusual. In adults, however, the disease persists and often progresses to SM.13 In some patients with CM, skin lesions are extensive and accompanied by symptoms which require management. This includes the use of antihistamines, application of skin care, and sometimes the use of psoralen-ultraviolet A (PUVA). In severe cases, topical glucocorticoids may be required.
Systemic mastocytosis (SM) may show either an indolent or aggressive clinical course. Aggressive systemic mastocytosis (ASM) is characterized by progressive organ infiltration by MCs with resulting impairment of organ-function.8,13 Respective findings are called “C-Findings”.8,13 MC infiltration with associated organomegaly should not automatically be assumed to be the same as organopathy.13 Rather, organomegaly without impairment of organ function is also found in patients with an indolent or an uncertain (smouldering) course, and there regarded as a “B-Finding.” B- and C-Findings are listed in Table 7 .
Indolent systemic mastocytosis
Indolent systemic mastocytosis (ISM) is the most frequently diagnosed variant of SM. ISM is associated with SM criteria,13 presence of urticaria pigmentosa (UP)-like skin lesions, and an indolent clinical course without significant organomegaly or organopathy. The prognosis appears to be good. Symptoms like flushing and abdominal cramping are often reported, and less frequently also episodes of hypotension. The bone marrow is almost invariably affected. In typical ISM, the grade of infiltration is rather low. Typically, MCs in bone marrow smears are spindle-shaped (Table 5 ). Patients with ISM are treated with “mediator-targeting” drugs including antihistamines as well as epinephrine for severe episodes of hypotension, but usually not with cytoreductive agents (Table 8 ). Skin lesions in ISM may require symptomatic therapy. In most cases, transient responses are seen after PUVA. Isolated bone marrow mastocytosis is a rare subentity of ISM characterized by the absence of skin lesions and lack of multiorgan involvement (Table 6 ). Smouldering systemic mastocytosis (SSM) is another subentity of ISM.17 In contrast to typical ISM, B-Findings (≥ 2) are noted in SSM (Table 6 ). These B-Findings reflect a high burden of MCs and extension of the clonal disease to several myeloid lineages.13,17 Clinically, the smouldering state has an uncertain prognosis and a variable clinical course. In some cases, the clinical course is long-lasting and silent. In other patients, an AHNMD or ASM develops after some time.17
SM-AHNMD
In these patients, criteria to diagnose an AHNMD as well as SM-criteria must be met. Patients with SM-AHNMD are categorized according to the AHNMD and the type of SM.13 In most cases, a myeloid neoplasm develops.13,18 In contrast, the occurrence of a lymphoid neoplasm is rare. In all cases, we recommend establishing separate treatment plans for SM and the AHNMD. In practice, SM should be treated as if no AHNMD is present, and AHNMD as if no SM had been diagnosed (Table 8 ).8,13,18
Aggressive systemic mastocytosis
This rare aggressive variant of SM (ASM) is characterized by organopathy caused by pathologic infiltration of various organs by neoplastic MCs and the resulting impairment of organ function.8,13 In contrast to MCL, the bone marrow smear shows less than 20% MCs. In contrast to ISM and smouldering SM, C-Finding(s) indicative of devastating organopathy due to MC infiltration is/are detectable (Table 7 ).8,13 The most commonly affected organs are the liver, bone marrow, and the skeletal system. UP-like skin lesions are usually absent. Patients with ASM are candidates for treatment with cytoreductive drugs (Table 8 ).8 Patients with a relatively slow progression are usually treated with glucocorticoids (prednisone) and IFN-α. We recommend initiating prednisone (50–75 mg p.o. daily) a few days before IFN-α is administered (3 million I.U. s.c. 3 times a week).8 During the first days of treatment, patients should be carefully monitored. After a few weeks, the IFN dosage can usually be escalated to 3–5 million units per day, and prednisone tapered to a low maintainance dose (12.5 mg/day or less) or even discontinued. In patients with severe osteoporosis, IFN-α can be administered without glucocorticoids. ASM patients with rapid disease progression, signs of MCL, or failure to respond to interferon-alpha, are candidates for 2CdA or other chemotherapy (Table 8 ).8,19,20 The use of targeted drugs has also been considered. Imatinib (STI571) has been described to be effective in patients with SM associated with eosinophilia and the FIP1L1/PDGFRA fusion gene.21 Patients with wild-type c-kit or c-kit mutations other than D816V, may also respond to Imatinib.9 However, most patients exhibit the D816V c-kit mutation which appears to confer relative resistance against Imatinib.3–,5,22 In addition, most patients lack the FIP1L1/PDGFRA fusion gene even if eosinophilia is present. Therefore, it can be expected that imatinb will not show significant beneficial effects in the majority of patients with ASM or MCL, at least when used as single agent. However, a number of drugs are currently being examined for their potential to overcome D816V-based resistance of Kit against STI571.
Mast cell leukemia and mast cell sarcoma (MCS)
Mast cell leukemia (MCL) is a rare subentity of SM characterized by circulating MCs and an aggressive clinical course.13,14 Patients typically suffer from rapidly progressive organopathy involving the liver, bone marrow, and other organs. The bone marrow typically shows diffuse and dense infiltration and ≥ 20% MCs on bone marrow smears.13,14 MCs are often immature with a blast-like morphology, and/or have polylobed nuclei (promastocytes). In typical MCL, MCs account for more than 10% of blood leukocytes. In a smaller group of patients, MCs account for less than 10% (aleukemic variant of MCL).13 The c-kit mutation D816V may be detected. The prognosis in MCL is poor. Most patients survive less than 1 year and respond poorly to cytoreductive drugs or chemotherapy. A curative therapy is currently not available.
Mast cell sarcoma (MCS) is an extremely rare form of mastocytosis. To date, we are aware of only 3 well-documented cases.13 The disease is defined by a local destructive sarcoma-like growth of a tumor consisting of highly atypical MCs. At initial diagnosis, no systemic involvement is found. However, the terminal phase may be indistinguishable from ASM or MCL. The prognosis in patients with MCS is grave.
Definition of “suboptimal” response. These criteria are based on imatinib monotherapy, 400 mg daily for 6 to 12 months.
| Abbreviations: RQ-PCR, real-time quantitative polymerase chain reaction | |
| Broadly applicable: | |
| 1. | Failure to achieve a complete hematological response after 3 months |
| 2. | Failure to achieve any significant cytogenetic response after 6 months |
| 3. | Failure to achieve a major cytogenetic response after 1 year |
| Applicable with access to reliable specialized facilities: | |
| 4. | Failure to achieve a 3-log reduction in BCR-ABL/ABL ratios compared to pre-treatment levels |
| 5. | Detection of ABL kinase domain mutations, particularly those in the P loop. Mutation analysis triggered by 2-fold rise in BCR-ABL/ABL ratio by RQ-PCR |
| Abbreviations: RQ-PCR, real-time quantitative polymerase chain reaction | |
| Broadly applicable: | |
| 1. | Failure to achieve a complete hematological response after 3 months |
| 2. | Failure to achieve any significant cytogenetic response after 6 months |
| 3. | Failure to achieve a major cytogenetic response after 1 year |
| Applicable with access to reliable specialized facilities: | |
| 4. | Failure to achieve a 3-log reduction in BCR-ABL/ABL ratios compared to pre-treatment levels |
| 5. | Detection of ABL kinase domain mutations, particularly those in the P loop. Mutation analysis triggered by 2-fold rise in BCR-ABL/ABL ratio by RQ-PCR |
Summary of novel antileukemic agents being evaluated in chronic myeloid leukemia (CML).a
| Agent . | Molecular mechanism(s) . | Ref. . |
|---|---|---|
| * See text for further details. | ||
| ** Data not yet published | ||
| a Modified from Druker BJ. Semin Hematol . 2003 ;40 :50 –58. | ||
| Abbreviations: As2O3, arsenic trioxide; hsp90, heat-shock protein 90; PS341, proteasome inhibitor 341; MAPK, mitogen-activated protein kinase; PI3-kinase, phosphatidylinositol-3-kinase; 17-AAG, allylamino-17-demethoxygeldanamycin; TRAIL, tumor necrosis factor α-related apoptosis-inducing ligand. | ||
| 17-AAG | hsp90 chaperone function inhibition | 35 |
| Adaphostin | Tyrosine kinase inhibition | 36 |
| AG490 | Tyrosine kinase inhibition | 37 |
| AMN107* | Abl kinase inhibitor | ** |
| AP23464 | Abl & src kinase inhibitor | 38 |
| As2O3 | Induction of mitochondrial damage, Bcr-Abl downregulation | 39,40 |
| Bestatin, actinonin | Aminopeptidase inhibition | 41 |
| BMS354825* | Abl & src kinase inhibitor | 34 |
| Decitabine | DNA hypomethylation | 42 |
| Homoharringtonine | Inhibition of protein synthesis | 43 |
| Leptomycin | Nuclear entrapment of Bcr-Abl | 44 |
| PD184352 | MAPK kinase inhibition | 45 |
| Wortmannin, LY294002 | PI3-kinase inhibition | 46,47 |
| PS341 | Proteasome inhibition | 48 |
| SCH66336, L-744,832 | Farnesyl transferase inhibition | 35,49,50 |
| TRAIL | Induction of apoptosis | 51 |
| Trichostatin A | Histone deacetylase inhibition | 42 |
| Agent . | Molecular mechanism(s) . | Ref. . |
|---|---|---|
| * See text for further details. | ||
| ** Data not yet published | ||
| a Modified from Druker BJ. Semin Hematol . 2003 ;40 :50 –58. | ||
| Abbreviations: As2O3, arsenic trioxide; hsp90, heat-shock protein 90; PS341, proteasome inhibitor 341; MAPK, mitogen-activated protein kinase; PI3-kinase, phosphatidylinositol-3-kinase; 17-AAG, allylamino-17-demethoxygeldanamycin; TRAIL, tumor necrosis factor α-related apoptosis-inducing ligand. | ||
| 17-AAG | hsp90 chaperone function inhibition | 35 |
| Adaphostin | Tyrosine kinase inhibition | 36 |
| AG490 | Tyrosine kinase inhibition | 37 |
| AMN107* | Abl kinase inhibitor | ** |
| AP23464 | Abl & src kinase inhibitor | 38 |
| As2O3 | Induction of mitochondrial damage, Bcr-Abl downregulation | 39,40 |
| Bestatin, actinonin | Aminopeptidase inhibition | 41 |
| BMS354825* | Abl & src kinase inhibitor | 34 |
| Decitabine | DNA hypomethylation | 42 |
| Homoharringtonine | Inhibition of protein synthesis | 43 |
| Leptomycin | Nuclear entrapment of Bcr-Abl | 44 |
| PD184352 | MAPK kinase inhibition | 45 |
| Wortmannin, LY294002 | PI3-kinase inhibition | 46,47 |
| PS341 | Proteasome inhibition | 48 |
| SCH66336, L-744,832 | Farnesyl transferase inhibition | 35,49,50 |
| TRAIL | Induction of apoptosis | 51 |
| Trichostatin A | Histone deacetylase inhibition | 42 |
Recognized gene defects, gene polymorphisms, and karyotype abnormalities in patients with (systemic) mastocytosis.
| Finding . | Reported in Patients with: . | Estimated Frequency in Patients with SM . |
|---|---|---|
| Abbreviations: CM, cutaneous mastocytosis; SM, systemic mastocytosis; SM-AHNMD, SM with an associated hematologic clonal non-mast cell lineage disease; HES, hypereosinophilic syndrome; n.k., not known | ||
| Gene defects | ||
| c-kit D816V | all variants of SM (rarely in CM) | > 80% |
| c-kit D816Y | CM, SM, SM-AHNMD | < 5% |
| c-kit D816F | CM, SM | < 5% |
| c-kit D816H | SM-AHNMD | < 5% |
| c-kit D820G | ASM | < 5% |
| c-kit V560G | SM | < 5% |
| c-kit F522C | SM | < 5% |
| c-kit E839K | CM | < 5% |
| c-kit V530I | SM-AML | < 5% |
| c-kit K509I | SM (familial type) | < 5% |
| FIP1L1/PDGFRA | SM-HES, SM with eosinophilia | < 5% |
| Gene polymorphisms | ||
| IL-4Rα Q576R | CM, indolent SM (ISM) | n.k. |
| Karyotype abnormalities | ||
| del 20(q12) | SM, SM-AHNMD | < 5% |
| +9 | SM, SM-AHNMD | < 5% |
| t(8;21) | SM-AML M2 | < 5% |
| Finding . | Reported in Patients with: . | Estimated Frequency in Patients with SM . |
|---|---|---|
| Abbreviations: CM, cutaneous mastocytosis; SM, systemic mastocytosis; SM-AHNMD, SM with an associated hematologic clonal non-mast cell lineage disease; HES, hypereosinophilic syndrome; n.k., not known | ||
| Gene defects | ||
| c-kit D816V | all variants of SM (rarely in CM) | > 80% |
| c-kit D816Y | CM, SM, SM-AHNMD | < 5% |
| c-kit D816F | CM, SM | < 5% |
| c-kit D816H | SM-AHNMD | < 5% |
| c-kit D820G | ASM | < 5% |
| c-kit V560G | SM | < 5% |
| c-kit F522C | SM | < 5% |
| c-kit E839K | CM | < 5% |
| c-kit V530I | SM-AML | < 5% |
| c-kit K509I | SM (familial type) | < 5% |
| FIP1L1/PDGFRA | SM-HES, SM with eosinophilia | < 5% |
| Gene polymorphisms | ||
| IL-4Rα Q576R | CM, indolent SM (ISM) | n.k. |
| Karyotype abnormalities | ||
| del 20(q12) | SM, SM-AHNMD | < 5% |
| +9 | SM, SM-AHNMD | < 5% |
| t(8;21) | SM-AML M2 | < 5% |
Criteria defining systemic mastocytosis (SM).
| * If at least 1 major and 1 one minor, or at least 3 minor criteria, are met, the diagnosis of SM can be established. | ||
| ** Activating mutations at codon 816; in most cases, c-kit D816V. | ||
| Abbreviations: MCs, mast cells; AHNMD, associated hematologic clonal non–mast cell lineage disease. | ||
| Major:* | Multifocal dense infiltrates of MCs in bone marrow or other extracutaneous organ(s) (>15 MCs in aggregate) | |
| Minor:* | a. | MCs in bone marrow or other extracutaneous organ(s) show an abnormal morphology (> 25%) |
| b. | c-kit mutation at codon 816** in extracutaneous organ(s) | |
| c. | MCs in bone marrow express CD2 and/or CD25 | |
| d. | Serum total tryptase > 20 ng/mL (does not count in patients who have AHNMD-type disease) | |
| * If at least 1 major and 1 one minor, or at least 3 minor criteria, are met, the diagnosis of SM can be established. | ||
| ** Activating mutations at codon 816; in most cases, c-kit D816V. | ||
| Abbreviations: MCs, mast cells; AHNMD, associated hematologic clonal non–mast cell lineage disease. | ||
| Major:* | Multifocal dense infiltrates of MCs in bone marrow or other extracutaneous organ(s) (>15 MCs in aggregate) | |
| Minor:* | a. | MCs in bone marrow or other extracutaneous organ(s) show an abnormal morphology (> 25%) |
| b. | c-kit mutation at codon 816** in extracutaneous organ(s) | |
| c. | MCs in bone marrow express CD2 and/or CD25 | |
| d. | Serum total tryptase > 20 ng/mL (does not count in patients who have AHNMD-type disease) | |
Subsets of mast cells defined by morphological criteria.
| Cell Type . | Morphological Properties . |
|---|---|
| Metachromatic blast | Blast-like, few metachromatic granules |
| Promastocyte = Atypical MC type II | Mostly immature mast cells with bi- or multi-lobed nuclei, often hypogranulated |
| Atypical mast cell type I | Mast cells in which 2 of the 3 following morphological aspects are found: a. cytoplasmic extensions (spindle shape) b. hypogranulated cytoplasm c. oval decentralized nucleus |
| Mature mast cell = typical tissue mast cell | Round cell with round central nucleusand well-granulated cytoplasm |
| Cell Type . | Morphological Properties . |
|---|---|
| Metachromatic blast | Blast-like, few metachromatic granules |
| Promastocyte = Atypical MC type II | Mostly immature mast cells with bi- or multi-lobed nuclei, often hypogranulated |
| Atypical mast cell type I | Mast cells in which 2 of the 3 following morphological aspects are found: a. cytoplasmic extensions (spindle shape) b. hypogranulated cytoplasm c. oval decentralized nucleus |
| Mature mast cell = typical tissue mast cell | Round cell with round central nucleusand well-granulated cytoplasm |
WHO classification of mastocytosis.*
| Variant - Term . | Abbreviation . | Subvariants . |
|---|---|---|
| * For details of the WHO classification of mastocytosis see refs #13 and #14. | ||
| ** In a few cases, the FIP1L1/PDGFRA-fusion gene may be detected | ||
| Abbreviations: WHO, World Health Organization; CM, cutaneous mastocytosis; SM, systemic mastocytosis; AML, acute myeloid leukemia; MDS, myelodysplastic syndrome; MPD, myeloproliferative disorder; CMML, chronic myelomonocytic leukemia; MCL, mast cell leukemia; NHL, non-Hodgkin’s lymphoma. | ||
| • Cutaneous Mastocytosis | CM | – Urticaria Pigmentosa (UP) = Maculopapular CM (MPCM) – Diffuse CM (DCM) – Mastocytoma of Skin |
| • Indolent Systemic Mastocytosis | ISM | – Smouldering SM (SSM) – Isolated bone marrow mastocytosis (BMM) |
| • Systemic Mastocytosis with an associated clonal hematologic non-mast cell lineage disease | SM- AHNMD | – SM-AML – SM-MDS – SM-MPD – SM-CMML – SM-NHL – SM-HES |
| • Aggressive Systemic Mastocytosis | ASM | – Lymphadenopathic SM with eosinophilia** |
| • Mast Cell Leukemia | MCL | – Aleukemic MCL |
| • Mast Cell Sarcoma | MCS | |
| • Extracutaneous Mastocytoma | ||
| Variant - Term . | Abbreviation . | Subvariants . |
|---|---|---|
| * For details of the WHO classification of mastocytosis see refs #13 and #14. | ||
| ** In a few cases, the FIP1L1/PDGFRA-fusion gene may be detected | ||
| Abbreviations: WHO, World Health Organization; CM, cutaneous mastocytosis; SM, systemic mastocytosis; AML, acute myeloid leukemia; MDS, myelodysplastic syndrome; MPD, myeloproliferative disorder; CMML, chronic myelomonocytic leukemia; MCL, mast cell leukemia; NHL, non-Hodgkin’s lymphoma. | ||
| • Cutaneous Mastocytosis | CM | – Urticaria Pigmentosa (UP) = Maculopapular CM (MPCM) – Diffuse CM (DCM) – Mastocytoma of Skin |
| • Indolent Systemic Mastocytosis | ISM | – Smouldering SM (SSM) – Isolated bone marrow mastocytosis (BMM) |
| • Systemic Mastocytosis with an associated clonal hematologic non-mast cell lineage disease | SM- AHNMD | – SM-AML – SM-MDS – SM-MPD – SM-CMML – SM-NHL – SM-HES |
| • Aggressive Systemic Mastocytosis | ASM | – Lymphadenopathic SM with eosinophilia** |
| • Mast Cell Leukemia | MCL | – Aleukemic MCL |
| • Mast Cell Sarcoma | MCS | |
| • Extracutaneous Mastocytoma | ||
B- and C-findings.
| B-Findings = | Indication of high burden of MCs, and expansion of the genetic defect into various myeloid lineages without impairment of organ function B = Borderline Benign |
| 1. | Infiltration grade (MCs) in bone marrow > 30% in histology and serum total tryptase level > 200 ng/mL |
| 2. | Hypercellular marrow with loss of fat cells, discrete signs of dysmyelopoiesis without substantial cytopenias or WHO criteria for an MDS or MPD |
| 3. | Organomegaly: palpable hepatomegaly, splenomegaly, or lymphadenopathy (on CT or US: > 2 cm) without impaired organ function |
| C-Findings = | Indication of impaired organ function due to MC infiltration (has to be confirmed by biopsy in most cases) C = Consider Cytoreduction |
| 1. | Cytopenia(s): ANC < 1000/μL or Hb < 10 g/dL or Plt < 100,000/μL |
| 2. | Hepatomegaly with ascites and impaired liver function |
| 3. | Palpable splenomegaly with hypersplenism |
| 4. | Malabsorption with hypoalbuminemia and weight loss |
| 5. | Skeletal lesions: large-sized osteolyses or/and severe osteoporosis causing pathologic fractures |
| 6. | Life-threatening organopathy in other organ systems that is definitively caused by an infiltration of the tissue by neoplastic MCs |
| B-Findings = | Indication of high burden of MCs, and expansion of the genetic defect into various myeloid lineages without impairment of organ function B = Borderline Benign |
| 1. | Infiltration grade (MCs) in bone marrow > 30% in histology and serum total tryptase level > 200 ng/mL |
| 2. | Hypercellular marrow with loss of fat cells, discrete signs of dysmyelopoiesis without substantial cytopenias or WHO criteria for an MDS or MPD |
| 3. | Organomegaly: palpable hepatomegaly, splenomegaly, or lymphadenopathy (on CT or US: > 2 cm) without impaired organ function |
| C-Findings = | Indication of impaired organ function due to MC infiltration (has to be confirmed by biopsy in most cases) C = Consider Cytoreduction |
| 1. | Cytopenia(s): ANC < 1000/μL or Hb < 10 g/dL or Plt < 100,000/μL |
| 2. | Hepatomegaly with ascites and impaired liver function |
| 3. | Palpable splenomegaly with hypersplenism |
| 4. | Malabsorption with hypoalbuminemia and weight loss |
| 5. | Skeletal lesions: large-sized osteolyses or/and severe osteoporosis causing pathologic fractures |
| 6. | Life-threatening organopathy in other organ systems that is definitively caused by an infiltration of the tissue by neoplastic MCs |
Cytoreductive treatment: options for patients with systemic mastocytosis (SM).
| Disease variant . | Treatment options . |
|---|---|
| Abbreviations: IFN, interferon; SM-AHNMD, systemic mastocytosis with an associated hematologic clonal non mast cell lineage disease. | |
| Typical indolent systemic mastocytosis (ISM) | Usually, no cytoreductive treatment is required. |
| Smouldering systemic mastocytosis (SSM) | Watch and wait in most cases. However, in select cases (rapidly progressive B-findings) IFN-α2b ± glucocorticoids or 2CdA can be considered. |
| SM-AHNMD | Treat AHNMD as if no SM is present and also treat SM as if no AHNMD is found. If splenomegaly and hypersplenism prohibit therapy, consider splenectomy. |
| Aggressive systemic mastocytosis (ASM) with slow progression | IFN-α2b ± glucocorticoids. If splenomegaly and hypersplenism prohibit therapy, consider splenectomy. In the absence of c-kit D816V, Imatinib may be considered. |
| ASM - rapid progression** and patients who do not respond to IFN-α2b | Polychemotherapy (± IFN-α2b); consider bone marrow transplantation in select cases. If splenomegaly and hypersplenism prohibit therapy, consider splenectomy. For select cases, cladribine (2CdA) or other cytoreductive drugs can be considered. Consider hydroxyurea as palliative drug. |
| Mast cell leukemia (MCL) | Polychemotherapy (± 2CdA; ± IFN-α2b). Consider bone marrow transplantation. If splenomegaly and hypersplenism prohibit therapy, consider splenectomy. Consider hydroxyurea as palliative drug. |
| Disease variant . | Treatment options . |
|---|---|
| Abbreviations: IFN, interferon; SM-AHNMD, systemic mastocytosis with an associated hematologic clonal non mast cell lineage disease. | |
| Typical indolent systemic mastocytosis (ISM) | Usually, no cytoreductive treatment is required. |
| Smouldering systemic mastocytosis (SSM) | Watch and wait in most cases. However, in select cases (rapidly progressive B-findings) IFN-α2b ± glucocorticoids or 2CdA can be considered. |
| SM-AHNMD | Treat AHNMD as if no SM is present and also treat SM as if no AHNMD is found. If splenomegaly and hypersplenism prohibit therapy, consider splenectomy. |
| Aggressive systemic mastocytosis (ASM) with slow progression | IFN-α2b ± glucocorticoids. If splenomegaly and hypersplenism prohibit therapy, consider splenectomy. In the absence of c-kit D816V, Imatinib may be considered. |
| ASM - rapid progression** and patients who do not respond to IFN-α2b | Polychemotherapy (± IFN-α2b); consider bone marrow transplantation in select cases. If splenomegaly and hypersplenism prohibit therapy, consider splenectomy. For select cases, cladribine (2CdA) or other cytoreductive drugs can be considered. Consider hydroxyurea as palliative drug. |
| Mast cell leukemia (MCL) | Polychemotherapy (± 2CdA; ± IFN-α2b). Consider bone marrow transplantation. If splenomegaly and hypersplenism prohibit therapy, consider splenectomy. Consider hydroxyurea as palliative drug. |
Potential pathogenetic mechanisms in myelofibrosis with myeloid metaplasia (MMM).
In mice, either thrombopoietin (TPO) overexposure or intrinsic GATA-1 underexpression results in megakaryocyte proliferation and the MMM phenotype. Megakaryocytes in such mice as well as in human MMM under-express the TPO receptor (c-mpl). This in turn might lead to decreased TPO clearance and local TPO excess. That might further contribute to megakaryocyte accumulation and stromal cell stimulation of cytokine production. Abnormal release of transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), and neutrophil elastase (NE) might result from pathologic interaction between MMM megakaryocytes and neutrophils. These cytokines, either directly or indirectly through vascular endothelial growth factor (VEGF) and osteoprotegerin (OPG), might contribute to several components of the stromal reaction in MMM.
Potential pathogenetic mechanisms in myelofibrosis with myeloid metaplasia (MMM).
In mice, either thrombopoietin (TPO) overexposure or intrinsic GATA-1 underexpression results in megakaryocyte proliferation and the MMM phenotype. Megakaryocytes in such mice as well as in human MMM under-express the TPO receptor (c-mpl). This in turn might lead to decreased TPO clearance and local TPO excess. That might further contribute to megakaryocyte accumulation and stromal cell stimulation of cytokine production. Abnormal release of transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), and neutrophil elastase (NE) might result from pathologic interaction between MMM megakaryocytes and neutrophils. These cytokines, either directly or indirectly through vascular endothelial growth factor (VEGF) and osteoprotegerin (OPG), might contribute to several components of the stromal reaction in MMM.
Transplant decision making in myelofibrosis with myeloid metaplasia.
Low-risk disease is defined by the absence of hemoglobin < 10 g/dL, circulating blasts, and severe constitutional symptoms. The presence of one or more of these features define intermediate-risk and high-risk disease, respectively.54
Transplant decision making in myelofibrosis with myeloid metaplasia.
Low-risk disease is defined by the absence of hemoglobin < 10 g/dL, circulating blasts, and severe constitutional symptoms. The presence of one or more of these features define intermediate-risk and high-risk disease, respectively.54