Abstract
Our appreciation of the roles that platelets play in vascular biology is constantly expanding. One of the major roles of platelets is in initiating and accelerating immune responses. Platelet transfusion may be associated with adverse inflammatory outcomes manifested as fever, discomfort, tachycardia, and respiratory issues. This may in part be due to immune mediators either expressed by activated platelets or released into the platelet media during platelet storage. This review will highlight some more recent knowledge gained regarding the platelet storage lesion and potential mediators of platelet transfusion reactions.
Introduction
Platelets are the cellular mediators of thrombosis, making normal platelet function essential for survival. Unwanted or excess platelet activation can result in thrombotic complications and too few platelets or platelets that are not responsive to vascular lesions can result in severe bleeding complications. Therefore, maintaining normal platelet numbers and function is essential. There are many disease states and conditions that can result in thrombocytopenia, including chemotherapy, aplastic anemia, HIV/AIDS, immune-mediated thrombocytopenia, sepsis, heparin-induced thrombocytopenia, excessive bleeding, toxic chemicals, viruses, pregnancy, and surgeries such as organ transplantation and cardiopulmonary bypass. Platelet transfusions are typically given when thrombocytopenia is severe or individuals have ongoing serious bleeding, but may also be given on a prophylactic basis to individuals who have a significant bleeding risk based on clinical presentation and platelet counts.1–3 The clinical indications for platelet transfusions, while based on medical evidence, can therefore be somewhat subjective when given on a prophylactic basis. Platelet transfusions are complicated by the unique storage conditions under which platelets must be kept, the need to keep platelets from activating during storage, and the diverse functions of platelets in health and disease.
Platelets must be stored at room temperature. To reduce the risk of bacterial growth and iatrogenic infections after transfusion, platelets have a shelf life of 5 days in the United States. Platelets stored in a manner similar to RBCs at colder temperatures have less contamination risk and improved in vitro function, but are rapidly cleared from the recipient's circulation.4 Chilling platelets results in irreversible clustering of the platelet glycoprotein Ibα (GPIbα) complex, leading to rapid platelet clearance when infused.5 Platelet clearance is primarily mediated by complement receptor 3 (CR3), a receptor expressed on hepatic macrophages that recognizes the clustered GPIbα complex, leading to rapid platelet removal from the circulation.5 Despite this important discovery, its clinical benefits have yet to be realized, but it does offer hope for therapeutic developments.
Although platelet transfusions are fairly common, it is not a benign procedure and is sometimes associated with transfusion reactions. One study reported that 26%-30% of transfusions were complicated by nonhemolytic reactions. Leukocyte or plasma reduction reduced the rate to 19%, still a fairly high number.6 Reactions to platelet transfusions are actually more common than with RBC transfusions and are typified by fever or allergic-type signs, more severe outcomes such as sepsis, and transfusion-related lung injury (TRALI).7 One cause of platelet transfusion reaction is the recipient immune system's response to “foreign” platelets. Platelet transfusions are blood group matched to reduce this risk, but this complication can still occur, especially in people who have had many blood and platelet transfusions.
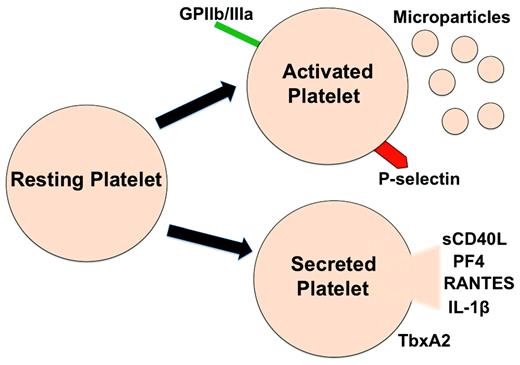
The other major cause of transfusion reactions is a response to what are often called “platelet storage lesions” (PSLs), which in many ways can be difficult to separate from TRALI. PSL refers to the changes in platelets that occur during their collection, processing, and storage before transfusion. During storage, there are changes both in the platelets themselves, which can result in altered in vivo function, and in the production and release of pro-inflammatory mediators that can also have untoward effects when platelets are infused (Figure 1).
Platelets become activated during storage, resulting in the expression of activated membrane receptors, microparticle production, and secretion of inflammatory mediators.
Platelets become activated during storage, resulting in the expression of activated membrane receptors, microparticle production, and secretion of inflammatory mediators.
Many platelet membrane–expressed proteins, granule constituents, and synthesized and secreted molecules have primary roles as immune mediators.8–11 Platelet-derived inflammatory mediators include surface adhesion molecules (eg, P-selectin and integrins), secreted small molecules (eg, ATP/ADP, serotonin, and glutamate), and chemokines and cytokines. A short list of platelet-derived inflammatory mediators found in the circulation includes platelet factor 4 (PF4/CXCL4), pro-platelet basic protein (ppbp and its breakdown product CXCL7/NAP-2), RANTES/CCL5, IL-1α/β, and sCD40L, all of which can recruit and activate leukocytes or endothelial cells both at the site of platelet deposition and at a distant site in the body.12 Other secreted mediators such as serotonin, ADP, and prostaglandins have short half-lives and tend to exert pro-inflammatory effects only in their local environment. Activated platelets also express exteriorized or activated adhesion molecules such as P-selectin and activated GPIIb/IIIa, which may have contact-dependent mechanisms for platelet and leukocyte interactions. Interactions between platelets and leukocytes can lead to leukocyte activation and exacerbated immune responses. For example, P-selectin engagement of P-selectin glycoprotein ligand (PSGL-1) facilitates rolling and integrin activation in neutrophils.13,14
Platelets are activated by multiple mechanisms in vivo, the most obvious being after endothelial layer denudation. However, platelets can also be activated by an intact and inflamed endothelium and deposit at the site of vascular inflammation without forming a complete obstructive thrombus.15 These events of platelet deposition and activation perpetuate the cycle of inflammatory interactions among platelets, endothelial cells, and leukocytes. This raises the possibility that not only can platelets activated during storage stimulate maladaptive immune responses, but interactions between infused platelets and an inflamed vascular bed may also lead to platelet activation and an exacerbation of the vascular injury. An appreciation for the concept that platelets are very pro-inflammatory and are an important part of immune responses leads to a better understanding of the idea that platelet transfusions are not a benign process. Rather, platelet activation during collection and storage can lead to changes in the platelet phenotype that may trigger inflammation upon infusion, and platelet-derived immune mediators secreted during storage may also drive recipient transfusion reactions.
Platelet changes associated with the PSL
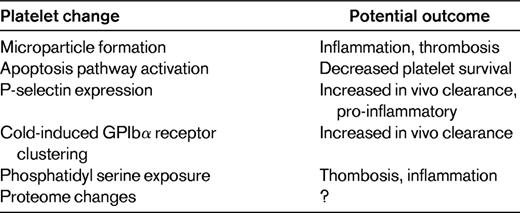
As indicated above, PSL refers to the changes in platelets that may occur during platelet collection, processing, and storage before transfusion. During storage, there are many changes in the platelets themselves that may contribute to transfusion-related inflammation (Table 1). Platelet storage can be associated with changes in platelet aggregation, granule release, cytoskeletal changes, and increased exposure to phosphatidyl serine.16 There have been recent studies using platelet proteomics as a means to identify platelet proteins that are changed during storage. This has been proposed as a starting point for subsequent mechanistic and therapeutic study. For example, separate studies by the Devine laboratory and the Phipps laboratory have each catalogued the changes in the platelet proteome during storage.17,18 A striking finding is that the expression of a large number of proteins is increased during storage. This reflects the fact that despite their anucleate status, platelets have the ability to synthesize protein de novo.19 Platelets not only have stored mRNA, but also pre-mRNA that they can splice and translate into protein. The quantitative changes in protein expression observed during storage likely represent new protein synthesis, but detailed studies have yet to be performed to clearly demonstrate this. These studies also raise the question of whether protein synthesis inhibitors may be beneficial in platelet storage.
Like pre-mRNA splicing, apoptosis has traditionally been thought of as a nucleated cell–restricted event. However, apoptosis also appears to have an important role in regulating platelet survival.20 Platelets have an intrinsic program for apoptosis that helps to control their circulation lifespan. A study by Mason et al demonstrated that mice lacking the pro-survival Bcl-x(L) gene or mice treated with Bcl-x(L) inhibitors had reduced platelet half-life and developed thrombocytopenia. Moreover, a lack of pro-apoptotic Bak corrected these defects, leading to their conclusion that platelets are genetically programmed to die by apoptosis.20 Because the lifespan of platelets in the circulation is on average only 6-10 days, there may be a significant loss of viable platelets during 5 days of storage. Before this elegant in vivo demonstration of platelet apoptosis, it had already been reported that platelet apoptosis occurs during storage. In 2000, Li et al showed that platelets contain apoptotic machinery and that activation of caspase-3 occurs during storage in a manner independent of platelet activation.21 Since that study, there have been other studies verifying apoptotic platelets in storage and attempting to prolong the life of stored platelets by inhibiting apoptosis pathways. In addition, Van der Wal et al have recently reported that cold-induced platelet GPIbα clustering resulted in a 14-3-3 protein–induced activation of apoptosis signaling.22
There are many other changes in platelets during storage that are more typical of an activated platelet phenotype. More than 15 years ago, it was noted that the number of CD62-, LAMP-1-, and LAMP-2–positive platelets all increased during platelet storage.23 Other typical markers of platelet activation, such as the active conformation of GPIIb/IIIa and the presence of platelet microparticles (PMPs) in storage, have also been described for more than 20 years. The increase in stored platelet PMPs occurs in the absence of agonist in a manner described to be the result of GPIIb/IIIa signaling to the actin cytoskeleton.24 PMPs may be beneficial in some cases because of their procoagulant role when platelet transfusions are given for active bleeding. PMPs may contribute to low level generation of thrombin in vivo,25 helping to support and sustain thrombosis.26 The role that PMPs may have in transfusion reactions has not been fully investigated. PMPs also contribute significantly to inflammation and may have a deleterious role in platelet transfusions given on a prophylactic basis. Much of platelet-derived IL-1α and IL-1β is on the surface of PMPs and PMP IL-1α may have an intriguing role in amplifying arthritis,27 demonstrating the importance of PMPs in common inflammatory disease processes. Sprague et al have also demonstrated that PMPs may be peripheral inflammatory messengers. PMPs expressing CD154 (CD40L) can deliver signals to B cells and stimulate antigen-specific IgG production and germinal center formation, demonstrating a broad potential role for PMPs in immune responses.28
Changes in platelets themselves may occur during storage, many of which are pro-inflammatory and potentially contribute to reduced platelet function and transfusion reactions in vivo.
Platelet storage releasate
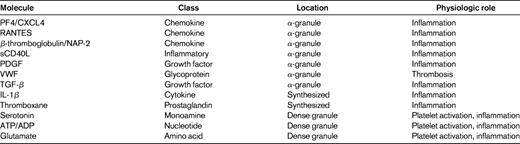
Platelets contain a large number and variety of inflammatory molecules, many in very high concentrations. Platelet-derived inflammatory mediators can either be secreted from stored vesicles or synthesized de novo by platelet-expressed enzymes (Table 2). A major class of platelet-synthesized mediators are prostaglandins and thromboxane, which have broad roles in thrombosis and inflammation. Platelets also have 3 types of granules: alpha, dense, and lysosomal granules. Alpha granules are primarily composed of proteins (eg, PF4, P-selectin, PDGF, and VWF), dense granules contain small molecules (eg, serotonin, glutamate, ADP, and ATP), and lysosomal granules contain degradative enzymes. Many of these stored mediators have important pro-inflammatory functions, and platelets have a major role in initiating and accelerating the immune response. This can include responses to many disease processes that have an important vascular inflammatory component, such as atherosclerosis, transplant rejection, arthritis, and malaria.29–34 Platelet granule release and product synthesis is initiated by platelet activation and, as noted above, there is significant evidence of platelet activation during storage.
Mediators of thrombosis and vascular inflammation increase in the supernatant of stored platelets. Platelet-derived factors such as β-thromboglobulin and PF4 accumulate in the storage medium over time, reflecting platelet activation and granule release.35,36 Recent work by Vlaar et al explored the role of stored platelet supernatants in transfusion-related lung injury using a rat model of syngeneic transfusion.37 In particular, they were interested in modeling whether lung injury was a result of a suggested “2-hit” hypothesis for lung injury: priming and then activation of neutrophils. In that study, they were able to demonstrate that aged platelets caused mild lung inflammation on their own, and this was made significantly worse by lipopolysaccharide pretreatment. Furthermore, supernatants of aged platelets resulted in lung injury in the lipopolysaccharide-pretreated rats. These investigators were able to show that the concentration of bioactive lipids such as lysophosphatidylcholine increased during blood storage, and that lysophosphatidylcholine in the platelet concentrate supernatant had neutrophil-priming capacity in vitro (and perhaps in vivo). The importance of this recent study is in its use of a novel animal model and the in vivo demonstration of the role that platelet-derived products can have in platelet transfusion–associated TRALI. In general, there have been few animal models used to explore transfusion-related injury, and the work of Vlaar et al has helped to establish a robust model. The use of genetically modified mice may also help in demonstrating mechanisms of PSL-associated inflammation in vivo.
A molecule that has received significant attention in the platelet transfusion literature is CD40L (CD154). T- and B-cell–derived CD40L is classically thought to bind CD40 on APCs and regulate adaptive immune cell function. CD40L is also expressed on activated platelets, and platelet-derived soluble CD40L (sCD40L) can be found in the circulation after platelet activation. The CD40L receptor CD40 is expressed by many non-APCs such as endothelial cells, and CD40-CD40L engagement can lead to endothelial chemokine and cytokine production and to adhesion molecule expression.38,39 CD40L-CD40 on neutrophils and macrophages also leads to their immune stimulation. Platelets also express CD40, and engagement of platelet CD40 may have an activating effect.40 sC40L is found at significantly higher concentrations in stored platelet units compared with fresh platelets, and has also been found to be higher in stored platelet concentrates implicated in TRALI reactions.41 Kaufaman et al confirmed that platelet concentrates prepared for transfusion can have high levels of membrane-bound CD40L and sCD40L, with maximum levels seen 72 hours after platelet collection, which highlights the potential importance of CD40L in transfusion reactions.42
A fairly complete study of stored platelet concentrate inflammatory mediators came in 2006 from Garraud et al, who prepared pooled platelet concentrates and measured the immune mediators IL-6, IL-8, PDGF-AA, sCD40L, RANTES, and TGF-beta on days 0, 1, 2, 3, and 5 of storage.43 That study found that the levels of RANTES, IL-8, and IL-6 were stable, but that the levels of PDGF-AA and sCD40L increased with time. Furthermore, the investigators determined that the concentration of sCD40L in the stored platelet concentrate may be sufficient to induce B-cell effects upon transfusion. This same group has also found the level of soluble P-selection increased significantly by day 3 and platelet membrane expression of P-selectin increased significantly after day 2 of platelet storage.44 These P-selectin changes were of a degree that may also affect leukocyte function in vivo and contribute to platelet transfusion reactions. Other studies have noted that platelet-derived cytokines such as RANTES, PF4, TGF-β, and IL-8 increase during storage.45
The above and many other studies indicate that physiologically relevant concentrations of platelet-derived cytokines and chemokines are found in stored platelets, and that these platelet-derived immune mediators may contribute greatly to adverse outcomes associated with platelet transfusions.
Summary
The physiologic role for platelets extends beyond their hemostatic function to an immune cell function. This important role for platelets is likely to exert an impact on platelet transfusion recipients and may be worsened during platelet storage. Careful consideration of platelets as immune cells may lead to novel strategies to reduce platelet transfusion reactions.
Disclosures
Conflict-of-interest disclosure: The author declares no competing financial interests. Off-label drug use: None disclosed.
Correspondence
Craig N. Morrell, Aab Cardiovascular Research Institute, Department of Medicine, University of Rochester School of Medicine, Box CVRI, 601 Elmwood Ave, Rochester, NY 14642; Phone: (585) 276-9921; Fax: 585-276-9829; e-mail: Craig_Morrell@URMC.Rochester.edu.