Abstract
Molecular monitoring of BCR-ABL1 transcripts for patients with chronic myeloid leukemia (CML) is now used to assess response to tyrosine kinase inhibitors (TKIs), including treatment failure that mandates a change of therapy. Therefore, many centers have adopted the molecular technique for measuring BCR-ABL1 and rely on conversion of values to the international reporting scale for appropriate clinical interpretation. However, the technique has a degree of inherent variability despite standardized procedures, which means care should be taken by the clinician when assessing response based on BCR-ABL1 cutoff limits. The last few years have witnessed the emergence of a new molecular response target, which is the achievement and maintenance of a deep molecular response. The ability to achieve treatment-free remission for some patients has shifted the relevant boundary for molecular response. However, the definitive safe BCR-ABL1 transcript level and length of the maintenance phase after which treatment cessation can be attempted has not yet been determined. For patients with TKI resistance, BCR-ABL1 kinase domain mutation analysis remains an essential assessment to guide therapy. Furthermore, low-level mutation detection is clinically relevant for response prediction to subsequent TKI therapy for some patients. Multiple low-level mutations may be a biomarker of a clonally diverse disease with the propensity for resistance evolution. Overall, molecular monitoring, including low-level monitoring is a fundamental component of management for patients with CML.
Learning Objectives
Understand that there will always be reduced confidence in the molecular response category around critical BCR-ABL1 cutoff values
Achieving a sustained deep molecular response is associated with safe and successful treatment-free remission in some patients but may take many years to achieve
Appreciate the clinically relevant role for low-level BCR-ABL1 mutation detection
Introduction
For newly diagnosed patients with chronic myeloid leukemia (CML), a major objective of tyrosine kinase inhibitor (TKI) therapy is achieving an optimal response in a timely manner. This offers the prospect of a good outcome and protection from progression to accelerated phase and blast crisis.1,2 In 2013, the European LeukemiaNet (ELN) and the National Comprehensive Cancer Network (NCCN) for the first time incorporated early molecular response levels into their recommendations and guidelines for treatment decisions. The prior focus was on cytogenetic response, but this has shifted in favor of using BCR-ABL1 transcript levels. Values achieved over the first 12 months of TKI therapy are strong predictors of outcome and are of particular significance for treatment intervention decisions. This is based on consistent data published over more than a decade for various patient cohorts treated with first- or second-line TKIs. More recent studies of imatinib-treated patients represent mature, long-term follow-up and have been highly influential for moving molecular monitoring into the mainstream.3-5
Because molecular monitoring has now taken on greater significance, many laboratories with varying expertise have adopted molecular techniques over the last few years. The requirement for peripheral blood rather than bone marrow as the tissue of choice for molecular monitoring has no doubt relieved a burden for patients. For laboratories, the availability of molecular kits standardized to the international reporting scale and recommendations published by the EUTOS group (European Treatment and Outcome Study for CML) has eased the burden of method development somewhat.6,7 Nevertheless, despite years working toward harmonized results, there is still work to be done as the clinical applications of the molecular assays expand. The new frontier of response is the achievement of a deep molecular response, with the promise of a trial of stopping TKI. These responses are substantially reduced below the currently defined optimal response, which is a major molecular response (MMR, BCR-ABL1 ≤0.10% on the international reporting scale [IS]), and can take many years to achieve in some patients.8,9 Clinical trial results expected over the next few years from large TKI cessation studies will inform and likely define the sensitivity boundary required of the assay for measuring BCR-ABL1 transcripts.
Clinicians may have adopted the recommendations and guidelines for treatment intervention using precisely defined cut points, but in reality the response is a continuum and, in general, patients fare better the more the BCR-ABL1 transcript level declines.4 Furthermore, the quantitative polymerase chain reaction (PCR) assay is an imprecise measurement with considerable variation around critical decision cutoff values. Education is required regarding this feature for the appropriate evaluation of molecular response and to aid clinical decisions.
How reliable is the molecular assay for measuring BCR-ABL1 transcripts?
The IS, harmonized procedures, standardized definitions for response, World Health Organization–certified reference reagents and secondary reference panels, and the recent availability of a certified plasmid calibrator have improved the comparability of BCR-ABL1 transcript values when generated by different laboratories.6,7,10-13 However, one cannot overlook the inherent variability of each molecular assay and its impact on the comparability of results and the interpretation of response for individual patients. The assay generates values over a 4- to 5-log range and the coefficient of variation diverges across the range, with lower values showing greater variation.
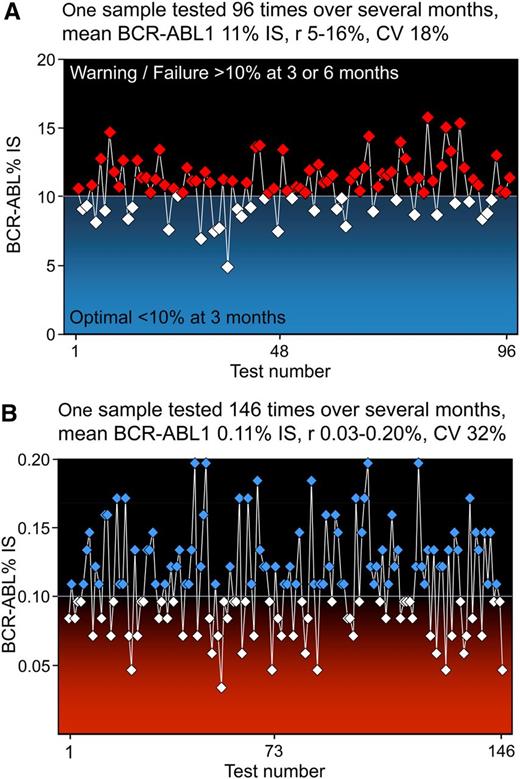
To demonstrate the inherent variability of a molecular assay, Figure 1 shows the range of BCR-ABL1 values generated by repeated measure over several months in our laboratory for 2 samples with high and low BCR-ABL1. The mean BCR-ABL1 value of the high level is 11% IS, close to the critical threshold of 10% IS that defines treatment failure.1,2 The coefficient of variation (CV) is 18%, and 31% of the values are within the optimal range for a measurement at 3 months of TKI, whereas the remainder are in the failure range for a measurement at 3 or 6 months of TKI. The low BCR-ABL1 level at a mean of 0.11% IS (close to MMR) displays greater variability with a CV of 32%. The purpose of the figure is to demonstrate that there will always be reduced confidence in the response category around critical BCR-ABL1 cutoff values. The ELN recognized that the studies that supported the prognostic value of the 10% cutoff were performed in a few reference laboratories, and the performance of these experienced centers may not represent the general standard of laboratories worldwide that have adopted molecular monitoring more recently.1 Hence, variability around the cutoff values may be greater in some centers. A robust quality control process to assess the degree of variability and to recognize failed technical procedures is an essential component of molecular techniques,14 just as it is for any clinical pathology test. This issue has taken on greater significance since the incorporation of molecular results for clinical decisions.1,2
The quantitative PCR assay generates variable BCR-ABL1 values. Quality control RNA samples with high and low BCR-ABL1 are included in every batch of patient samples in our laboratory to determine the run acceptability. The graphs demonstrate the BCR-ABL1 values recovered for the high (A) and low (B) controls analyzed over several months by different operators. These controls were manufactured in-house from BCR-ABL1–positive and –negative cell lines, and cells were stored frozen in RNA stabilization solution in large batches. Each control undergoes reverse transcription followed by quantitative polymerase chain reaction (PCR) in every batch of patient samples. Each value on the graphs represents the BCR-ABL1 result recovered from separate reverse transcription and quantitative PCRs. Reverse transcription is the process that introduces the greatest degree of variability. To appropriately evaluate the variability of an assay and to mimic the variability observed for patient BCR-ABL1 values, it is essential to include this process in the evaluation.
The quantitative PCR assay generates variable BCR-ABL1 values. Quality control RNA samples with high and low BCR-ABL1 are included in every batch of patient samples in our laboratory to determine the run acceptability. The graphs demonstrate the BCR-ABL1 values recovered for the high (A) and low (B) controls analyzed over several months by different operators. These controls were manufactured in-house from BCR-ABL1–positive and –negative cell lines, and cells were stored frozen in RNA stabilization solution in large batches. Each control undergoes reverse transcription followed by quantitative polymerase chain reaction (PCR) in every batch of patient samples. Each value on the graphs represents the BCR-ABL1 result recovered from separate reverse transcription and quantitative PCRs. Reverse transcription is the process that introduces the greatest degree of variability. To appropriately evaluate the variability of an assay and to mimic the variability observed for patient BCR-ABL1 values, it is essential to include this process in the evaluation.
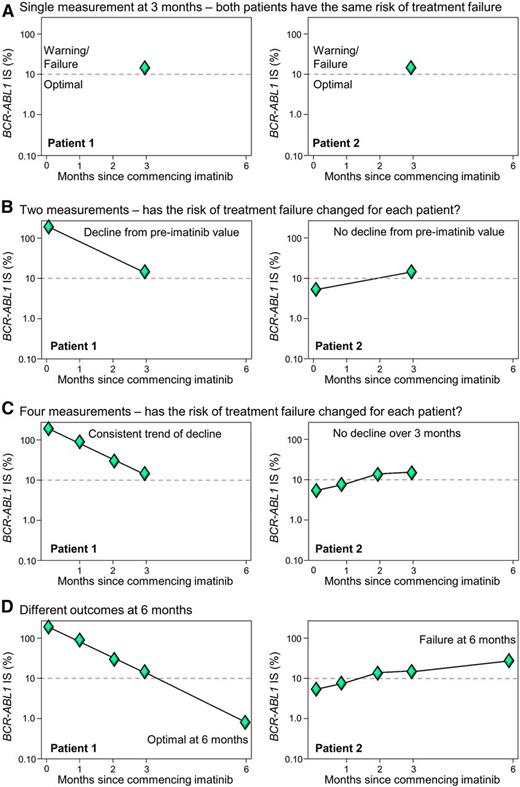
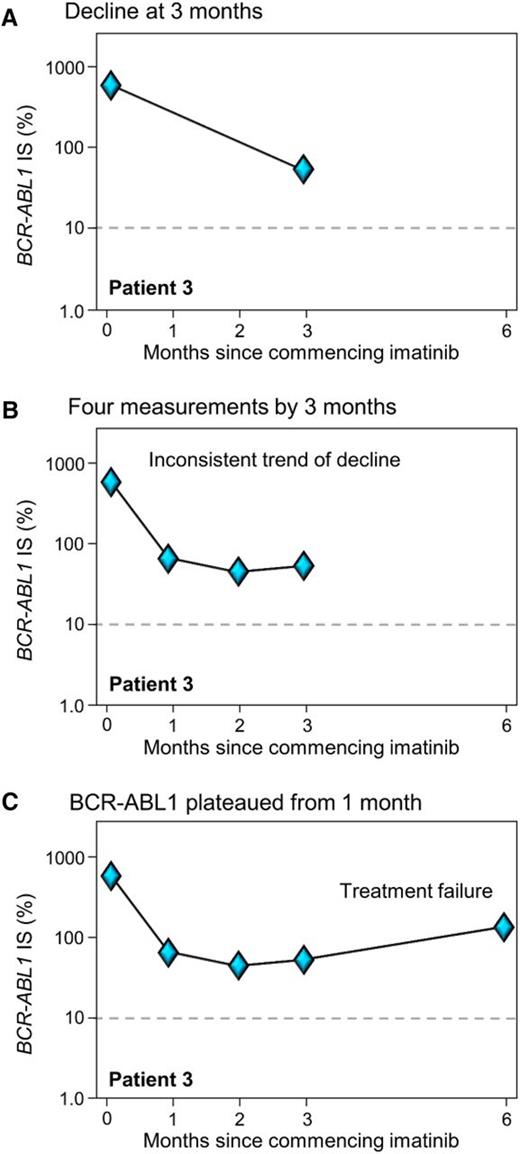
The ELN considered that one BCR-ABL1 measurement at 3 months of TKI was insufficient for decisions regarding treatment change to limit the risk of death or progression.1 Two or more measurements from 3 to 6 months may be a sounder basis for therapeutic intervention decisions. The ELN recommend that a change of therapy should proceed if BCR-ABL1 is still >10% at 6 months. However, a proportion of patients have disease progression early, and a delay in treatment decisions may not be desirable. This has prompted recent investigation into whether additional BCR-ABL1 measurements could provide extra information to guide early therapy decisions. A number of groups have incorporated the pre-TKI BCR-ABL1 value to assess the trend of decline over the first 3 months of therapy and have demonstrated the clinical utility of this approach.15-19 In effect, these studies incorporated the recommendation of the ELN for additional measurements to aid clinical decisions but applied the concept to earlier time points. Essentially, if there has been no or minimal BCR-ABL1 reduction from the pre-TKI value, it may support an early change of therapy. Assessment of the kinetics of response could be particularly useful for patients in whom the BCR-ABL1 value is close to the cutoff (Figure 2). Additional measurements in the critical first and second months of therapy may also prove beneficial. Figure 3A illustrates an example in which the decline measured using just 2 time points, pre-imatinib and at 3 months, indicates a substantial decline and response to imatinib. However, by including the measurements performed at 1 and 2 months (Figure 3B), it is evident that BCR-ABL1 plateaued early into therapy and the patient clearly had no response after 1 month of imatinib. The initial BCR-ABL1 values are highly predictive of long-term outcome and can identify the patients at risk of achieving less than optimal responses, including deep molecular responses.5 Two recent reviews have included tables that succinctly summarize outcomes according to landmark molecular responses.20,21
Multiple measurements in the critical first months of TKI therapy can aid response prediction and therapeutic decisions. (A) Two patients with the same BCR-ABL1 value >10% IS at 3 months of imatinib, indicating a warning (ELN) or treatment failure (NCCN). A single BCR-ABL1 measurement at 3 months may be insufficient for some patients to predict response. (B-D) Additional measurements over the first 3 months may identify a trend in response. Patients with no decline over the first 3 months may be at greater risk of treatment failure.
Multiple measurements in the critical first months of TKI therapy can aid response prediction and therapeutic decisions. (A) Two patients with the same BCR-ABL1 value >10% IS at 3 months of imatinib, indicating a warning (ELN) or treatment failure (NCCN). A single BCR-ABL1 measurement at 3 months may be insufficient for some patients to predict response. (B-D) Additional measurements over the first 3 months may identify a trend in response. Patients with no decline over the first 3 months may be at greater risk of treatment failure.
Two BCR-ABL1 measurements may be insufficient to assess the trend of decline in some patients. (A) At 3 months of imatinib, BCR-ABL1 had declined and the trend suggested the value may decline out of the failure zone at 6 months. (B-C) After an initial reduction at 1 month, BCR-ABL1 reached a plateau and the patient clearly failed therapy at 3 and 6 months. A kinase domain mutation was not detected at 1 month and the patient possibly acquired a BCR-ABL1–independent resistance mechanism.
Two BCR-ABL1 measurements may be insufficient to assess the trend of decline in some patients. (A) At 3 months of imatinib, BCR-ABL1 had declined and the trend suggested the value may decline out of the failure zone at 6 months. (B-C) After an initial reduction at 1 month, BCR-ABL1 reached a plateau and the patient clearly failed therapy at 3 and 6 months. A kinase domain mutation was not detected at 1 month and the patient possibly acquired a BCR-ABL1–independent resistance mechanism.
Why is a deep molecular response a new frontier?
A deep molecular response is commonly defined as BCR-ABL1 values of ≤0.01% IS and is described as various BCR-ABL1 cutoff values, where molecular response 4 (MR4) is ≤0.01% IS, MR4.5 ≤0.0032% IS, and MR5 <0.001%.6 Standard quantitative PCR assays aim to measure reliably to at least MR4, depending on the quality of the sample received for testing, whereas recent innovations have seen the sensitivity level extended to MR4.5 for some methods (with 95% confidence interval).22,23 From the first trial of imatinib in newly diagnosed patients (IRIS trial), there has been interest in the achievement of a deep molecular response. Allogeneic hematopoietic stem cell transplant rapidly induces undetectable levels of BCR-ABL1 in most patients, whereas only ∼3% achieved undetectable levels with imatinib using strict sensitivity criteria. This was consistent with in vitro data suggesting imatinib would not readily eradicate the leukemic clone. More recently, reaching a deep molecular response has amassed clinical significance, most prominently because it is associated with treatment-free remission.24,25 These studies have demonstrated that cessation for eligible patients is safe, and most patients rapidly regain response when imatinib is restarted after molecular relapse.24,25
The primary requirement for a drug cessation trial is reaching and maintaining a deep molecular response. The success of the initial imatinib cessation studies (STIM24 and TWISTER25 ), where ∼40% of patients remained in remission after cessation, has led to a plethora of follow-up studies. The initial cessation studies required at least 2 years of undetectable BCR-ABL1 using strict PCR sensitivity criteria before stopping (equivalent to MR4.5 or MR5).24,25 This length of time and PCR sensitivity were considered reasonable at the time,24 but new studies have introduced less stringent criteria to test the boundaries and thresholds for successful cessation.26-28
The Pan European Stop Tyrosine Kinase Inhibitor Trial in CML (EURO-SKI study) aims to identify prognostic markers to increase the rate of treatment-free remission after stopping TKI therapy. There are important differences in the enrollment criteria from the earlier studies, where 2 years undetectable BCR-ABL1 was required before stopping (at MR4.5 or MR5). For the EURO-SKI study, the less stringent response level of MR4 (detectable or undetectable BCR-ABL1) needs to be maintained for a minimum of only 1 year. An interim analysis of the first 200 patients observed no significant difference for molecular recurrence after TKI cessation according to the depth of response at the time of stopping therapy: MR4 vs MR4.5 vs MR5.26 Furthermore, savings at the time of analysis were estimated at €7 million. The EURO-SKI study preliminary data are very encouraging and suggest that less stringent criteria for discontinuation are possible, which has the potential to increase the number of patients meeting the criteria for a cessation study. With imatinib-based therapy and 8 years of follow-up, it was estimated that only 9% to 15% of patients would achieve treatment-free remission using the stringent criteria of the initial imatinib cessation studies.8
There is a clear and consistent association between a sustained deep molecular response and treatment-free remission. Some reports suggest that deep molecular response may also provide an advantage in terms of long-term clinical outcomes,9,29,30 as outlined in an excellent review by Mahon and Etienne.31 The German CML-Study IV assessed 1104 patients treated with front-line imatinib and found a survival advantage for those who had a confirmed MR4.5 at 4 years compared with patients with BCR-ABL1 values 0.1% to 1% IS (basically equivalent to complete cytogenetic response [CCyR] but no MMR): 8-year overall survival 92% vs 83%, P = .047. There was no statistical difference between MR4.5 and MR4 or between MR4 and MMR.9 It has been suggested by Kantarjian and Cortes32 that it is the inability to achieve CCyR or loss of CCyR that is the most important adverse factor for survival and an indication to change therapy.1,2 The molecular equivalent of CCyR is 1% BCR-ABL1,3 a value that is recognized by the ELN1 and the NCCN and incorporated into the recommendations and guidelines for decisions regarding therapy change.2
How to reach a deep molecular response
The response in the first few months of TKI therapy may be the most critical for long-term response. A factor strongly correlated with the highest rates of deep molecular response is reaching a BCR-ABL1 level of <1% IS by 3 months, irrespective of the starting TKI.5,8,9,33 Furthermore, failure to reach an MMR by the 12-month time point is associated with low rates of deep response.8,9 This is reflected in the recommendations for defining an optimal response, which assigns an MMR at 12 months as optimal.1 In imatinib-treated patients, we found that the highest rate of undetectable BCR-ABL1 MR4.5 was achieved by patients who reached an MMR at 3 months: 78% with 8 years of follow-up compared with 53% for patients with BCR-ABL1 >0.1% to 1% IS.8 A similar finding was reported for the CML-Study IV, where MMR at 3 months was associated with the highest MR4.5 level: 83% at 5 years vs 53% for patients with BCR-ABL1 >0.1% to 1% IS.9 For both of these evaluations of patients treated with imatinib-based therapy, only a minority group achieved MMR at 3 months: 9%8 and 4%9 of evaluable patients. However, the CML-Study IV demonstrated that the potency of kinase inhibition was associated with the time to achieve MMR: patients treated with a tolerability-adapted imatinib dose of 800 mg per day reached MMR at a median of 10 months compared with 18 months for those treated with imatinib 400 mg per day.34 Similarly, more patients reached deep responses with first-line use of nilotinib or dasatinib compared with imatinib 400 mg per day.33,35
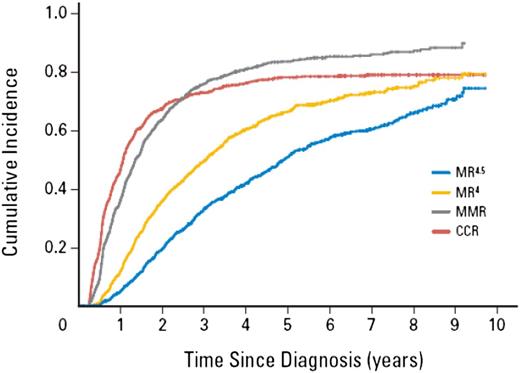
If reaching a deep molecular response is a goal of therapy, then minimizing the time to reach milestone responses may be a critical determinant of treatment choice, and more potent kinase inhibition may aid in the achievement of this goal. Of course, TKI-related toxicity is an essential consideration for treatment decisions.36 Nevertheless, sustained deep molecular responses in general require many years of therapy, which is related to the slow kinetics of BCR-ABL1 decline. Mathematical modeling suggests there is a triphasic exponential decline representing the turnover rate of different cell populations in response to TKI therapy.37 Evidence for the differing rates of decline in the clinical setting over the time course of imatinib is shown in Figure 4, which is the cumulative incidence of molecular response levels in the CML-Study IV.9 An initial rapid decline over the first 3 to 12 months was followed by a more gradual decline as MR4 was achieved and indicated by the median time to the responses. The rate of decline may slow again as evidenced by the longer time to reach MR4.5 from MR4 (a 3.2-fold difference) compared with the time to reach MR4 from MMR (a tenfold difference).
Deep molecular response can take many years to achieve because of the molecular response kinetics. The slow response kinetics was demonstrated in the German CML-Study IV. The graph shows the cumulative incidence of response for all patients treated in the imatinib-based study (>1300 patients). The estimated median time to reach MMR from diagnosis was rapid, at 1.4 years (∼300-fold reduction from the pre-TKI value). The estimated median time to reach the next response level MR4 (tenfold BCR-ABL1 reduction from MMR) was 3.1 years, a difference of 1.7 years. The median time to reach the next response level MR4.5 (3.2-fold BCR-ABL1 reduction from MR4) was 4.9 years, a difference of 1.8 years. This suggests the decline is not constant. The decline slows after reaching MMR and may slow again after declining to MR4. Reprinted from Hehlmann et al9 with permission. ©2014 American Society of Clinical Oncology. All rights reserved.)
Deep molecular response can take many years to achieve because of the molecular response kinetics. The slow response kinetics was demonstrated in the German CML-Study IV. The graph shows the cumulative incidence of response for all patients treated in the imatinib-based study (>1300 patients). The estimated median time to reach MMR from diagnosis was rapid, at 1.4 years (∼300-fold reduction from the pre-TKI value). The estimated median time to reach the next response level MR4 (tenfold BCR-ABL1 reduction from MMR) was 3.1 years, a difference of 1.7 years. The median time to reach the next response level MR4.5 (3.2-fold BCR-ABL1 reduction from MR4) was 4.9 years, a difference of 1.8 years. This suggests the decline is not constant. The decline slows after reaching MMR and may slow again after declining to MR4. Reprinted from Hehlmann et al9 with permission. ©2014 American Society of Clinical Oncology. All rights reserved.)
The slow kinetics of response is an important factor when considering molecular end points for clinical trials. Indeed, the ENESTcmr trial selected the rate of confirmed undetectable BCR-ABL1 by 12 months as the primary end point for patients who switched to nilotinib while in a CCyR compared with patients who continued on imatinib.38 The study failed to meet its primary end point, although the difference was significant at 24 months. The lack of statistical significance at 12 months may be related to the slow kinetics of decline in this patient population.
Molecular monitoring: how low do you need to go?
Evidence is now strong in support of reaching deep molecular response levels for certain patient populations. Toxicity, patient age, and other considerations will no doubt dictate the goals of therapy for individual patients. The question remains whether BCR-ABL1 detection limits of MR4 to MR5 are adequate to meet the clinical requirements. In other words, is there any evidence that the detection of BCR-ABL1 at levels <0.001% IS are of any clinical benefit? I suggest it would be extremely unlikely that a survival advantage or a difference in the rate of failure events could be demonstrated compared with MR4.5. Therefore, any clinical benefit of enhanced molecular sensitivity would relate to TKI cessation studies.
The first imatinib cessation studies used undetectable BCR-ABL1 as the molecular qualification criterion,24,25 and a reasonable hypothesis was that patients with detectable BCR-ABL1 at the time of ceasing imatinib using more sensitive techniques are at a higher risk of molecular recurrence. However, this was not the case.39,40 Ross et al39 determined the patient-specific genomic DNA BCR-ABL1 breakpoints in patients with known outcome after ceasing imatinib. Patient-specific, enhanced-sensitivity PCR assays were used to evaluate BCR-ABL1 DNA levels at the time of cessation. Five of 7 patients who maintained treatment-free remission and 8 of 9 patients with molecular recurrence had detectable BCR-ABL1 DNA before ceasing imatinib. Therefore, there was no discriminating power for the enhanced-sensitivity DNA PCR assay. Mahon et al40 performed a similar analysis using an enhanced-sensitivity PCR for BCR-ABL1 transcripts with replicate analysis. Again, the assay provided no discriminating power for the prediction of relapse. The DNA and RNA enhanced-sensitivity techniques achieved improved detection limits of ∼tenfold compared with the standard methods that were used for these cessation trials.24,25 Importantly, the enhanced sensitivity analyses provided evidence of TKI-free leukemic suppression. An area of active research continues to be the identification of patients at risk for molecular recurrence after TKI cessation.
Whether there is a BCR-ABL1 level below which no patient will have molecular recurrence after stopping TKI is unknown. Next-generation sequencing combined with digital DNA BCR-ABL1 PCR is a recent innovation that could be the most sensitive currently available technique.41 In the future, this or other novel techniques may be used to revisit the relevance of the BCR-ABL1 detection limit for patient selection for TKI cessation studies.
It is clear that currently available technology could be of immediate clinical benefit for monitoring the molecular response of many patients in low-resource regions or for laboratories that lack relevant molecular biology expertise for adequate BCR-ABL1 method development. The Cepheid GeneXpert BCR-ABL monitor is an automated cartridge-based microfluidic system that combines RNA extraction, reverse transcription, and quantitative PCR. The GeneXpert instrument has an onboard computer to calculate BCR-ABL1 IS values on a cartridge lot-to-lot basis and provides the sensitivity achieved for each sample. An enhanced sensitivity cartridge (MR4.5) has recently been described.23 Furthermore, a novel use of the cartridge system has recently been demonstrated in which comparable GeneXpert BCR-ABL1 values were recovered from dried blood that was spotted onto a paper template and posted by regular mail internationally between centers.42 The success of this study paves the way to improve the accessibility of molecular testing for more patients by the affordable transport of blood samples to specialized centers for testing, while ensuring RNA integrity.
BCR-ABL1 mutation analysis: how low do you need to go?
Point mutations in the BCR-ABL1 kinase domain remain the major known mechanism of acquired TKI resistance. Standard Sanger direct sequencing is the mainstay of mutation detection, but the technique has a limited detection range and can only detect mutations if they represent at least 10% to 20% of cells. This is usually adequate for characterizing the type of mutation associated with clinical resistance of individual patients and for choice of the appropriate rescue therapy according to the sensitivity profile of the mutation. Mutations expand very rapidly with resistance and are usually accompanied by a rise in BCR-ABL1 transcript level. By direct sequencing, mutations are frequently detectable in the sample collected just before the BCR-ABL1 rise, but rarely before this. Therefore, routine testing for resistant mutations for patients treated with first-line TKI plays no role in patient monitoring because of the relative rarity of mutation emergence and the rapidity of BCR-ABL1 mutation emergence.
More sensitive BCR-ABL1 mutation detection is clinically relevant in some situations.43 Using a mass spectrometry technique for a selection of 31 mutations and a detection sensitivity of ∼0.2%, low-level mutations detected before commencing nilotinib or dasatinib after imatinib resistance rapidly expanded with resistance. This was for mutations such as low-level T315I, which is known to confer resistance to both inhibitors, or F317L or Y253H, known to confer clinical resistance to dasatinib and nilotinib, respectively. Furthermore, the detection of more than one low-level mutation was associated with poor outcome to subsequent TKI therapy after resistance, irrespective of the resistance profile of the mutations.44 Therefore, subclonal diversity could be an important prognostic marker of the capacity of leukemic cells to evolve TKI resistance. Interestingly, using the mass spectrometry sensitive mutation technique, Parker et al45 demonstrated in chronic- and advanced-phase patients treated with ponatinib in the PACE study that the association between multiple low-level mutations and poor outcome was not found with ponatinib therapy. Therefore, ponatinib may be beneficial for patients with multiple mutations after prior TKI. However, T315I with additional mutations was associated with poorer outcome for the chronic-phase patients compared with those who only had the T315I mutation.
Next-generation sequencing (NGS) has the capacity for low-level mutation detection and, unlike mass spectrometry in which only selected mutations are sequenced, allows an unbiased analysis of the entire BCR-ABL1 kinase domain.46 With a detection limit of 1% mutant, Soverini et al46 used amplicon NGS to investigate the clonal structure and evolution of BCR-ABL1 mutations over time during therapeutic intervention for 33 patients with TKI resistance. Low-level mutants were detected in 55% of patients and a complex clonal architecture was revealed. However, the sensitivity of NGS coupled with PCR amplicon analysis may expose PCR-mediated recombination events and these need to be taken into account when assessing results.47 NGS was coupled with direct sequencing to assess the impact of low-level and compound mutations (where mutations occur in the same BCR-ABL1 molecule) in chronic-phase patients treated with ponatinib.47 These were heavily pretreated patients from the PACE trial. Preclinical studies suggested that certain compound mutations would lead to ponatinib resistance. This was not the case for these patients: no single or compound mutation was a major driver of resistance. Similar to other studies, low-level mutations were detected (27% of patients).
Conclusion
Low-level molecular analysis has become an important component of the monitoring strategy for patients with CML. For those who remain on TKI long term, particularly patients who do not experience resistance, it seems that the majority will achieve MR4.5. Whether there will be any clinical benefit for more sensitive BCR-ABL1 transcript detection remains to be established. However, new technology may expand the boundaries of BCR-ABL1 detection to allow further clinical evaluation, principally for those patients who enter TKI cessation studies. The EURO-SKI TKI cessation study should provide important data and could redefine the threshold where cessation could be attempted.
Standard Sanger sequencing remains an important assessment for patients experiencing resistance or loss of any response. Sensitive mutation analysis at the time of resistance can distinguish the patients with multiple mutations, and in certain situations these patients have an inferior outcome with subsequent TKI. Therefore, there is a clinically relevant role for low-level BCR-ABL1 mutation detection.
Correspondence
Susan Branford, Department of Genetics and Molecular Pathology, SA Pathology, PO Box 14 Rundle Mall, Adelaide, SA 5000, Australia; e-mail: susan.branford@sa.gov.au.
References
Competing Interests
Conflict-of-interest disclosure: The author is on the Board of Directors or an advisory committee for Qiagen; has received research funding from Ariad, Novartis, and Otsuka; has consulted for Cepheid; has received honoraria from Qiagen, Bristol-Myers Squibb, and Novartis; and has been affiliated with the Speakers Bureau for Novartis.
Author notes
Off-label drug use: None disclosed.