Key Points
Long-lived plasma cells secreting vaccinia-specific antibodies are detected in human bone marrow >35 years after the eradication of smallpox.
Long-lived plasma cells secreting vaccinia-specific antibodies are still able to express the B-lymphocyte antigen CD19.
Introduction
One of the most important achievements in medicine is the eradication of smallpox in 1979. Before that time, smallpox outbreaks were common and left a trail of death, blindness, and disfigurement. High titers of smallpox-specific antibodies are still detected in the blood of individuals who were vaccinated decades ago. The fact that more than 35 years have passed since the eradication of smallpox suggests that vaccine-specific antibody production has been maintained without restimulation by a specific antigen after vaccination. Moreover, smallpox-specific antibody levels are maintained in serum for more than 70 years.1 The mechanism behind high antibody titers that persist for decades despite the absence of the inducing antigen is poorly understood.
Viral infection models have shown that the initial antibody response occurs in the spleen. However, after clearing the initial infection, and for the remainder of the animal’s life, the bone marrow (BM) takes over antibody production and becomes the major organ for long-term production of virus-specific antibodies.2 In addition, a large fraction of plasma cells in murine BM is able to survive and produce antibodies for more than a year without any detectable memory B cells.3 Indeed, long-lived plasma cells (LLPCs) can be found in the BM where they reside in a complex survival niche for an undefined length of time and secrete antigen-specific antibodies that protect us from infections.2-4 Importantly, the majority of the knowledge on LLPCs has been gained through mouse models. Data from human BM has been scarce until recently, when it was suggested that the expression of CD19 is downregulated on LLPCs.5,6
Methods
Patients
Patients undergoing a primary hip arthroplasty were recruited at Sahlgrenska University Hospital, Gothenburg, Sweden. In all, 36 patients were recruited: 26 females (median age, 74.5 years; range, 50-86 years) and 10 males (median age, 71 years; range, 49-91 years). Patients were asked whether or not they had received vaccinia vaccine: females (yes, 17; unsure, 9; no, 0) and males (yes, 4; unsure, 5; no, 1). Patients were also examined for a vaccine-related scar. The study was approved by the regional ethical committee (permit 627-14) and conducted in accordance with the Declaration of Helsinki.
BM processing
BM was homogenized, and mononuclear cells were isolated by Ficoll density gradient (GE Healthcare Life Sciences, Little Chalfont, UK) before staining for flow cytometry. Details on processing are provided in the supplemental Methods.
Flow cytometry
Cells were stained with combinations of fluorescent antibodies and analyzed by using an LSR II or an ARIA IIIμ cell sorter (BD Biosciences, Oxford, UK). Data were analyzed by using FlowJo software (Tree Star, Ashland, OR). See supplemental Methods for further details.
ELISPOT
The Enzyme-Linked ImmunoSpot (ELISPOT) assay was performed essentially as previously described.7 Further details are given in the supplemental Methods.
Statistical analysis
Statistical analysis was performed with GraphPad Prism 6.0 (GraphPad Software, La Jolla, CA). For comparison of 2 independent groups, the Mann-Whitney U test was used. A P value below .05 was considered statistically significant.
Results and discussion
Peripheral blood mononuclear cells from an individual vaccinated with vaccinia virus 39 years ago (1978) were stimulated with a B-cell activation cocktail.7 This induced memory B cells in peripheral blood to differentiate into cells secreting vaccinia-specific antibodies (supplemental Figure 1). We next investigated BM to address whether B cells secreting vaccinia-specific antibodies were present in the BM of individuals vaccinated >40 years ago and more than 35 years after the eradication of smallpox. In unstimulated BM mononuclear cells, B cells secreting vaccinia-specific immunoglobulin G (IgG) were detected (Figure 1A). The presence of cells secreting vaccinia-specific antibodies in the absence of stimulation suggests that they are LLPCs.
Plasma cells in human bone marrow. (A) Vaccinia-specific IgG-secreting cells (top) and total IgG-secreting cells (bottom) of total BM mononuclear cells compared with control are shown (50 000 cells per well were seeded). The cells are from the same patient. The plot shows the frequency of vaccinia-specific IgG-secreting cells among total BM mononuclear cells of all patients examined (n = 26). (B) Gating strategy to sort plasma cells into CD19+ (left) and CD19– populations (right). (C) The percentage of plasma cells of 100 000 live BM mononuclear cells (left) and the percentage of CD19+ and CD19– plasma cells of total plasma cells (right), gated as in panel B, are shown (n = 27). (D) CD19+ and CD19– plasma cells secreting vaccinia-specific antibodies (top) with matching wells showing the secretion of total IgG (bottom). All wells are from the same patient and 5000 cells of each type were seeded. The plot shows paired CD19+ and CD19– vaccinia-specific IgG+ plasma cells from the same patient (n = 19). *P < .05; **P < .01; ***P < .001.
Plasma cells in human bone marrow. (A) Vaccinia-specific IgG-secreting cells (top) and total IgG-secreting cells (bottom) of total BM mononuclear cells compared with control are shown (50 000 cells per well were seeded). The cells are from the same patient. The plot shows the frequency of vaccinia-specific IgG-secreting cells among total BM mononuclear cells of all patients examined (n = 26). (B) Gating strategy to sort plasma cells into CD19+ (left) and CD19– populations (right). (C) The percentage of plasma cells of 100 000 live BM mononuclear cells (left) and the percentage of CD19+ and CD19– plasma cells of total plasma cells (right), gated as in panel B, are shown (n = 27). (D) CD19+ and CD19– plasma cells secreting vaccinia-specific antibodies (top) with matching wells showing the secretion of total IgG (bottom). All wells are from the same patient and 5000 cells of each type were seeded. The plot shows paired CD19+ and CD19– vaccinia-specific IgG+ plasma cells from the same patient (n = 19). *P < .05; **P < .01; ***P < .001.
CD19 expression has been shown to be downregulated on LLPCs specific for measles and mumps.5,6 We thus examined CD19 expression on vaccinia-specific LLPCs. CD138+CD38high plasma cells were sorted as either CD19+ or CD19– (Figure 1B). LLPCs secreting vaccinia-specific IgG+ antibodies were detected in both the CD19+ and CD19– fractions (Figure 1C). The presence of C19+ vaccinia-specific LLPCs suggests that the generation of LLPCs is an ongoing process. It also raises the possibility that the vaccinia-specific LLPCs expressing CD19 have recently been generated and have not yet downregulated CD19. Detecting LLPCs secreting vaccinia-specific antibodies that have not downregulated CD19 might be explained by the possibility that the vaccinia vaccine given to these individuals decades ago was a more potent vaccine than those for measles, mumps, or tetanus.
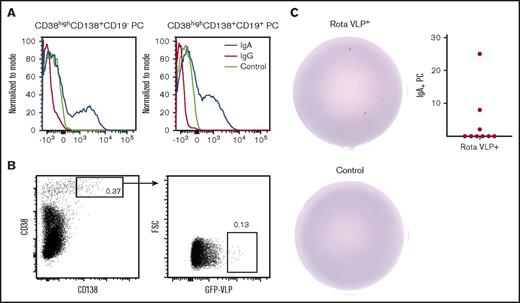
While investigating plasma cells in the BM, we found that expression of IgA on CD19+ and CD19– plasma cells was not downregulated but surface IgG was (Figure 2A). A similar observation was previously reported for antibody-secreting cells in the human gut8 and for plasma cells.9 To further examine surface IgA on LLPCs, we turned to a mucosal disease that affects nearly all children, namely, rotavirus. During the first years of life, nearly all children will experience a rotavirus infection at least once.10,11 Mucosal priming has been shown to induce LLPCs secreting IgA+ antigen-specific antibodies in murine BM.12 We thus examined whether rotavirus-specific LLPCs could be detected in human BM.
Surface IgA expression is maintained on plasma cells. (A) Surface expression of IgA and IgG on CD19+ and CD19– plasma cells from BM. Control is isotype control. (B) The gating strategy for sorting rotavirus-specific plasma cells. (C) Sorted rotavirus-specific cells secrete rotavirus-specific IgA (top) compared with control (bottom) (200 rotavirus-specific cells were seeded). The graph depicts the frequency of IgA+ rotavirus-specific plasma cells of total plasma cells (n = 9).
Surface IgA expression is maintained on plasma cells. (A) Surface expression of IgA and IgG on CD19+ and CD19– plasma cells from BM. Control is isotype control. (B) The gating strategy for sorting rotavirus-specific plasma cells. (C) Sorted rotavirus-specific cells secrete rotavirus-specific IgA (top) compared with control (bottom) (200 rotavirus-specific cells were seeded). The graph depicts the frequency of IgA+ rotavirus-specific plasma cells of total plasma cells (n = 9).
CD138+CD38+ plasma cells expressing a B-cell receptor specific for rotavirus were sorted from BM by using rotavirus virus-like particles (VLPs) expressing GFP.13 The VLPs have previously been used for sorting rotavirus-specific plasma cells.8 GFP+ events were collected and seeded on ELISPOT plates coated with rotavirus VLPs (Figure 2B). LLPCs secreting rotavirus-specific IgA were detected in 3 of 9 patients (Figure 2C). This indicates that rotavirus-specific IgA+ plasma cells can maintain their B-cell receptors. Considering that the BM was obtained from elderly individuals exposed to the virus many decades ago, the rotavirus-specific plasma cells are likely LLPCs. However, we cannot rule out that the individuals have encountered rotavirus again during their daily life.
It has been hypothesized that CD19+ plasma cells generated by vaccination migrate to the BM where the majority of cells undergo apoptosis. A few cells escape apoptotic death and are able to mature into LLPCs, a transition that includes downregulation of CD19 expression.5,6 That both vaccination to and eradication of smallpox occurred decades ago suggest that the vaccinia-specific plasma cells detected in the BM have not migrated there recently. This raises the possibility that differentiation of new vaccinia-specific plasma cells may occur in the BM. In support of this, we detected both CD19+ and CD19– vaccinia-specific plasma cells in the BM, suggesting that the LLPC pool may be upheld by a process in which vaccinia-specific B cells differentiate into LLPCs in the BM. Whether CD19+ vaccinia-specific LLPCs are generated from vaccinia-specific memory B cells or by some other mechanism and with time downregulate CD19 expression and mature into LLPCs, is presently not known.
Taken together we have shown that both CD19+ and CD19– vaccinia-specific LLPCs are present in BM decades after vaccination and eradication of smallpox. Moreover, we have demonstrated that rotavirus-specific IgA-producing LLPCs in the BM are still able to express their B-cell receptors on the cell surface. These results underscore the complexity of the BM as a survival niche for immunity and its maintenance, which is highly important for the design of more potent vaccines capable of inducing life-long protection against infectious diseases.
The full-text version of this article contains a data supplement.
Acknowledgments
The authors thank Didier Poncet and Annie Charpilienne (Gif-sur-Yvette, France) for providing the rotavirus VLPs; Elias Guðbrandsson, Peter Grant, Christofer Ståhlgren, Kamal Kadum, and Ola Rolfson for assisting with collection of samples; and all the medical personnel in Departments 232 and 233 at Mölndals Hospital who provided assistance.
Authorship
Contribution: M.M. and J.K. provided human bone marrow; S.F.B. performed the experiments; and S.F.B. and M.-J.W. designed the research, analyzed the data, and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Siggeir F. Brynjolfsson, Department of Microbiology and Immunology, University of Gothenburg, Box 435, 405 30 Gothenburg, Sweden; e-mail: siggeir.brynjolfsson@gu.se.