Abstract
Bone marrow (BM) constitutes one of the largest organs in mice and humans, continuously generating, in a highly regulated manner, red blood cells, platelets, and white blood cells that together form the majority of cells of the body. In this review, we provide a quantitative overview of BM cellular composition, we summarize emerging knowledge on its structural organization and cellular niches, and we argue for the need of multidimensional approaches such as recently developed imaging techniques to uncover the complex spatial logic that underlies BM function in health and disease.
Since Neumann and Bizzozero independently proposed in 1868 that the origin of blood cells was to be found in soft tissues contained inside bone cavities, the bone marrow (BM) has continued to fascinate scientists and clinicians alike.1,2 Yet more than a century later, our understanding of how the hematopoietic, immune, and bone-forming tasks of the BM are tightly controlled and integrated in the context of one common anatomical space is still incomplete.
Methodological approaches and advances to study BM tissues
Early studies on BM cellular composition and microarchitecture date back to the 19th century when the first biopsies of marrow content were performed in living individuals.3 Microscopic observation of BM smears and examination of histological sections became the standard technique, which is still used to this day to study, diagnose, and classify hematologic disorders. The realization in the post–World War II era that transplantation of BM cells from healthy individuals could rescue the lethal effects of high-dose irradiation on hematopoiesis galvanized scientific interest in BM and lead to the development of methods to detect and measure hematopoietic stem and progenitor cell (HSPC) activity, absolute cellularity, lineage-specific half-lives, and kinetics of cell production in the BM of humans and in laboratory animals.4,5 Gradual improvements in light and electron microscopy further refined the understanding of the quantitative composition and ultrastructural features of BM tissues.6 The biggest leap in the fields of immunology and hematology came with the advent of recombinant antibody technology and flow cytometry in the 1960s. These breakthroughs permitted the identification, quantification, and isolation of diverse populations from highly heterogeneous cell suspensions through detection of phenotypic traits.5 Indeed, to this day, flow cytometry remains the technological mainstay for the study and dissection of the hematopoietic system.
Immunolabeling methods were further adopted in modern fluorescence-based microscopy to discriminate cellular phenotypes in tissue sections and study cellular organization in the BM. In recent years, confocal microscopy has become the most popular modality to define spatial relationships and study developmental-stage specific localization patterns, with a keen interest of many groups in the dissection of HSPC niches.7 In addition, intravital multiphoton microscopy is employed to obtain dynamic images of the most superficial layers of BM beneath bone surfaces, leading to seminal discoveries on cellular interplay and trafficking.7,8 A spectacular enhancement in the quantitative power of microscopy techniques is currently being driven by technological advances, which include the development of (1) protocols that render organs almost completely transparent for volumetric imaging9 ; (2) novel modalities of optical imaging such as light sheet microscopy or optical projection tomography for faster, automated acquisition of multidimensional images of large tissue regions or entire organs10,11 ; (3) more sophisticated excitation and detection systems that enable increased multiplexing; and (4) bioimage-based analysis software tools to facilitate the unbiased extraction of cytometric measurements and computation of quantitative spatial information from complex, high-dimensional data sets.12 Thus, it is now becoming possible to generate and analyze organ-wide three-dimensional (3D) reconstructions of a variety of tissues including BM and spleen with cellular and subcellular resolution (Figure 1).13-15 Collectively, multidimensional microscopy techniques hold great promise to take our understanding of the global microanatomy of hematopoiesis to the next level, that is, one in which distinct stages of development along the hematopoietic continuum are precisely resolved and mapped into a panoramic view of BM topology.
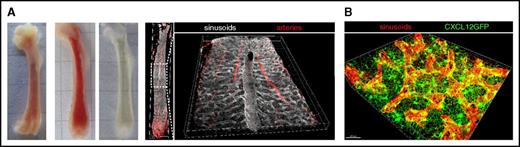
Three-dimensional imaging of BM. (A) Representative stages of murine femoral bones during processing for 3D imaging as described in Nombela-Arrieta et al.13 From left to right: unsectioned, sliced unprocessed, and optically cleared BM slices. The last 2 images to the right correspond to an example of 3D reconstruction of the entire BM microvascular system of the femur (arteries and sinusoids) using multidimensional confocal imaging of a cleared BM slice. A detailed view of the central sinus running along the longitudinal axis of the marrow is provided. (B) High-resolution 3D imaging of a reduced field of view showing sinusoidal vessels (red) and the network of perivascular bodies of CAR cells (green), forming a dense matrix through the emission of abundant cytoplasmic projections.
Three-dimensional imaging of BM. (A) Representative stages of murine femoral bones during processing for 3D imaging as described in Nombela-Arrieta et al.13 From left to right: unsectioned, sliced unprocessed, and optically cleared BM slices. The last 2 images to the right correspond to an example of 3D reconstruction of the entire BM microvascular system of the femur (arteries and sinusoids) using multidimensional confocal imaging of a cleared BM slice. A detailed view of the central sinus running along the longitudinal axis of the marrow is provided. (B) High-resolution 3D imaging of a reduced field of view showing sinusoidal vessels (red) and the network of perivascular bodies of CAR cells (green), forming a dense matrix through the emission of abundant cytoplasmic projections.
Cellular content of BM tissues in mice and humans
Intraosseous marrow tissue masses distributed inside the different long, short, and flat bones of the organism constitute one of the largest organs in humans, accounting for 4% to 5% of total body weight,16,17 therefore exceeding other major vital organs such as brain (2%), liver (2.6%),18 heart (0.5%),19 or the entire network of secondary lymphoid organs (1% to 1.5%).18 In a “standard” 70 kg adult human, BM tissues weigh ∼3 kg and occupy a volume of 1.5 to 3 L (Table 1). BM is the most prominent source of de novo cellular generation, reaching rates of 4 × 1011 to 5 × 1011 cells per day in adult humans (0.5-1.5 × 109 in mice),20-23 which outnumber those of other tissues of high turnover such as intestinal epithelium (1011 cells per day)24 or testis (100-300 × 106 spermatozoids per day). To achieve this, it is estimated that BM tissues collectively deliver daily to systemic circulation the equivalent of half of their total cellular content, although only approximately one-fourth of produced cells are nucleated. Remarkably, at any given moment, 90% of total cell numbers of the organism originates from or resides in the BM, with anuclear erythrocytes and platelets accounting for the majority of it.25
A detailed census of BM hematopoietic cellular constituents in humans and animal models has been generated through the use of flow cytometry (Table 2). Despite the transient and dynamic nature of its hematopoietic components, which results in an ever-changing cellular landscape, the relative proportions of different lineages remain consistently uniform between different bone cavities, strongly arguing for the existence of robust systemic signals that control BM cellular content.26 Rare, self-renewing HSPCs lie at the apex of the system’s hierarchy and give rise to myriads of oligopotent, highly proliferative progenitors that sequentially progress along the hematopoietic hierarchy.27 Collectively, the most primitive compartment including HSPCs and multipotent cells only comprises a minor part of the cellular content of the marrow (Table 2). Otherwise, BM spaces team with differentiating and fully mature cells, predominantly of the myeloid lineage, which mostly includes immature and mature granulocytes. Myeloid prevalence is significantly more accentuated in human compared with murine BM, where the B-cell lineage represents a more sizeable fraction of BM content. In both systems, however, aging results in an increased myeloid content at the expense of lymphoid lineages through a lineage-biased molecular program, which is activated already at the level of the most primitive HSC subsets.28,29
Notably, flow cytometric analyses are typically restricted to BM mononuclear cells, thus neglecting the contribution of some of the most frequent cell types, such as erythrocytes, platelets, and the rarer but still relatively frequent megakaryocytes.30 Although flow cytometry has started to be applied to study the composition of densely packed nonhematopoietic stromal meshworks formed by endothelial, mesenchymal, and neural cells, these are hard to dissociate into liquid suspensions of viable cells. It is therefore unclear to what extent stromal subsets extracted by enzymatic digestion quantitatively and qualitatively relate to the overall composition of the BM microenvironment in vivo. For instance, a number of studies using fluorescent reporters or immunophenotypical signatures have indicated that in mouse BM, mesenchymal, and vascular cells account for <1% of total mononuclear cellular pool.31-35 However, by direct comparison with imaging data14,31 (Figure 1B) it becomes unequivocally obvious that such quantifications result in a vast underestimation of these cell types, a limitation worth taking into account when drawing conclusions on the physiological relevance of stromal subsets.
The 3D organization of BM tissues and cell-specific niches
Beyond the enumeration of cellular components, in depth knowledge on tissue structural assembly and 3D microarchitecture is essential for the detailed understanding of multicellular organ function.36 The definition of a BM microstructural standard is confounded by the fact that BM tissues are dispersed throughout bone cavities that greatly differ in size and morphology. Following general macroscopic criteria of bone morphology, 2 types of BM regions are distinguished: (1) Trabecular or cancellous bone regions, mostly present in flat and irregular bones, as well as the epiphysis and metaphysis of long bones, contain BM islands interspersed within a labyrinth of boney trabeculae.37 (2) Cortical bone is found in the diaphysis or shafts of long bones, which is predominant in human and mouse skeletal systems. Trabecular regions are most active in terms of bone remodeling, contain an increased endosteal surface/BM volume ratio, and are more densely vascularized. Despite obvious anatomical differences, only minor variations in hematopoietic content and physiology between both types of BM spaces have been documented.14,26,38
Stromal scaffolds in BM.
Given the rapid turnover of most hematopoietic cells, stromal scaffolds provide the immanent 3D infrastructure for BM tissue integrity.39 Vascular networks, aside from essential functions of nutrient delivery, waste removal, cellular trafficking, and angiocrine-mediated regulation of hematopoiesis, fulfill important structural roles.40 Using 3D imaging, we and others have provided a detailed 3D overview of the microarchitecture of the microvascular system in different bones, which is composed of arterial and sinusoidal vessels (the venous equivalent of the BM), interconnected through a recently defined intermediate vascular type.13,14,32,41,42 Collectively, the pervading BM vascular tree occupies 15% to 30% of the volume of BM, most of which corresponds to the wide, highly branched, labyrinth-like sinusoidal network32 (C.N.-A., unpublished data) (Figure 1A). In essence, the vast majority of BM cells lie proximal to sinusoidal surfaces,14 thereby illustrating the potentially massive influence of endothelial components in the regulation of hematopoiesis in steady state as well as during adaptation to stress conditions. Indeed, dynamic reactive changes in vascular morphology and diameter correlate with drastic shifts in blood and BM cellular content43,44 (C.N.-A., unpublished data).
The mesenchymal compartment is thought to organize as a developmental hierarchy, which according to recent data, could be as diversified as its hematopoietic counterpart.45,46 The largest fraction of the BM mesenchymal component presents in the form of a dense weblike network of perivascular cell bodies interconnected through large cytoplasmic projections that pervades entire BM tissues (Figure 1B). These fibroblast-like reticular cells have been generically defined in mice by expression of common markers (leptin receptor and CD140a), and the secretion of large amounts, at least by a substantial fraction of this population, of regulatory factors such as interleukin-7 (IL-7), stem cell factor, and CXCL12.31,47-50 For this feature, they were first visualized in the CXCL12-GFP knock-in mouse model and termed CXCL12-abundant reticular (CAR) cells.51 Multiple studies have now shown that CAR cells are fundamental players in the regulation of specific and very distinct stages of hematopoiesis. How they exert such seemingly diverse regulatory actions is poorly understood and points to the presence of a high heterogeneity within this population that is yet to be dissected. The CAR cell network is also an essential component of the skeletal system as it includes bipotent progenitor cells, which are the source of newly formed osteoblasts and adipocytes during adulthood.31,52 Terminal osteoblastic differentiation is proposed to take place in a continuous fashion along inner bone surfaces where mature osteoblasts reside,53 but the precise cellular intermediates through which osteo-and adipogenic lineage differentiation progress are not well defined, and thus their spatial localization in BM tissues has not been mapped in detail. Although incompletely defined, a human counterpart of CAR cells, with similar osteoprogenitor and hematopoietic supporting potential, has been described.54 Mesenchymal progenitor cells have also been reported to include a subset of rare, Nestin-GFPhi NG2+ elongated cells, which run along the trajectory of the arterial network, adjacent to nonmyelinating Schwann cells and sympathetic nerves, forming anatomically distinct neurovascular structures.32,55 On the basis of expression of distinct phenotypic signatures, the presence in BM of additional cellular subsets of mesenchymal origin have been reported.34,56 Yet the quantitative and structural contribution of such populations to BM tissues, their potential role in hematopoietic regulation, as well as their specific developmental position in current models of mesenchymal differentiation are still to be dissected.
BM adipogenesis from reticular cells takes place gradually and asymmetrically in different bone locations during physiological aging, a process that is more conspicuous in humans than mice. Adipocytic infiltration is prominently induced in stress conditions such as irradiation, and correlates with a degeneration of vascular structures, lower bone formation rates,57 and reduced hematopoietic activity.58 The mechanisms driving spatial patterning of adipogenesis and its impact in the integrity of neighboring vascular and mesenchymal structures are poorly understood. Of note, multiple studies suggest that marrow adipose tissue is heterogeneous in terms of adipocytic and lipid content, regional distribution, and regulatory function.59,60 Finally, stromal cell networks are built around the proteic acellular backbone created by fibers of extracellular matrix (ECM). Studies in BM and other lymphoid organs indicate that highly plastic ECM networks could play vital roles in steady state as well as pathological hematopoiesis.61,62 However, not much is known to date on the heterogeneity, biochemical composition, and topology of BM ECM.
Spatial organization of hematopoiesis.
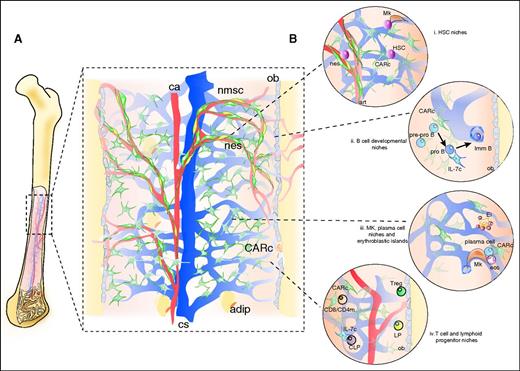
How the succession of orderly events that leads to continuous hematopoiesis develops within the described tissue microfabric remains poorly understood. Unlike other hematolymphatic organs such as thymus and lymph nodes,36 no conspicuous regionalization of lineages, developmental stages and/or cellular processes has been described in the BM. Rather than stable, well-demarcated zones, studies on BM organization have revealed the presence of microdomains defined by interactions of 2 or more cell types, which associate into functional units in which exchange of key signals leads to maintenance or differentiation of specific developmental stages. Cellular “niches” in BM have been reported for HSPCs, common lymphoid progenitors (CLPs), B-cell progenitors, erythrocytes, regulatory T cells, CD4/CD8 memory and plasma B cells, among others (summarized in Figure 2 and described in detail by Mercier et al63 ). Many of the niches are constructed around diverse microvessel types, which distinctly regulate access to nutrients, oxygen, and metabolites derived from circulating blood.64-66
Structural organization of BM tissues. (A) BM stromal microarchitecture. The microarchitecture of the vascular system has been studied in detail in the mouse femoral cavity. A central artery (ca) penetrates the cavity through the nutrient canal and splits into ascending and descending branches, which run longitudinally, arborizing into smaller radial arteries that migrate toward endosteal regions. In the proximity of endosteum, arteries give rise to a dense plexus of arterioles that travel along the cortical bone area and eventually develop into venous vessels, of fenestrated endothelium, termed sinusoidal. Sinusoids form a labyrinth that extends inward and merges in a big central collecting sinus (cs) that drains into peripheral circulation (see also Figure 1). Thin periosteal arteries also penetrate the bone and merge with arteriolar vessels in endosteal regions, connecting BM and bone circulation. In trabecular bone areas, multiple smaller arteries enter the marrow cavities and give rise to sinusoidal networks along the endosteal surface of trabeculae. CXCL12-abundant fibroblastic reticular cells (CARc) extend throughout the entire cavity in the form of dense networks. Nestin-GFPhi NG2+ (nes) elongated cells run adjacent to arteries and arterioles and bundles of nonmyelinating Schwann cells (nmsc), which ensheath adrenergic nerves. Mature osteoblasts (ob) line endosteal surfaces and are mostly derived from reticular progenitors of osetoadipogenic potential. The emergence of adipocytes (adip) takes place gradually during aging and can be abrupt and prominent in certain pathological conditions. (B) Schematic representation of cell or developmental stage-specific niches. (i) HSC niches: a number of studies have determined that HSCs are found scattered throughout BM tissues adjoining the extraluminal surface of sinusoidal endothelial cells and in contact with stem cell factor–producing, LepR+ CAR cells13,14,50,90 and, in some cases, to megakaryocytes.103,104 In addition, a minor fraction enriched in quiescent HSCs has been reported to localize in a protective niche adjacent to nonpermeable arterioles, under the regulatory influence of neighboring Nestin-GFPhi, NG2+ mesenchymal cells (nes) and nonmyelinating Schwann cells (in yellow).32,55,65 (ii) B-cell progenitors have been suggested to sequentially migrate along different niches as they progress through maturation. Early stage pre-pro B cells are mostly found adjacent to the cell bodies of CAR cells and migrate toward IL-7–expressing CAR cells as they enter the pro B cell stage. Mature B220+IgM+ B cells, in turn, move away from both stromal cell types.105 The latest stages of B-cell maturation occur intravascularly, within sinusoids, where the dynamic behavior of immature B cells has been visualized in vivo.106 In addition, early pro B cells (B220+CD43+) have been shown to accumulate in endosteal zones and gradually decrease in frequency toward bone-distal marrow regions.68 (iii) Megakaryocytes (Mk) are found in close adjacency to the endothelial surface of sinusoidal vessel wall, which they traverse in the form of protrusions from which proplatelets are continuously shedded into the venous circulatory system.107 Red blood cell development takes place in so-called erythoblastic islands (Ei), where erythroid precursors proliferate, enucleate, and terminally differentiate into reticulocytes around a central macrophage.108 Plasma B cells are long-lived antibody-secreting cells that have been found in physical association to CAR cells, megakaryoctes, and eosinophils (eos).109-111 (iv) Although a significant fraction (30%) of early lymphoid progenitors (Lin−IL7-rα+) has been shown to lie proximal to mature, bone-lining osteoblasts,48 the vast majority of phenotypically defined CLPs are in contact with IL-7–expressing CAR cells.50 Quiescent CD4+ memory T cells are found scattered throughout the BM in contact with perisinusoidal IL-7–secreting stromal cells.112 Regulatory T cells (Tregs) have been reported to lodge in close proximity to the endosteum of trabecular bone.113
Structural organization of BM tissues. (A) BM stromal microarchitecture. The microarchitecture of the vascular system has been studied in detail in the mouse femoral cavity. A central artery (ca) penetrates the cavity through the nutrient canal and splits into ascending and descending branches, which run longitudinally, arborizing into smaller radial arteries that migrate toward endosteal regions. In the proximity of endosteum, arteries give rise to a dense plexus of arterioles that travel along the cortical bone area and eventually develop into venous vessels, of fenestrated endothelium, termed sinusoidal. Sinusoids form a labyrinth that extends inward and merges in a big central collecting sinus (cs) that drains into peripheral circulation (see also Figure 1). Thin periosteal arteries also penetrate the bone and merge with arteriolar vessels in endosteal regions, connecting BM and bone circulation. In trabecular bone areas, multiple smaller arteries enter the marrow cavities and give rise to sinusoidal networks along the endosteal surface of trabeculae. CXCL12-abundant fibroblastic reticular cells (CARc) extend throughout the entire cavity in the form of dense networks. Nestin-GFPhi NG2+ (nes) elongated cells run adjacent to arteries and arterioles and bundles of nonmyelinating Schwann cells (nmsc), which ensheath adrenergic nerves. Mature osteoblasts (ob) line endosteal surfaces and are mostly derived from reticular progenitors of osetoadipogenic potential. The emergence of adipocytes (adip) takes place gradually during aging and can be abrupt and prominent in certain pathological conditions. (B) Schematic representation of cell or developmental stage-specific niches. (i) HSC niches: a number of studies have determined that HSCs are found scattered throughout BM tissues adjoining the extraluminal surface of sinusoidal endothelial cells and in contact with stem cell factor–producing, LepR+ CAR cells13,14,50,90 and, in some cases, to megakaryocytes.103,104 In addition, a minor fraction enriched in quiescent HSCs has been reported to localize in a protective niche adjacent to nonpermeable arterioles, under the regulatory influence of neighboring Nestin-GFPhi, NG2+ mesenchymal cells (nes) and nonmyelinating Schwann cells (in yellow).32,55,65 (ii) B-cell progenitors have been suggested to sequentially migrate along different niches as they progress through maturation. Early stage pre-pro B cells are mostly found adjacent to the cell bodies of CAR cells and migrate toward IL-7–expressing CAR cells as they enter the pro B cell stage. Mature B220+IgM+ B cells, in turn, move away from both stromal cell types.105 The latest stages of B-cell maturation occur intravascularly, within sinusoids, where the dynamic behavior of immature B cells has been visualized in vivo.106 In addition, early pro B cells (B220+CD43+) have been shown to accumulate in endosteal zones and gradually decrease in frequency toward bone-distal marrow regions.68 (iii) Megakaryocytes (Mk) are found in close adjacency to the endothelial surface of sinusoidal vessel wall, which they traverse in the form of protrusions from which proplatelets are continuously shedded into the venous circulatory system.107 Red blood cell development takes place in so-called erythoblastic islands (Ei), where erythroid precursors proliferate, enucleate, and terminally differentiate into reticulocytes around a central macrophage.108 Plasma B cells are long-lived antibody-secreting cells that have been found in physical association to CAR cells, megakaryoctes, and eosinophils (eos).109-111 (iv) Although a significant fraction (30%) of early lymphoid progenitors (Lin−IL7-rα+) has been shown to lie proximal to mature, bone-lining osteoblasts,48 the vast majority of phenotypically defined CLPs are in contact with IL-7–expressing CAR cells.50 Quiescent CD4+ memory T cells are found scattered throughout the BM in contact with perisinusoidal IL-7–secreting stromal cells.112 Regulatory T cells (Tregs) have been reported to lodge in close proximity to the endosteum of trabecular bone.113
Nevertheless, for the most part, available data on BM subcompartments remain fragmentary, and a broader picture integrating comprehensive spatial patterns and number of niches in an organ-wide context is lacking. In fact, only a limited number of studies have analyzed cell subset localization at a global scale and thus explored the hypothesis that hematopoietic cells distribute in a nonarbitrary fashion in accordance to a defined zonation of the BM landscape. In early work, Lord et al employed functional assays to define the frequency of primitive progenitor cells in regions of tissue that were microdissected along the longitudinal axis of the diaphysis of femoral cavities. These studies revealed that progenitor cells endowed with hematopoietic colony-forming capacity were enriched in the proximity of endosteal zones.67 Using quantitative imaging of entire BM sections, we confirmed a tendency of phenotypically defined progenitors to spatially distribute along gradients, accumulating in bone-proximal regions and gradually decreasing in frequency toward central areas.13 Moreover, early lymphoid progenitor IL-7 receptor–expressing cells have been observed to preferentially distribute toward the proximity of endosteal surfaces and functionally depend on osteoblast-derived signals.48,68 Collectively, these data point to the possibility that progression downstream of the hematopoietic hierarchy could correlate with a centripetal movement away from bone surfaces toward deeper regions of the BM. However, this conceptually attractive hypothesis is challenged by 2 recent studies. First, a 2D spatial analysis of the tissue distribution adopted by CLPs suggests these progenitors rather localize in bone-distal regions, in close interaction with IL-7–expressing CAR cells50 . Second, the first global mapping of thousands of genetically labeled HSPCs within intact 3D marrow plugs using deep tissue imaging clearly uncovered their significant accumulation in central BM zones.14 Thus, whether some sort of spatial correlate of the hierarchical organization of the hematopoietic compartment can be found in its microanatomical organization in the BM remains to be defined.
Outlook and future challenges
The notion that structure and function are inextricably linked is a fundamental tenet, which applies to all scales of biological systems. Although the hematopoietic hierarchy has been hailed as the best characterized somatic stem cell–based system, an integrated model of how the system functions in the defined structural context of BM microanatomy is largely lacking. Just as the quest for the identification of HSCs has historically been the driving force for the dissection of the hematopoietic system, much of what has been learned on BM microarchitecture has been gleaned from formative studies in search of the HSC niche.69 Indeed, significant progress has been achieved. However, beyond the precise nature of HSC niches, much is still to be learned on the spatiotemporal dynamics of hematopoiesis. A comprehensive systems-based approach will be necessary to understand how the plethora of individual components assemble to form the integrated cellular wiring required for hematopoietically competent BM tissues. Key unresolved questions relate to the global distribution of oligocellular niches described so far, how they are built and dismantled, and the hierarchical and functional relationships established by their components. For their ability to generate global, high-resolution, and multidimensional data, quantitative 3D imaging techniques will be vital when addressing these questions and determining how disturbances of canonical 3D organization patterns relate to disease states in BM. This will also be of key importance in light of studies that demonstrate that functional and structural remodeling of the 3D tissue microenvironment are essential and necessary for malignant transformation and tumor progression in certain cancers, including those of the hematopoietic system.70-72 The knowledge gained will have further implications for antineoplastic therapies and regenerative medicine. The full recapitulation of the conditions of differentiation and maintenance of HSCs in vitro, and the design of ideal strategies for hematopoietic tissue engineering, will inevitably require the complementation of comprehensive molecular and cellular data with essential information on native tissue microarchitecture.
Acknowledgments
CXCL12-GFP mice were kindly provided by Takashi Nagasawa (Osaka University). The authors thank Stephan Isringhausen for the images depicted in Figure 1.
This work was supported by the Swiss National Science Foundation (31003A_159597/1), the Vontobel Foundation (Zurich, Switzerland) and the Helmut Horten Foundation (Lugano, Switzerland) (C.N.-A.); and the Swiss National Science Foundation (310030_146528/1) and the Clinical Research Priority Program “Human Hemato-Lymphatic Diseases” of the University of Zurich (M.G.M.).
Authorship
Contribution: C.N.-A. and M.G.M. wrote the manuscript and prepared the figures.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Cesar Nombela-Arrieta, Hematology, University Hospital and University of Zurich, Häldeliweg 4, CH-8044 Zurich, Switzerland; e-mail: cesar.nombelaarrieta@usz.ch.