Key Points
Bendamustine with rituximab was the preferred frontline regimen, while BTK inhibitors were preferred in second or later lines of therapy.
The presence of chromosome 17p deletion or TP53 mutation did not shorten the overall survival in BTK inhibitor– treated patients.
Abstract
Mantle cell lymphoma (MCL) is considered incurable with the available chemoimmunotherapy approaches, and therefore, newer effective targeted therapies such as Bruton tyrosine kinase (BTK) inhibitors are increasingly used in MCL as chronic suppressive therapy, especially in the elderly. We aimed to describe the treatment patterns in MCL at different lines of therapy with a focus on BTK inhibitor use and compare outcomes with known prognostic factors using a nationwide Flatiron Health electronic health record–derived de-identified database. We analyzed patient-level data from the period of 2011 to 2021. In this study of 4336 patients with MCL, we found that bendamustine plus rituximab chemotherapy was the most commonly used frontline regimen (42%). Maintenance rituximab or consolidative autologous stem cell transplant (ASCT) was administered to 31% of all patients. Also, for patients who received ASCT as consolidation therapy, only 34% subsequently received rituximab maintenance. BTK inhibitors were the most preferred agents in second or later lines of therapy (n = 933, 57%), followed by bortezomib, lenalidomide, and venetoclax, respectively. Among patients treated with BTK inhibitors, the median real-world overall survival (rwOS) was 35 months (95% confidence interval [CI], 27-50), 24 months (95% CI, 22-30), and 18 months (95% CI, 14-21) for first line, second line, and third or later line of therapy, respectively. Patients with a deletion 17p/TP53 mutation and blastoid variant MCL had poor outcomes; however, BTK inhibitors appeared to mitigate the negative influence of del17p/TP53-mutated MCL with a hazard ratio of 1.17 (95% CI, 0.88-1.55) on multivariable analysis.
Introduction
Mantle cell lymphoma (MCL) is a unique subtype of non-Hodgkin lymphoma, accounting for 5% to 10% of all non-Hodgkin lymphomas.1,2 Its clinical course is heterogeneous, and outcomes are dependent on various clinical and biological prognostic factors and treatment approaches. Currently, induction chemoimmunotherapy with or without consolidative autologous stem cell transplantation is the standard of care treatment for newly diagnosed MCL.3-5 Despite these treatments, MCL is considered incurable. Therefore, relapse is inevitable for most, requiring multiple lines of treatment.6
Over the last decade, our understanding of the pathophysiology of MCL has improved, leading to the development and use of effective targeted therapies such as Bruton tyrosine kinase (BTK) inhibitors, B-cell lymphoma-2 inhibitors, immunomodulatory agents, and proteasome inhibitors in the relapsed or refractory setting.7-10 Despite using these targeted agents, patients with relapsed or refractory MCL have a median overall survival (OS) of approximately 3 to 4 years.11 The survival duration decreases with progressive lines of therapy. Published data of prospective clinical trials using BTK inhibitors generally report short-term outcomes for fit and eligible patients for participation in clinical trials. There is a paucity of real-world data for utilization and outcomes of BTK inhibitors in routine clinical practice outside of clinical trials. In our study, with the use of a nationwide electronic health record (EHR)-derived de-identified database, we described the treatment patterns of MCL and the utilization patterns, patient characteristics, and survival outcomes of patients with relapsed MCL treated with different BTK inhibitors either as a single agent or in combination in predominantly community-based oncology practices. We also evaluated the influence of disease-specific factors such as blastoid variant MCL and presence of deletion of chromosome 17p and/or TP53 mutation (del17p/TP53) status on real-world OS (rwOS) among patients treated with BTK inhibitors.
Methods
Data source
We used the nationwide Flatiron Health EHR-derived de-identified database. The Flatiron Health database is a longitudinal database comprising de-identified patient-level structured and unstructured data, curated via technology-enabled abstraction.12,13 Most patients in the database originate from community oncology settings; relative community/academic proportions may vary depending on the study cohort. The institutional review board approved the study (IRB-300004501) and allowed a waiver of informed consent.
Population
The inclusion criteria included patients over 18 years with a diagnosis of MCL from 1 January 2011 until February 2021. The diagnosis of MCL was captured from the pathology records in the EHR. Patients were required to have at least 2 EHR-documented visits on or after the date of diagnosis, with at least 3 months of follow-up from data cutoff in February 2021. Patients were excluded from the analysis if they had no structured activity (vital signs, laboratory measurement, documented office visit, or medication administration) within 90 days from diagnosis.
Objectives
The study’s primary objectives were to determine the treatment patterns in MCL at different lines of therapy and to describe the utilization patterns of BTK inhibitors compared with other targeted agents. The study’s secondary objective was to determine the influence of prognostic factors such as blastoid variant MCL and TP53 mutation/del17p status on rwOS from the time of diagnosis. Other secondary objectives included determining the time to next treatment (TTNT) and rwOS for BTK inhibitors stratified for lines of therapy.
Variables
The type of therapy received was the exposure variable. The targeted agents were grouped into the following categories: BTK inhibitors ± chemoimmunotherapy, bortezomib ± chemoimmunotherapy, lenalidomide ± chemoimmunotherapy, venetoclax ± chemoimmunotherapy, and venetoclax + BTK inhibitors. The chemotherapy regimens were grouped into the following categories: BR (bendamustine + rituximab), R-CHOP (rituximab in combination with cyclophosphamide, doxorubicin, vincristine, and prednisone), R-CHOP/R-DHAP (rituximab in combination with dexamethasone, cytarabine, and platinum chemotherapy), HyperCVAD (combination chemotherapy containing cyclophosphamide, vincristine, doxorubicin, methotrexate, and cytarabine) and BR/HiDAC (BR with sequential high-dose cytarabine).
Covariates included demographics, disease, and treatment-specific variables. Information regarding the demographics, disease, and treatment-specific covariates of the study population such as age at diagnosis, sex, practice type, disease subtype (blastoid, leukemic, or not otherwise specified [NOS]), date of diagnosis, Ann Arbor stage, documented del17p/TP53 mutation status, Ki-67% staining at diagnosis, oncologist-defined lines of therapy, the start date for BTK inhibitor, type of BTK inhibitor, treatment regimen, date of stem cell transplant, type of stem cell transplant, date of last follow-up, and date of death were extracted from the database. For analysis at the time of initiation of BTK inhibitors, the variables used were the most recent lactate dehydrogenase (LDH), the upper limit of normal for LDH, white blood cell (WBC) count, and Eastern Cooperative Oncology Group (ECOG) performance status within 90 days before the start date of BTK inhibitor. The TP53 mutation and/or deletion 17p status was available from the fluorescent in situ hybridization (FISH) report, and the next-generation sequencing (NGS) report was captured from the EHR. MCL international prognostic index (MIPI) original score was calculated using the formula MIPI formula = [0.03535 × age (years)] + 0.6978 (if ECOG 2-4) + [1.367 × log10(LDH/upper limit of normal)] + [0.9393 × log10(WBC)].14 The data regarding response to therapy and disease status at the time of transplantation were not available.
Statistical analysis
The primary objective was descriptive. The results were tabulated and reported as a total number of patients and percentage of patients for the respective line of therapy for targeted agents.
The time-to-event end points of rwOS and TTNT were described using the method of Kaplan-Meier plots, and a comparison of survival functions was performed with the log-rank test. For rwOS analysis, patients were followed up until the relevant event of interest (death) or censored at their last activity in the EHR, defined as either a documented office visit, vital status measurement, laboratory test, or medication administration.15 For TTNT analysis, TTNT was defined as the duration between the start of the first BTK inhibitor treatment to the start of the subsequent treatment or death as measured in months. Patients were followed until initiation of the next treatment or death (event) or censored at last confirmed activity if they were alive and did not have documented the next line of treatment. For patients retreated with BTK inhibitors, only the first instance was used for all analysis. Survival analyses for BTK inhibitors were stratified by line of therapy (first line, second line, and ≥third line), and comparisons between the presence or absence of del17p/TP53 mutation status and blastoid vs nonblastoid subtype of MCL were adjusted for covariates of the line of therapy, age, WBC count, LDH, and ECOG performance status using Cox proportional hazards model multivariable analysis. The proportional hazards assumption was assessed via a test of the Schoenfeld residuals. Statistical significance was defined as a 2-tailed P ≤ .05 for all analyses. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. The reverse Kaplan-Meier method was used to calculate the median follow-up duration.
To account for missing data of ECOG, LDH, and WBC count, we used multiple imputations by chained equation to create and analyze 50 multiply imputed datasets. Incomplete variables were imputed under fully conditional specification, using the default settings of the mice 3.13 package.16 The WBC count, LDH, and ECOG performance status were estimated in each imputed dataset separately and combined using Rubin’s rules. The combined dataset was used to compare survival analysis among del17p/TP53 mutation statuses and disease subtypes (blastoid vs nonblastoid variant) of MCL treated with BTK inhibitors. For comparison, we also performed the analysis on the dataset of complete cases. All analyses were conducted using RStudio (Version 1.4.1717 ©2009-2021 RStudio, PBC RStudio).
Results
Patient characteristics
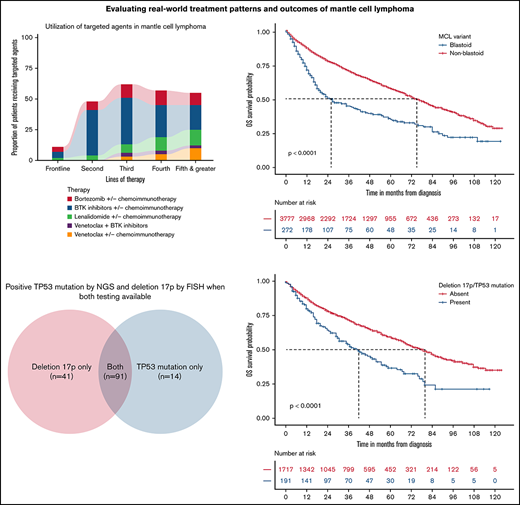
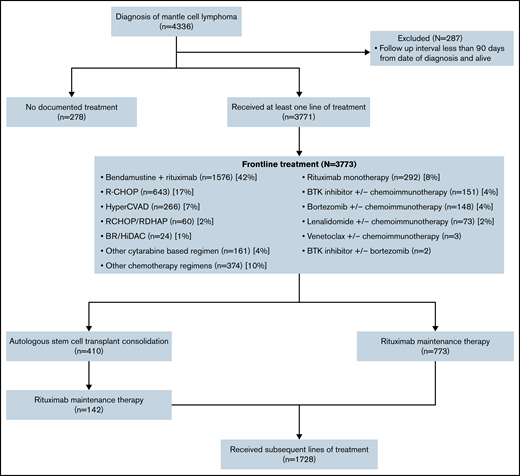
Among 4336 patients with MCL from the database, 287 patients were excluded as they had no structured activity within 90 days of their clinical eligibility date (Figure 1). Characteristics of the remaining 4049 patients are described in Table 1. Briefly, the median age at diagnosis was 69 years (interquartile range [IQR], 62-76) with a male predominance (71%). Eighty-five percent of patients received care in community-based practices. Most of the patients had advanced-stage disease (70%), with approximately 7% of patients with blastoid variant MCL and 5% with del17p/TP53 mutation. The median duration of follow-up was 51 months (IQR, 26-35). A higher proportion of patients with del17p/TP53 mutation in the blastoid variant of MCL vs other subtypes (23% vs 9%) was observed. For patients who had both deletions of chromosome 17p (del17p) by FISH and TP53 mutation by NGS data available (n = 1773), the double positivity (presence of del17p and TP53 mutation) rate was 62%. For discordant results, del17p by FISH was more prevalent and was seen in 75% of cases.
Treatment patterns in MCL
Of 4049 patients, 3771 patients received frontline treatment, whereas 278 (7%) patients did not receive any treatment documented in the database at the last follow-up. Chemo-immunotherapy with BR (n = 1576, 42%) was used most commonly, followed by R-CHOP only (n = 643, 17%) and HyperCVAD (n = 266, 7%). Cytarabine-based induction chemotherapy was administered to 41% of patients younger than 65 years of age vs only 5% of patients who were 65 or older. A total of 1183 (31%) patients received consolidation therapy after frontline induction, with the majority receiving maintenance rituximab only (n = 773, 65%), whereas the remaining underwent autologous stem cell transplant (ASCT; n = 410, 35%). Among patients who received ASCT as consolidation therapy, only one-third of patients (n = 142) subsequently received rituximab maintenance (Figure 1).
A total of 1728 (49%) patients received second or later lines of therapy. Chemo-immunotherapy remained the dominant treatment used in the second line (52%), with 139 patients (7%) receiving ASCT after salvage chemotherapy. Other cellular therapies were infrequent, with 49 patients receiving allogeneic stem cell transplants and 22 patients receiving chimeric antigen receptor T-cell therapy in the third or later lines of therapy.
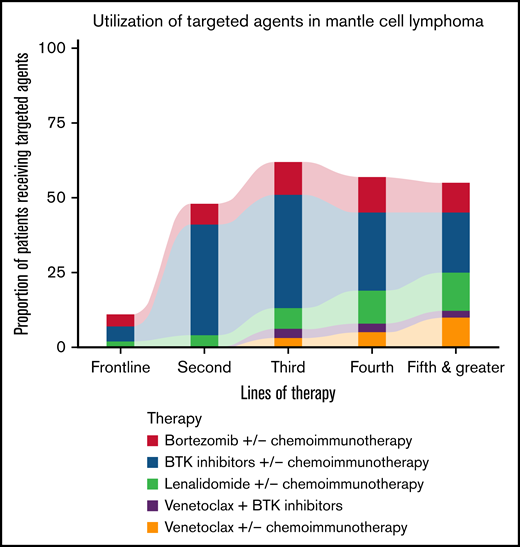
The use of BTK inhibitors, bortezomib, lenalidomide, and venetoclax as monotherapy or in combination with chemoimmunotherapy for different lines of therapy is described in Table 2. Targeted agents, either used alone or combined with chemoimmunotherapy, represent approximately 10% of all treatment regimens used in the frontline setting, as illustrated in Figure 2. For subsequent lines of treatment, there was a predominant use of BTK inhibitors followed by bortezomib and lenalidomide. A combination of different targeted agents was not routinely used and represented less than 1% for all lines of therapy.
Proportion of patients receiving targeted agents for different lines of therapy.
Proportion of patients receiving targeted agents for different lines of therapy.
Utilization patterns of BTK inhibitors in MCL
BTK inhibitors were the most frequently used targeted agents in frontline and relapsed MCL primarily as monotherapy (Figure 2). Of the 1614 patients who received a second or later line of therapy after December 2013 (US Food and Drug Administration approval for first BTK inhibitor ibrutinib), 933 patients (57%) received a BTK inhibitor at least once. Among BTK inhibitors, most patients received ibrutinib (78%), followed by acalabrutinib (20%) and zanubrutinib (2%). There were 105 patients who received more than 1 type of BTK inhibitor, of which 89 patients switched from ibrutinib to acalabrutinib, 11 patients switched from ibrutinib to zanubrutinib, and 5 patients switched from acalabrutinib to zanubrutinib. A total of 35 patients retreated with the same BTK inhibitor at later lines of therapy in combination with chemotherapy. For combination therapy with chemotherapy, no particular pattern could be observed. Among patients treated with BTK inhibitors in the frontline setting as monotherapy, 85% were 65 or older, with 25% with ECOG 2 or higher.
Outcomes for deletion 17p/TP53 mutated and blastoid variant MCL
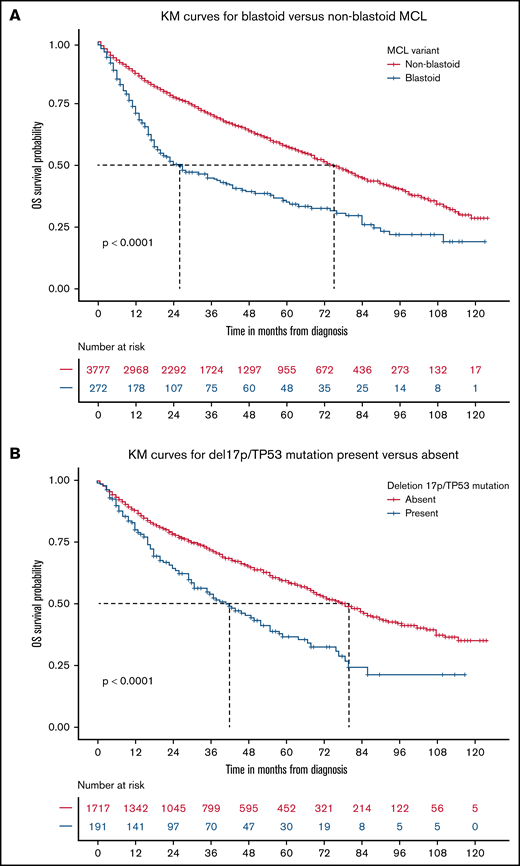
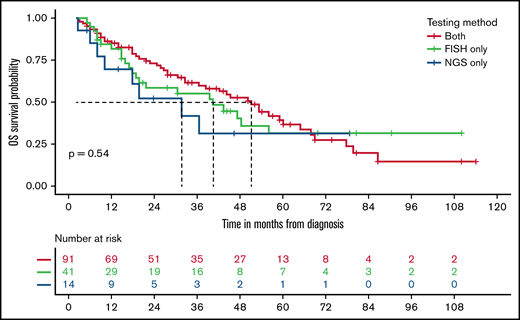
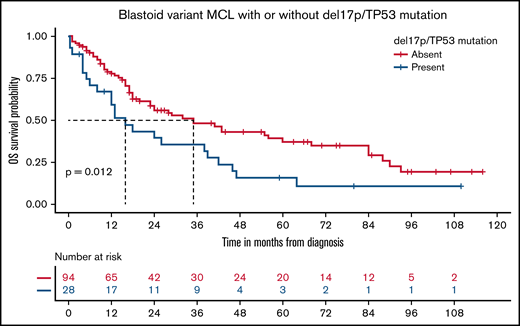
The blastoid variant of MCL was rare and noted in only 7% of patients. The rwOS was shorter for blastoid variant MCL compared with other subtypes, with a median rwOS of 26 months (95% CI, 20-41) vs 75 months (95% CI, 72-80; P < .0001; Figure 3A). Similarly, the presence of del17p/TP53 mutation occurred in 5% of patients, which resulted in an inferior rwOS of 42 months (95% CI, 31-55) vs 80 months (95% CI, 72-86) in those without such abnormalities (P < .0001; Figure 3B). There was no difference in outcomes with del17p/TP53 mutated MCL between those with del17p on FISH analysis compared with the presence of TP53 mutation by NGS only (Figure 4). Also, the presence of del17p/TP53 mutation and blastoid variant MCL had a very short median rwOS of 16 months compared with 35 months for blastoid variant MCL without del17p/TP53 mutation (P = .01; Figure 5).
Kaplan-Meier plots for rwOS for blastoid variant MCL with and without concurrent del17p/TP53 mutation.
Kaplan-Meier plots for rwOS for blastoid variant MCL with and without concurrent del17p/TP53 mutation.
Outcomes with BTK inhibitors in MCL
BTK inhibitors were the most common targeted agent used to treat MCL. Baseline characteristics of patients who received ibrutinib, acalabrutinib, or zanubrutinib and characteristics at the time of BTK initiation are described in Table 3. The median age of patients receiving BTK inhibitors at the time of starting treatment was 72 years (IQR, 64-78), with a median use as second line of therapy with 114 patients receiving prior ASCT. Among BTK inhibitors, ibrutinib was used in 843 patients, followed by acalabrutinib in 211 patients and zanubrutinib in 22 patients. The median time to BTK initiation from the date of diagnosis was 19 months (IQR, 6-38). When analyzing the entire cohort irrespective of lines of therapy, the median TTNT was 13 months (95% CI, 11-15), with a median rwOS from initiation of BTK inhibitors of 23 months (95% CI, 21-27) after a median follow-up of 35 months (IQR, 15-59). On stratifying based on prior lines of therapy, the median TTNT for BTK inhibitors used as frontline therapy, second-line therapy, and third or later lines of therapy was 17 months (95% CI, 13-24), 13 months (95% CI, 11-16), and 9 months (95% CI, 8-12), respectively. The rwOS for frontline therapy, second-line therapy, and third or later lines of therapy was 35 months (95% CI, 27-50), 24 months (95% CI, 22-30), and 18 months (95% CI, 14-21), respectively.
For comparing the rwOS among del17p/TP53 mutational statuses and disease subtypes of MCL treated with BTK inhibitors, we adjusted for known covariates of line of therapy, age, ECOG, LDH, and WBC count. The missing values across 7 variables varied between 0% and 44%. Therefore, we analyzed imputed datasets and complete cases. In the imputed dataset, blastoid variant MCL predicted inferior rwOS with an HR of 1.56 (95% CI, 1.21-2.02), whereas the negative influence of del17p/TP53 mutation status was not present on treatment with BTK inhibitors with an HR of 1.17 (95% CI, 0.88-1.55). In the complete case analysis of 232 of 1076 patients, such findings were not seen, and blastoid variant MCL and the presence of del17p/TP53 mutation did not confer an inferior rwOS.
Discussion
Over the last decade, our understanding of the pathophysiology of MCL has improved, and there have been advances in the available treatment options for MCL. In addition to the development of alternative induction chemotherapy regimens, various newer targeted therapies have improved outcomes in MCL. However, relapses occur frequently, and survival decreases with subsequent lines of therapy. With MCL representing only 5% to 10% of all non-Hodgkin lymphomas, real-world observational studies like ours complement clinical trials to make meaningful observations regarding treatment patterns and outcomes of treatment regimens.
To our knowledge, this is the largest real-world study evaluating treatment patterns and outcomes of patients with MCL predominantly treated at community-based practices. The median age in our study was 69 years, with male predominance and advanced stage at diagnosis, which is representative of the US population.17 The presence of deletion of chromosome 17p on FISH or TP53 mutation on NGS testing was seen in 5% of patients with available data (n = 1908), with a double positivity rate of 62% between FISH for deletion 17p and NGS for TP53 mutation. The incidence is lower than that observed in other prior smaller studies (16% vs 5%); however, with a large sample size (176 vs 1908), we believe our estimates are closer to the true incidence of the presence of del17p/TP53 mutation in MCL.18 The incidence of blastoid MCL in our study was 7%, which is similar to prior studies.19,20
BR chemotherapy (42%) was the most common regimen used in the frontline setting, followed by R-CHOP (17%) and HyperCVAD (7%). Prior studies showed that the addition of cytarabine to the induction regimen improved outcomes.19,21,22 However, only 41% of patients younger than 65 received cytarabine-based induction chemotherapy in our study. This lower use of cytarabine-based induction therapy could be attributed to factors not captured in our study, such as physicians’ assessment of patients' fitness and concurrent comorbidities. Consolidation therapy with either rituximab or autologous stem cell transplant was administered to only one-third of all patients. Also, for patients who received autologous stem cell transplantation as consolidation therapy, only one-third of patients subsequently received rituximab maintenance. With a well-designed phase 3 study showing an improvement in OS with maintenance rituximab after ASCT, we anticipate that more patients will receive rituximab maintenance in the future.3
BTK inhibitors were the most used targeted therapies in the relapsed setting and represented 57% of all therapies received. Even in the frontline setting, BTK inhibitors, predominantly as monotherapy, were preferred over lenalidomide or bortezomib. With multiple ongoing clinical trials incorporating BTK inhibitors in the frontline setting (#NCT04566887, #NCT03282396, #NCT04002297), the use of BTK inhibitors in the frontline setting has also percolated into routine clinical practice for the elderly and unfit patients.
In terms of outcomes for MCL, blastoid variant MCL and the presence of deletion 17p/TP53 mutation confer a poor outcome. Both together are associated with an extremely short median rwOS of 16 months. On evaluating the outcomes of all 1076 patients treated with BTK inhibitors, the rwOS in our study was 23 months (95% CI, 21-27), which is numerically lower than the median OS of 30.3 months observed in a phase 3 clinical trial of 139 patients treated with ibrutinib in relapsed MCL with a median of 2 prior lines of therapy.23 In a more extensive study of 370 patients pooled from 3 open-label studies of ibrutinib in relapsed MCL, the median OS observed was similar to that seen in our study (25 vs 23 months), suggestive of concordance of findings with the increase in the number of patients.24 Although blastoid variant MCL continues to be a negative prognostic indicator on treatment with BTK inhibitors, the negative influence of del17p/TP53 mutation was negated, which has been seen in studies with chromosome 17p deleted chronic lymphocytic leukemia treated with BTK inhibitors.25
Our study has a few limitations. The data are abstracted from the EHR of academic and private oncology practices in the United States. Once these data are abstracted and de-identified, re-evaluation of the medical record for additional data cannot be conducted. Second, data such as WBC count, LDH value, and ECOG performance status were dependent on practice patterns for requesting diagnostic testing and documentation practices by physicians in the medical records and were highly variable, leading to missing data. Nearly half of the patients had their data missing for at least 1 of the known prognostic factors such as LDH, Ki-67, and deletion/TP53 mutations status, which might lead to a selection bias. This limitation, although significant, is challenging to overcome in retrospective studies. Our study used multiple imputations by chained equation to impute missing data. Methodologists currently regard multiple imputations as a state-of-the-art technique because it improves accuracy and statistical power relative to other missing data techniques, including complete case analysis.16 Similarly, obtaining accurate records on the mortality of patients in real-world studies is difficult and often inaccurate. The database uses a composite of structured and unstructured EHR-derived data, obituary data, and the social security death index to obtain information on mortality which has been shown to have a high sensitivity, specificity, positive predictive value, and negative predictive value compared with the national death index.26 Last, we did not have data on disease progression or tumor response. Instead, TTNT was used as a proxy for progression with an event defined as advancing the line of therapy or death. Lack of data on the reason for discontinuation of therapy limited our evaluation of the efficacy and toxicities of therapies. However, this limitation should have minimal influence on robust clinical end points such as OS.
In conclusion, in this real-world cohort of patients with MCL predominantly treated at community-based practices, we demonstrate that chemoimmunotherapy with BR was the most preferred frontline regimen, with only a fraction of patients receiving either maintenance rituximab, or consolidative ASCT. Among targeted agents, BTK inhibitors were preferred and used in 57% of patients who relapsed after frontline therapy. The deletion 17p/TP53 mutation and blastoid variant MCL were associated with shorter rwOS. The lack of negative influence of del17/TP53 mutation status on rwOS with BTK inhibitors was an interesting observation in our study, which will need to be validated in prospective studies. With longer OS with BTK inhibitors in the frontline setting (vs subsequent lines) and without the negative influence of del17p/TP53 mutations, our study provides preliminary evidence supporting the use of BTK inhibitors in frail patients who are unable to tolerate intensive frontline chemoimmunotherapy.
Authorship
Contribution: M.N. designed research, collected data, analyzed and interpreted data, and performed statistical analysis; and all authors wrote the manuscript.
Conflict-of-interest disclosure: M.N. has institutional research funding from Gilead/Forty seven, TG Therapeutics, Roche/Genentech, and EUSA Pharmaceuticals. A.M. has a consulting or advisory role in Aileron Therapeutics, Bristol Myers Squibb, Seattle Genetics, Kite Pharma, Carevive, AstraZeneca, Pharmacyclics, TG therapeutics, Incyte, Kyowa Kirin International, Rigel, Novartis, Daiichi Sankyo/Lilly. Speakers Bureau for AstraZeneca, Kite Pharma, Gilead Sciences, Seattle Genetics, Incyte, Beigene, and Epizyme; research funding from Innate Pharma, TG Therapeutics, Affimed Therapeutics, ADC Therapeutics, and Rhizen Pharmaceuticals; and institutional research funding from Incyte, Roche/Genentech, Merck, Bristol Myers Squibb, Juno Therapeutics, Gilead Sciences, Forty Seven, Takeda, Astex Pharmaceuticals, Pharmacyclics, Epizyme, Aileron Therapeutics, Carevive, Oncotartis, ADC therapeutics, and Seattle Genetics. S.G. has institutional research funding from Carevive, Sanofi. and Janssen. The remaining authors declare no competing financial interests.
Correspondence: Mayur Narkhede, Division of Hematology and Oncology, Department of Medicine, O’Neal Comprehensive Cancer Center, University of Alabama at Birmingham, Birmingham 35223, AL; e-mail: msnarkhede@uabmc.edu.
References
Author notes
The data that support the findings of this study have been originated by Flatiron Health, Inc. These de-identified data may be made available upon request and are subject to a license agreement with Flatiron Health; interested researchers should contact dataaccess@flatiron.com to determine licensing terms.