Abstract
COVID-19–related acute illness is associated with an increased risk of venous thromboembolism (VTE).
These evidence-based guidelines of the American Society of Hematology (ASH) are intended to support patients, clinicians, and other health care professionals in decisions about the use of anticoagulation for thromboprophylaxis in patients with COVID-19 who do not have confirmed or suspected VTE.
ASH formed a multidisciplinary guideline panel, including 3 patient representatives, and applied strategies to minimize potential bias from conflicts of interest. The McMaster University GRADE Centre supported the guideline development process, including performing systematic evidence reviews (up to March 2021). The panel prioritized clinical questions and outcomes according to their importance for clinicians and patients. The panel used the grading of recommendations assessment, development, and evaluation (GRADE) approach to assess evidence and make recommendations, which were subject to public comment.
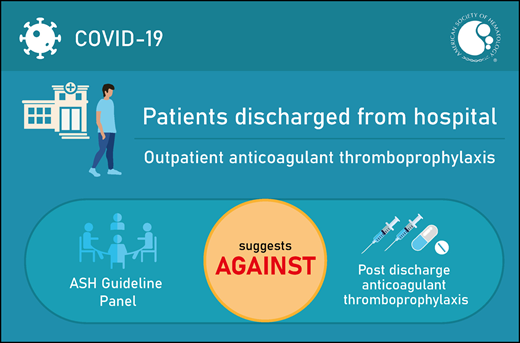
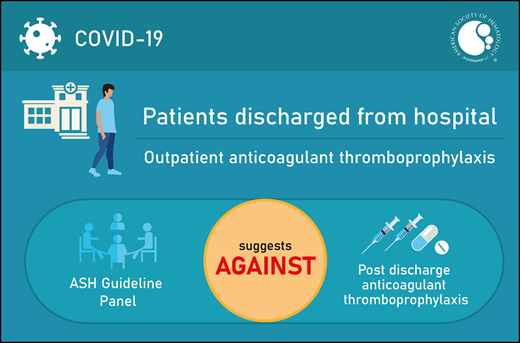
The panel agreed on 1 additional recommendation. The panel issued a conditional recommendation against the use of outpatient anticoagulant prophylaxis in patients with COVID-19 who are discharged from the hospital and who do not have suspected or confirmed VTE or another indication for anticoagulation.
This recommendation was based on very low certainty in the evidence, underscoring the need for high-quality randomized controlled trials assessing the role of postdischarge thromboprophylaxis. Other key research priorities include better evidence on assessing risk of thrombosis and bleeding outcomes in patients with COVID-19 after hospital discharge.
Summary of recommendations
Recommendation 3
The ASH guideline panel suggests that outpatient anticoagulant thromboprophylaxis not be used for patients with COVID-19 who are being discharged from the hospital and do not have suspected or confirmed venous thromboembolism (VTE) or another indication for anticoagulation (conditional recommendation based on very low certainty in the evidence about effects ⊕◯◯◯).
Remarks:
An individualized assessment of the patient’s risk of thrombosis and bleeding and shared decision making are important when deciding on whether to use postdischarge thromboprophylaxis. Prospectively validated risk assessment models to estimate thrombotic and bleeding risk in COVID-19 patients after hospital discharge are not available.
The panel acknowledged that postdischarge thromboprophylaxis may be reasonable for patients judged to be at high risk of thrombosis and low risk of bleeding.
Background
Hospitalization for acute medical illness is a common risk factor for venous thromboembolism (VTE), with most such events occurring after hospital discharge.1,2 Meanwhile, there is a high incidence of thrombotic complications in patients hospitalized for COVID-19–related acute illness or critical illness.3,4 However, there are limited reports regarding rates of VTE after discharge for patients who have been hospitalized for COVID-19.5,6 Thus, there has been much interest in establishing whether postdischarge pharmacological thromboprophylaxis in this population is warranted.
These guidelines are based on systematic reviews of evidence conducted under the direction of the McMaster University GRADE (grading of recommendations, assessment, development and evaluations) Centre with international collaborators. This is an update of the previous American Society of Hematology (ASH) guideline published in February 20217 and focuses on the role of thromboprophylaxis for patients discharged from the hospital after COVID-19. The panel followed best practice for guideline development recommended by the Institute of Medicine and the Guidelines International Network (GIN).8-10 The panel used the GRADE approach11-17 to assess the certainty of the evidence and formulate recommendations. The recommendation is listed in Table 1.
Values and preferences
The guideline panel identified all-cause mortality, pulmonary embolism (PE), deep vein thrombosis (DVT), and major bleeding as critical outcomes and placed a high value on avoiding these outcomes with the interventions assessed.
Panel members noted that there was possible uncertainty and variability in the relative value that patients place on avoiding major bleeding events compared with reducing thrombotic events.
Explanations and other considerations
Please refer to the original ASH guidelines on the use of thromboprophylaxis for patients with COVID-19.7
Interpretation of strong and conditional recommendations
Please refer to the original ASH guidelines on the use of thromboprophylaxis for patients with COVID-19.7
Introduction
Aims of these guidelines and specific objectives
Description of the health problem
The COVID-19 pandemic has had a significant public health impact. As of 21 September 2021, over 229 million cases and 4.5 million deaths have been attributed to COVID-19–related illness globally.19 It is estimated that 5% to 20% of infected patients require hospital admission, of whom 5% to 15% may develop critical illness requiring intensive care support.20-22
Thrombosis has emerged as an important complication of patients hospitalized with COVID-19–related acute or critical illness, with VTE occurring in up to 8% and 23% of such patients, respectively.4 This result aligns with the recognition that patients hospitalized for other acute medical illnesses, such as pneumonia, stroke, and heart failure, are at increased risk of VTE. Predictors of VTE in such medically ill inpatients include reduced mobility, advanced age, active cancer, and prior VTE.23,24 Previously published ASH guidelines provided focused recommendations for the prevention of VTE in hospitalized patients with COVID-19, and in non-COVID-19 medical inpatients.7,25,26
In non–COVID-19 medical inpatients, most hospitalization-related thrombotic events occur after discharge; heightened VTE risk may extend until at least 1 month after discharge.1,27 Several risk assessment models (RAMs) in patients without COVID-19 have been developed to assess the risk of VTE after hospitalization.24,28,29 However, randomized trials of postdischarge prophylaxis in patients without COVID-19 have not shown a substantial absolute benefit in VTE reduction, and the 2018 ASH guidelines recommended against postdischarge prophylaxis in medically ill patients who did not have COVID-19, owing to a low absolute benefit offset by an increase in bleeding risk.26,30 -32
There is a belief that patients with COVID-19 may have a higher risk of VTE after discharge than patients without COVID-19. However, available COVID-19–specific estimates do not bear this out. Although there remains considerable uncertainty, published estimates of postdischarge VTE in COVID-19 patients generally range from 0.5% to 1.5%,5,6,33 comparable to the baseline risk of postdischarge VTE in the non-COVID-19 population. Several clinical risk factors have been found to be independently associated with postdischarge VTE after COVID-19, including advanced age, cardiovascular disease, chronic kidney disease, and intensive care unit (ICU) admission.5 However, there are no RAMs that have been specifically derived and prospectively validated in patients with COVID-19 thus far, although non-COVID RAMs have been externally validated in hospitalized patients with COVID-19, and the COVID-thromboembolism score was derived specifically from patients with COVID-19 with concomitant malignancy.34-36 Moreover, although the recent CORE-19 registry demonstrated a reduction in the composite outcome of VTE, arterial thromboembolism (ATE), and all-cause mortality with postdischarge anticoagulation, there remains a paucity of high-quality prospective and randomized data. As such, the relative risks and benefits of postdischarge thromboprophylaxis remain uncertain in this population. In this living guideline update, the role of postdischarge thromboprophylaxis after hospitalization for COVID-19 is addressed.
Description of the target populations
The target population included patients discharged from the hospital after admission for COVID-19–related illness.
Methods
This new guideline recommendation on the use of prophylactic-intensity anticoagulation for patients being discharged after admission for COVID-19–related illness was developed in the living phase of the ASH 2021 Living Guidelines on the Use of Anticoagulation for Thromboprophylaxis for Patients with COVID-19. The ASH guideline panel added this question as a priority in January 2021 and generated recommendation 3 on 14 April 2021 before asking for public comments.
We followed the same methods as published in the initial guideline,7 with the following important updates and differences for the recommendation reported herein:
Organization, panel composition, planning, and coordination: with 1 exception, we retained the same panel members; no conflicts of interest emerged that would require exclusion of panel members.
Guideline funding and management of conflicts of interest: Supplement 4 provides updated Participant Information Forms for all panel members, detailing financial and nonfinancial interests, as well as the ASH conflict of interest policies agreed to by each individual. Supplement 5 provides the updated complete Participant Information Forms of researchers on the systematic review team who contributed to these guidelines.
Formulating specific clinical questions and determining outcomes of interest: this update of the guidelines focuses on 1 question: for patients with COVID-19 who are being discharged from the hospital and who do not have suspected or confirmed VTE or another indication for anticoagulation, should we use prophylactic-intensity direct oral anticoagulants, low-molecular-weight heparin, unfractionated heparin, or fondaparinux vs no anticoagulation?
We originally intended to include antiplatelet agents in the question, but we ultimately chose not to because of the lack of published evidence in COVID-19 patients. However, antiplatelet agents may be included in living updates.
Evidence review and development of recommendations
An evidence-to-decision framework was created for recommendation 3 (see “Recommendations”), by using the same methods as the initial guideline.6 The systematic review to identify comparative anticoagulation studies for the entire guideline was updated until 5 March 2021. The initial guideline’s literature search strategy (Supplement 6) was modified to add search terms for antiplatelet agents for this guideline question. The protocol (Supplement 9) was modified to focus on inclusion of only randomized controlled trials for other previously published recommendations. Baseline risk estimates for outcomes of patients with COVID-19 who were discharged from the hospital were based on studies identified using the same overall systematic review as reported in the initial guideline, updated until 24 March 2021.
Document review
The draft recommendation was reviewed by all members of the panel and made available online from 8 to 19 July 2021 for external review by stakeholders, including allied organizations, other medical professionals, patients, and the public. As part of the public comment, there were 143 views; 2 individuals or organizations submitted responses that did not require changes to the document. On 10 August 2021, the ASH Guideline Oversight Subcommittee and the ASH Committee on Quality assured that the defined guideline development process was followed, and on 12 August 2021, the officers of the ASH Executive Committee approved submission of the updated guideline manuscript for publication under the imprimatur of ASH. The updated guideline was then subjected to peer review by Blood Advances.
How to use these guidelines
Recommendations
Patients being discharged from the hospital after admission for COVID-19
Should prophylactic-intensity direct oral anticoagulants, low-molecular-weight heparin, unfractionated heparin, or fondaparinux vs no anticoagulation be used for postdischarge thromboprophylaxis for patients with COVID-19 who are discharged from the hospital and who do not have suspected or confirmed VTE or another indication for anticoagulation?
Recommendation 3
The ASH guideline panel suggests that outpatient anticoagulant thromboprophylaxis not be used for patients with COVID-19 who are discharged from the hospital and do not have suspected or confirmed VTE or another indication for anticoagulation (conditional recommendation based on very low certainty in the evidence about effects ⊕◯◯◯).
Remarks:
An individualized assessment of the patient’s risk of thrombosis and bleeding and shared decision making are important when deciding on whether to use postdischarge thromboprophylaxis. Prospectively validated risk assessment models to estimate thrombotic and bleeding risk of patients with COVID-19 after hospital discharge are not available.
The panel acknowledged that postdischarge thromboprophylaxis may be reasonable for patients judged to be at high risk of thrombosis and low risk of bleeding.
Summary of the evidence.
We rated the certainty in the evidence as low for the outcome of major bleeding in patients without COVID-19, owing to very serious indirectness, and as very low for all other outcomes, mainly owing to (very) serious risk of bias (see evidence profile and evidence to decision [EtD] framework online at https://guidelines.ash.gradepro.org/profile/uEXUjtWgVAQ). We found no systematic reviews that addressed this question. Three observational studies provided evidence related to the question. Supplement 10 presents the characteristics of the included studies.
One prospective registry study reported the effect of postdischarge prophylactic-intensity anticoagulation on the outcomes of any VTE or ATE, mortality, and bleeding5 ; 1 retrospective cohort study on the outcome of any VTE or ATE38 ; and 1 matched case-control study on the outcome of readmission.39
Benefits.
The evidence was very uncertain for all of the considered outcomes leading to the following interpretation. Postdischarge prophylactic-intensity anticoagulation may reduce the risk of mortality (odds ratio [OR] 0.55; 95% confidence interval [CI], 0.37-0.83), which corresponds to 5 fewer (from 7 fewer to 2 fewer) deaths per 1000 patients; very low certainty. Postdischarge prophylactic-intensity anticoagulation may reduce the risk of PE (OR, 0.76; 95% CI, 0.46-1.25), which corresponds to 1 fewer (from 3 fewer to 1 more) PEs per 1000 patients; very low certainty. Postdischarge prophylactic-intensity anticoagulation may reduce the risk of VTE (OR, 0.76; 95% CI, 0.46-1.25), which corresponds to 4 fewer (from 9 fewer to 4 more) VTEs per 1000 patients; very low certainty. Postdischarge prophylactic-intensity anticoagulation may reduce the risk of readmission (OR, 0.92; 95% CI, 0.41-2.05), which corresponds to 5 fewer (from 35 fewer to 57 more) readmissions per 1000 patients; very low certainty. No benefit or harm was observed for the outcomes of DVT, ischemic stroke, and ST-elevation myocardial infarction in terms of absolute effect, but there was very low certainty in the evidence.
Harms and burden.
Indirect evidence from acutely ill patients who do not have COVID-19 indicated that postdischarge prophylactic-intensity anticoagulation after hospitalization may increase the risk of major bleeding (relative risk, 2.09; 95% CI, 1.33-3.27), but the evidence is uncertain; this corresponds to 4 more (from 1 more to 9 more) major bleeding episodes per 1000 patients at lower risk of bleeding, and to 13 more (from 4 more to 27 more) major bleeding episodes per 1000 patients at higher risk of bleeding; low certainty. Direct evidence from patients acutely ill with COVID-19 indicates that postdischarge prophylactic-intensity anticoagulation may increase the risk of major bleeding but the evidence is very uncertain (OR, 1.52; 95% CI, 0.86-2.67). This result corresponds to 1 more (from 0 to 2 more) major bleeding episode per 1000 patients; very low certainty.
Other EtD criteria and considerations.
The guideline panel noted that there was possible uncertainty and variability in the relative value patients place on reducing thrombotic events compared with avoiding major bleeding events. The panel agreed that the use of postdischarge anticoagulant thromboprophylaxis would be acceptable to patients and health care providers. In addition, the panel acknowledged that patients who are not insured may have reduced access to postdischarge anticoagulant therapy, which could adversely affect health care equity. There would be a potential for increased costs for those paying out of pocket for postdischarge prophylaxis.
Conclusions for this recommendation.
The panel judged both the benefits and harms of postdischarge thromboprophylaxis to be trivial in terms of absolute effects. Although there was a trivial mortality benefit and reduction in VTE with postdischarge anticoagulant therapy, there was very low certainty in the evidence.
Meanwhile, there was less uncertainty in the potential undesirable effects of anticoagulant therapy in increasing the risk of major bleeding complications. Although there was no direct high-quality evidence available for patients with COVID-19, the panel considered that there was higher quality indirect evidence for patients without COVID-19 that indicated an increase in the risk of major bleeding when postdischarge anticoagulation was used.32,40,41
The panel judged that the major bleeding complications outweigh the potential benefits (particularly given the relatively low baseline risk of postdischarge VTE) and that, overall, the undesirable consequences outweigh the desirable consequences. On the basis of these judgments, the panel suggests that postdischarge thromboprophylaxis not be used.26 However, the panel emphasized the importance of an individualized decision for each patient based on an assessment of thrombosis and bleeding risks.
What are others saying and what is new in these guidelines?
There are multiple other guidance documents on the use of anticoagulation in patients with COVID-19. These include the 2020 CHEST COVID-19 Guidelines, Anticoagulation Forum interim clinical guidance, International Society on Thrombosis and Haemostasis (ISTH) Scientific and Standardization Committee (SSC) COVID-19 clinical guidance, National Institutes of Health (NIH) COVID-19 treatment guidelines, and American College of Cardiology (ACC) clinical guidance.42-46
Major differences between the current ASH guidelines and these other documents include use in ASH guidelines of high-quality systematic reviews and ED frameworks, which increase transparency, along with the use of marker states to estimate the relative importance to patients of key outcomes of treatment. The present ASH guideline is also unique in its “living” format, though other guidance documents may also be updated.
All 5 of these other guidance documents do not recommend routine use of postdischarge pharmacological thromboprophylaxis after hospitalization for COVID-19–related illness. However, given the lack of high-quality evidence, they all suggest that an individualized decision be made taking into account the patient’s thrombosis risk factors and bleeding risk at the time of discharge.
The CHEST guideline suggests that postdischarge thromboprophylaxis would result in net clinical benefit only if the risk of symptomatic VTE were found to be >1.8% within 35 to 42 days after release from the hospital.44 Meanwhile, the ISTH guidance document suggests that postdischarge thromboprophylaxis be considered for all patients hospitalized with COVID-19 who meet high-risk VTE criteria, including advanced age, ICU admission, cancer, prior VTE history, thrombophilia, severe immobility, elevated D-dimer, or an IMPROVE VTE score of 4 or more.42 The guidance from AC Forum, NIH, and ACC all suggest that postdischarge prophylaxis be considered in light of thrombotic risk, bleeding risk, and access to medication.
At the time of this writing, there have been no published randomized trials examining the efficacy and safety of postdischarge thromboprophylaxis in patients with COVID-19. However, there are several ongoing or recently completed clinical studies examining this question.47 These prospective studies include the MICHELLE (registered at https://clinicaltrial.gov, as NCT04662684)48 and ACTIV-4c trials (NCT04650087),49 which examine the use of prophylactic-intensity direct oral anticoagulants.
Limitations of these guidelines
The limitations of these guidelines are inherent in the low certainty in the evidence we identified for the research questions. In addition, the use of treatments other than anticoagulants for management of COVID-19–related acute and critical illness (eg, corticosteroids, anticytokine therapies, ventilatory support), the affected patient population, as well as the emergence of different viral variants has changed over the course of the pandemic. These changes may affect the baseline risk of VTE. Evidence collected earlier in the pandemic and included in our systematic reviews may not fully reflect the baseline risk of VTE or the effect of postdischarge thromboprophylaxis in the current phase of the pandemic.
Plans for updating these guidelines
These recommendations will be updated based on a living review of evolving evidence, including data from randomized trials. See the initial guideline publication for methods of living systematic reviews and recommendations, including considerations for deciding when to reassess and update recommendations.7
Updating or adapting recommendations locally
Adaptation of these guidelines will be necessary in many circumstances. These adaptations should be based on the associated EtD frameworks.15
Priorities for research
On the basis of gaps in evidence identified during the guideline development process, the panel identified the following urgent research priorities in this patient population:
Studies assessing baseline VTE risk after hospitalization in patients with COVID-19–related illness.
Randomized controlled trials comparing antithrombotic therapy (including anticoagulants and antiplatelet agents) for thromboprophylaxis after hospitalization.
Identification of predictors of thrombosis and bleeding in patients discharged after COVID-19–related illness.
Development and validation of risk assessment models for thrombosis and bleeding risk in patients discharged after COVID-19–related illness.
Studies examining the impact of nonanticoagulant interventions (eg, anticomplement therapy, corticosteroids, antiviral therapies, anticytokine therapies, antiplatelet therapies, monoclonal antibody therapy, convalescent plasma) on risk of thrombosis after hospital discharge.
Studies examining the impact of anticoagulant therapy on thrombosis and bleeding outcomes after hospitalization in patients of different racial and ethnic backgrounds.
Acknowledgments
The authors thank Rob Kunkle, Eddrika Russell, and Kendall Alexander for overall coordination of the guideline panel and Emma Cain, a member of the knowledge synthesis team, for contributions to this work.
D.R.T. was supported by a career development award from the National Institutes of Health, National Heart, Lung, and Blood Institute, grant 1K01HL135466.
Authorship
Contribution: E.K.T., R.N., A.C., R.A.M., and H.J.S. wrote the manuscript; all other authors contributed to critical revisions of the manuscript and approved the content; members of the knowledge synthesis team, R.N., R.N.A.J., Y.N.A.J., A.M.B., A.B., I.B.A., M.B., R.B.-P., R.C., M.C., L.E.C.-L., K.D., A.J.D., H.H., S.G.K., P.K., R.M., G.P.M., R.Z.M., G.M.-S., M.K.N., A.N., B.A.P., T.P., Y.Q., Y.R.B., F.S., A.S., K.S., and W.W. searched the literature, extracted data from eligible studies, analyzed the data, and prepared evidence summaries and evidence-to-decision tables; panel members A.C., E.K.T., P.A., C.B., K.D., J.D., M.T.D., D.D., D.O.G., S.R.K., F.A.K., A.I.L., I.N., A.P., M.R., K.M.S., D.S., M.S., D.R.T., K.T., R.A.M., and H.J.S. assessed the evidence, voted and made judgments within the evidence-to-decision framework, and discussed and issued the recommendations; the methods leadership team, R.N., R.B.-P., K.D., A.S., K.S., A.C., E.A.A., W.W., R.A.M., and H.J.S. developed the methods and provided guidance to the knowledge synthesis team and guideline panel; and A.C., R.A.M., and H.J.S. were the cochairs of the panel and led the panel meetings.
Conflict-of-interest disclosure: All authors were members of the guideline panel or members of the systematic review team or both. As such, they completed a disclosure of interest form, which was reviewed by ASH and is available as Supplements 4 and 5.
Correspondence: Adam Cuker, Hospital of the University of Pennsylvania, 3400 Spruce St, Philadelphia, PA 19104; e-mail: adam.cuker@pennmedicine.upenn.edu.
References
Author notes
A.C. and E.K.T. are joint first authors.
Requests for data sharing may be submitted to Adam Cuker: adam.cuker@pennmedicine.upenn.edu
The full-text version of this article contains a data supplement.