In this issue of Blood Advances, Fortmann et al1 have identified a subpopulation of proinflammatory megakaryocytes in the peripheral blood of patients with COVID-19. Specifically, the megakaryocytes contain calprotectin heterodimers (S100A8/A9) and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) protein and RNA and can transfer SARS-CoV-2 antigens to their daughter platelets. Notably, in a cohort of 218 patients, the authors found that the number of SARS-CoV-2–containing megakaryocytes strongly correlates with mortality and multiorgan injury.
Although megakaryocytes have classically been viewed as platelet progenitor cells, accumulating evidence suggests that megakaryocytes play important roles in both innate and adaptive immunity.2 Furthermore, various studies report the presence of extramedullary megakaryocytes in autopsies of patients with COVID-19.3 Moreover, multiple independent studies have used single-cell RNA-sequencing to identify discreet megakaryocyte cell populations within the bone marrow; the so-called “immune megakaryocyte” population is consistent with the megakaryocytes that were found to increase in the peripheral blood of patients with COVID-19.4
This study by Fortmann et al builds on the previous studies by further characterizing the circulating megakaryocytes in patients with COVID-19 and expands on them by determining that elevated circulating megakaryocytes correlate with poor disease outcomes. For this, the authors used publicly available single-cell RNA sequencing data sets and identified that interferon induced transmembrane protein 3 (IFTM3), interferon alfa–inducible protein 27 (IFI27), and both subunits of calprotectin, S100A8 and S100A9, transcripts were upregulated in circulating megakaryocytes. Of note, the calprotectin heterodimer was only upregulated during active infection and early recovery, and its levels returned to normal afterward. Thus, the authors used S100A8/A9 to identify a subpopulation of megakaryocytes by flow cytometry, and further characterized megakaryocytes in a cohort of 218 patients with COVID-19. Intriguingly, both the SARS-CoV-2 genome and spike protein were found in this subpopulation of megakaryocytes, mainly in the perinuclear space, whereas this was never found in megakaryocytes that lacked expression of S100A8/A9. These cells also expressed higher levels of toll-like receptor (TLR)-2 and TLR-4 and had a proinflammatory phenotype, characterized by the upregulation of an NF-kB signature and the cytokines IL-1, IL-8, and TNF. Moreover, the activated form of GPIIb/IIIa was specifically induced in the subpopulation of megakaryocytes that contained viral molecules and expressed S100A8/A9. Ultimately, the authors showed that the disease severity scores assessed at the time of blood collection or through the inpatient hospitalization directly correlated with the amount of circulating, proinflammatory megakaryocytes.
This study helps to reconcile previous reports aiming to understand the higher risk of thrombosis in patients with COVID-19. For instance, they revealed that megakaryocytes can transfer viral molecules to daughter platelets in vitro, which may explain the reported presence of SARS-CoV-2 RNA molecules platelets in patients with COVID-19.5,6 The enrichment of type I interferon signatures and S100A8/A9 in a subpopulation of megakaryocytes mirrors the observations made in platelets in COVID-19.5,7 Furthermore, they identified proteins implicated in SARS-CoV-2 infection, notably angiotensin-converting enzyme 2 (ACE2) and transmembrane protease serine 2 (TMPRSS2), on megakaryocytes. These proteins were only significantly expressed by the subpopulation of S100A8/A9-expressing megakaryocytes that contained viral molecules. These observations suggest that ACE2 and TMPRSS2 are variably expressed by megakaryocytes and may explain why they were inconsistently identified in human platelets in previously published work.8
One notable finding was the low frequency of megakaryocytes in the blood of healthy individuals. This is consistent with prior findings; pioneer investigations had estimated the number of megakaryocytes in the pulmonary artery blood to be ∼5.0 megakaryocytes/mL, whereas <0.5 megakaryocytes/mL exit the lungs with partial cytoplasmic content.9 It is thus suggested that megakaryocytes that developed in the bone marrow environment ingress the central venous blood and reach the lungs, where they are filtered and produce platelets. These investigations took place several decades before the advent of single-cell RNA sequencing approaches and therefore mainly relied on microscopic assessment of cells in the blood of individuals or animals with cardiopulmonary bypass. With the use of modern OMICS approaches, however, data must be cautiously interpreted. Indeed, platelet-leukocyte aggregates, which are reported in numerous pathological conditions including COVID-19, as well as platelet aggregates, may be mistaken for megakaryocytes by single-cell RNA sequencing or flow cytometry approaches. The discernment of circulating megakaryocytes is, therefore, not trivial.
Fortmann et al undertook significant efforts to validate their findings. Because S100A8/A9 is typically expressed by neutrophils and monocytes, they confirmed the absence of numerous leukocyte markers on peripheral megakaryocytes. Moreover, they determined megakaryocyte polyploidy, and used imaging flow cytometry to visualize the cells and exclude the presence of cell aggregates. From their calculations, they estimated that platelets contain 1000 times less RNA than leukocytes and, thereby, aggregates comprising hundreds to a few thousand platelets would be required to provide sufficient messenger RNA for droplet-based single-cell sequencing. Thus, the authors used multiple techniques to substantiate their claims. We propose that moving forward, the same standard is upheld to identify circulating megakaryocytes, including but not limited to the following: (1) identification of megakaryocyte markers and exclusion leukocyte markers by flow cytometry; (2) imaging flow cytometry and/or immunofluorescence imaging of samples; (3) exclusion of platelet aggregates by DNA or RNA analysis; and (4) analysis of ploidy by flow cytometry.
But what explains the larger number of megakaryocytes in the peripheral blood of patients with COVID-19? Perhaps an inflamed or damaged vasculature filters out megakaryocytes less efficiently, which combines with increased megakaryopoiesis, as seen in inflammation,10 or additional sites of megakaryopoiesis help explain the findings (see figure). These events can take place in various pathogenic conditions, in addition to COVID-19, and may shape megakaryocyte and, thus, platelet functions. Considering the large set of immune molecules expressed by peripheral megakaryocytes, future investigations have the potential to reveal novel roles for megakaryocytes in other inflammatory diseases.
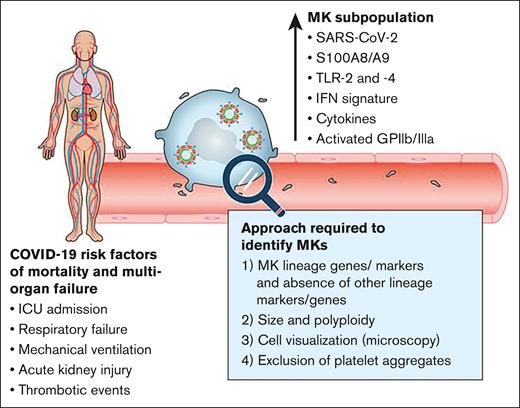
Circulating SARS-CoV-2+ megakaryocytes associate with severe viral infection in COVID-19. A subpopulation of proinflammatory megakaryocytes (MK) is identified in the peripheral blood of patients with COVID-19 with severe manifestations. These cells contain calprotectin heterodimers (S100A8/A9), present interferon response and inflammatory cytokines signatures, and are activated. SARS-CoV-2 can be found in megakaryocytes. SARS-CoV-2 protein and RNA (a whole virus is illustrated, although the presence of actual virions in circulating megakaryocytes is hypothetical and is not shown in this study). The number of SARS-CoV-2-containing megakaryocytes strongly correlates with risk factors such as mortality and multiorgan injury. A combination of experimental approaches is needed to confirm the presence of megakaryocytes. Professional illustration by ScEYEnce Studios.
Circulating SARS-CoV-2+ megakaryocytes associate with severe viral infection in COVID-19. A subpopulation of proinflammatory megakaryocytes (MK) is identified in the peripheral blood of patients with COVID-19 with severe manifestations. These cells contain calprotectin heterodimers (S100A8/A9), present interferon response and inflammatory cytokines signatures, and are activated. SARS-CoV-2 can be found in megakaryocytes. SARS-CoV-2 protein and RNA (a whole virus is illustrated, although the presence of actual virions in circulating megakaryocytes is hypothetical and is not shown in this study). The number of SARS-CoV-2-containing megakaryocytes strongly correlates with risk factors such as mortality and multiorgan injury. A combination of experimental approaches is needed to confirm the presence of megakaryocytes. Professional illustration by ScEYEnce Studios.
Conflict-of-interest disclosure: The authors declare no competing financial interests.