In PWH, CD4 nadir associates with clonal hematopoiesis, a risk factor of atherosclerosis and cancer.
Clonal hematopoiesis is less common outside of North America among PWH.
Visual Abstract
Clonal hematopoiesis of indeterminate potential (CHIP), the clonal expansion of myeloid cells with leukemogenic mutations, results in increased coronary artery disease (CAD) risk. CHIP is more prevalent among people with HIV (PWH), but the risk factors are unknown. CHIP was identified among PWH in REPRIEVE (Randomized Trial to Prevent Vascular Events in HIV) using whole-exome sequencing. Logistic regression was used to associate sociodemographic factors and HIV-specific factors with CHIP adjusting for age, sex, and smoking status. In the studied global cohort of 4486 PWH, mean age was 49.9 (standard deviation [SD], 6.4) years; 1650 (36.8%) were female; and 3418 (76.2%) were non-White. CHIP was identified in 223 of 4486 (4.97%) and in 38 of 373 (10.2%) among those aged ≥60 years. Age (odds ratio [OR], 1.07; 95% confidence interval [CI], 1.05-1.09; P < .0001) and smoking (OR, 1.37; 95% CI, 1.14-1.66; P < .001) associated with increased odds of CHIP. Globally, participants outside of North America had lower odds of CHIP including sub-Saharan Africa (OR, 0.57; 95% CI, 0.4-0.81; P = .0019), South Asia (OR, 0.45; 95% CI, 0.23-0.80; P = .01), and Latin America/Caribbean (OR, 0.56; 95% CI, 0.34-0.87; P = .014). Hispanic/Latino ethnicity (OR, 0.38; 95% CI, 0.23-0.54; P = .002) associated with significantly lower odds of CHIP. Among HIV-specific factors, CD4 nadir <50 cells/mm3 associated with a 1.9-fold (95%CI, 1.21-3.05; P = .006) increased odds of CHIP, with the effect being significantly stronger among individuals with short duration of antiretroviral therapy (ART; OR, 4.15; 95% CI, 1.51-11.1; P = .005) (Pinteraction= .0492). Among PWH at low-to-moderate CAD risk on stable ART, smoking, CD4 nadir, North American origin, and non-Hispanic ethnicity associated with increased odds of CHIP. This trial was registered at www.ClinicalTrials.gov as NCT02344290.
Introduction
People with HIV (PWH) have an increased risk of atherosclerotic cardiovascular disease (ASCVD),1 and a consistent body of evidence demonstrates a 50% to 100% increased relative risk.2-4 The excess ASCVD risk among PWH is out of proportion to the increased prevalence of recognized coronary artery disease (CAD) risk factors, including smoking, dyslipidemia, and type 2 diabetes, even among virally suppressed individuals.5 The excess risk is at least partly believed to be related to HIV’s activation of the immune system with resultant increased levels of proinflammatory proatherogenic cytokines, such as interleukin-6 (IL-6).6,7
Clonal hematopoiesis of indeterminate potential (CHIP) is a recently recognized risk factor for both hematologic malignancy and CAD.8-11 CHIP is the presence of leukemogenic somatic mutations (typically in DNMT3A, TET2, ASXL1, JAK2, and TP53) in hematopoietic stem cells with resultant clonal expansion. In addition to increased risks for hematologic malignancy and all-cause mortality, CHIP is associated with a twofold increased risk of CAD and a fourfold increased risk of early onset myocardial infarction.12 The excess ASCVD risk associated with TET2 CHIP is believed to be at least partly mediated through heightened inflammation via the NLRP3/IL-1β/IL-6 pathway.13,14 More recently, smaller studies have revealed a higher prevalence of CHIP among PWH, particularly in older age groups,15-18 which has been proposed as a predictive marker for future ASCVD events among PWH without traditional risk factors.7
The REPRIEVE (Randomized Trial to Prevent Vascular Events in HIV) trial was launched in April 2015 and is the largest clinical trial assessing an ASCVD prevention strategy among PWH, representing a unique opportunity to investigate ASCVD risk in PWH.19 REPRIEVE enrolled PWH with no known history of ASCVD and with low-to-moderate CAD risk using traditional risk factors. The trial is conducted in collaboration with the AIDS Clinical Trials Group (ACTG) network, across >100 sites in 12 countries, spanning 5 continents. Here, we report on the prevalence of CHIP in the REPRIEVE cohort, as well as the association of sociodemographic, clinical cardiovascular, and HIV-specific risk factors with CHIP prevalence.
Methods
Study participants
PWH aged 40 to 75 years without known ASCVD, on stable antiretroviral therapy (ART), not receiving statins, and with low-to-moderate estimated ASCVD risk (as calculated with the 2013 American College of Cardiology/American Heart Association Pooled Cohort Equations) were enrolled in the REPRIEVE trial as previously described.19,20 Enrollment occurred from May 2015 to February 2018, participants were blindly randomized to pitavastatin vs placebo, and though the trial was closed due to observed efficacy of the study treatment in April 2023, there will be continued follow-up until termination visits. This cross-sectional study focuses on parameters at time of enrollment in the trial, including baseline blood DNA. Participants’ countries of residence were grouped using the World Health Organization Global Burden of Disease scheme as previously described.21 The coordinating centers and sites obtained institutional review board and other applicable regulatory entity approvals. All participants were provided with study information, including discussion of risks and benefits, and signed the approved declaration of informed consent. The Massachusetts General Hospital Institutional Review Board approved this secondary data analyses.
Phenotypes (exposures)
At enrollment, participants provided clinical history and blood samples for standard laboratory investigations (supplemental Table 1). Data on demographic parameters, medical history, lifestyle behaviors, and HIV-specific parameters were collected as part of REPRIEVE, as previously described. Natal sex, race, ethnicity, and CD4 nadir were self-reported. CD4 nadir is defined as the lowest CD4 count identified for the participant, typically at the time of HIV diagnosis, and is regarded as a measure of infection severity. The trial collected information on the current ART regimen, duration of ART use and exposure to selected medications (eg, abacavir [ABC], tenofovir disoproxil fumarate [TDF], thymidine analogs [zidovudine, stavudine, and didanosine], and protease inhibitors). Regimens were classified by the presence of ≥1 potent antiretrovirals including nonnucleoside reverse transcriptase inhibitors, integrase strand transfer inhibitors, nucleoside reverse transcriptase inhibitors, and protease inhibitors.
WES
Participants recruited at accredited ACTG partner sites were consented for genetic analyses. Baseline characteristics for REPRIEVE participants recruited at ACTG sites vs non-ACTG sites are presented in supplemental Table 2. Whole-exome sequencing (WES) from whole blood samples in 4557 REPRIEVE participants was performed by the Broad Institute’s Genomics Platform using the Germline Exome v6 Plus GSA v3 Array on the NovaSeqS4 (Illumina, Inc). The Germline Exome v6 combines human WES product with an Infinium Global Screening plus Multi-Disease Array (GSA v3). The hybrid selection libraries typically meet or exceed 85% of targets at 20×, comparable to ∼55× mean coverage. The array consists of a 24-sample Infinium array with ∼245 000 fixed genome-wide markers, designed by the Broad.
CHIP genotyping (outcome)
Somatic mutations were called from WES data using Mutect2 software22 in the Terra platform (https://app.terra.bio/), as previously described.13,23 A panel-of-normal (the 1000 Genomes PON; gs://gatk-best-practices/somatic-hg38/1000g_pon.hg38.vcf.gz) and the Genome Aggregation Database (gnomAD) were used to filter germ line variants from the putative somatic mutation calls.24 Mutect2 calls were further filtered and variants were retained if (1) total depth of coverage ≥20; (2) number of reads supporting the alternate allele ≥3; (3) ≥1 read in both forward and reverse direction supporting the alternate allele; (4) variant allele fraction (VAF) ≥0.02; and (5) gnomAD allele frequency ≤0.001. To classify CHIP, pathogenic variants were queried in genes known to drive clonal hematopoiesis and myeloid malignancies.10,23,25 The detailed CHIP calling pipeline was previously reported.23
Statistical analysis
Descriptive statistics are reported using means with standard deviations (SDs) or medians with interquartile range (IQRs) for normal and non-normal distributions for continuous variables, respectively, as assessed by exploratory plotting and Shapiro-Wilk testing. Categorical variables are reported using absolute counts and relative frequencies. Comparisons between groups were performed with a t test, Wilcoxon rank sum test, or χ2 test as appropriate. Adjusted logistic regression models were used to associate presence of CHIP with sociodemographic, clinical, and HIV-specific risk factors.
For HIV-specific factors, the primary outcome variable was presence of CHIP, and the primary exposures were CD4 nadir and ART duration. Models were adjusted for age, natal sex, and smoking status. Secondary exploratory analyses were conducted associating sociodemographic risk factors, clinical ASCVD risk factors, and HIV-specific therapies with CHIP. All exploratory associations were adjusted for multiple comparisons using the Benjamini-Hochberg method with a false discovery rate of 0.05. Both adjusted and unadjusted P values and Benjamini-Hochberg thresholds for interpretation are presented. All statistical analyses were performed using R version 4.0.5 and RStudio v2021.09.1 Build 372.
Results
Study population
The main REPRIEVE study included 7769 participants, 4837 of whom were recruited from ACTG sites, and of these, 4807 provided consent for genotyping. Among those, 250 participants were excluded for missing data or nongenetics consent. WES was performed on the 4557 who consented for genetic analyses, and among these individuals, 4486 passed quality control filters and were included in this study (supplemental Figure 1). The mean age was 49.9 (SD, 6.38) years, 1650 of 4486 (36.8%) had female sex at birth, 2089 of 4486 (46.6%) were of Black or African American race, and 353 of 1878 (18.8%) of those in North America were of Hispanic or Latino ethnicity (Table 1). A total of 1878 (41.9%) were from the United States or Canada, 587 (13.1%) from Thailand, 475 (10.6%) from South Africa, 447 (10.0%) from India, 277 (6.2%) from Botswana, 231 (5.1%) from Brazil, 180 (4.0%) from Uganda, 146 (3.3%) from Peru, 140 (3.1%) from Haiti, and 125 (2.8%) from Zimbabwe. Median body mass index was 25.3 (IQR, 22.2-29.0), and 980 of 4486 (21.8%) reported current smoking, 964 of 4486 (21.5%) reported former smoking, and 2537 of 4486 (56.6%) reported never smoking. Mean total cholesterol level was 185 (SD, 36.3) mg/dL, high-density lipoprotein cholesterol level was 51.5 (16.9) mg/dL, low-density lipoprotein cholesterol level was 107 (30.9) mg/dL, and median triglycerides level was 108 (IQR, 78.0-159) mg/dL. The median 10-year estimated ASCVD Risk Score was 3.9% (IQR, 1.7%-6.6%). The mean CD4 count at entry was 621 (SD, 291) cells per mm3. Plots of the distribution of age, entry CD4 count, and nadir CD4 count are presented in supplemental Figure 2. Demographics of individuals recruited from ACTG and non-ACTG sites are available in supplemental Table 2.
CHIP prevalence
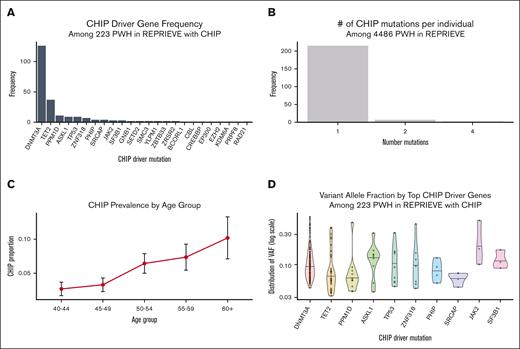
Overall, 223 of 4486 individuals (4.97%) were identified to have CHIP with a VAF ≥2%, and 87 of 4486 (1.94%) had large CHIP clones (VAF ≥ 10%). A list of all CHIP variants and VAF are noted in supplemental Table 3. CHIP prevalence increased with age group for both VAF ≥2% and VAF ≥10% (supplemental Table 4). The most common CHIP driver mutations were DNMT3A and TET2, followed by PPM1D, ASXL1, and TP53 (Figure 1A). The top genes were similar to non-HIV cohorts with the exception of the relative enrichment of PPM1D and TP53. The majority had 1 CHIP mutation (215/223; 96.4%); 7 had 2 CHIP mutations (7/223; 3.1%), and 1 individual (0.5%) had 4 mutations (Figure 1B). Consistent with prior reports, CHIP prevalence increased with advancing age (Figure 1C). VAF did not vary significantly across the top ten CHIP driver genes (Figure 1D).
Prevalence of CHIP in REPRIEVE. (A) Relative frequency of CHIP driver mutations in known CHIP genes; (B) frequency of participants carrying 1 or greater number of CHIP mutations; (C) CHIP prevalence by age; (D) VAF does not vary significantly by the top 10 CHIP driver mutations.
Prevalence of CHIP in REPRIEVE. (A) Relative frequency of CHIP driver mutations in known CHIP genes; (B) frequency of participants carrying 1 or greater number of CHIP mutations; (C) CHIP prevalence by age; (D) VAF does not vary significantly by the top 10 CHIP driver mutations.
Sociodemographic and clinical cardiovascular risk factors and CHIP prevalence
CHIP was found to be associated with select sociodemographic and clinical cardiovascular risk factors that were collected as part of REPRIEVE (Table 2). Age and cigarette smoking were demonstrated to be highly significant predictors of CHIP, and all other models were adjusted for age, male sex, and cigarette smoking status. Lipids and calculated 10-year ASCVD risk were not found to be associated with CHIP. The most significant findings were decreased risk of CHIP among individuals of Hispanic/Latino ethnicity in North America (odds ratio [OR], 0.38; 95% confidence interval [CI], 0.23-0.54; P = .0017) and lower odds of CHIP among participants outside of high income North American countries including sub-Saharan Africa (OR, 0.57; 95% CI, 0.4-0.81; P = .0019), South Asia (OR, 0.45; 95% CI, 0.23-0.80; P = .01), and Latin America/Caribbean (OR, 0.56; 95% CI, 0.34-0.87; P = .014).
HIV-specific factors and CHIP prevalence
After adjustment for age, natal sex, and smoking status, CD4 nadir was significantly associated with presence of CHIP. When compared with a reference of 350+ cells per mm3, a CD4 nadir of <50 cells per mm3 had a 90% increased odds of CHIP (OR, 1.91; 95% CI, 1.21-3.05; P = .006), and individuals with a CD4 nadir of 200 to 349 cells per mm3 had a 63% increased odds of CHIP (OR, 1.63; 95% CI, 1.06-2.55; P = .03) (Table 3). When modeled as an ordinal categorical variable, there was a linear relationship across CD4 nadir group (350+, 200-349, 50-199, and <50 cells per mm3) and odds of CHIP with a 49% increase in odds of CHIP for every decrement in nadir CD4 level (OR, 1.49; 95% CI, 1.09-2.06; P = .014). Total ART duration (<5 years, 5-10 years, and ≥10 years) was not significantly associated with presence of CHIP (Figure 2). However, a significant interaction effect (pinteraction= 0.049) was present between CD4 nadir and ART duration when the strength of the association between CD4 nadir <50 cells per mm3 and presence of CHIP (when compared with those with high CD4 nadirs [≥350 cells per mm3]) was stronger among individuals with short ART duration <5 years (OR, 4.15; 95% CI, 1.51-11.1; P = .005) than among individuals with long ART duration ≥10 years (OR, 1.26; 95% CI, 00.66-2.53; P = .50).
HIV-Specific risk factors and odds of CHIP. Forest plot of multiple regression models for odds of CHIP, adjusting for age, natal sex, and smoking status. INSTI, integrase strand transfer inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor; TDF, tenofovir disoproxil fumarate.
HIV-Specific risk factors and odds of CHIP. Forest plot of multiple regression models for odds of CHIP, adjusting for age, natal sex, and smoking status. INSTI, integrase strand transfer inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor; TDF, tenofovir disoproxil fumarate.
Entry CD4 count modeled as a standardized continuous variable was not associated with presence of CHIP after adjustment for age, natal sex, and smoking status (supplemental Table 5). A significant interaction effect was found between entry CD4 count and total ART duration <5 years (OR, 0.56; Pinteraction = .006), in which for every 1 SD increase in entry CD4 count, the odds of CHIP presence was 40% lower (OR, 0.60; 95% CI, 0.39-0.88; P = .01) if individuals had been on ART for a short duration (<5 years); compared with no significant effect of increasing entry CD4 count among those who were on ART for long durations (≥10 years) (OR, 1.15; 95% CI, 0.96-1.37; P = .14). Notably, the lowest entry CD4 count in the data set was 92 cells per mm3 as these were stable individual at low risk. To probe whether there was a relationship to HIV viral load, a supplementary analysis was performed using 3011 of 4486 (67.1%) who had HIV viral load data at study entry available. Of these individuals, 2652 of 3011 (88.1%) individuals had undetectable viral load. We did not observe a significant association between viral load and odds of CHIP (supplemental Table 6).
No specific ART regimen was significantly associated with increased CHIP presence (supplemental Table 7). Additionally, exposure or duration of exposure to specific ART medications were not associated with the presence of CHIP (supplemental Table 7).
Given prior reports that mutations in DNA-damage response (DDR) genes (ie, PPM1D, TP53, and RAD21) were enriched among individuals with exposure to chemotherapy administration, we performed secondary analyses for this CHIP subset. No HIV-specific regimens were found to be significantly associated with presence of CHIP with DDR driver genes (supplemental Table 8). Given that 1% to 3% of individuals in the study cohort were known to previously have a cancer diagnosis (supplemental Table 2), we performed additional supplementary analyses to associate presence of cancer with odds of CHIP and found no significant relationship given the rarity of cancer in this cohort. No gene-specific enhancement was seen by any cancer type.
Discussion
The REPRIEVE trial provides a unique opportunity to study CHIP among a global cohort of ART-treated PWH with low-to-moderate traditional ASCVD risk. Even in this group, a high prevalence of CHIP was still identified, with 1 in 20 affected overall (mean age, 50 years) and 1 in 10 affected among those aged >60 years. Global variation in CHIP prevalence among PWH has not previously been explored, and here, we observed that CHIP prevalence was highest in North America by 56%. Among participants in North America, those of Hispanic/Latino ethnicity had a 62% lower odds of CHIP presence. This is consistent when examining race/ethnicity differences among residents of North America in non-HIV studies.10,23 Consistent with the literature, we found strong relationships with age and smoking status for presence of CHIP and adjusted all other analyses for age, sex, and smoking status. Furthermore, we found that lower CD4 nadir at time of HIV diagnosis was associated with a higher odds of CHIP presence, in which those with the lowest CD4 nadir (<50 cells per mm3) had a nearly doubled odds of CHIP compared with those with CD4 nadirs ≥350 cells per mm3. Although no significant associations were identified with a particular drug treatment group, most participants had been exposed to numerous classes of therapies, especially as first-line therapeutic options have evolved over time. These observations inform our understanding of the factors influencing CHIP presence as well as HIV-associated adverse clinical outcomes.
First, prevalence of CHIP is relatively high among PWH in REPRIEVE, who, based on enrollment criteria, were early middle-aged, stably treated with ART, and presenting with low-to-moderate traditional ASCVD risk. Consistent with smaller geographically restricted studies, we observe an acceleration of CHIP incidence in later middle-life. This observation and others support the scientific premise that there are unique selective features among PWH that accelerate clonal proliferation later in life.15,16,18 However, this larger study found a lower CHIP prevalence than prior studies among PWH potentially due to the younger age as well as the lower prevalence of clinical comorbidities and risk factors relative of the REPRIEVE population. Interpreting these data in the context of other studies indicating an exponential rise in CHIP prevalence among PWH later in life; one possibility is that influences on clonal fitness among individuals with HIV may occur decades after HIV infection. Further longitudinal and controlled studies are needed to confirm this hypothesis.
A surprising unique observation in this study was the increased prevalence of CHIP driver mutations in DDR genes. Because DDR driver mutations has been previously associated with exposure to cancer therapies,26 ASCVD, and peripheral arterial disease,27 we sought to investigate whether the same was true of ART in REPRIEVE, but our exploratory analyses lacked sufficient power given the tremendous diversity of specific ART regimens and the numerous regimens to which each individual was exposed over the course of their diagnosis. Further investigations of ART regimen and influence on CHIP is an important future direction for study as it may influence choice of ARTs for PWH given the high rate of cardiovascular morbidity among PWH with longstanding infection.
We observed that the severity of HIV infection, as marked by CD4 nadir at time of diagnosis (and not current CD4 count), was associated with increased odds of CHIP. This finding among 4486 PWH confirms an initial similar observation among 200 PWH.28 This raises questions about whether features intrinsic to chronic HIV infection increase the odds of CHIP or accelerates clonal expansion. Furthermore, we now observe a synergistic effect of low CD4 nadir and more recent initiation of ART for increased CHIP prevalence. The residual inflammation demonstrated in virally suppressed PWH has long-term health implications29,30 and has recently been demonstrated to be affected by host tumor suppressor, innate immune, and inflammasome responses. Resistance to chronic inflammation has been demonstrated to be a critical element of clonal selection and survival advantage in CHIP. Thereby, these data lend further support to the hypothesis that the residual inflammation latent even in treated PWH may contribute to high CHIP prevalence. Although ART treatment was not randomized, these observations support a hypothesis that early HIV-related factors may have long-term inflammatory effects.31-33 CD4 nadir is directly linked to the size of the latent HIV reservoir, even in virally suppressed individuals.34 The residual inflammation demonstrated in virally suppressed PWH has long-term health implications35,29 and has recently been demonstrated to be affected by host tumor suppressor, innate immune, and inflammasome responses.30 Resistance to chronic inflammation has been demonstrated to be a critical element of clonal selection and survival advantage in CHIP.36 Thereby, these data lend further support to the hypothesis that the residual inflammation latent even in treated PWH may contribute to high CHIP prevalence. Although residual inflammation due to HIV infection leading to clonal selection is perhaps more plausible, an alternate mechanism could be that ART treatment and suppression of viral replication might decrease CHIP clone size; future studies with serial CHIP acquisition will be necessary to assess this.
This study findings must be interpreted in the context of its limitations. First, the REPRIEVE cohort consists of PWH with low-to-moderate risk from traditional cardiovascular risk factors, thus perhaps, limiting generalizability. However, given the global study population, this study provides important data on diverse populations of PWH. Second, given the younger age of the cohort than prior studies of CHIP in PWH, the prevalence of CHIP in REPRIEVE is lower, thus limiting gene-specific analyses. Third, despite being 10- to 20-fold larger in size with a more diverse population than other CHIP and HIV studies to date, we cannot rule out the possibility of reduced power in examining HIV-related risk factors of CHIP. Nevertheless, given the sample size, to our knowledge, our study has greater power than prior studies in addition to lower false discovery rates.
In conclusion, among a global cohort of ART-treated PWH, CHIP presence is related to intrinsic HIV-related factors, including low CD4 nadir, especially in those who more recently initiated ART. Additionally, global variation in the prevalence of CHIP is more profound than previously known, including interethnicity variation. No specific ART regimen was noted to be associated with a greater odds of CHIP prevalence.
Acknowledgments
The study investigators thank the study participants, site staff, and study-associated personnel for their ongoing participation in the trial. In addition, the authors thank the following: the AIDS Clinical Trial Group (ACTG) for clinical site support; ACTG Clinical Trials Specialists (Laura Moran and Jhoanna Roa) for regulatory support; the data management center, Frontier Science Foundation, for data support; the Center for Biostatistics in AIDS Research for statistical support; and the Community Advisory Board for input for the community.
This study is supported through National Institutes of Health (NIH) grants U01HL123336 to the Clinical Coordinating Center, and U01HL123339 to the Data Coordinating Center as well as funding from Kowa Pharmaceuticals America, Inc, Gilead Sciences, and ViiV Healthcare. The NIAID supported this study through grants UM1 AI068636 which supports the ACTG Leadership and Operations Center; and UM1 AI106701, which supports the ACTG Laboratory Center.
The views expressed in this manuscript are those of the authors and do not necessarily represent the views of the National Heart, Lung, and Blood Institute or the National Institute of Allergy and Infectious Diseases; the NIH; or the US Department of Health and Human Services.
Authorship
Contribution: R. Bhattacharya performed primary analysis; R. Bhattacharya and P.N. composed the initial manuscript draft; M.M.U. and A.N. performed CHIP calling and curation; P.F., R. Bernardo, and W.H. supported data collection, transfer and analysis; A.P.P., K.V.F., M.T.L., G.S.B., C.M., J.A.A., C.J.F., H.J.R., M.V.Z., P.L., B.L.E., and P.S.D. performed critical manuscript review and revision; and S.K.G. and P.N. oversaw conception, funding, and execution of the project.
Conflict-of-interest disclosure: R.B. has served as a medical adviser to Casana Care Inc, unrelated to present work. M.T.L. reports research support to his institution from the American Heart Association, AstraZeneca, Ionis, Johnson & Johnson Innovation, Kowa Pharmaceuticals America, MedImmune, the National Academy of Medicine, and the National Institutes of Health (NIH)/ National Heart, Lung, and Blood Institute (NHLBI) and the Risk Management Foundation of the Harvard Medical Institutions Incorporated outside of the submitted work. C.M. reports research support to his institution from Lilly and serves on the advisory boards for ViiV Healthcare, Gilead Sciences, and Pfizer unrelated to the present work. J.A.A. reports institutional research support for clinical trials from Emergent Biosolutions, Frontier Technologies, Gilead Sciences, GlaxoSmithKline (GSK), Janssen, Merck, Pfizer, Regeneron, and ViiV Healthcare; personal fees for advisory boards from GSK/ViiV and Merck; and participation on data safety monitoring Board for Kintor Pharmaceuticals, all outside the submitted work. C.J.F. reports grant support through his institution from Gilead Sciences, ViiV Healthcare, GSK, Janssen, AbbVie, Merck, Amgen, and Cytodyn; personal fees from Theratechnologies and ViiV for consulting and participation on advisory board unrelated to REPRIEVE; and data safety monitoring board Chair for Intrepid Study, all outside the submitted work. H.J.R. reports receiving grants from NIH/NHLBI and Kowa Pharmaceuticals during the conduct of the study, as well as grants from NIH/ National Institute of Allergy and Infectious Diseases (NIAID), NIH/NHLBI, NIH/National Institute of Diabetes and Digestive and Kidney Diseases, and NIH/National Institute on Aging, outside the submitted work. P.L. is an unpaid consultant to or involved in clinical trials for Amgen, AstraZeneca, Baim Institute, Beren Therapeutics, Esperion Therapeutics, Genentech, Kancera, Kowa Pharmaceuticals, Medimmune, Merck, Moderna, Novo Nordisk, Novartis, Pfizer, and Sanofi-Regeneron; is a member of the scientific advisory board for Amgen, Caristo Diagnostics, Cartesian Therapeutics, CSL Behring, DalCor Pharmaceuticals, Dewpoint Therapeutics, Eulicid Bioimaging, Kancera, Kowa Pharmaceuticals, Olatec Therapeutics, Medimmune, Novartis, PlaqueTec, TenSixteen Bio, Soley Thereapeutics, and XBiotech, Inc; and P.L.’s laboratory has received research funding in the last 2 years from Novartis, Novo Nordisk, and Genentech; P.L. is on the board of directors of XBiotech, Inc; has a financial interest in XBiotech, a company developing therapeutic human antibodies, in TenSixteen Bio, a company targeting somatic mosaicism and clonal hematopoiesis of indeterminate potential (CHIP) to discover and develop novel therapeutics to treat age-related diseases, and in Soley Therapeutics, a biotechnology company that is combining artificial intelligence with molecular and cellular response detection for discovering and developing new drugs, currently focusing on cancer therapeutics; and P.L.’s interests were reviewed and are managed by Brigham and Women’s Hospital and Mass General Brigham in accordance with their conflict-of-interest policies. B.L.E. has received research funding from Celgene, Deerfield, Novartis, and Calico; consulting fees from GRAIL; and is a member of the scientific advisory board and shareholder for Neomorph Inc, TenSixteen Bio, Skyhawk Therapeutics, and Exo Therapeutics. M.V.Z. reports grant support through her institution from NIH/NIAID and Gilead Sciences, Inc, relevant to the conduct of the study, as well as grants from NIH/NIAID and NIH/NHLBI outside the submitted work. S.K.G. reports grant support through his institution from NIH/NIDDK, Kowa Pharmaceuticals America, Inc, Gilead Sciences, Inc, and ViiV Healthcare for the conduct of the study; personal fees from Theratechnologies and ViiV; and service on the scientific advisory board of Marathon Asset Management, all outside the submitted work. P.N. reports research grants from Allelica, Apple, Amgen, Boston Scientific, Genentech/Roche, and Novartis; personal fees from Allelica, Apple, AstraZeneca, Blackstone Life Sciences, Foresite Labs, Genentech/Roche, GV, HeartFlow, Magnet Biomedicine, and Novartis; scientific advisory board membership of Esperion Therapeutics, Preciseli, and TenSixteen Bio; and is a scientific cofounder of TenSixteen Bio, equity in Preciseli and TenSixteen Bio, and spousal employment at Vertex Pharmaceuticals, all unrelated to the present work. The remaining authors declare no competing financial interests.
Correspondence: Pradeep Natarajan, Massachusetts General Hospital, Medicine/Cardiology, 185 Cambridge St, CPZN 3.184, Boston, MA 02114; email: pnatarajan@mgh.harvard.edu.
References
Author notes
Access to original REPRIEVE data are available upon reasonable request from the corresponding author, Pradeep Natarajan (sgrinspoon@mgh.harvard.edu).
The full-text version of this article contains a data supplement.