Key Points
In low-risk BMT for thalassemia, ATG-BuCy seems as effective as Tt-BuCy in the prevention of rejection and may decrease transplant-related mortality.
Abstract
Matched-related bone marrow transplantation (BMT) may cure >80% of low-risk children with severe thalassemia (ST). Very long-term follow-up studies have shown how the standard busulfan-cyclophosphamide (BuCy) regimen may be associated with normalization of health-related quality of life, no second malignancies in the absence of chronic graft-versus-host disease, and fertility preservation in many patients. However, because BuCy may be associated with high rejection rates, some centers incorporate thiotepa (Tt) in busulfan- or treosulfan-based regimens, a combination that may increase the risk of permanent infertility. This study retrospectively compares matched-related BMT outcomes in 2 groups of low-risk ST patients conditioned with either Tt or anti–thymocyte globulin (ATG) in addition to BuCy. A total of 81 consecutive first BMTs were performed in 5 collaborating startup BMT centers in the Indian subcontinent between January 2009 and January 2016; 30 patients were transplanted after conditioning with Tt-BuCy between January 2009 and July 2013, whereas between August 2013 and January 2016, 51 patients received ATG-BuCy. All patients were <15 years and had no hepatomegaly (liver ≤2 cm from costal margin). Actuarial overall survival in the Tt-BuCy and ATG-BuCy groups was 87% and 94% and thalassemia-free survival was 80% and 85% at a median follow-up of 37 and 17 months, respectively, with no significant differences by log-rank statistics. Substituting Tt with ATG in the standard BuCy context seems safe and effective and may decrease transplant-related mortality. Higher fertility rates are expected for patients who received ATG-BuCy.
Introduction
Severe thalassemia (ST) syndromes are the most prevalent life-threatening noncommunicable disorder of childhood1 in Southeast Asia and a major financial burden.2-4 In spite of improved iron chelation therapy and safer transfusions,5 ST remains a severe disease, particularly in developing countries, where often state-of-the-art care is not affordable and/or accessible. ST is also a major indication for blood and marrow transplantation (BMT) in children in the Indian subcontinent.6-8 BMT is currently the only definitive curative option with success rates >80%9 and potential normalization of health-related quality of life (HRQoL)10 in low-risk children with a compatible related donor. The cost of BMT is equivalent to that of a few years of noncurative supportive care.3,11,12 The standard combination of busulfan and cyclophosphamide (BuCy) is associated with rejection rates ranging from 12% to 35%,13,14 which prompted the addition of other engraftment-enhancing agents such as thiotepa (Tt)15,16 or anti–thymocyte globulin (ATG)13,17-19 to busulfan- or treosulfan-containing regimens. Very long-term follow-up studies have shown that BuCy may be associated with normalization of HRQoL, no second malignancies in the absence of chronic graft-versus-host disease (cGVHD), and preservation of fertility in a significant number of patients transplanted prior to age 15 years.10 The combination of 2 myeloablative alkylating agents such as Tt and busulfan or treosulfan may result in a very high infertility rate,20,21 which is increasingly unacceptable in the new thalassemia era of effective chelation, where a substantial proportion of individuals with ST may bear children.22
This study compares retrospectively 2 standard preparative regimens based on BuCy in addition to either Tt or ATG for matched-related low-risk BMTs for ST. Moreover, all BMTs were performed in homogeneous lower-risk patients directly in startup BMT centers in lower middle income countries (LMICs)23 where this disease is most prevalent.
Patients and methods
Study design and procedures
This analysis retrospectively compared 2 widely used preparative drug combinations, Tt-BuCy and ATG-BuCy, in a homogeneous group of low-risk patients with ST directly in LMICs. All BMTs were carried out in 5 newly developed BMT units in the Indian subcontinent: The Children’s Hospital of the Pakistan Institute of Medical Sciences (CHPIMS) in Islamabad, Pakistan; the South East Asia Institute for Thalassemia in (SEAIT) in Jaipur, India; the Asiri Central Hospital (ACH) in Colombo, Sri Lanka; the Nawaloka Hospital (NH) in Colombo, Sri Lanka; and People Tree Hospital (PTH) in Bangalore, India. Institutional partnerships and collaboration methodology are summarized elsewere.24 The great majority of patients included in this report were fully supported by organizations mentioned in the Acknowledgments, and patients were purely selected on medical grounds rather than funding availability. Once enrolled in the program, BMT was free of cost as well as family support.
An information technology platform facilitating interaction among BMT professionals and promoting continuing quality improvement was employed by each participating center, and data were entered prospectively on a daily basis by local physicians and nurses (BMTPlus; Jagrity Innovations, Bangalore, India).25 Computer-generated (Microsoft Excel) individualized treatment plans developed according to good clinical practices and providing clear and simple operating instructions to point-of-care professionals were generated for each patient and double checked by BMT consultants. Written informed consent was obtained from parents or donors >18 years, including sharing data on the online database, the use of which was approved by institutional review boards at each participating center.
All patients and their caretakers were hosted in single rooms with split air conditioning but no centralized HEPA filtration with positive pressure gradients; strict hand washing and sanitation of all personnel and visitors was enforced, and BMT units were thoroughly cleaned daily by dedicated personnel.
Neutrophil recovery was defined as the first of three consecutive days with an absolute neutrophil count (ANC) >500/µL and platelet recovery as the first day of a stable platelet count >20 000/µL without platelet transfusions in the preceding week. Toxicity was graded according to Eastern Cooperative Oncology Group (ECOG) common toxicity criteria.26 Hypertension was categorized using standard criteria.27 Sinusoidal obstructive syndrome was defined and graded according to the Settle criteria,28 acute graft-versus-host disease (aGVHD) according to standard criteria,29 and cGVHD according to National Institutes of Health guidelines.30 Mixed chimerism was categorized in 3 levels according to percentage of donor cells (level 1, 90% to 99%; level 2, 75% to 89%; and level 3, 5% to 74%).31
Patient characteristics and transplant procedure
Inclusion criteria were (a) a diagnosis of severe thalassemia defined as a thalassemia syndrome with the inability to spontaneously maintain hemoglobin levels ≥7 g/dL; (b) liver ≤2 cm below the costal margin on abdominal palpation; (c) availability of a suitable HLA genoidentical related donor; (d) clear understanding of BMT risks and benefits, including parental/care taker informed consent for transplantation; (e) patients <15 years, with no active severe infectious diseases (HIV, hepatitis B, tuberculosis, malaria) or other conditions affecting transplant outcome; (f) creatinine, bilirubin, and serum glutamic-pyruvic transaminase (SGPT) less than three times normal values (SGPT up to 10 times normal values was accepted in case of hepatitis C virus positivity), normal chest radiograph and echocardiogram (shortening fraction ≥35%), and normal age-appropriate performance scale; and (g) institutional commitment to sharing patient data according to privacy regulations.
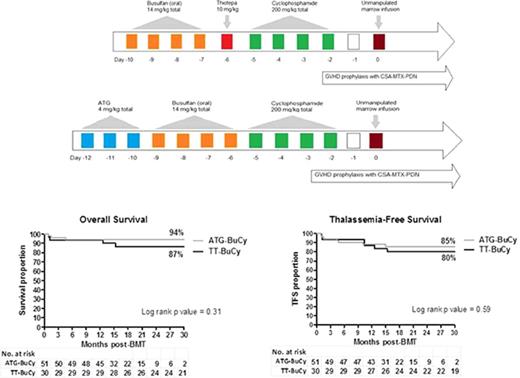
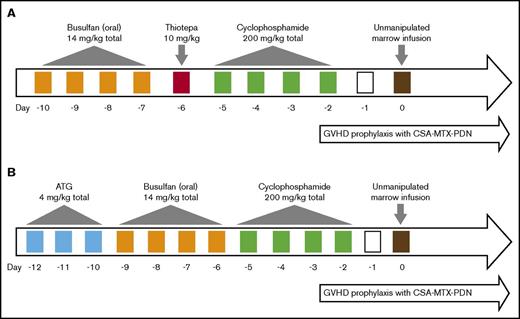
A total of 81 consecutive patients received first BMT from January 2009 to July 2016, 30 patients were transplanted after conditioning with Tt, busulfan (oral), and cyclophosphamide (Tt-BuCy) (Figure 1A) between January 2009 and July 2013, whereas between August 2013 and January 2016, 51 patients were conditioned with ATG, busulfan (oral), and cyclophosphamide (ATG-BuCy) (Figure 1B). Single-lumen central venous catheters, which were tunneled in 49 patients and nontunneled in 31 patients, were employed; 1 patient (who was hepatitis C positive) was managed with peripheral vein cannulas throughout BMT. Tt-BuCy consisted of oral busulfan 3.5 mg/kg day in 4 divided doses on days −10 to −7 (total dose 14 mg/kg), Tt 10 mg/kg per day in 2 divided doses on day −6 (total dose 10 mg/kg), and cyclophosphamide 50 mg/kg per day once daily on days −5 to −2 (total dose 200 mg/kg) followed by the infusion of freshly harvested HLA-compatible marrow on day 0. ATG-BuCy consisted of ATG (rabbit thymoglobulin) 4 mg/kg total dose on days −12 to −10 (at CHPIMS, rabbit-Fresenius was used at 16 mg/kg total dose, because thymoglobulin was not available), busulfan and cyclophosphamide were given as in Tt-BuCy, except busulfan administration was started 1 day later (days −9 to −6). The busulfan dose was not adjusted to blood levels. On day 0, both groups received exclusively bone marrow, which was unprimed in the Tt-BuCy group but granulocyte colony-stimulating factor (G-CSF) primed in 65% of the ATG-BuCy group by treating the donor with filgrastim 5 μg/kg per dose twice daily for either 3 (19 patients) or 5 days (14 patients) prior to harvest. After both regimens, GVHD prophylaxis consisted of cyclosporin A in 2 daily doses aiming at trough levels of 150 to 250 ng/mL for up to day +180, after which it was tapered by −5%/week and discontinued at 12 months post-BMT unless otherwise indicated. A “short” methotrexate course consisting of 10 mg/m2 IV on day +1 (24 hours after marrow infusion) and 8 mg/m2 IV +3, +6, and +11 (the latter dose only in the absence of severe mucositis) with folinic acid rescue at 24 hours after each methotrexate with 3 doses of 10 mg/m2 IV at 8-hour intervals. Methylprednisolone at 0.5 mg/kg per day IV from day −1 to +30, tapered by −1/3 every 5 days over 15 days and stopped on day +45, was also used. Erythrocyte depletion from marrow harvest was required only in cases of major ABO mismatches with isohemagglutinin titers >1:32. All patients received mebendazole 100 mg twice daily for 3 days before conditioning for antihelminthic empirical therapy regardless of stool examination result. No antibacterial prophylaxis was employed. Antifungal prophylaxis consisted of fluconazole 6 mg/kg once daily from day +4 until ANC >500/µL. Acyclovir 250-500 mg/m2 per dose 3 times daily from day +1 to +90 was used for herpes varicella-zoster and simplex prophylaxis. Peripheral blood weekly cytomegalovirus (CMV) DNA copy was monitored by qualitative polymerase chain reaction from day +30 to day +100 in uncomplicated cases; qualitative real-time polymerase chain reaction was employed if the former result was positive, and ganciclovir pre-emptive therapy was administered for DNA copies >1000/mL. For Pneumocystis jiroveci prophylaxis, cotrimoxazole at 5 mg/kg per dose twice daily on alternate days 3 times a week was administered from the day the total leukocyte count reached >1000/µL to day +100.
Conditioning regimens. (A) Tt-BuCy. (B) ATG-BuCy. CSA, cyclosporin A; MTX, methotrexate; PDN, prednisone.
Conditioning regimens. (A) Tt-BuCy. (B) ATG-BuCy. CSA, cyclosporin A; MTX, methotrexate; PDN, prednisone.
Post-BMT, all blood products except for the allograft were irradiated at ≥25 Gy. Immunoglobulin prophylaxis was not employed. No autologous backup marrow was collected. Engraftment was monitored at least at 1, 2, 4, and 8 months by molecular (short tandem repeats) analysis, Y chromosome cytogenetics, or fluorescent in situ hybridization in case of sex mismatch. Table 1 summarizes patient distribution by center and treatment regimen, and Table 2 summarizes patient characteristics by treatment regimen. All patients had at least 6 months of follow-up post-BMT. None of these patients regularly received leukodepleted erythrocyte transfusions prior to BMT.
Patient distribution by center
| Center . | Date of first BMT . | Total BMTs . | Tt-Bu-Cy . | ATG-Bu-Cy . |
|---|---|---|---|---|
| CHPIMS | January 2009 | 41 | 21 | 19 |
| SEAIT | February 2012 | 28 | 9 | 19 |
| ACH | June 2014 | 7 | 0 | 7 |
| NHC | June 2015 | 2 | 0 | 2 |
| PTH | July 2015 | 4 | 0 | 4 |
| Center . | Date of first BMT . | Total BMTs . | Tt-Bu-Cy . | ATG-Bu-Cy . |
|---|---|---|---|---|
| CHPIMS | January 2009 | 41 | 21 | 19 |
| SEAIT | February 2012 | 28 | 9 | 19 |
| ACH | June 2014 | 7 | 0 | 7 |
| NHC | June 2015 | 2 | 0 | 2 |
| PTH | July 2015 | 4 | 0 | 4 |
Patient characteristics
| . | Tt-BuCy . | ATG-BuCy . | P . |
|---|---|---|---|
| No. of patients | 30 | 51 | |
| Follow up, mo* | 36.8 (17.3-80.5) | 16.9 (7.4-30.7) | <.001 |
| Age, y* | 3.1 (0.9-6.4) | 4.8 (1.4-14.5) | .002 |
| Sex ratio, male/female | 23/7 | 31/20 | .222 |
| Height-for-age z-score* | −1.085 (−2.90 to 0.93) | −1.380 (−3.08 to 1.33) | .328 |
| Body mass index-for-age z-score* | −0.29 (−2.85 to 1.58) | −0.85 (−3.88 to 1.32) | .110 |
| Consanguinity | 13 of 27 (3 NA) (48%) | 25 of 46 (5 NA) (54%) | .636 |
| No. of previous transfusions | 30 (6-128) | 42 (3-338) | .026 |
| Donor age | 7.1 (1.5-22.3) | 6.8 (1.1-37.4) | .907 |
| Matched-related donor type | Sibling in 29 of 30 (1 first-degree cousin) | Sibling in 50 of 51 (1 mother) | 1.000 |
| No sibling sex mismatch | 12 of 29 (41%) | 21 of 50 (42%) | 1.000 |
| Sister donor to brother | 13 of 29 (45%) | 18 of 50 (36%) | .480 |
| Brother donor to sister | 4 of 29 (14%) | 11 of 50 (22%) | .553 |
| Donor thalassemia minor | 23 of 30 (77%) | 37 of 51 (73%) | .796 |
| Major ABO mismatch | 7 of 30 (23%) | 8 of 51 (16%) | .394 |
| Ferritin at BMT, ng/mL* | 2282 (591-8008) | 1920 (376 to 6171) | .354 |
| Hydroxyurea therapy prior to BMT | 1 of 30 (3%) | 21 of 51 (41%) | <.001 |
| Liver size, cm from costal margin | all ≤2 cm | all ≤2 cm | |
| Spleen size, cm from costal margin | 28 ≤ 2 cm, 1 = 3 cm, 1 NA | 49 ≤ 2 cm, 1 = 3 cm, 1 = 4 cm | |
| Splenectomy | 1 (3%) | 0 | |
| SGPT, IU/L (HCV-negative patients)* | 49.5 (12-149) | 32 (11 to 151) | .008 |
| HCV positive | 7 of 30 (23%) | 5 of 51 (10%) | .108 |
| SGPT, IU/L (HCV-positive patients)* | 161 (23-537) | 90 (35 to 118) | .268 |
| . | Tt-BuCy . | ATG-BuCy . | P . |
|---|---|---|---|
| No. of patients | 30 | 51 | |
| Follow up, mo* | 36.8 (17.3-80.5) | 16.9 (7.4-30.7) | <.001 |
| Age, y* | 3.1 (0.9-6.4) | 4.8 (1.4-14.5) | .002 |
| Sex ratio, male/female | 23/7 | 31/20 | .222 |
| Height-for-age z-score* | −1.085 (−2.90 to 0.93) | −1.380 (−3.08 to 1.33) | .328 |
| Body mass index-for-age z-score* | −0.29 (−2.85 to 1.58) | −0.85 (−3.88 to 1.32) | .110 |
| Consanguinity | 13 of 27 (3 NA) (48%) | 25 of 46 (5 NA) (54%) | .636 |
| No. of previous transfusions | 30 (6-128) | 42 (3-338) | .026 |
| Donor age | 7.1 (1.5-22.3) | 6.8 (1.1-37.4) | .907 |
| Matched-related donor type | Sibling in 29 of 30 (1 first-degree cousin) | Sibling in 50 of 51 (1 mother) | 1.000 |
| No sibling sex mismatch | 12 of 29 (41%) | 21 of 50 (42%) | 1.000 |
| Sister donor to brother | 13 of 29 (45%) | 18 of 50 (36%) | .480 |
| Brother donor to sister | 4 of 29 (14%) | 11 of 50 (22%) | .553 |
| Donor thalassemia minor | 23 of 30 (77%) | 37 of 51 (73%) | .796 |
| Major ABO mismatch | 7 of 30 (23%) | 8 of 51 (16%) | .394 |
| Ferritin at BMT, ng/mL* | 2282 (591-8008) | 1920 (376 to 6171) | .354 |
| Hydroxyurea therapy prior to BMT | 1 of 30 (3%) | 21 of 51 (41%) | <.001 |
| Liver size, cm from costal margin | all ≤2 cm | all ≤2 cm | |
| Spleen size, cm from costal margin | 28 ≤ 2 cm, 1 = 3 cm, 1 NA | 49 ≤ 2 cm, 1 = 3 cm, 1 = 4 cm | |
| Splenectomy | 1 (3%) | 0 | |
| SGPT, IU/L (HCV-negative patients)* | 49.5 (12-149) | 32 (11 to 151) | .008 |
| HCV positive | 7 of 30 (23%) | 5 of 51 (10%) | .108 |
| SGPT, IU/L (HCV-positive patients)* | 161 (23-537) | 90 (35 to 118) | .268 |
Bold type indicates statistical significance.
HCV, hepatitis C virus; NA, not available.
Median (range).
Statistics
Data were collected and analyzed in December 2016. Patient characteristics were summarized through the use of medians and ranges. Fisher’s exact tests were used to compare proportions, and Mann-Whitney nonparametric tests were used to compare continuous distribution values. Kaplan-Meier survival curves were compared using the log-rank (Mantel-Cox) test. All P values are 2 tailed. Statistical analysis was performed using GraphPad Prism software version 5 (www.graphpad.com/prism/Prism.htm), except for contingency table Fisher exact test P values, which were generated at www.graphpad.com/quickcalcs/contingency1.cfm. Height-for-age and body mass index-for-age z-scores were calculated based on World Health Organization anthropometric data.32
Results
Patients in the Tt-BuCy group had a median age of 3.1 years, and no patient was >7 years of age. In the ATG-BuCy group, median age was 4.8 years, and 15 patients (29%) were >7 years and 3 (6%) were between 12 and 15 years of age. Follow-up was significantly longer in the Tt-BuCy group, with a median of 36.8 months (vs 16.9 months in the ATG-BuCy group). Pre-BMT ferritin values were slightly but not significantly higher in the Tt-BuCy than in the ATG-BuCy group (median 2282 ng/mL vs 1920 ng/mL); in fact, the latter group received more aggressive pre-BMT chelation and hydroxyurea (Hu) therapy (Table 2), and 10 patients had liver size between 2.5 to 5 cm, which decreased to ≤2 cm prior to BMT. SGPT median value was slightly higher in the hepatitis C–negative Tt-BuCy group but still within 3 times the normal value. There was no difference in cell dose between cohorts. Median day to neutrophil engraftment seemed moderately but significantly delayed (ie, 15 days in the Tt-BuCy vs 18 days in the ATG-BuCy group); however, since in the latter group the ANC nadir was reached a few days later, the difference in total number of neutropenic days was less evident (ie, 14 vs 16 days with ANC <500/µL and 11 and 12 days with ANC <200/µL, respectively). There was no statistically significant difference in platelet count recovery, number of platelet or erythrocyte transfusions, or day of last erythrocyte transfusion as well as day of discharge (Table 3).
Engraftment data
| . | Tt-BuCy . | ATG-BuCy . | P . |
|---|---|---|---|
| No. of patients | 30 | 51 | |
| Cell dose, 108 nucleated cells/kg* | 5.4 (3.1-16.8) | 5.5 (2.6-18.9) | .587 |
| G-CSF marrow priming (5 µg/kg × 3-5 d) | 0 | 32 of 51 (63%) | <.001 |
| Days to ANC >500/µL* | 15 (12-28) | 18 (13-32) | <.001 |
| Total days of neutropenia <500/µL* | 14 (10-27) | 15 (8-28) | .092 |
| Days to ANC >200/µL* | 12 (10-28) | 16 (11-29) | <.001 |
| Total days of neutropenia <200/µL* | 11 (8-27) | 12 (6-24) | .489 |
| Use of G-CSF post-BMT | 2 (7%) | 7 (14%) | .473 |
| Days to platelet count >20 000/µL* | 20 (13-68) | 21 (13-38) | .429 |
| Total red cell transfusions* | 2 (0-9) | 3 (0 to 9) | .2392 |
| Last day of red cell transfusion* | 11 (−14 to 152) | 12 (−3 to 58) | .643 |
| Total platelet transfusions* | 4 (0-19) | 4 (0-28) | .945 |
| Discharge day* | 39 (21-165) | 46 (23-99) | .235 |
| . | Tt-BuCy . | ATG-BuCy . | P . |
|---|---|---|---|
| No. of patients | 30 | 51 | |
| Cell dose, 108 nucleated cells/kg* | 5.4 (3.1-16.8) | 5.5 (2.6-18.9) | .587 |
| G-CSF marrow priming (5 µg/kg × 3-5 d) | 0 | 32 of 51 (63%) | <.001 |
| Days to ANC >500/µL* | 15 (12-28) | 18 (13-32) | <.001 |
| Total days of neutropenia <500/µL* | 14 (10-27) | 15 (8-28) | .092 |
| Days to ANC >200/µL* | 12 (10-28) | 16 (11-29) | <.001 |
| Total days of neutropenia <200/µL* | 11 (8-27) | 12 (6-24) | .489 |
| Use of G-CSF post-BMT | 2 (7%) | 7 (14%) | .473 |
| Days to platelet count >20 000/µL* | 20 (13-68) | 21 (13-38) | .429 |
| Total red cell transfusions* | 2 (0-9) | 3 (0 to 9) | .2392 |
| Last day of red cell transfusion* | 11 (−14 to 152) | 12 (−3 to 58) | .643 |
| Total platelet transfusions* | 4 (0-19) | 4 (0-28) | .945 |
| Discharge day* | 39 (21-165) | 46 (23-99) | .235 |
Bold type indicates statistical significance.
Median (range).
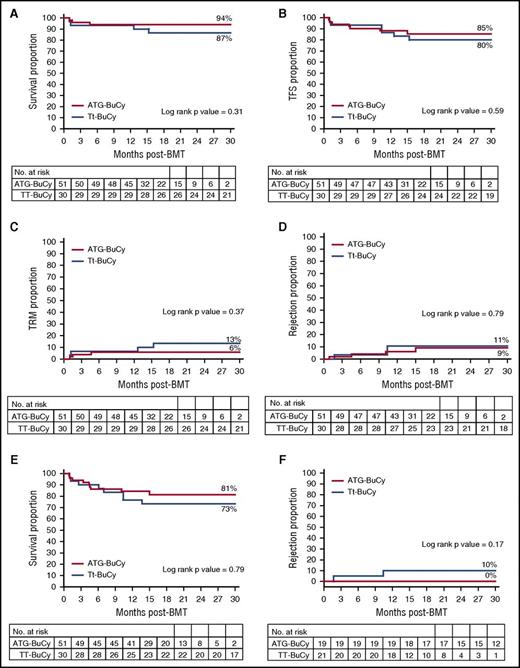
Actuarial rejection rates (defined as <5% donor DNA in peripheral blood) did not seem to differ between the Tt-BuCy and ATG-BuCy groups (11% vs 9%, respectively, with a log-rank P value = .79). In the ATG-BuCy group, in which most patients received Hu prior to BMT (as opposed to the Tt-BuCy group), of the 4 patients who rejected the transplant, 2 received Hu prior to BMT and 2 did not. Transplant-related mortality was higher, albeit not significantly, in the Tt-BuCy group than in the ATG-BuCy group (13% vs 6%, respectively; P = .37) and overall survival lower (87% vs 94%; P = .31), thalassemia-free survival was similar with 80% vs 85% (P = .59). Moderate to severe cGVHD could affect HRQoL as much as ST itself, and thus its occurrence might be considered a treatment failure. Composite cGVHD-free and thalassemia-free survival was 73% in the Tt-BuCy and 81% in the ATG-BuCy group (P = .79). In the 40 children <4 years at BMT (ie, the age group in which the addition of Tt to BuCy has been advocated because of higher rejection risk),15 the rejection rate was 10% in the 21 patients in the Tt-BuCy group and 0% in the 19 ATG-BuCy patients (P = .17). None of these outcome differences approached statistical significance, and both groups had a comparable number of patients at risk 18 months post-BMT (Figure 2). Mixed chimerism was observed in 80% of patients in the Tt group and 87% of patients in the ATG-BuCy group; all patients in the former group had chimerism >90% (level 1), while in the ATG-BuCy group, 7 patients (16%) had level 2 or 3 chimerism (Table 3). As outlined in Table 4, immunosuppression duration in thalassemia-free patients was significantly longer in the Tt-BuCy group, partly because donor mixed chimerism of <90%, which was observed only in the ATG-BuCy group, often prompted early immunosuppression taper, so that in patients with level 2 or 3 chimerism, the median (range) day of immunosuppression discontinuation was 257 (97-562) as opposed to 374 (158-497) in those patients with chimerism persistently ≥90% (P = .01). Patients with >8 months of follow-up had chimerism at least at 8 months, a time after which mixed chimerism tends to be stable.33,34 For cost-containment reasons, most children with a stable hemoglobin >9 g/dL after that time point did not have further chimerism testing, because it was not deemed clinically relevant. Only 1 patient in the Tt-BuCy group received a second transplant and is thalassemia-free at 46.5 months from the second BMT (described elsewere35 ); no child in the ATG-BuCy group has received a second BMT at the time of this analysis. Donors were all harvested under general anesthesia with no adverse events. All recipients tolerated the conditioning regimen well. One patient had an anaphylactic reaction to intravenous cyclosporine that did not recur after switching to the oral formulation. BMT-related complications are summarizes in Table 4. A total of 4 patients (13%) in the Tt-BuCy group and 3 patients (6%) in the ATG-BuCy group died. Mucositis ECOG score >2 (ie, severe enough to cause inability to eat) was more frequent in the former group (23% vs 2%), while the frequency of hemorrhagic cystitis ECOG score >2 (ie, gross hematuria with clots) was similar. Acute GVHD >2 occurred in 3% of patients in the Tt-BuCy group and 8% of patients in the ATG-BuCy group, with 2 GVHD-related deaths in the latter group. The incidence of moderate to severe cGVHD seemed to be higher, albeit not significantly, in the Tt-BuCy group, with 3 cases (10%) of bronchiolitis obliterans (BO), while in the ATG-BuCy, it occurred in 2 cases (4%) (1 BO and 1 nephrotic syndrome). In the latter group, there was 1 case of immune thrombocytopenia at day +75, which resolved. Two patients (both in the ATG-BuCy group) experienced intracranial hemorrhage, one with visual cortex involvement and decreased sight (which has been improving) and the other in the context of sepsis and disseminated intravascular coagulation. Two patients died of cGVHD (both BO) in the Tt-BuCy group, while both patients in the ATG-BuCy are alive and had normal performance scores at the last follow-up. All recipients were CMV positive at BMT, and in spite of comparable proportions of CMV-negative donors, CMV reactivation was seen significantly more often in the Tt-BuCy group (ie, 43% vs 13% in the ATG-BuCy), all patients received ganciclovir, and none had overt CMV disease. One patient in each group had a diagnosis of tuberculosis post-BMT (on day +190 [pulmonary] in the Tt-BuCy group and on day +406 [vertebral body] in the ATG-BuCy group); both patients have been treated and are doing well. One patient in the Tt-BuCy group died of encephalitis, the etiological cause of which could not be ascertained.
Survival curves. (A) Overall survival; (B) thalassemia-free survival; (C) transplant-related mortality; (D) rejection; (E) composite GVHD and thalassemia-free survival; and (F) rejection in patients <4 years of age.
Survival curves. (A) Overall survival; (B) thalassemia-free survival; (C) transplant-related mortality; (D) rejection; (E) composite GVHD and thalassemia-free survival; and (F) rejection in patients <4 years of age.
Complications
| . | Tt-BuCy . | ATG-BuCy . | P . |
|---|---|---|---|
| No. of patients | 30 | 51 | |
| Transplant-related deaths | 4 (13%) (2 BO, 1 encephalitis, 1 TMA) | 3 (6%) (2 aGVHD, 1 sepsis) | .414 |
| Severe mucositis* | 7 (23%) | 1 (2%) | .003 |
| Severe hemorrhagic cystitis* | 3 (10%) | 7 (14%) | .737 |
| Hypertension stage 2 | 13 (43%) | 26 (51%) | .646 |
| aGVHD > grade 2 | 1 (3%) (grade 3) | 4 (8%) (3 grade 3, 1 grade 4) | .648 |
| cGVHD > mild | 3 (10%) (all BO) | 2 (4%) (1 BO, 1 NS) | .350 |
| Immune cytopenias | 0 | 1 (2%) (ITP) | 1.000 |
| Sinusoidal obstructive syndrome | 2 (7%) | 1 (2%) | .551 |
| Thrombotic microangiopathy | 3 (10%) | 1 (2%) | .136 |
| CMV-positive recipient | 100% | 100% | 1.000 |
| CMV-negative donor | 5 (17%) | 7 (14%) | .753 |
| CMV reactivation | 13 (43%) | 7 (14%) | .006 |
| Central venous line infection | 5 (17%) | 17 (33%) | .126 |
| Clinical sepsis | 2 (7%) | 7 (13%) | .473 |
| Fungal infection (proven or probable) | 0 | 0 | 1.000 |
| Empirical use of antifungals | 3 (10%) | 5 (10%) | 1.000 |
| Tuberculosis | 1 (3%) | 1 (2%) | 1.000 |
| Malaria | 1 (3%) | 0 | .370 |
| Other | 2 intracranial hemorrhage, 1 encephalitis | 1 cutaneous leishmaniasis, 1 endocarditis, 1 Salmonella bacteremia, 1 HAV infection | |
| Immunosuppression withdrawal | 409 (158-562) | 320 (101-419) | .013 |
| Mixed chimerism (5%-99%) with transfusion independence | 20 of 25 evaluable cases (80%) | 39 of 44 evaluable cases (87%) | .478 |
| Level 1 (90%-99% donor) | 20 | 32 | .506 |
| Level 2 (75%-89% donor) | 0 | 3 | .549 |
| Level 3 (5%-74% donor) | 0 | 4 | .289 |
| . | Tt-BuCy . | ATG-BuCy . | P . |
|---|---|---|---|
| No. of patients | 30 | 51 | |
| Transplant-related deaths | 4 (13%) (2 BO, 1 encephalitis, 1 TMA) | 3 (6%) (2 aGVHD, 1 sepsis) | .414 |
| Severe mucositis* | 7 (23%) | 1 (2%) | .003 |
| Severe hemorrhagic cystitis* | 3 (10%) | 7 (14%) | .737 |
| Hypertension stage 2 | 13 (43%) | 26 (51%) | .646 |
| aGVHD > grade 2 | 1 (3%) (grade 3) | 4 (8%) (3 grade 3, 1 grade 4) | .648 |
| cGVHD > mild | 3 (10%) (all BO) | 2 (4%) (1 BO, 1 NS) | .350 |
| Immune cytopenias | 0 | 1 (2%) (ITP) | 1.000 |
| Sinusoidal obstructive syndrome | 2 (7%) | 1 (2%) | .551 |
| Thrombotic microangiopathy | 3 (10%) | 1 (2%) | .136 |
| CMV-positive recipient | 100% | 100% | 1.000 |
| CMV-negative donor | 5 (17%) | 7 (14%) | .753 |
| CMV reactivation | 13 (43%) | 7 (14%) | .006 |
| Central venous line infection | 5 (17%) | 17 (33%) | .126 |
| Clinical sepsis | 2 (7%) | 7 (13%) | .473 |
| Fungal infection (proven or probable) | 0 | 0 | 1.000 |
| Empirical use of antifungals | 3 (10%) | 5 (10%) | 1.000 |
| Tuberculosis | 1 (3%) | 1 (2%) | 1.000 |
| Malaria | 1 (3%) | 0 | .370 |
| Other | 2 intracranial hemorrhage, 1 encephalitis | 1 cutaneous leishmaniasis, 1 endocarditis, 1 Salmonella bacteremia, 1 HAV infection | |
| Immunosuppression withdrawal | 409 (158-562) | 320 (101-419) | .013 |
| Mixed chimerism (5%-99%) with transfusion independence | 20 of 25 evaluable cases (80%) | 39 of 44 evaluable cases (87%) | .478 |
| Level 1 (90%-99% donor) | 20 | 32 | .506 |
| Level 2 (75%-89% donor) | 0 | 3 | .549 |
| Level 3 (5%-74% donor) | 0 | 4 | .289 |
Bold type indicates statistical significance.
HAV, hepatitis A virus; ICH, intracranial hemorrhage; ITP, immune thrombocytopenic purpura; NS, nephritic syndrome.
Greater than ECOG grade 2.
Discussion
This study is the first multinational study in startup BMT services in LMICs comparing homogeneous patient groups and BMT regimens for ST. All centers had never performed BMT before, had an almost exclusive focus on matched-related BMT for thalassemia, and extensively employed information technology tools for daily interaction with experienced BMT professionals. In this selected but relatively common group of low-risk patients, outcomes were comparable to those reported in high-income countries.9 Our retrospective comparison suggests that the addition of a second myeloablative agent may not be warranted and that the ATG-BuCy combination can be quite tolerable and associated with low rejection rates.
We used the term severe thalassemia instead of thalassemia major to encompass higher-risk thalassemia intermedia patients and also bypass a possible confusion related to the Western working definition of thalassemia major (ie, requiring >8 erythrocyte transfusions per year).36 In fact, pretransfusion hemoglobin is often much lower in lower income countries than in the West, and thus transfusion frequency may not necessarily reflect disease severity. We also did not apply the Pesaro classification relying on liver biopsy,37 because the combination of liver size and age have been shown to be reliable predictors of transplant-associated risk in thalassemia.38,39 All children in this report had a liver ≤2 cm from costal margin, so they might be considered Pesaro class I or II. The age cutoff we adopted was extended from 8 to 15 years in the ATG-BuCy group. A high level of ferritin is known to be correlated with increased transplant-related mortality.40 The majority of our patients were aggressively chelated prior to BMT, and median serum ferritin values were <2500 ng/mL, a possible relevant cutoff in terms of transplant-related morbidity and mortality.39,41,42 Many patients, particularly in the ATG-BuCy group, were treated with Hu in order to minimize both liver and spleen size as well as extramedullary hematopoiesis.43,44
There is a general perception that BMT centers need complex engineering standards requiring undue investments, even more so in developing countries, where the risk of infection might be greater. In fact, there is no evidence that the latter is true.45,46 On the contrary, local physicians might be in the best position to identify and treat locally prevalent diseases (eg, the patients with tuberculosis, malaria, and leishmaniasis in our series are doing well). There is increasing evidence that allogeneic transplant patients may be safely cared for in regular hospital rooms46 or even as outpatients,47,48 and many widely held practices calling for “sterile” BMT units are being reconsidered. In fact, international guidelines do not mandate admission in HEPA-filtered or positive pressure gradient rooms, at least for low-risk BMTs.49 The human development index does not seem to correlate with non–relapse mortality after BMT for acute leukemia,50 suggesting that the quality of remission assessment, an issue that does not pertain to ST, might be more relevant than where the BMT procedure itself is carried out. The infectious complications we observed did not seem to be substantially different from those observed internationally.19,51
The chronic nature of ST, which allows to perform BMT in patients when conditions are optimal, combined with their lack of prior exposure to chemotherapy, low mucositis, and GVHD rates, as well as relative short duration of profound neutropenia, most likely played a role in the very low rate of fungal infections we observed, and the same may not apply to higher-risk diseases such as leukemias or severe aplastic anemias.
The 2 initial centers of this project, CHPIMS and SEAIT, were both staffed by professionals with little or no BMT experience who relied on on-site, whole-team–focused training by experienced BMT physicians followed by daily interaction via video conferencing and web-based medical records.24 The centers in Sri Lanka (ACH and NH) were run by adult hematologists with some BMT experience in collaboration with pediatricians, as well as on-site and online consultation with pediatric transplant physicians. This is the first report on a large series of thalassemia patients undergoing BMT exclusively in startup centers and directly in LMICs and might provide a relevant precedent to address the dire shortage of such services in emerging countries,52 where hemoglobinopathies are the most frequent life-threatening noncommunicable disease among children1 and a major indication for BMT.6,7,53
Adding Tt54 or ATG13,17-19 to the BuCy backbone has substantially reduced rejection rates. In the age group <4 years of age, where Tt seems particularly important to reduce rejection,15 we observed no significant rejection differences between the ATG and Tt groups. A combination of treosulfan, Tt, and fludarabine has also been advocated as the preferable BMT conditioning for patients with thalassemia, particularly those with higher transplantation risk55-57 ; however, this recommendation is not based on prospective studies, and the combined use of 2 myeloablative alkylating agents may be associated with high infertility rates20 as well as substantial increases in costs. A formal cost analysis could not be performed, but the average all-inclusive cost per BMT was ∼12 000 US dollars and that of family support was ∼2000 US dollars. We also believe that in the current era of effective chelation, it might no longer be justified to take higher-risk patients directly to transplant without aggressive chelation and possibly Hu prior to BMT. In our series, 10 patients (all in the ATG-BuCy group) had an initial liver size >2 cm from costal margin and could thus have been considered higher-risk patients, but they were downstaged into a lower risk category by aggressive supportive care prior to BMT.
In long-term follow-up, BuCy has been shown to be associated with birth rates comparable to the control population in former ST patients.10 However, our current report does not have sufficient follow-up to directly assess the impact of substituting Tt with ATG on pubertal development or fertility.
In our experience with Tt-BuCy, severe GVHD (grade III-IV) was quite rare (in the 3% range). For this reason, we opted for early and low-dose ATG administration (days −12 to −10) with little or no ATG carryover at marrow infusion, thus favoring engraftment and immune reconstitution as opposed to the standard day −5 to −1 ATG timing geared also toward in vivo donor T-cell depletion and GVHD prevention. Moreover, all our patients received bone marrow rather than mobilized leukapheresis, the latter having been associated with an increased risk of extensive cGVHD,58-60 a major factor affecting HRQoL and risk of secondary malignancies post-BMT.10 Most patients in the ATG-BuCy group received G-CSF–primed marrow, which may have further contributed to decreased GVHD occurrence.61 None had autologous backup marrow stored prior to transplant, since its indication is questionable.62 No formal immune reconstitution studies were performed, but the decreased frequency of CMV reactivation in the ATG-BuCy group suggests that the ATG approach employed in this study might be less immunosuppressive than Tt-BuCy. This might also be reflected by an increased rate of stable mixed chimerism in the ATG-BuCy group, although rejection rates were comparable. Persistent mixed chimerism with transfusion independence is a well-known occurrence after BMT for thalassemia33 and might actually be an advantage given the reduced risk of GVHD associated with mixed chimerism.63
The major limitation of this report is its retrospective nature, which makes it difficult to tease out whether the apparent, albeit not statistically significant, improvement in thalassemia-free and overall survival observed with ATG-BuCy over Tt-BuCy is related to differences in duration of follow-up and other confounding factors. However, the number of patients at risk at 18 months (a relevant time point after which rejection is rarely observed and mixed chimerism is stable in most patients31 ) was comparable. Also, median age was significantly higher in the ATG-BuCy group, with 8 patients being older than 7 years as opposed to none in the Tt-BuCy group, which might have skewed outcomes in favor of the latter,39 while pre-BMT therapy with Hu and increased use of G-CSF priming of marrow might have favored the ATG-BuCy group, as well as a time-period effect as the startup centers gained more experience.
In conclusion, the ATG-BuCy combination seems to be as effective as, and possibly safer than, Tt-BuCy. Within structured and intensive cooperation programs with experienced BMT professionals focusing on a single disease and a low-risk transplant procedure, it is possible to obtain high success rates directly in LMICs, even without highly trained physicians and nurses.
Acknowledgments
The authors thank the patients, their parents, and all the nurses and physicians at collaborating institutions.
This work was partially supported by Cure2Children Foundation (Florence, Italy), Sankalp India Foundation (Bangalore, India), Heartfile (Islamabad, Pakistan), Fondazione Monte dei Paschi di Siena (Siena, Italy), Cassa di Risparmio di Firenze (Florence, Italy), and The Italian and Pakistani Governments (Cooperazione Italiana allo Sviluppo through the Pakistan-Italy Dept for Development Swap Agreement). The authors are also grateful to DKMS (Tübingen, Germany) for providing free HLA typing.
Authorship
Contribution: L.F. designed and performed the research, collected the data, performed the statistical analyses, interpreted the data, and wrote the manuscript; R.K.A., A.S., and R. Dhanya collected the data and performed the statistical analyses; L.F, C.U., S.K., P.M., R.S., N.Y., S.A., I.F., S.K.G., T.Z., S.R., L.G., R. Dissanayake, S.W., W.R., and R.S. participated in patient management; R.K.A., A.S., R.S., R. Dhanya, L.P., and A.K. coordinated patient registration, data management, and HLA typing; and all authors critically reviewed and approved the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Lawrence Faulkner, Medical Coordinator Cure2Children Foundation, via Marconi 30, 50131 Florence, Italy; e-mail: lawrence.faulkner@cure2children.org.