Background
Tanzania has the third highest number of infants in the world born annually with sickle cell disease (SCD). Tanzania lacks a national newborn screening program, and a large majority of the clinical care, resources, and research for SCD are located in the coastal city of Dar es Salaam. Previous studies have suggested that the northwestern area of the country around Lake Victoria served by Bugando Medical Centre (BMC) has a particularly high incidence of SCD. BMC also houses the HIV early infant diagnosis (EID) laboratory. All dried blood spots (DBS) from children born to HIV-infected mothers in the northwestern portion of the country are shipped to BMC for polymerase chain reaction (PCR) testing and are afterward stored on site.
Objectives
To build local capacity for accurate diagnosis and appropriate care for children born with SCD in northwestern Tanzania.
To determine the prevalence of sickle cell trait (SCT) and SCD by region and district in northwestern Tanzania.
To explore the associations between SCT, SCD, malaria, and HIV.
To characterize regional hemoglobin variants.
Hypotheses
Collaborative research-based investigation of locally important health conditions is an effective method of capacity building with sustainable goals.
SCT and SCD prevalence will be highest in regions and districts closest to Lake Victoria.
Historical malaria endemic areas will have the highest prevalence of SCT and SCD.
HIV is a lethal comorbidity of SCD, so the prevalence of SCD will be lower in HIV-infected infants.
Methods
Study design
The Tanzania Sickle Surveillance Study is a year-long, cross-sectional study among HIV-exposed infants born in northwestern Tanzania, whose DBS collected by the EID program are tested at BMC and available for further testing of SCT and SCD. Samples collected from children <24 months of age will be included.
Laboratory analysis
Isoelectric focusing (IEF) is used to determine sickle cell status, and specifically sickle trait vs disease. Further identification of other hemoglobin variants will be conducted using high-performance liquid chromatography, DNA-based techniques, such as PCR, and sequencing of the α- and β-globin genes.
Capacity building
A new IEF system was provided by the Association of Public Health Laboratories to the BMC laboratory for high-volume SCD testing.
Two laboratory technicians completed a 2-day course provided by PerkinElmer on IEF operation and interpretation.
One clinician performed a 2-month observership in hematology at Cincinnati Children’s Hospital.
All IEF testing is performed on site at the BMC laboratory.
IEF gels are run on a daily basis and interpreted by 2 Tanzanian staff members with independent scoring.
All completed IEF gels and interpretation results are scanned using a smartphone application and sent to collaborators in the United States for discussion and continued education.
A US-based laboratory collaborator assesses each gel for overall quality and serves as an independent scorer for the gels.
Biweekly Skype calls allow for discussion of quality and interpretation as well as resolution of any discordant scoring.
Results
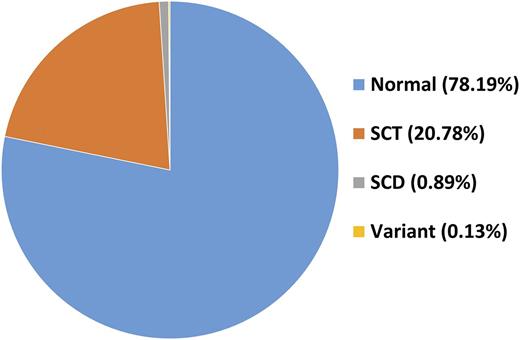
Over the course of 6 months, 91 gels have been run. A total of 6105 DBS samples have been scored, and 5941 were collected from children <24 months old. Only 13 DBS yielded uninterpretable IEF results. All samples with SCD or variant hemoglobin have been confirmed on a repeat gel. The overall prevalence of SCT is 20.78%, and the prevalence of SCD is 0.89%. The prevalence of variants is 0.13%. Malaria and HIV correlations will be performed at the study end.
Conclusions
Collaborative research partnerships provide an excellent mechanism for building local capacity to diagnose and improve care for SCD. The prevalence of SCT and SCD is high in northwestern Tanzania.
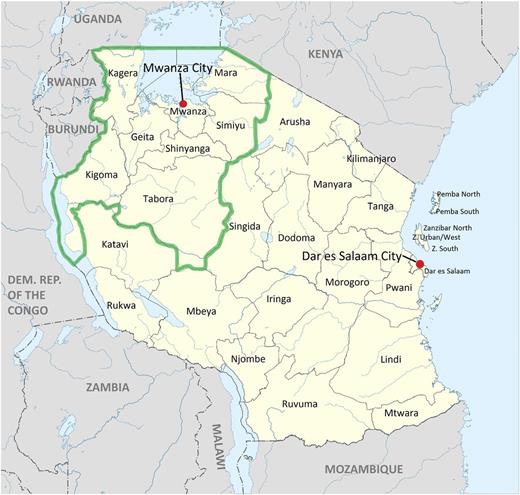
Map of Tanzania. The catchment area for the HIV EID program serviced by BMC PCR laboratory is outlined in green. Derived from https://commons.Wikimedia.org/wiki/File:Tanzania location map.svg. © Sémhur/Wikimedia Commons/CC-BY-SA-3.0 (or Free Art License).
Map of Tanzania. The catchment area for the HIV EID program serviced by BMC PCR laboratory is outlined in green. Derived from https://commons.Wikimedia.org/wiki/File:Tanzania location map.svg. © Sémhur/Wikimedia Commons/CC-BY-SA-3.0 (or Free Art License).
(A) IEF gel preparation at BMC. (B) Completed gel that was scanned and e-mailed to collaborators.
(A) IEF gel preparation at BMC. (B) Completed gel that was scanned and e-mailed to collaborators.
Acknowledgments:
Alan Anderson and Jelili Ojodu provided the IEF equipment, and Petri Huhtinen organized the in-country training.
Conflict-of-interest disclosure: R.W.: Nova Laboratories (consultancy), Agios (membership on an entity's Board of Directors or advisory committees), Bristol-Myers Squibb (research funding), Addmedica (research funding), Global Blood Therapeutics (consultancy). Other authors: no competing financial interests declared.
Correspondence: Emmanuela E. Ambrose; e-mail: emmanuelaambrose@gmail.com.