Key Points
Children receiving BuFlu for nonmalignant conditions experienced less toxicity than those receiving BuCy, but survival was comparable.
Children with malignancy had shorter postrelapse survival with BuFlu than BuCy; transplant-related mortality and relapse were similar.
Abstract
Busulfan combined with cyclophosphamide (BuCy) has long been considered a standard myeloablative conditioning regimen for allogeneic hematopoietic cell transplantation (HCT), including both nonmalignant conditions and myeloid diseases. Substituting fludarabine for cyclophosphamide (BuFlu) to reduce toxicity without an increase in relapse has been increasingly performed in children, but without comparison with BuCy. We retrospectively analyzed 1781 children transplanted from 2008 to 2014 to compare the effectiveness of BuCy with BuFlu. Nonmalignant and malignant disease populations were analyzed separately. Overall mortality was comparable for children with nonmalignant conditions who received BuFlu or BuCy (relative risk [RR], 1.14, P = .52). Lower incidences of sinusoidal obstruction syndrome (P = .04), hemorrhagic cystitis (P = .04), and chronic graft-versus-host disease (P = .02) were observed after BuFlu, but the influence of the conditioning regimen could not be assessed by multivariate analysis because of the low frequency of these complications. Children transplanted for malignancies were more likely to receive BuFlu if they had higher hematopoietic cell transplantation-comorbidity index scores (P < .001) or their donor was unrelated and HLA-mismatched (P = .004). Nevertheless, there were no differences in transplant toxicities and comparable transplant-related mortality (RR, 1.2; P = .46), relapse (RR, 1.2; P = .15), and treatment failure (RR, 1.2; P = .12). BuFlu was associated with higher overall mortality (RR, 1.4; P = .008) related to inferior postrelapse survival (P = .001). Our findings demonstrated that outcomes after BuFlu are similar to those for BuCy for children, but for unclear reasons, those receiving BuFlu for malignancy may be at risk for shorter postrelapse survival.
Introduction
Allogeneic hematopoietic cell transplantation (HCT) is a potentially curative therapy for a variety of hematologic diseases. For many years, the standard conditioning regimen administered for patients with myeloid malignancies and nonmalignant diseases (such as sickle cell disease) was myeloablative busulfan combined with high-dose cyclophosphamide (BuCy). Although producing reliable engraftment, this regimen results in high rates of regimen-related toxicities, including sinusoidal obstruction syndrome (SOS) at 20% to 50%1,2 and hemorrhagic cystitis (5%-15%).3 The substitution of fludarabine, a nucleoside analog with potent immunosuppressive properties and antileukemic synergy with alkylating agents,4 for cyclophosphamide (BuFlu) resulted in less toxicity and better outcomes for adult patients.5-8 There are few reports, however, comparing myeloablative BuCy with BuFlu in children.9 Despite the lack of pediatric-specific literature, the experience with BuFlu in adults prompted several pediatric centers to use this regimen, and the Children’s Oncology Group adopted it for the most recent treatment protocol for de novo acute myeloid leukemia (AML; NCT01371981).
To better compare the toxicities and outcomes for BuCy to BuFlu conditioning in the pediatric population, we performed a retrospective observational study of patients receiving these 2 regimens who were reported to the Center for International Blood and Marrow Transplant Research (CIBMTR).
Patients and methods
Data sources
The CIBMTR is a voluntary working group of more than 450 international transplantation centers that contribute data from allogeneic and autologous stem cell transplants. Participating centers are required to report all transplantations consecutively; patients are followed longitudinally, and compliance is monitored by on-site audits. Computerized checks for discrepancies, physicians’ review of submitted data, and on-site audits of participating centers ensure data quality. Observational studies conducted by the CIBMTR are performed in compliance with all applicable federal regulations pertaining to the protection of human research participants.
The CIBMTR collects data at 2 levels: Transplant Essential Data (TED) and Comprehensive Report Form (CRF) data. TED data include disease type, age, sex, pre-HCT disease stage and chemotherapy-responsiveness, date of diagnosis, graft type (bone marrow- and/or blood-derived stem cells), conditioning regimen, posttransplant disease progression and survival, development of a new malignancy, and cause of death. All CIBMTR centers contribute TED-level data. More detailed disease and pre- and posttransplant clinical information are collected on a subset of registered patients selected for CRF data by a weighted randomization selection. TED and CRF-level data are collected pretransplant, 100 days and 6 months post-HCT, and annually thereafter or until death. Data for the current analysis were retrieved from both TED and CRF CIBMTR databases.
Patients
The study population consisted of all patients at least 18 years of age who received a first allogeneic transplant for any indication between 2008 and 2014 and who received myeloablative doses of busulfan, defined as a planned dose of at least 9 mg/kg oral or 7.2 mg/kg IV,10 combined with either cyclophosphamide or fludarabine. The CIBMTR uses 2 approaches to determine conditioning intensity: whether the intent of the conditioning regimen is myeloablative, as reported by the transplant center; and the calculated dose based on total dose given, as collected on the CRFs plus dosing weight. Patients receiving additional chemotherapy agents (eg, melphalan) or total body irradiation were excluded, but those who received serotherapy (eg, antithymocyte globulin or alemtuzumab) were included. Patients receiving umbilical cord blood grafts or transplants from donors with less than a 7/8 match at HLA-A, HLA-B, HLA-C, and HLA-DR were also excluded. Busulfan pharmacokinetic data were not available for the study cohort.
Study end points and definitions
The primary end points were transplant-related mortality (TRM) and disease-free survival (DFS) for patients with malignancy, and overall survival (OS) for all patients. Patients receiving a second transplant were not censored for survival analyses. Secondary end points included neutrophil and platelet engraftment, SOS, noninfectious hemorrhagic cystitis and pulmonary toxicity, graft failure, infections, and acute and chronic graft-versus-host disease (GVHD). Patient data were censored at the time of death or last follow-up. TRM was defined as any death that occurred in the absence of disease relapse; relapse was a competing risk for this event. Relapse was defined as hematologic, cytogenetic, or molecular evidence of disease; patients without relapse information were considered to have relapse if they received posttransplant therapy with a reported intent of treatment of relapse (ie, donor leukocyte infusion, second transplant, or chemotherapy). DFS was defined as time to treatment failure (either death or disease recurrence). Neutrophil and platelet engraftment were calculated as time from transplantation to the first of 3 consecutive days with neutrophils ≥0.5 × 109/L and platelets ≥20 × 109/L without transfusion in the preceding 7 days. Acute GVHD was diagnosed and graded per standard criteria,11 and chronic GVHD was defined as the development of any chronic GVHD based on standard clinical criteria.12,13 Death was treated as a competing risk for relapse, engraftment, SOS, noninfectious pulmonary toxicity, and hemorrhagic cystitis. Relapse and death were competing risks for acute and chronic GVHD.
Statistical methods
Patient-, disease-, and transplant-related variables were compared using Mann-Whitney U tests (continuous variables) and Fisher’s exact/χ2 analyses (categorical variables). Survival outcomes were computed using the Kaplan-Meier method, and comparisons were performed using log-rank tests. Cumulative incidences were calculated for relapse, TRM, acute and chronic GVHD, SOS, pulmonary toxicity, and hemorrhagic cystitis after accounting for competing risks. Patients transplanted for malignancies and nonmalignant diseases were analyzed separately for all outcomes. Cox proportional hazards regression models were built using a forward stepwise selection procedure. The proportional hazards assumptions were tested. The interaction between the main effect and the other significant covariates (α < 0.05) was examined. Multivariate models were built to test disease relapse, TRM, treatment failure (1-DFS), and overall mortality for patients with malignancies, as well as overall mortality in patients with nonmalignant diseases. Covariates tested in all models were age (0-9 vs 10-18 years), sex, performance score (≥90% vs <90%), disease status for malignancies (early, intermediate, and advanced), HCT comorbidity index (HCT-CI; 0, 1-2, or >3), donor type (HLA-matched related, HLA-matched unrelated, or single-allele mismatched unrelated), graft source (bone marrow or peripheral blood), GVHD prophylaxis (tacrolimus or cyclosporine and methotrexate, tacrolimus or cyclosporine and mycophenolate mofetil, or other strategies), and use of serotherapy (antithymocyte globulin/alemtuzumab; yes vs no). Adjusted curves were created on the basis of the final model for each outcome. Comparison of these regimens was also performed on a subset of patients with CRF-level data for univariate analysis of neutrophil and platelet recovery, graft failure, infections, SOS, hemorrhagic cystitis, and acute and chronic GVHD. Subset analyses using CRF-level data were planned at time of protocol development. Post hoc analyses of survival after disease relapse and subset analyses comparing these 2 regimens among patients with AML in complete remission as a homogenous population were performed to explore the reasons for differences in OS between the regimens among patients with malignant diseases. All analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC).
Results
A total of 1781 patients met eligibility that received BuCy (N = 1400) or BuFlu (N = 381) (Table 1). Eight-hundred three patients received transplants for nonmalignant conditions (BuCy, n = 627; BuFlu, n = 176), whereas 978 patients (BuCy, n = 773; BuFlu, n = 205) were transplanted for malignancies. Comprehensive-level data required for secondary end point analyses were available for 571 patients (BuCy, n = 399; BuFlu, n = 172; supplemental Tables 1 and 2). Only a small number of patients (n = 148; 8%) received oral busulfan in either conditioning regimen, although patients with nonmalignant diagnoses were more likely to receive oral busulfan as part of BuCy conditioning (12% vs <1%; P < .001). With regard to patients for whom CRF-level data were available (n = 571), a majority (n = 438; 77%) of subjects had pharmacokinetic-directed busulfan dosing, although patients with nonmalignant conditions who received BuFlu were less likely to have pharmacokinetic-directed dosing than those who received BuCy (65% vs 85%; P = .003) and had a lower median busulfan dose (12.87 vs 15.42 mg/kg; P < .001; supplemental Table 1). Busulfan doses were comparable between those receiving BuFlu and BuCy (13.02 vs 14.1 mg/kg; P = .52; supplemental Table 2).
Patient baseline characteristics
| Variable . | Malignancies . | Nonmalignant . | ||||
|---|---|---|---|---|---|---|
| Bu + Cy . | Bu + Flu . | P . | Bu + Cy . | Bu + Flu . | P . | |
| Number of enrolled patients | 773 | 205 | 627 | 176 | ||
| Number of centers | 105 | 64 | 66 | 45 | ||
| Age at transplant, y | .43 | .95 | ||||
| Median (range) | 10 (<1-18) | 11 (<1-18) | 5 (<1-18) | 3 (<1-18) | ||
| 1-9 | 371 (48) | 92 (45) | 501 (80) | 141 (80) | ||
| 10-20 | 402 (52) | 113 (55) | 126 (20) | 35 (20) | ||
| Sex | .06 | .15 | ||||
| Male | 414 (54) | 125 (61) | 361 (58) | 112 (64) | ||
| Female | 359 (46) | 80 (39) | 266 (42) | 64 (36) | ||
| Lansky performance score | .41 | .02 | ||||
| ≥90% | 672 (87) | 173 (84) | 499 (80) | 150 (85) | ||
| <90% | 89 (12) | 30 (15) | 119 (19) | 20 (11) | ||
| Missing | 12 (2) | 2 | 9 (1) | 6 (3) | ||
| Primary disease | .10 | .10 | ||||
| AML | 515 (67) | 138 (67) | ||||
| ALL | 37 (5) | 15 (7) | ||||
| CML | 77 (10) | 13 (6) | ||||
| MDS | 117 (15) | 33 (16) | ||||
| MPN | 14 (2) | 0 | ||||
| Other acute leukemia | 13 (2) | 6 (3) | ||||
| Hemoglobinopathies | 322 (51) | 48 (27) | ||||
| Primary Immunodeficiencies | 195 (31) | 86 (49) | ||||
| Inherited abnormalities of platelets | 10 (2) | 2 (1) | ||||
| Inherited disorder of metabolism | 65 (10) | 24 (14) | ||||
| Histiocytic disorders | 35 (6) | 16 (9) | ||||
| HCT-CI | <.001 | <.001 | ||||
| 0 | 511 (66) | 109 (53) | 347 (55) | 98 (56) | ||
| 1-2 | 107 (14) | 35 (17) | 60 (10) | 29 (16) | ||
| ≥3 | 61 (8) | 36 (18) | 37 (6) | 22 (13) | ||
| Missing | 94 (12) | 25 (12) | 183 (29) | 27 (15) | ||
| Disease status* | .036 | |||||
| Early | 433 (56) | 95 (46) | ||||
| Intermediate | 125 (16) | 49 (24) | ||||
| Advanced | 106 (14) | 29 (14) | ||||
| Unknown | 109 (16) | 32 (16) | ||||
| Donor type | .004 | <.001 | ||||
| HLA-matched related | 444 (57) | 95 (46) | 503 (80) | 82 (47) | ||
| 8/8 URD | 239 (31) | 71 (35) | 96 (15) | 72 (41) | ||
| 7/8 URD | 90 (12) | 39 (19) | 28 (4) | 22 (13) | ||
| Graft type | <.001 | .004 | ||||
| Bone marrow | 686 (89) | 136 (66) | 614 (98) | 165 (94) | ||
| Peripheral blood stem cells | 87 (11) | 69 (34) | 13 (2) | 11 (6) | ||
| Route of busulfan administration | .35 | <.001 | ||||
| Oral | 63 (8) | 11 (5) | 73 (12) | 1 (<1) | ||
| IV | 709 (92) | 194 (95) | 552 (88) | 175 (99) | ||
| Unknown | 1 (<1) | 0 | 2 (<1) | 0 | ||
| GVHD prophylaxis | <.001 | <.001 | ||||
| TAC+MMF | 27 (3) | 21 (10) | 30 (5) | 10 (6) | ||
| TAC+MTX | 180 (23) | 94 (46) | 67 (11) | 33 (19) | ||
| CSA+MMF | 26 (3) | 16 (8) | 33 (5) | 27 (15) | ||
| CSA+MTX | 455 (59) | 65 (32) | 418 (67) | 85 (48) | ||
| Others | 85 (11) | 9 (4) | 79 (13) | 21 (12) | ||
| Antithymocyte globulin/alemtuzumab | <.001 | <.001 | ||||
| No | 529 (68) | 90 (44) | 194 (31) | 41 (23) | ||
| Yes | 219 (28) | 106 (52) | 396 (63) | 94 (53) | ||
| Missing | 25 (3) | 9 (4) | 37 (6) | 41 (23) | ||
| Year of transplant | <.001 | <.001 | ||||
| 2008 | 130 (17) | 8 (4) | 100 (16) | 7 (4) | ||
| 2009 | 128 (17) | 10 (5) | 103 (16) | 7 (4) | ||
| 2010 | 129 (17) | 16 (8) | 89 (14) | 17 (10) | ||
| 2011 | 107 (14) | 27 (13) | 102 (16) | 19 (11) | ||
| 2012 | 98 (13) | 38 (19) | 106 (17) | 27 (15) | ||
| 2013 | 88 (11) | 40 (20) | 67 (11) | 46 (26) | ||
| 2014 | 93 (12) | 66 (32) | 60 (10) | 53 (30) | ||
| Median follow-up of survivors, months (range) | 60 (3-94) | 26 (6-86) | 62 (6-79) | 24 (3-96) | ||
| Variable . | Malignancies . | Nonmalignant . | ||||
|---|---|---|---|---|---|---|
| Bu + Cy . | Bu + Flu . | P . | Bu + Cy . | Bu + Flu . | P . | |
| Number of enrolled patients | 773 | 205 | 627 | 176 | ||
| Number of centers | 105 | 64 | 66 | 45 | ||
| Age at transplant, y | .43 | .95 | ||||
| Median (range) | 10 (<1-18) | 11 (<1-18) | 5 (<1-18) | 3 (<1-18) | ||
| 1-9 | 371 (48) | 92 (45) | 501 (80) | 141 (80) | ||
| 10-20 | 402 (52) | 113 (55) | 126 (20) | 35 (20) | ||
| Sex | .06 | .15 | ||||
| Male | 414 (54) | 125 (61) | 361 (58) | 112 (64) | ||
| Female | 359 (46) | 80 (39) | 266 (42) | 64 (36) | ||
| Lansky performance score | .41 | .02 | ||||
| ≥90% | 672 (87) | 173 (84) | 499 (80) | 150 (85) | ||
| <90% | 89 (12) | 30 (15) | 119 (19) | 20 (11) | ||
| Missing | 12 (2) | 2 | 9 (1) | 6 (3) | ||
| Primary disease | .10 | .10 | ||||
| AML | 515 (67) | 138 (67) | ||||
| ALL | 37 (5) | 15 (7) | ||||
| CML | 77 (10) | 13 (6) | ||||
| MDS | 117 (15) | 33 (16) | ||||
| MPN | 14 (2) | 0 | ||||
| Other acute leukemia | 13 (2) | 6 (3) | ||||
| Hemoglobinopathies | 322 (51) | 48 (27) | ||||
| Primary Immunodeficiencies | 195 (31) | 86 (49) | ||||
| Inherited abnormalities of platelets | 10 (2) | 2 (1) | ||||
| Inherited disorder of metabolism | 65 (10) | 24 (14) | ||||
| Histiocytic disorders | 35 (6) | 16 (9) | ||||
| HCT-CI | <.001 | <.001 | ||||
| 0 | 511 (66) | 109 (53) | 347 (55) | 98 (56) | ||
| 1-2 | 107 (14) | 35 (17) | 60 (10) | 29 (16) | ||
| ≥3 | 61 (8) | 36 (18) | 37 (6) | 22 (13) | ||
| Missing | 94 (12) | 25 (12) | 183 (29) | 27 (15) | ||
| Disease status* | .036 | |||||
| Early | 433 (56) | 95 (46) | ||||
| Intermediate | 125 (16) | 49 (24) | ||||
| Advanced | 106 (14) | 29 (14) | ||||
| Unknown | 109 (16) | 32 (16) | ||||
| Donor type | .004 | <.001 | ||||
| HLA-matched related | 444 (57) | 95 (46) | 503 (80) | 82 (47) | ||
| 8/8 URD | 239 (31) | 71 (35) | 96 (15) | 72 (41) | ||
| 7/8 URD | 90 (12) | 39 (19) | 28 (4) | 22 (13) | ||
| Graft type | <.001 | .004 | ||||
| Bone marrow | 686 (89) | 136 (66) | 614 (98) | 165 (94) | ||
| Peripheral blood stem cells | 87 (11) | 69 (34) | 13 (2) | 11 (6) | ||
| Route of busulfan administration | .35 | <.001 | ||||
| Oral | 63 (8) | 11 (5) | 73 (12) | 1 (<1) | ||
| IV | 709 (92) | 194 (95) | 552 (88) | 175 (99) | ||
| Unknown | 1 (<1) | 0 | 2 (<1) | 0 | ||
| GVHD prophylaxis | <.001 | <.001 | ||||
| TAC+MMF | 27 (3) | 21 (10) | 30 (5) | 10 (6) | ||
| TAC+MTX | 180 (23) | 94 (46) | 67 (11) | 33 (19) | ||
| CSA+MMF | 26 (3) | 16 (8) | 33 (5) | 27 (15) | ||
| CSA+MTX | 455 (59) | 65 (32) | 418 (67) | 85 (48) | ||
| Others | 85 (11) | 9 (4) | 79 (13) | 21 (12) | ||
| Antithymocyte globulin/alemtuzumab | <.001 | <.001 | ||||
| No | 529 (68) | 90 (44) | 194 (31) | 41 (23) | ||
| Yes | 219 (28) | 106 (52) | 396 (63) | 94 (53) | ||
| Missing | 25 (3) | 9 (4) | 37 (6) | 41 (23) | ||
| Year of transplant | <.001 | <.001 | ||||
| 2008 | 130 (17) | 8 (4) | 100 (16) | 7 (4) | ||
| 2009 | 128 (17) | 10 (5) | 103 (16) | 7 (4) | ||
| 2010 | 129 (17) | 16 (8) | 89 (14) | 17 (10) | ||
| 2011 | 107 (14) | 27 (13) | 102 (16) | 19 (11) | ||
| 2012 | 98 (13) | 38 (19) | 106 (17) | 27 (15) | ||
| 2013 | 88 (11) | 40 (20) | 67 (11) | 46 (26) | ||
| 2014 | 93 (12) | 66 (32) | 60 (10) | 53 (30) | ||
| Median follow-up of survivors, months (range) | 60 (3-94) | 26 (6-86) | 62 (6-79) | 24 (3-96) | ||
Values are n (%) unless otherwise noted.
ALL, acute lymphoblastic leukemia; CML, chronic myelogenous leukemia; CSA, cyclosporine A; MDS, myelodysplasia; MMF, mycophenolate mofetil; MPN, myeloproliferative neoplasm; MTX, methotrexate; URD, unrelated donor.
Disease status definition: early, AML/ALL in first complete remission, CML in chronic phase or MDS excluding refractory anemia with excess blasts; intermediate, AML/ALL in any complete remission beyond first or CML in accelerated phase any time before transplant; and advanced, AML/ALL with primary induction failure or relapse, CML in blastic crisis any time before transplant or MDS with refractory anemia with excess blasts.
Patients with nonmalignant diseases
Patient, disease, and transplant characteristics for those receiving transplants for nonmalignant diseases are listed in Table 1. There was no difference in age, sex, or indication for transplant between the 2 conditioning regimens. The BuFlu cohort had a lower proportion of patients with a Lansky performance score less than 90% (11% vs 19%; P = .02), but a higher proportion of patients with HCT-CI scores greater or equal to 3 (13% vs 6%; P < .001) compared with the BuCy cohort. In addition, children receiving BuFlu were less likely to receive allografts from HLA-matched related donors (47% vs 80%; P < .001), less likely to receive cyclosporine or tacrolimus with methotrexate for GVHD prophylaxis (67% vs 78%; P = .008), and more likely to receive HCT later in the 2008 to 2014 period (P < .001).
Engraftment.
There were no differences in neutrophil recovery at post-HCT days 28 (P = .7) and 100 (P = .13) between the 2 regimens. A higher proportion of BuFlu recipients recovered the platelet counts by day 28 (59% vs 40%; P < .001); however, this difference was no longer observed at day 100 (P = .41; supplemental Table 3).
Toxicities and GVHD.
Patients receiving BuFlu experienced fewer toxicities than those receiving BuCy. Cumulative incidences by day 100 of SOS were 7% and 16% (P = .04), and incidences of hemorrhagic cystitis were 2% and 9% (P = .04) for BuFlu and BuCy, respectively. Idiopathic pneumonia syndrome was rare, but comparable (2% and 1%; P = .80) between the regimens. The frequency of these toxicities was too low to perform a multivariate analysis to determine whether the observed differences in these toxicities could be attributed to the conditioning regimen and/or other factors. Six-month cumulative incidences of grade II-IV and III/IV acute GVHD were 16% and 25% (P = .13) and 8% and 14% (P = .18) for BuFlu and BuCy, respectively. Patients receiving BuFlu had a lower incidence of chronic GVHD by 1 year (11% vs 22%; P = .02). Rates of graft failure and infection were not different between the regimens (supplemental Table 4).
Survival.
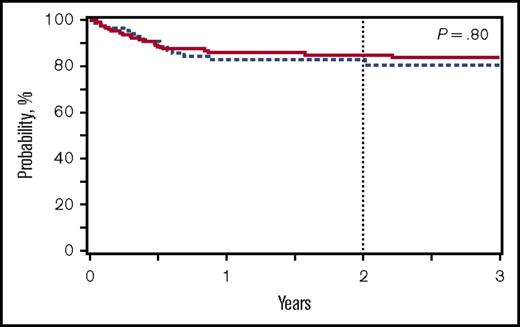
Despite the decreased incidences of SOS, hemorrhagic cystitis and chronic GVHD observed for patients receiving BuFlu, the 2-year OS was 85% for both cohorts (P = .85), with a RR for BuFlu of 1.14 (P = .52) on multivariate analysis (supplemental Table 5). Cytomegalovirus serologic status was associated with OS on multivariate analysis, mainly driven by donor-recipient cytomegalovirus serologic mismatch compared with both donor and recipient cytomegalovirus seronegative (supplemental Table 5). Most deaths were a result of GVHD, infection, or organ failure, and these did not differ between the conditioning regimen groups (supplemental Table 4).
Patients with malignancies
Patient, disease, and transplant characteristics for those receiving transplant for malignancy categorized by conditioning regimen are listed in Table 1. There was no difference in age, sex, underlying malignancy, donor-recipient sex match, or CMV status between regimens. The presence of risk factors for inferior HCT outcomes were not evenly distributed between groups. The BuFlu cohort had a higher proportion of patients with HCT-CI score ≥3 (18% vs 8%; P < .001) and was less likely to receive allografts from HLA-matched related donors (46% vs 57%; P = .004). In addition, children receiving BuFlu were more likely to receive peripheral blood stem cell grafts (34% vs 11%; P < .001) and serotherapy with antithymocyte globulin or alemtuzumab (52% vs 28%; P < .001). The use of cyclosporine or tacrolimus with methotrexate for GVHD prophylaxis was comparable between patients receiving BuFlu and those receiving BuCy (78% vs 82%; P = .16), and patients receiving BuFlu were more likely to be transplanted later in the 2008 to 2014 period (P < .001).
Engraftment.
Consistent with the increased use of peripheral blood stem cell grafts, more children receiving BuFlu engrafted neutrophils (94% vs 91%; P = .002) and platelets (67% vs 43%; P < .001) by post-HCT day 28. The differences in hematologic recovery were no longer observed by day 100 for both neutrophils (P = .19) and platelets (P = .1; supplemental Table 3).
Toxicities and GVHD.
Day 100 cumulative incidences of SOS were 13% and 15% (P = .66), and instances of hemorrhagic cystitis were 7% and 8% (P = .75) for BuFlu and BuCy, respectively. Cumulative incidences of idiopathic pneumonia syndrome were also comparable (5% and 2%; P = .66). Again, the low frequencies of these events did not permit for multivariate analysis. Similarly, graft failure, infections, and causes of death were not different between the regimens (supplemental Table 6). No difference was observed in the 6-month cumulative incidences of grade II-IV (31% vs 26%; P = .35) and grade III/IV (18% vs 11%; P = .13) acute GVHD for BuFlu and BuCy, respectively; incidences for chronic GVHD by 1 year were also similar (28% vs 23%; P = .37).
TRM, disease relapse, DFS, OS.
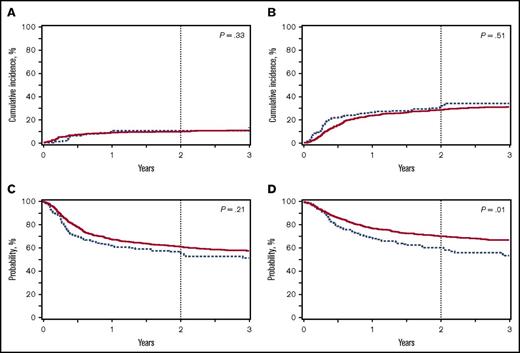
Unlike nonmalignant disease, in which survival is largely dependent on the incidence of TRM, survival for patients with malignant disease is determined by the incidence of TRM and relapse. For BuFlu and BuCy, the 2-year cumulative incidences of TRM were 13% and 10% (P = .33; Figure 2A), and disease relapse incidences were 31% and 28% (P = .51; Figure 2B), respectively. There was a trend to more frequent relapse at 3 months post-BMT after BuFlu conditioning (12% vs 7%; P = .05), but this difference decreased at subsequent points. The comparable TRM and relapse rates unsurprisingly resulted in a similar 2-year DFS (56% vs 62%, P = .21; Figure 2C). The 2-year OS (61% vs 71%, P = .01; Figure 2D), however, was significantly lower for children who received BuFlu when compared with those who received BuCy.
OS for patients transplanted for nonmalignant conditions receiving BuCy (n = 627; solid red line) or BuFlu (n = 176; dashed blue line).
OS for patients transplanted for nonmalignant conditions receiving BuCy (n = 627; solid red line) or BuFlu (n = 176; dashed blue line).
Outcomes for patients transplanted for malignancy receiving BuCy (n = 773; solid red line) or BuFlu (n = 205; dashed blue line). (A) Transplant-related mortality. (B) Disease relapse. (C) Disease-free survival. (D) OS.
Outcomes for patients transplanted for malignancy receiving BuCy (n = 773; solid red line) or BuFlu (n = 205; dashed blue line). (A) Transplant-related mortality. (B) Disease relapse. (C) Disease-free survival. (D) OS.
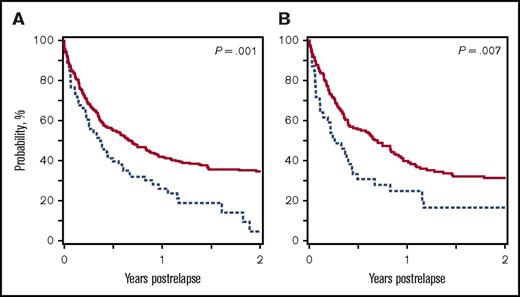
Multivariate analyses were performed for each outcome to better understand the influence of the conditioning regimen and other covariates on these findings (Table 2). Multiple risk factors influenced the RR for TRM (older age, male sex, donor type) and relapse (younger age, disease status at time of transplant), but conditioning regimen did not (TRM RR, 1.2 [P = .46]; relapse RR, 1.2 [P = .15]). Similarly, treatment failure appears to be a result of unfavorable donor type, HLA mismatch, and more advanced disease, and not the choice of conditioning regimen (RR, 1.2; P = .12). Donor type and disease status also contributed to worse OS, but after adjusting for these variables, the use of BuFlu conditioning still significantly increased the likelihood for mortality (RR, 1.4; P = .008). A post hoc analysis revealed that a major contributor to differences in OS was shorter survival after disease relapse for patients receiving BuFlu (2-year postrelapse survival, 35% vs 5%; P = .001; Figure 3A).
Multivariate analysis of outcomes for patients with malignant disease
| Effect . | Transplant-related mortality . | Relapse . | Treatment failure (1-DFS) . | Overall mortality . | ||||
|---|---|---|---|---|---|---|---|---|
| RR (95% CI) . | P . | RR (95% CI) . | P . | RR (95% CI) . | P . | RR (95% CI) . | P . | |
| Conditioning regimen | .46 | .15 | .12 | .008 | ||||
| BuCy | 1 | 1 | 1 | 1 | ||||
| BuFlu | 1.2 (0.8-1.9) | 1.2 (0.9-1.6) | 1.2 (1-1.5) | 1.4 (1.1-1.8) | ||||
| Age, y | <.001 | .0002 | ||||||
| 0-9 | 1 | 1 | ||||||
| 10-18 | 2.5 (1.6-3.9) | 0.6 (0.5-0.8) | ||||||
| Sex | .044 | |||||||
| Male | 1 | |||||||
| Female | 0.7 (0.4-1) | |||||||
| Donor type | .0002 | .019 | .006 | |||||
| HLA-identical sibling | 1 | 1 | 1 | |||||
| 8/8 unrelated | 1.85 (1.2-2.8) | 1.2 (1-1.5) | 1.3 (1-1.6) | |||||
| 7/8 unrelated | 2.74 (1.7-4.5) | 1.5 (1.1-2) | 1.6 (1.2-2.2) | |||||
| Disease status | <.001 | <.001 | <.001 | |||||
| Early | 1 | 1 | 1 | |||||
| Intermediate | 1.3 (1-1.8) | 1.2 (0.9-1.6) | 1.3 (1-1.7) | |||||
| Advanced | 2.5 (1.9-3.4) | 2.1 (1.6-2.7) | 2.3 (1.7-3) | |||||
| Unknown | 0.9 (0.6-1.4) | 1 (0.7-1.3) | 0.9 (0.6-1.2) | |||||
| Effect . | Transplant-related mortality . | Relapse . | Treatment failure (1-DFS) . | Overall mortality . | ||||
|---|---|---|---|---|---|---|---|---|
| RR (95% CI) . | P . | RR (95% CI) . | P . | RR (95% CI) . | P . | RR (95% CI) . | P . | |
| Conditioning regimen | .46 | .15 | .12 | .008 | ||||
| BuCy | 1 | 1 | 1 | 1 | ||||
| BuFlu | 1.2 (0.8-1.9) | 1.2 (0.9-1.6) | 1.2 (1-1.5) | 1.4 (1.1-1.8) | ||||
| Age, y | <.001 | .0002 | ||||||
| 0-9 | 1 | 1 | ||||||
| 10-18 | 2.5 (1.6-3.9) | 0.6 (0.5-0.8) | ||||||
| Sex | .044 | |||||||
| Male | 1 | |||||||
| Female | 0.7 (0.4-1) | |||||||
| Donor type | .0002 | .019 | .006 | |||||
| HLA-identical sibling | 1 | 1 | 1 | |||||
| 8/8 unrelated | 1.85 (1.2-2.8) | 1.2 (1-1.5) | 1.3 (1-1.6) | |||||
| 7/8 unrelated | 2.74 (1.7-4.5) | 1.5 (1.1-2) | 1.6 (1.2-2.2) | |||||
| Disease status | <.001 | <.001 | <.001 | |||||
| Early | 1 | 1 | 1 | |||||
| Intermediate | 1.3 (1-1.8) | 1.2 (0.9-1.6) | 1.3 (1-1.7) | |||||
| Advanced | 2.5 (1.9-3.4) | 2.1 (1.6-2.7) | 2.3 (1.7-3) | |||||
| Unknown | 0.9 (0.6-1.4) | 1 (0.7-1.3) | 0.9 (0.6-1.2) | |||||
CI, confidence interval.
Postrelapse OS of patients receiving BuCy (solid red line) or BuFlu (dashed blue line). (A) Patients with malignancies. (B) Patients with AML in complete remission.
Postrelapse OS of patients receiving BuCy (solid red line) or BuFlu (dashed blue line). (A) Patients with malignancies. (B) Patients with AML in complete remission.
Subset analysis: AML in complete remission
Given the unexpected finding that postrelapse survival was related to conditioning regimen, a subset analysis was performed for children with AML who received HCT in complete remission, the leading indication for HCT in this study, to evaluate a group with more homogenous disease. Outcomes were compared in patients with AML in first complete remission receiving BuFlu (n = 115) or BuCy (n = 454) to further explore the effect of the conditioning regimen. Similar to the main cohort, there were no differences in 2-year incidences of TRM (P = .84) and relapse (P = .67) or 2-year probabilities of DFS (P = .79) between the cohorts (supplemental Figure 1). Unlike in the main cohort, the 2-year OS probabilities were not statistically different for BuFlu and BuCy cohorts (63% vs 71%; P = .12). Children in this subset who relapsed after BuFlu conditioning again had shorter survival after leukemic relapse (2-year postrelapse survival, 17% vs 31%; P = .007; Figure 3B).
Discussion
A substantial body of medical literature supports the transition from BuCy to BuFlu conditioning for adult patients resulting from decreased TRM and toxicity, and particularly hepatotoxicity, without an increase in relapse for patients transplanted for malignancy.14-17 These findings were mirrored in a pediatric prospective study, although melphalan was added to BuCy conditioning for malignancy, and patients received the different regimens consecutively rather than concurrently and in a randomized fashion.9
In the current study, children with nonmalignant diseases who received BuFlu experienced fewer serious toxicities than those who received BuCy, but the low frequency of these events after either conditioning regimen precluded multivariate analysis to determine whether the difference in toxicity could be attributed to the conditioning regimen and/or other factors. Importantly, there was no difference in survival between regimens.
In contrast to other studies,9,18 no difference was observed in the incidences of SOS or hemorrhagic cystitis for patients with malignant diseases. The heterogeneity of the underlying malignancies complicates interpretation of relapse, DFS, and OS results. Minimal residual disease testing before transplant, which is increasingly used to determine disease risk,19 was unavailable for patients in this report. Cytogenetic risk, another important disease risk factor,20 was only available for the subset of patients with CRF-level data, and was comparable between patients receiving either conditioning regimen for whom these data were available. No difference was observed in relapse rates, however, suggesting comparable disease control from both conditioning regimens. Although DFS was also similar between regimens, OS was inferior on for patients receiving BuFlu on multivariate analysis. This result was surprising given the lack of difference in TRM, relapse, or DFS, and appears to be a result of short postrelapse survival in the BuFlu cohort. It is unclear why the choice of conditioning appeared to influence survival for patients who relapse posttransplant. Although uncommon after either conditioning regimen, there was a trend toward increased early relapse after BuFlu, which may have influenced the decision to provide postrelapse treatment of some patients; the small numbers with early relapse following either regimen are unlikely to account for the observed differences in postrelapse survival. Another possible explanation is that the patients who received BuFlu had a higher HCT-CI score, which suggests bias in regimen selection by treating physicians. This bias may have also influenced decisions about how postrelapse was managed. Alternatively, it is possible that subjects who received BuFlu had higher-risk disease for reasons not readily apparent in the available data, such as detectable pretransplant minimal residual disease, and thus had fewer treatment options available. The difference in postrelapse survival may also reflect a center effect related to some centers more aggressively managing relapse than others. Unfortunately, information on postrelapse treatment was not available to help explain this observation.
The heterogeneity of the underlying malignancies prompted analysis of the subset of patients with AML in complete remission. This patient subset is of particular interest because of the change by many programs from BuCy to BuFlu as the assigned regimen for patients with de novo AML who receive allogeneic HCT. BuFlu recipients again experienced shorter postrelapse survival compared with BuCy recipients. The reason for such a difference in postrelapse survival remains unclear, but recent changes in patient selection for transplant may play a role. For example, the indication for allogeneic BMT for AML has changed from availability of a sibling donor regardless of relapse risk21,22 to the presence of very high risk features such as minimal residual disease after induction or high allelic ratio FLT3/ITD (Children’s Oncology Group AAML 1031; NCT01371981). Patients with lower-risk disease may have been more likely to survive after relapse, regardless of conditioning regimen.
Interestingly, significantly greater use of unrelated and HLA-mismatched donors for patients receiving BuFlu did not result in more acute GVHD in any of the patient subsets evaluated, and patients transplanted for nonmalignant conditioning experienced less chronic GVHD despite these differences. The increased use of serotherapy in patients with malignancies receiving BuFlu may have helped mitigate GVHD rates for these patients.23
As with any retrospective study, this study has limitations. Children who were less likely to survive as a result of more comorbidities or the use of HLA-mismatched donors disproportionately received BuFlu conditioning, which may influence the findings of this retrospective study. Differences in patient groups can be addressed through multivariate analysis, but this approach can be hampered when a high-risk variable is uncommon, as was the case with high HCT-CI score in this study or when an event of interest occurs infrequently. The increased use of BuFlu in more recent years may also make comparisons difficult because of changes in supportive care, increased sensitivity of disease detection (eg, the use of minimal residual disease testing), and changes to patient selection for transplant. In addition, busulfan exposure and pharmacokinetics are known to influence transplant outcomes,18,24-28 but these data were not available for many patients, and thus we were unable to evaluate the effect of busulfan exposure on outcomes. Last, it is important to note that differences may exist between regimens with respect to late effects, which were not evaluated in this study.
In conclusion, our data suggest that BuFlu is comparable to BuCy among children with nonmalignant diseases and may reduce the risk for SOS and hemorrhagic cystitis. However, the evidence of shorter survival with BuFlu among patients with malignancies needs to be interpreted with caution, given their shorter postrelapse survival, and requires further analysis in a randomized setting. Even though there were no differences in any other outcome comparing BuFlu with BuCy, it is plausible that the OS difference observed is related to factors other than the conditioning regimen. This study emphasizes that changes in TRM, relapse, DFS, and OS must be monitored carefully when implementing new conditioning strategies. This study also demonstrates the importance of careful analysis of newer regimens before extrapolating results from adult patients to the pediatric population.
The full-text version of this article contains a data supplement.
Acknowledgments
The CIBMTR is supported primarily by Public Health Service Grant/Cooperative Agreement (5U24CA076518) from the National Institutes of Health, National Cancer Institute, National Institute of Allergy and Infectious Diseases, and National Heart, Lung, and Blood Institute; a grant/cooperative agreement (4U10HL069294) from the National Institutes of Health, National Heart, Lung, and Blood Institute and National Cancer Institute; a contract (HHSH250201200016C) with Health Resources and Services Administration (Health Resources and Services Administration/US Department of Health and Human Services); 2 grants (N00014-17-1-2388 and N0014-17-1-2850) from the Office of Naval Research; and grants from *Actinium Pharmaceuticals, Inc.; *Amgen, Inc.; *Amneal Biosciences; *Angiocrine Bioscience, Inc.; Anonymous donation to the Medical College of Wisconsin; Astellas Pharma US; Atara Biotherapeutics, Inc.; Be the Match Foundation; *bluebird bio, Inc.; *Bristol-Myers Squibb Oncology; *Celgene Corporation; Cerus Corporation; *Chimerix, Inc.; Fred Hutchinson Cancer Research Center; Gamida Cell Ltd.; Gilead Sciences, Inc.; HistoGenetics, Inc.; Immucor; *Incyte Corporation; Janssen Scientific Affairs, LLC; *Jazz Pharmaceuticals, Inc.; Juno Therapeutics; Karyopharm Therapeutics, Inc.; Kite Pharma, Inc.; Medac, GmbH; MedImmune; The Medical College of Wisconsin; *Mediware; *Merck & Co, Inc.; *Mesoblast; MesoScale Diagnostics, Inc.; Millennium, the Takeda Oncology Co.; *Miltenyi Biotec, Inc.; National Marrow Donor Program; *Neovii Biotech NA, Inc.; Novartis Pharmaceuticals Corporation; Otsuka Pharmaceutical Co, Ltd.–Japan; PCORI; *Pfizer, Inc; *Pharmacyclics, LLC; PIRCHE AG; *Sanofi Genzyme; *Seattle Genetics; Shire; Spectrum Pharmaceuticals, Inc.; St. Baldrick’s Foundation; *Sunesis Pharmaceuticals, Inc.; Swedish Orphan Biovitrum, Inc.; Takeda Oncology; Telomere Diagnostics, Inc.; and University of Minnesota. *Corporate members.
The views expressed in this article do not reflect the official policy or position of the National Institutes of Health, the Department of the Navy, the Department of Defense, Health Resources and Services Administration or any other agency of the US Government.
Authorship
Contribution: A.C.H., J.J.B., M.C.P., and J.E.L. contributed the study design, analysis plan, interpretation of the analysis, and writing of the manuscript; K.W.A. and M.F. contributed statistical analysis and generation of figures; and M.F., A. Abraham, A. Artz, C.D., H.F., C.F., R.P.G., S.H., H.M.L., A.L., S.M., T.N., T.O., and K.W. reviewed and provided input on the study design and contributed to interpretation of the analysis and writing of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Andrew C. Harris, Department of Pediatric Hematology-Oncology, Blood and Marrow Transplant Program, University of Utah–Primary Children’s Hospital, 100 N. Mario Capecchi Dr, Suite 4100, Salt Lake City, UT 84113; e-mail: andrew.harris@hsc.utah.edu.
References
Author notes
A.C.H. and J.J.B. contributed equally to this study.
M.C.P. and J.E.L. contributed equally to this study.