Key Points
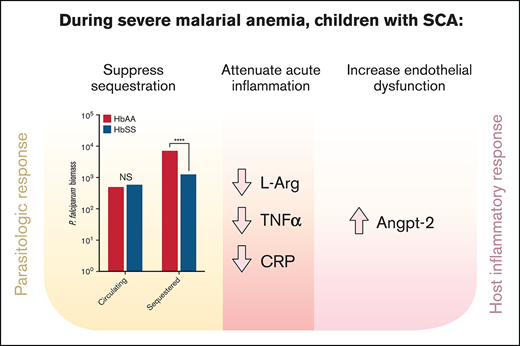
During severe anemia with malaria, children with SCA have decreased parasite burden and inflammation but more endothelial dysregulation.
In children with SCA, host response to malaria with severe anemia may reduce acute disease severity but increase long-term sequelae risk.
Abstract
Plasmodium falciparum malaria causes morbidity and mortality in African children with sickle cell anemia (SCA), but comparisons of host responses to P falciparum between children with SCA (homozygous sickle cell disease/hemoglobin SS [HbSS]) and normal hemoglobin genotype/hemoglobin AA (HbAA) are limited. We assessed parasite biomass and plasma markers of inflammation and endothelial activation in children with HbAA (n = 208) or HbSS (n = 22) who presented with severe anemia and P falciparum parasitemia to Mulago Hospital in Kampala, Uganda. Genotyping was performed at study completion. No child had known SCA at enrollment. Children with HbSS did not differ from children with HbAA in peripheral parasite density, but had significantly lower sequestered parasite biomass. Children with HbSS had greater leukocytosis but significantly lower concentrations of several plasma inflammatory cytokines, including tumor necrosis factor α (TNF-α). In contrast, children with HbSS had threefold greater concentrations of angiopoietin-2 (Angpt-2), a marker of endothelial dysregulation associated with mortality in severe malaria. Lower TNF-α concentrations were associated with increased risk of postdischarge mortality or readmission, whereas higher Angpt-2 concentrations were associated with increased risk of recurrent clinical malaria. Children with SCA have decreased parasite sequestration and inflammation but increased endothelial dysregulation during severe anemia with P falciparum parasitemia, which may ameliorate acute infectious complications but predispose to harmful long-term sequelae.
Introduction
Sickle cell anemia (SCA) affects 25 million people worldwide, including >300 000 newborn children each year.1-3 Most of these children live in sub-Saharan Africa. SCA is highly prevalent in sub-Saharan Africa because of the protective effect of the sickle cell trait/hemoglobin AS (HbAS), the heterozygous sickle gene mutation, against malaria,4-6 particularly, severe malaria.4,6-10 Thus, SCA primarily affects African regions with historically high malaria transmission.
Malaria is considered a major cause of morbidity and death in African children with SCA, but data on the incidence of malaria and of deaths from malaria in children with SCA are limited. Two studies with small numbers of children with SCA and severe malaria in Kenya and Tanzania (n = 5 and n = 21, respectively) suggested that children with SCA have a high mortality rate with severe malaria.11,12 However, a recent systematic review found that children with homozygous sickle cell disease/hemoglobin SS (HbSS) do not have an increased risk of developing uncomplicated malaria,13 and recent studies of children with SCA and severe malaria in Kenya (38 children with severe malarial anemia [SMA]14 ) and Uganda (5 children with severe malaria15 and separately 22 children with SMA16 ) all showed no mortality.
The controversy surrounding risk of mortality in children with HbSS and severe malaria underscores how little is currently known of the pathogenesis and host responses in severe malaria in individuals with HbSS compared with normal hemoglobin genotype/hemoglobin AA (HbAA). Specific factors associated with severe disease and mortality in severe malaria, including the proinflammatory cytokine tumor necrosis factor α (TNF-α) and components of the angiopoietin (Angpt)–Tie-2 system, are also associated with clinical severity in SCA.17-23 However, there have been no studies to date comparing these and other host responses in children with HbSS vs HbAA during episodes of severe anemia with Plasmodium falciparum parasitemia, or how the host response may alter parasite sequestration and density. In the present study, we assessed differences in children with HbSS vs HbAA in parasite biomass and markers of inflammation and endothelial activation during severe anemia with P falciparum parasitemia to better determine how SCA affects the host response to P falciparum during severe malaria.
Methods
Study participants
The study was performed at Mulago Hospital, Kampala, Uganda. Participants were enrolled in a study assessing neurodevelopmental impairment in severe malaria, conducted from November 2008 to December 2013. Children 18 months to 12 years of age with cerebral malaria (CM) or SMA meeting study criteria were enrolled from the Acute Care Unit at Mulago Hospital. Community children were recruited from the extended family or nearby neighborhood of children with CM or SMA. A total of 267 children with CM, 232 children with SMA, and 216 community children with no acute illness were enrolled in the study. The age, sex, and presence of HbAA, HbAS, or HbSS in each group is summarized in supplemental Table 1. Enrollment criteria for children with CM and the community children were previously published.24 In this study, we focus on the children with SMA because 22 children with SMA had HbSS compared with 1 with CM and no community children. SMA was defined per the Uganda Ministry of Health National Guidelines25 as P falciparum parasitemia on blood smear in a child with a hemoglobin level ≤ 5.0 g/dL. Children with SMA were excluded if they had impaired consciousness at the time of examination, other clinical evidence of central nervous system disease, or if they had >1 seizure before admission. Exclusion criteria for all groups included known chronic illness requiring medical care; developmental delay; prior history of coma, head trauma, or cerebral palsy; or prior hospitalization for malnutrition. Twenty-five children were excluded for prior diagnosis of SCA at the time of admission for SMA or CM.
Clinical management
Children were managed according to Ugandan Ministry of Health treatment guidelines for malaria, which at time included IV quinine or artesunate followed by oral quinine or artemether-lumefantrine. All children with SMA received a blood transfusion (20 mL/kg whole blood or 10 mL/kg packed red blood cells [RBCs]).
Hemoglobin S testing
β-globin genotyping was performed on stored samples after study completion. Genomic DNA was isolated from whole-blood samples using the DNeasy Blood and tissue kit (Qiagen, Valencia, CA). The locus of interest was amplified using specific primers.26,27 Children with HbSS were referred to the Mulago Hospital Sickle Cell Clinic, where HbSS was confirmed by hemoglobin electrophoresis.
Blood chemistries and immunoassays
Plasmodium infection was diagnosed by microscopy on Giemsa-stained peripheral blood smears; parasitemia was counted using standard procedures. Blood chemistries were performed by the Advanced Research and Diagnostic Laboratory at the University of Minnesota. Complete blood counts were performed by the clinical laboratory at Mulago Hospital. Biomarkers were detected using enzyme-linked immunosorbent assays (ELISAs) using plasma isolated from whole blood collected in EDTA or lithium-heparin tubes. Prior to analysis, samples were stored at −80°C. Plasma P falciparum histidine-rich protein-2 (PfHRP-2) was quantitated using the Malaria Ag CELISA (Cellabs, Brookvale, Australia), total parasite biomass was estimated as Ptot = 7.3 × PfHRP-2 [g/L] × (1 – hematocrit) × body weight [kg] × 1013; circulating biomass was estimated as Pcirc = parasites/µL × 106 × 0.08 L/kg × body weight (blood volume); and sequestered biomass was calculated as Pseq = Ptot – Pcirc as previously described.28 Plasma TNF-α and interleukin-6 (IL-6) concentrations were determined using a 2-plex assay from EMD Millipore (Billerica, MA). All other cytokines were measured using a Bio-Plex Human Cytokine 17-plex Assay (Bio-Rad, Hercules, CA). von Willebrand Factor (VWF) was measured using the READDS VWF:Ag ELISA (Corgenix Inc, Broomfield, CO) and expressed as a relative percentage concentration as derived from reference plasma. Angpt-1 and Angpt-2 were detected in plasma diluted 1/5 using DuoSet ELISA (R&D Systems, Minneapolis, MN). Soluble adhesins (sICAM-1, sVCAM-1, P-selectin, sE-Selectin) were measured by cytometric bead assay (R&D Systems) using plasma diluted 1/300. Soluble endothelial protein C receptor (sEPCR) was detected via Asserachrom immunoassay against the sEPCR PC-binding site (Stago, France). C-reactive protein (CRP) and α-1-acid glycoprotein (AGP) were measured by the Milliplex MAP human cytokine magnetic bead kit (EMD Millipore) and Quantikine ELISA (R&D Systems), respectively. Plasma asymmetric dimethylarginine (ADMA) was measured by ELISA (DLD Diagnostika GmbH, Hamburg, Germany), l-arginine by the Hitachi Amino Acid Analyzer (Hitachi High-Tech Co, Nashville, TN), and total nitric oxide (NO) was measured by fluorimetric analysis (Calbiochem, San Diego, CA) via treatment with nitrate reductase and fluorescence conversion and detection of total nitrite (total NO) via 2,3-diaminonapthalene.
Statistical analysis
Statistical analysis was performed in STATA 15 (Stata Corporation, College Station, TX). Categorical variables and continuous parametric and nonparametric continuous variables were compared with the χ2 test, Student t test, and Wilcoxon rank-sum test, respectively. Linear regression models were used to adjust for age, with log-transformation (base 10) of variables not normally distributed. The false discovery rate (FDR) was set at 0.05. The relationship between Angpt-2 or TNF-α and risk of readmission or death or risk of clinical malaria was analyzed using Cox proportional hazards regression, with adjustment for age and hemoglobin genotype.
Ethical review
Written informed consent was obtained from parents or guardians of study participants. Institutional review boards for human studies at Makerere University and the University of Minnesota granted ethical approval for the study. Additional regulatory approval was obtained from the Uganda National Council of Science and Technology.
Results
Demographic and clinical characteristics
Among the 232 children admitted for Uganda Ministry of Health–defined SMA (severe anemia [hemoglobin, ≤5 g/dL] with P falciparum parasitemia) who had samples available for genotyping, 208 children had HbAA and 22 had the SCA HbSS genotype. Two children with SMA had HbAS and were omitted from this analysis. Demographic characteristics are summarized in Table 1. Children with HbSS were significantly older than children with HbAA, so all comparisons were adjusted for age. Clinical characteristics of children with HbSS and HbAA were similar (Table 1). During admission, there were no deaths in the 22 children with HbSS, and 1 death in the 208 children with HbAA. No child with HbSS was taking hydroxyurea.
Clinical and demographic characteristics of children with severe anemia and P falciparum parasitemia, by hemoglobin genotype
| Baseline characteristics . | HbSS, n = 22 . | HbAA, n = 208 . | P*,† . |
|---|---|---|---|
| Demographic | |||
| No. of male patients (%) | 13 (59.1) | 127 (61.1) | .86 |
| Median age (IQR), y | 4.6 (3.7, 5.7) | 2.7 (2.0, 4.1) | <.001 |
| Mean height-for-age z-score (±SD) | −0.51 ± 0.85 (n = 21) | −1.2 ± 1.4 (n = 205) | .03 |
| Mean weight-for-age z-score (±SD) | −1.0 ± −0.9 | −1.5 ± 1.1 | .04 |
| Clinical | |||
| Median admission temperature (IQR), °C | 37.7 (37.0, 38.4) | 37.7 (36.8, 38.5) | .83 |
| Mean heart rate ± SD, beats/min | 144 ± 23.7 | 149 ± 20.9 | .27 |
| Mean systolic blood pressure ± SD, mm Hg | 95.2 ± 10.7 | 90.5 ± 13.3 | .05 |
| Median diastolic blood pressure (IQR), mm Hg | 56 (46, 66) | 55 (45, 61) | .51 |
| Mean respiratory rate ± SD, breaths/min | 43.8 ± 13.7 | 45.8 ± 13.4 (n = 207) | .50 |
| Mean O2 saturation‡ ± SD, % | 96.2 ± 2.6 | 96.4 ± 3.6 (n = 207) | .80 |
| Respiratory distress,§ % (95% CI) | 27.3 (8.7, 45.9) | 27.9 (21.7, 34.0) | .96 |
| Deep acidotic breathing, % (95% CI) | 9.1 (0.0, 22) | 6.7 (3.3, 10) | .70 |
| Capillary refill > 2 s, % (95% CI) | 9.1 (0.0, 22) | 19.7 (14.3, 25.2) | .22 |
| Palpable spleen (%) | 11 (50.0) | 136 (65.4) | .15 |
| Baseline characteristics . | HbSS, n = 22 . | HbAA, n = 208 . | P*,† . |
|---|---|---|---|
| Demographic | |||
| No. of male patients (%) | 13 (59.1) | 127 (61.1) | .86 |
| Median age (IQR), y | 4.6 (3.7, 5.7) | 2.7 (2.0, 4.1) | <.001 |
| Mean height-for-age z-score (±SD) | −0.51 ± 0.85 (n = 21) | −1.2 ± 1.4 (n = 205) | .03 |
| Mean weight-for-age z-score (±SD) | −1.0 ± −0.9 | −1.5 ± 1.1 | .04 |
| Clinical | |||
| Median admission temperature (IQR), °C | 37.7 (37.0, 38.4) | 37.7 (36.8, 38.5) | .83 |
| Mean heart rate ± SD, beats/min | 144 ± 23.7 | 149 ± 20.9 | .27 |
| Mean systolic blood pressure ± SD, mm Hg | 95.2 ± 10.7 | 90.5 ± 13.3 | .05 |
| Median diastolic blood pressure (IQR), mm Hg | 56 (46, 66) | 55 (45, 61) | .51 |
| Mean respiratory rate ± SD, breaths/min | 43.8 ± 13.7 | 45.8 ± 13.4 (n = 207) | .50 |
| Mean O2 saturation‡ ± SD, % | 96.2 ± 2.6 | 96.4 ± 3.6 (n = 207) | .80 |
| Respiratory distress,§ % (95% CI) | 27.3 (8.7, 45.9) | 27.9 (21.7, 34.0) | .96 |
| Deep acidotic breathing, % (95% CI) | 9.1 (0.0, 22) | 6.7 (3.3, 10) | .70 |
| Capillary refill > 2 s, % (95% CI) | 9.1 (0.0, 22) | 19.7 (14.3, 25.2) | .22 |
| Palpable spleen (%) | 11 (50.0) | 136 (65.4) | .15 |
For tests that did not include all participants, the n is listed next to the result.
IQR, interquartile range.
Determined by use of χ2 for categorical data, Student t test for parametric continuous data, and Wilcoxon rank-sum test for nonparametric continuous data, as appropriate.
P < .05 regarded as significant. Bold P value represents significant differences.
O2 saturation is peripheral capillary oxygen saturation.
Respiratory distress includes nasal flaring, intercostal recessions, chest indrawing, and/or deep acidotic breathing.
Clinical laboratory values
Hemoglobin concentrations were similar in children with SMA regardless of hemoglobin genotype (Table 2). The white blood cell count (WBC) in children with HbSS was almost threefold higher than in HbAA (mean WBC [standard deviation (SD)], 32.5 × 103/µL [16.0] compared with 12.9 × 103/µL [8.2]; P < .001). Other clinical laboratory differences included increased mean corpuscular volume (MCV) and RBC distribution width (RDW); higher platelet counts, plasma bilirubin, and albumin; and lower lactate dehydrogenase (LDH), creatinine, and blood urea nitrogen (BUN) concentrations in children with HbSS compared with children with HbAA (Table 2). Among 18 children with SCA who had blood cultures drawn during admission, 1 (5.6%) had concomitant bacteremia, compared with 22 of 178 children (12.3%) without SCA. No child with SCA had evidence of helminthic infection, whereas 10 of 208 children (4.8%) without SCA did. HIV was detected in 6 of 205 children without SCA (2.9%) and no child with SCA (Table 2).
Clinical laboratory values in children with severe anemia and P falciparum parasitemia by sickle cell genotype
| . | HbSS, n = 22 . | HbAA, n = 208 . | P*,† . | β coefficient‡,§ (95% CI) . | P,† adjusted for age . |
|---|---|---|---|---|---|
| Complete blood count (mean ± SD) | |||||
| Hemoglobin, g/dL | 3.7 (0.6) | 3.7 (0.9) (n = 208) | .71 | −0.04 (−0.4, 0.4) | .85 |
| Hematocrit, % | 12.5 (2.5) | 13.7 (5.8) (n = 206) | .35 | −1.7 (−4.2, 0.9) | .21 |
| MCV, fL | 96.9 (11.7) | 82.4 (10.3) (n = 206) | <.001 | 14.4 (9.5, 19.2) | <.001 |
| RDW, % | 21.4 (5.17) | 16.4 (3.7) (n = 202) | <.001 | 6.2 (4.4, 7.9) | <.001 |
| WBC count, ×103/μL | 32.5 (16.0) (n = 21) | 12.9 (8.2) (n = 206) | <.001 | 20.1 (15.8, 24.5) | <.001 |
| % Neutrophils | 40.5 (12.8) (n = 20) | 46.8 (13.7) (n = 206) | .05 | −9.2 (−15.5, −2.8) | .005 |
| % Lymphocytes | 48.7 (15.1) (n = 20) | 41.0 (12.4) (n = 206) | .01 | 10.2 (4.2, 16.1) | .001 |
| % Monocytes | 8.7 (4.0) (n = 20) | 10.4 (5.5) (n = 206) | .16 | −1.3 (−3.9, 1.3) | .32 |
| % Basophils | 1.7 (1.4) (n = 14) | 1.2 (1.3) (n = 161) | .14 | 0.5 (−0.2, 1.2) | .18 |
| % Eosinophils | 1.3 (2.1) (n = 14) | 1.1 (1.1) (n = 161) | .48 | 0.3 (−0.4, 1.0) | .39 |
| Platelet count, x103/μL | 273 (203) | 175 (126) (n = 206) | .002 | 86.7 (24.1, 149.4) | .007 |
| Plasma markers of hemolysis (median, IQR) | |||||
| LDH, U/L | 639 (469, 769) (n = 21) | 772 (639, 995) (n = 194) | .02 | −0.1 (−0.2, −0.004)§ | .04 |
| Haptoglobin, mg/dL | 0.1 (0.1, 0.1) (n = 21) | 0.1 (0.1, 0.1) (n = 195) | .64 | 0.2 (−0.2, 0.4)§ | .52 |
| Total bilirubin, mg/dL | 2.7 (1.6, 5) (n = 21) | 1.2 (0.7, 1.9) (n = 195) | <.001 | 0.3 (0.2, 0.5)§ | <.001 |
| Renal function (median, IQR) | |||||
| Creatinine, mg/dL | 0.35 (0.27, 0.43) (n = 21) | 0.35 (0.28, 0.47) (n = 196) | .73 | −0.08 (−0.16, −0.004)§ | .04 |
| BUN, mg/dL | 9.0 (7.0, 13) | 14 (9.0, 20) (n = 204) | .007 | −0.2 (−0.3, −0.1)§ | .001 |
| Other clinical laboratory values (median, IQR) | |||||
| Albumin, g/dL | 3.3 (2.9, 3.5) | 2.6 (2.3, 2.9) (n = 203) | <.001 | 0.09 (0.05, 0.12)§ | <.001 |
| Lactate, mmol/L | 3.9 (2.2, 7.0) (n = 20) | 5.0 (3.0, 8.3) (n = 194) | .21 | −0.7 (−2.5, 1.0) | .43 |
| Blood culture positivity, n (%) | 1 (5.5) (n = 18) | 22 (12.4) (n = 178) | .64 | −0.03 (−0.2, 0.1) | .75 |
| Stool helminthic infection, n (%) | 0 (0) (n = 160 | 9 (6.8) (n = 132) | .60 | −0.08 (−0.2, 0.05) | .22 |
| HIV positivity, n (%) | 0 (0) (n = 22) | 6 (2.9) (n = 205) | 1.0 | −0.04 (−0.1, 0.04) | .33 |
| Parasite density and PfHRP-2 concentration (median, IQR) | |||||
| P falciparum HRP-2, ng/mL | 197 (6, 469) | 1050 (468, 2840) (n = 205) | <.001 | −1.0§ (−1.3, −0.6)§ | <.001 |
| Parasite density in peripheral blood, parasites ×103/μL | 42 (3.0, 141) (n = 21) | 50 (15, 230) (n = 205) | .18 | −0.3 (−0.7, 0.2)§ | .29 |
| Estimated parasite biomass | |||||
| Circulating (×109) | 60 (3.00, 189) (n = 21) | 51 (14, 207) (n = 205) | .42 | −0.3 (−0.7, 0.2) | .31 |
| Sequestered (×109) | 130 (0.00, 325) (n = 21) | 731 (190, 1921) (n = 202) | <.001 | −0.5 (−0.9, −0.1) | .01 |
| Total (×109) | 238 (6.00, 445) | 835 (327, 2043) (n = 203) | <.001 | −0.6 (−0.9, −0.3) | <.001 |
| . | HbSS, n = 22 . | HbAA, n = 208 . | P*,† . | β coefficient‡,§ (95% CI) . | P,† adjusted for age . |
|---|---|---|---|---|---|
| Complete blood count (mean ± SD) | |||||
| Hemoglobin, g/dL | 3.7 (0.6) | 3.7 (0.9) (n = 208) | .71 | −0.04 (−0.4, 0.4) | .85 |
| Hematocrit, % | 12.5 (2.5) | 13.7 (5.8) (n = 206) | .35 | −1.7 (−4.2, 0.9) | .21 |
| MCV, fL | 96.9 (11.7) | 82.4 (10.3) (n = 206) | <.001 | 14.4 (9.5, 19.2) | <.001 |
| RDW, % | 21.4 (5.17) | 16.4 (3.7) (n = 202) | <.001 | 6.2 (4.4, 7.9) | <.001 |
| WBC count, ×103/μL | 32.5 (16.0) (n = 21) | 12.9 (8.2) (n = 206) | <.001 | 20.1 (15.8, 24.5) | <.001 |
| % Neutrophils | 40.5 (12.8) (n = 20) | 46.8 (13.7) (n = 206) | .05 | −9.2 (−15.5, −2.8) | .005 |
| % Lymphocytes | 48.7 (15.1) (n = 20) | 41.0 (12.4) (n = 206) | .01 | 10.2 (4.2, 16.1) | .001 |
| % Monocytes | 8.7 (4.0) (n = 20) | 10.4 (5.5) (n = 206) | .16 | −1.3 (−3.9, 1.3) | .32 |
| % Basophils | 1.7 (1.4) (n = 14) | 1.2 (1.3) (n = 161) | .14 | 0.5 (−0.2, 1.2) | .18 |
| % Eosinophils | 1.3 (2.1) (n = 14) | 1.1 (1.1) (n = 161) | .48 | 0.3 (−0.4, 1.0) | .39 |
| Platelet count, x103/μL | 273 (203) | 175 (126) (n = 206) | .002 | 86.7 (24.1, 149.4) | .007 |
| Plasma markers of hemolysis (median, IQR) | |||||
| LDH, U/L | 639 (469, 769) (n = 21) | 772 (639, 995) (n = 194) | .02 | −0.1 (−0.2, −0.004)§ | .04 |
| Haptoglobin, mg/dL | 0.1 (0.1, 0.1) (n = 21) | 0.1 (0.1, 0.1) (n = 195) | .64 | 0.2 (−0.2, 0.4)§ | .52 |
| Total bilirubin, mg/dL | 2.7 (1.6, 5) (n = 21) | 1.2 (0.7, 1.9) (n = 195) | <.001 | 0.3 (0.2, 0.5)§ | <.001 |
| Renal function (median, IQR) | |||||
| Creatinine, mg/dL | 0.35 (0.27, 0.43) (n = 21) | 0.35 (0.28, 0.47) (n = 196) | .73 | −0.08 (−0.16, −0.004)§ | .04 |
| BUN, mg/dL | 9.0 (7.0, 13) | 14 (9.0, 20) (n = 204) | .007 | −0.2 (−0.3, −0.1)§ | .001 |
| Other clinical laboratory values (median, IQR) | |||||
| Albumin, g/dL | 3.3 (2.9, 3.5) | 2.6 (2.3, 2.9) (n = 203) | <.001 | 0.09 (0.05, 0.12)§ | <.001 |
| Lactate, mmol/L | 3.9 (2.2, 7.0) (n = 20) | 5.0 (3.0, 8.3) (n = 194) | .21 | −0.7 (−2.5, 1.0) | .43 |
| Blood culture positivity, n (%) | 1 (5.5) (n = 18) | 22 (12.4) (n = 178) | .64 | −0.03 (−0.2, 0.1) | .75 |
| Stool helminthic infection, n (%) | 0 (0) (n = 160 | 9 (6.8) (n = 132) | .60 | −0.08 (−0.2, 0.05) | .22 |
| HIV positivity, n (%) | 0 (0) (n = 22) | 6 (2.9) (n = 205) | 1.0 | −0.04 (−0.1, 0.04) | .33 |
| Parasite density and PfHRP-2 concentration (median, IQR) | |||||
| P falciparum HRP-2, ng/mL | 197 (6, 469) | 1050 (468, 2840) (n = 205) | <.001 | −1.0§ (−1.3, −0.6)§ | <.001 |
| Parasite density in peripheral blood, parasites ×103/μL | 42 (3.0, 141) (n = 21) | 50 (15, 230) (n = 205) | .18 | −0.3 (−0.7, 0.2)§ | .29 |
| Estimated parasite biomass | |||||
| Circulating (×109) | 60 (3.00, 189) (n = 21) | 51 (14, 207) (n = 205) | .42 | −0.3 (−0.7, 0.2) | .31 |
| Sequestered (×109) | 130 (0.00, 325) (n = 21) | 731 (190, 1921) (n = 202) | <.001 | −0.5 (−0.9, −0.1) | .01 |
| Total (×109) | 238 (6.00, 445) | 835 (327, 2043) (n = 203) | <.001 | −0.6 (−0.9, −0.3) | <.001 |
For tests that did not include all participants, the n is listed next to the result.
HRP-2, histidine-rich protein-2; IQR, interquartile range.
Determined by use of χ2 or Fisher exact test for categorical data where appropriate, Student t test for parametric continuous data, and Wilcoxon rank-sum test for nonparametric continuous data, as appropriate.
Significance determined with FDR = 0.05 correction for multiple comparisons. Bold P values represent significant differences.
β coefficient of linear regression controlling for age.
Log10-transformed values used for linear regression.
Risk of postdischarge clinical malaria, readmission, or death
There was no difference in the risks of postdischarge clinical malaria or postdischarge readmission or death between children with HbSS vs HbAA (hazard ratio [HR] [95% confidence interval (CI)], malaria: 1.13 [0.60, 2.10]; readmission or death: 1.65 [0.78, 3.47]).
Peripheral parasite density and estimated total, circulating, and sequestered parasite biomass
Children with HbSS had similar levels of peripheral parasitemia and estimated circulating P falciparum biomass to children with HbAA, but lower P falciparum HRP-2 concentrations (197 vs 1051 ng/mL; P < .001) and estimated total (238 × 109 vs 835 × 109; P < .001) and sequestered (130 × 109 vs 731 × 109; P < .001) parasite biomass (Table 2; supplemental Figure 1). Hemoglobin concentration inversely correlated with total parasite biomass (log10-transformed) in children with HbAA (β = 0.33 [95% CI, 0.18, 0.48]; P < .001), but was unrelated to total parasite biomass in children with HbSS (β = 0.05 [95% CI, −0.12, 0.22]; P = .5).
Cytokines and inflammatory cytokines
Although children with SCA presented with greater leukocytosis than children with HbAA, children with SCA had significantly lower levels of the inflammatory biomarkers AGP (1350 vs 2128 μg/mL; P < .001) and CRP (25.0 vs 44.0 mg/dL; P < .001) than children with HbAA (Table 2). Similarly, concentrations of the immune activator TNF-α and the proinflammatory chemokines monocyte chemoattract protein 1 (MCP-1) and macrophage inflammatory protein 1β (MIP-1β) were lower in children with HbSS compared with children with HbAA (Table 3). PfHRP-2, a marker of total parasite biomass, correlated strongly with CRP concentrations (β = 3.14 [95% CI, 1.94, 4.34]; P < .001), as well as the 3 proinflammatory cytokines and chemokines (TNF-α, MCP-1, MIP-1β, all P < .001).
Comparison of host immune and inflammatory response to P falciparum in children with severe anemia and P falciparum parasitemia by sickle cell genotype
| Cell biomarker . | HbSS, Median (IQR), n = 22 . | HbAA, Median (IQR), n = 208 . | P*,† . | β coefficient‡ (95% CI), adjusted for age . | P† . |
|---|---|---|---|---|---|
| Markers of inflammation | |||||
| C-reactive protein, mg/dL | 25.0 (16.7, 34.5) | 44.0 (25.2, 64.7) (n = 192) | <.001 | −0.2 (−0.4, 0.0) | .03 |
| α-1 glycoprotein, μcg/mL | 1350 (1048, 1887) (n = 19) | 2128 (1726, 2601) (n = 170) | <.001 | −0.2 (−0.2, −0.1) | <.001 |
| Plasma concentrations of pro- and anti-inflammatory cytokines and chemokines | |||||
| G-CSF, pg/mL | 39.8 (17.3, 74.1) (n = 19) | 60.0 (37.4, 102.3) (n = 172) | .02 | −0.2 (−0.4, 0.0) | .10 |
| Interferon-γ, pg/mL | 115 (67.9, 178) (n = 19) | 91.7 (57.5, 158) (n = 172) | .29 | 0.1 (−0.2, 0.3) | .60 |
| IL-1β, pg/mL | 2.5 (0.5, 4.4) (n = 19) | 2.9 (1.9, 4.1) (n = 172) | .75 | −0.1 (−0.3, 0.1) | .38 |
| IL-1 receptor α, pg/mL | 506 (263, 811) (n = 19) | 607 (323, 1610) (n = 172) | .12 | −0.1 (−0.4, 0.1) | .40 |
| IL-4, pg/mL | 2.8 (1.3, 6.0) (n = 19) | 2.5 (1.1, 4.4) (n = 172) | .60 | 0.0 (−0.3, 0.3) | .96 |
| IL-6, pg/mL | 36.8 (5.4, 68.9) (n = 21) | 40.2 (13.7, 105) (n = 195) | .39 | −0.2 (−0.5, 0.2) | .36 |
| IL-8, pg/mL | 22.9 (15.8, 37.0) (n = 19) | 30.5 (19.8, 44.9) (n = 172) | .14 | −0.1 (−0.3, 0.1) | .29 |
| IL-10, pg/mL | 53.7 (16.2, 118) (n = 19) | 102 (35.3, 263) (n = 172) | .05 | −0.2 (−0.5, 0.2) | .27 |
| IL-12 p70, pg/mL | 17.5 (9.8, 26.3) (n = 19) | 22.2 (13.3, 35.8) (n = 172) | .11 | −0.1 (−0.4, 0.1) | .23 |
| MCP-1, pg/mL | 15.4 (11.1, 25.8) (n = 19) | 33.4 (18.2, 87.9) (n = 172) | <.001 | −0.4 (−0.7, −0.1) | .02 |
| MIP-1α, pg/mL | 6.7 (4.1, 11) (n = 19) | 6.4 (4.2, 10) (n = 172) | .90 | 0.0 (−0.3, 0.2) | .75 |
| MIP-1β, pg/mL | 181 (71.9, 313) (n = 19) | 285 (184, 425) (n = 172) | .004 | −0.2 (−0.4, −0.1) | <.001 |
| Platelet factor 4 (platelet-free plasma), ng/mL | 878 (381, 2100) (n = 19) | 709 (318, 1530) (n = 166) | .20 | 0.2 (−0.1, 0.5) | .14 |
| RANTES, pg/mL | 3790 (1530, 5470) (n = 19) | 3480 (1750, 6860) (n = 172) | .61 | −0.1 (−0.3, 0.1) | .48 |
| TNF-α, pg/mL | 41.7 (21.5, 64.1) (n = 21) | 87.6 (51.1, 141) (n = 195) | <.001 | −0.3 (−0.6, −0.1) | .006 |
| Plasma concentrations of markers of endothelial activation | |||||
| VWF, % of normal | 104 (72.6, 198) (n = 19) | 137 (69.2, 239) (n = 146) | .29 | −0.1 (−0.3, 0.1) | .41 |
| sVCAM-1, ng/mL | 3050 (2070, 5720) (n = 19) | 3260 (2550, 4800) (n = 171) | .51 | 0.0 (−0.1, 0.1) | .78 |
| sICAM-1, ng/mL | 370 (230, 1230) (n = 19) | 727 (300, 1400) (n = 172) | .20 | 0.0 (−0.4, 0.3) | .84 |
| sE-selectin, ng/mL | 184 (117, 245) (n = 19) | 190 (135, 258) (n = 172) | .88 | 0.0 (−0.1, 0.1) | .54 |
| P-selectin, ng/mL | 58.3 (47.9, 85.8) (n = 9) | 45.1 (35.4, 66.3) (n = 99) | .22 | 0.1 (−0.1, 0.3) | .23 |
| sEPCR, ng/mL | 68.9 (60.1, 102) (n = 20) | 97.2 (72.5, 250) (n = 187) | .02 | −0.1 (−0.2, 0.00) | .06 |
| Plasma concentrations of markers of angiogenesis | |||||
| Angpt-1, ng/mL | 5.4 (2.8, 7.6) (n = 18) | 4.2 (1.8, 7.5) (n = 172) | .28 | 0.2 (−0.1, 0.4) | .24 |
| Angpt-2, ng/mL | 5.0 (3.6, 6.7) (n = 18) | 1.7 (0.9, 2.8) (n = 172) | <.001 | 0.3 (0.1, 0.5) | .01 |
| VEGF, pg/mL | 45.0 (24.3, 138) (n = 19) | 62.3 (34.1, 93.6) (n = 172) | .67 | 0.0 (−0.2, 0.2) | .92 |
| PDGF-bb, pg/mL | 2080 (617, 2930) (n = 19) | 899 (446, 1730) (n = 172) | .04 | 0.2 (0.0, 0.5) | .04 |
| Plasma concentrations of NO and regulators | |||||
| NO concentration, μmol/L | 44.1 (31.4, 87.6) (n = 18) | 53.1 (31.7, 90.6) (n = 164) | .88 | 0.0 (−0.2, 0.2) | .59 |
| ADMA, μmol/L | 0.9 (0.7, 1.1) (n = 22) | 0.7 (0.6, 0.9) (n = 204) | .005 | 0.07 (0.0, 0.1) | .06 |
| l-arginine, µmol/L | 20 (20, 30) (n = 18) | 30 (20, 30) (n = 153) | .05 | −0.1 (−0.2, 0.0) | .007 |
| l-arginine/ADMA§ | 24.2 (21.6, 30.3) (n = 18) | 34.7 (25.9, 47.6) (n = 149) | .002 | −13.5 (−22.6, −4.4) | .004 |
| Cell biomarker . | HbSS, Median (IQR), n = 22 . | HbAA, Median (IQR), n = 208 . | P*,† . | β coefficient‡ (95% CI), adjusted for age . | P† . |
|---|---|---|---|---|---|
| Markers of inflammation | |||||
| C-reactive protein, mg/dL | 25.0 (16.7, 34.5) | 44.0 (25.2, 64.7) (n = 192) | <.001 | −0.2 (−0.4, 0.0) | .03 |
| α-1 glycoprotein, μcg/mL | 1350 (1048, 1887) (n = 19) | 2128 (1726, 2601) (n = 170) | <.001 | −0.2 (−0.2, −0.1) | <.001 |
| Plasma concentrations of pro- and anti-inflammatory cytokines and chemokines | |||||
| G-CSF, pg/mL | 39.8 (17.3, 74.1) (n = 19) | 60.0 (37.4, 102.3) (n = 172) | .02 | −0.2 (−0.4, 0.0) | .10 |
| Interferon-γ, pg/mL | 115 (67.9, 178) (n = 19) | 91.7 (57.5, 158) (n = 172) | .29 | 0.1 (−0.2, 0.3) | .60 |
| IL-1β, pg/mL | 2.5 (0.5, 4.4) (n = 19) | 2.9 (1.9, 4.1) (n = 172) | .75 | −0.1 (−0.3, 0.1) | .38 |
| IL-1 receptor α, pg/mL | 506 (263, 811) (n = 19) | 607 (323, 1610) (n = 172) | .12 | −0.1 (−0.4, 0.1) | .40 |
| IL-4, pg/mL | 2.8 (1.3, 6.0) (n = 19) | 2.5 (1.1, 4.4) (n = 172) | .60 | 0.0 (−0.3, 0.3) | .96 |
| IL-6, pg/mL | 36.8 (5.4, 68.9) (n = 21) | 40.2 (13.7, 105) (n = 195) | .39 | −0.2 (−0.5, 0.2) | .36 |
| IL-8, pg/mL | 22.9 (15.8, 37.0) (n = 19) | 30.5 (19.8, 44.9) (n = 172) | .14 | −0.1 (−0.3, 0.1) | .29 |
| IL-10, pg/mL | 53.7 (16.2, 118) (n = 19) | 102 (35.3, 263) (n = 172) | .05 | −0.2 (−0.5, 0.2) | .27 |
| IL-12 p70, pg/mL | 17.5 (9.8, 26.3) (n = 19) | 22.2 (13.3, 35.8) (n = 172) | .11 | −0.1 (−0.4, 0.1) | .23 |
| MCP-1, pg/mL | 15.4 (11.1, 25.8) (n = 19) | 33.4 (18.2, 87.9) (n = 172) | <.001 | −0.4 (−0.7, −0.1) | .02 |
| MIP-1α, pg/mL | 6.7 (4.1, 11) (n = 19) | 6.4 (4.2, 10) (n = 172) | .90 | 0.0 (−0.3, 0.2) | .75 |
| MIP-1β, pg/mL | 181 (71.9, 313) (n = 19) | 285 (184, 425) (n = 172) | .004 | −0.2 (−0.4, −0.1) | <.001 |
| Platelet factor 4 (platelet-free plasma), ng/mL | 878 (381, 2100) (n = 19) | 709 (318, 1530) (n = 166) | .20 | 0.2 (−0.1, 0.5) | .14 |
| RANTES, pg/mL | 3790 (1530, 5470) (n = 19) | 3480 (1750, 6860) (n = 172) | .61 | −0.1 (−0.3, 0.1) | .48 |
| TNF-α, pg/mL | 41.7 (21.5, 64.1) (n = 21) | 87.6 (51.1, 141) (n = 195) | <.001 | −0.3 (−0.6, −0.1) | .006 |
| Plasma concentrations of markers of endothelial activation | |||||
| VWF, % of normal | 104 (72.6, 198) (n = 19) | 137 (69.2, 239) (n = 146) | .29 | −0.1 (−0.3, 0.1) | .41 |
| sVCAM-1, ng/mL | 3050 (2070, 5720) (n = 19) | 3260 (2550, 4800) (n = 171) | .51 | 0.0 (−0.1, 0.1) | .78 |
| sICAM-1, ng/mL | 370 (230, 1230) (n = 19) | 727 (300, 1400) (n = 172) | .20 | 0.0 (−0.4, 0.3) | .84 |
| sE-selectin, ng/mL | 184 (117, 245) (n = 19) | 190 (135, 258) (n = 172) | .88 | 0.0 (−0.1, 0.1) | .54 |
| P-selectin, ng/mL | 58.3 (47.9, 85.8) (n = 9) | 45.1 (35.4, 66.3) (n = 99) | .22 | 0.1 (−0.1, 0.3) | .23 |
| sEPCR, ng/mL | 68.9 (60.1, 102) (n = 20) | 97.2 (72.5, 250) (n = 187) | .02 | −0.1 (−0.2, 0.00) | .06 |
| Plasma concentrations of markers of angiogenesis | |||||
| Angpt-1, ng/mL | 5.4 (2.8, 7.6) (n = 18) | 4.2 (1.8, 7.5) (n = 172) | .28 | 0.2 (−0.1, 0.4) | .24 |
| Angpt-2, ng/mL | 5.0 (3.6, 6.7) (n = 18) | 1.7 (0.9, 2.8) (n = 172) | <.001 | 0.3 (0.1, 0.5) | .01 |
| VEGF, pg/mL | 45.0 (24.3, 138) (n = 19) | 62.3 (34.1, 93.6) (n = 172) | .67 | 0.0 (−0.2, 0.2) | .92 |
| PDGF-bb, pg/mL | 2080 (617, 2930) (n = 19) | 899 (446, 1730) (n = 172) | .04 | 0.2 (0.0, 0.5) | .04 |
| Plasma concentrations of NO and regulators | |||||
| NO concentration, μmol/L | 44.1 (31.4, 87.6) (n = 18) | 53.1 (31.7, 90.6) (n = 164) | .88 | 0.0 (−0.2, 0.2) | .59 |
| ADMA, μmol/L | 0.9 (0.7, 1.1) (n = 22) | 0.7 (0.6, 0.9) (n = 204) | .005 | 0.07 (0.0, 0.1) | .06 |
| l-arginine, µmol/L | 20 (20, 30) (n = 18) | 30 (20, 30) (n = 153) | .05 | −0.1 (−0.2, 0.0) | .007 |
| l-arginine/ADMA§ | 24.2 (21.6, 30.3) (n = 18) | 34.7 (25.9, 47.6) (n = 149) | .002 | −13.5 (−22.6, −4.4) | .004 |
For tests that did not include all participants, the n is listed next to the result.
Determined by use of Wilcoxon rank-sum test.
Significant P values in bold, determined by FDR = 0.05 correction for multiple comparisons.
Denotes linear regression β coefficient for log10-transformed values, adjusted for age.
Denotes linear regression performed using non-transformed values.
Markers of endothelial activation and angiogenesis
Endothelial activation occurs in SCA and severe malaria.1,19,23,29-33 Recent data have implicated the Angpt–Tie-2 system in the pathogenesis of SCA and severe malaria, where elevated Angpt-2 and decreased Angpt-1 correlate with disease severity.17-22 We quantitated levels of these and other biomarkers of endothelial activation as well as angiogenic cytokines vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF) in this study population (Table 2). Median Angpt-2 was threefold higher in the children with HbSS compared with HbAA (5.0 vs 1.7 ng/mL; P = .01; Table 2). Median plasma concentrations of sEPCR, the soluble form of Plasmodium falciparum erthrocyte membrane protein-1 (PfEMP1) binding receptors important in microvascular sequestration and the pathogenesis of CM,30,34,35 were lower in children with HbSS (adjusted P = .05).
In addition to VEGF, NO responds to tissue hypoxia, inducing vasodilation and angiogenesis.36-39 NO production requires l-arginine, and ADMA is a negative regulator of NO production. The ratio of l-arginine and ADMA reflects NO homeostasis.40 Lower l-arginine and higher ADMA levels in children with HbSS would be expected with the hypoxia and tissue damage caused by vaso-occlusion in SCA. Children with HbSS had lower l-arginine and higher ADMA concentrations (lower l-arginine/ADMA ratio) than children with HbAA (Table 3), reflecting dysregulated NO homeostasis in response to P falciparum infection in children with severe malaria and SCA.
Association of endothelial and proinflammatory factors with long-term outcomes
Severe anemia confers increased risk of death or readmission after discharge,41 and children with SMA are at risk of readmission or uncomplicated malaria after discharge.42,43 Given the differences in TNF-α, Angpt-2, and l-arginine/ADMA ratios noted here among children with SMA, and previous data linking these factors with adverse sequelae including death,44-47 we assessed associations between these factors and postdischarge death or readmission or uncomplicated malaria incidence during the 24-month follow-up period of the study (Table 4; supplemental Tables 2 and 3 ). Contrary to our expectations, higher TNF-α concentrations during SMA were associated with a lower risk of postdischarge death or readmission (HR, 0.55 [95% CI, 0.36-0.83], P = .004). Angpt-2 concentrations, also elevated in the children with SCA, were not significantly associated with altered risk for readmission or death after discharge (Table 4). However, Angpt-2 concentrations were associated with significantly increased risk of uncomplicated malaria (HR, 2.03 [95% CI, 1.14-3.59], P = .016). There was no interaction between hemoglobin genotype and TNF-α or Angpt-2 concentration on the risk of postdischarge uncomplicated malaria, readmission, or mortality.
Association of markers of inflammation and endothelial activation across all children with severe anemia and P falciparum parasitemia
| . | N obs, n event . | HR . | 95% CI . | P* . | HR, adjusted for age and Hb genotype . | 95% CI, adjusted for age and Hb genotype . | P* . |
|---|---|---|---|---|---|---|---|
| Risk of death or readmission† | |||||||
| Angpt-2, pg/mL | 186, 49 | 1.45 | 0.77, 2.72 | .25 | 1.48 | 0.75, 2.92 | .25 |
| TNF-α, pg/mL | 212, 57 | 0.54 | 0.36, 0.81 | .003 | 0.55 | 0.36, 0.83 | .004 |
| l-arg/ADMA ratio | 165, 48 | 2.24 | 0.56, 8.91 | .25 | 2.67 | 0.64, 11.1 | .18 |
| Risk of malaria outpatient visit† | |||||||
| Angpt-2, pg/mL | 186, 81 | 1.83 | 1.09, 3.06 | .02 | 2.03 | 1.14, 3.59 | .016 |
| TNF-α, pg/mL | 212, 88 | 1.29 | 0.81, 2.06 | .28 | 1.33 | 0.82, 2.15 | .25 |
| l-arg/ADMA ratio | 165, 75 | 1.44 | 0.46, 4.47 | .53 | 1.77 | 0.55, 5.72 | .34 |
| . | N obs, n event . | HR . | 95% CI . | P* . | HR, adjusted for age and Hb genotype . | 95% CI, adjusted for age and Hb genotype . | P* . |
|---|---|---|---|---|---|---|---|
| Risk of death or readmission† | |||||||
| Angpt-2, pg/mL | 186, 49 | 1.45 | 0.77, 2.72 | .25 | 1.48 | 0.75, 2.92 | .25 |
| TNF-α, pg/mL | 212, 57 | 0.54 | 0.36, 0.81 | .003 | 0.55 | 0.36, 0.83 | .004 |
| l-arg/ADMA ratio | 165, 48 | 2.24 | 0.56, 8.91 | .25 | 2.67 | 0.64, 11.1 | .18 |
| Risk of malaria outpatient visit† | |||||||
| Angpt-2, pg/mL | 186, 81 | 1.83 | 1.09, 3.06 | .02 | 2.03 | 1.14, 3.59 | .016 |
| TNF-α, pg/mL | 212, 88 | 1.29 | 0.81, 2.06 | .28 | 1.33 | 0.82, 2.15 | .25 |
| l-arg/ADMA ratio | 165, 75 | 1.44 | 0.46, 4.47 | .53 | 1.77 | 0.55, 5.72 | .34 |
Association of markers of inflammation and endothelial activation associated with SCA and risk of readmission or death and risk of malarial outpatient visit within 24 months across all children with severe anemia and P falciparum parasitemia.
N obs, number observed.
Significant P values in bold, determined by FDR = 0.05.
Cox regression is based on log10 transformation of biomarkers, so HR is increase in outcome for each log10 increase in biomarker.
Discussion
Understanding differences in pathogenesis and host responses to P falciparum is critical to determining how malaria affects children with SCA and may provide insights into how to prevent acute and long-term complications of malaria infection in children with SCA. In the present study, children with severe anemia (hemoglobin, ≤5 g/dL) and P falciparum parasitemia differed in their level of parasite sequestration, proinflammatory response, and level of endothelial cell children dysfunction according to whether they had HbSS or HbAA. Children with severe anemia and P falciparum parasitemia were enrolled and classified in the study as having SMA based on Uganda Ministry of Health criteria. However, because we cannot completely exclude other causes of anemia in children with SCA, we have termed the clinical condition “severe anemia with P falciparum parasitemia” rather than “severe malarial anemia.”
Children with severe anemia and P falciparum parasitemia, and without SCA, had similar clinical characteristics during severe malaria, but children with SCA broadly attenuated parasite biomass and inflammatory mediators. Children with SCA had higher WBCs compared with children without SCA, but concentrations of CRP48,49 and multiple proinflammatory cytokines, including TNF-α, were significantly decreased. Pronounced leukocytosis and thrombocytosis are known features of steady-state SCA, as seen in the NOHARM trial also conducted at Mulago Hospital, and thus do not directly reflect the inflammatory response due to malaria.15 Similarly, Angpt-2 was elevated and the Arg/ADMA ratio lower in children with SCA, also features of steady-state SCA that may be exacerbated by malarial infection. Across the cohort as a whole, decreased TNF-α was associated with increased risk of postadmission readmission or death, whereas increased Angpt-2 was associated with increased risk of subsequent uncomplicated clinical malaria. The findings suggest 2 possibilities: children with HbSS may express symptoms of illness at a lower parasite biomass and with less inflammation than children with HbAA, or that SCA-related mechanisms, still not fully characterized, may lead to attenuation of parasite sequestration and sequestration-driven inflammation in children with SCA compared with children without SCA. The data also hint at potential long-term consequences from severe malaria, if the associated endothelial and NO dysregulation persist after the malaria episode.
The reduction in sequestered but not circulating P falciparum biomass and concomitant decrease in proinflammatory factors in children with SCA during severe anemia with P falciparum parasitemia was surprising. A number of studies suggest that peripheral P falciparum density is lower in children with SCA than in children with HbAA.5,11,14,50-52 In 3 studies describing their calculations, each applied a standard value (ranging from an arbitrary 8 × 103/µL to the observed population mean WBC, 14.8 × 103/µL) to all patients in the cohort.11,14,50 However, during SMA, children with SCA have higher and more variable WBCs (mean [SD], 32.5 × 103/µL [16.0] in the present study). When we used a standard WBC for all children, peripheral parasite density was lower in children with SCA, but when we used individual WBC determined on the day of diagnosis, peripheral parasite density did not differ significantly. Thus, measurement and incorporation of exact WBCs into parasitemia calculations on a per-participant basis is essential.
Insights from studies of hemoglobin AC (HbAC) trait and hemoglobin C (HbC) disease, hemoglobinopathies caused by a similar mutation in the β-globin chain encoding E6K (HbC) rather than E6V hemoglobin S (HbS), may shed light on our novel observations in children with HbSS/SCA.7,53-55 In particular, HbC expression also provides substantial protection from severe malarial infection hemoglobin CC (HbCC greater than HbAC), and the mechanism of this protection stems from impaired PfEMP1 export and reduced cytoadherence of parasitized heterozygous HbAC and homozygous HbCC red cells.53,56-58 Impaired PfEMP1 export in these experiments impairs rosetting, the clinically important process of clumping of infected RBCs in the microvasculature, with predicted reductions in inflammation.56-58 HbCC disease has thus been associated with protection from CM.53 Subsequent studies with HbS-containing red cells found a similar reduction in cytoadherence in HbSS compared with HbAA cells in vitro, suggesting commonalities between HbSS and HbCC disease on a cellular level.57,59,60 Molecular studies link this abnormal EMP1 display to inhibition of RBC actin remodeling by HbS oxidation products, which disrupts Mauer cleft biogenesis.61 Interestingly, heterozygous HbAS individuals may not exhibit the same protection from sequestration,62 though this remains understudied in vivo. Reduced PfEMP-1 export in individuals with HbSS could explain decreased sequestration in children with SCA, and decreased binding of infected RBCs to endothelial cells may reduce the likelihood of vaso-occlusive crisis during the malaria episode in these children.
The impaired cytoadherence demonstrated in these studies and the HbS-mediated antiplasmodial cytotoxicity demonstrated by others63-65 may underlie the unexpected molecular and parasitologic response to severe malaria observed here in children with SCA. During the intraerythrocytic cycle, some sequestered parasites are likely killed in the hypoxic environment of the HbSS red cell and venous microvasculature, reducing the overall parasite burden, whereas others slip back into circulation due to impaired cytoadherence, reducing sequestered biomass and normalizing the peripheral parasitemia between patients with and without SCA. Increased phagocytosis of sickled, parasitized red cells in children with SCA may also contribute to globally reduced parasitemia.
In the present study cohort, HbSS was a strong risk factor for severe anemia with P falciparum parasitemia, with a prevalence of 9.6%, whereas HbSS was not present in a parallel cohort of asymptomatic children in the same community.24,66 Children with HbSS have baseline anemia, so are at higher risk of developing severe anemia with P falciparum parasitemia during malarial infection. Thus, in SCA, while the decrease in parasite sequestration does not protect from malaria manifested as severe anemia, it may have contributed to the lack of mortality or signs of additional disease severity in these children. For example, decreased sequestration may explain why in other studies, children with SCA experience fewer neurovascular complications of severe malaria such as convulsions and impaired consciousness.67 Importantly, although plasma HRP-2 allows mathematical calculation of sequestered biomass, some isolates display variation in HRP-2 secretion, and HRP-2 half-life, related to acquired immunity, may vary by patient,68,69 so parasite biomass must be viewed as a crude estimate in any individual study participant.
Additionally, children with SCA may be protected from mortality during an acute episode of SMA by broadly reduced inflammation, which is intimately linked with parasite sequestration. Specifically, the interaction of ICAM-1 and PfEMP1 is a major molecular event driving sequestration and major proinflammatory event that drives the release of TNF-α, which triggers further luminal display of ICAM-1 on endothelial cells.34,70-72 In our cohort, children with SCA had significantly lower levels of TNF-α and trended toward lower concentrations of sEPCR, another substrate for PfEMP1 binding. Other vascular adhesins ICAM-1, VCAM-1, and E- and P-selectins were not significantly increased or decreased in children with SCA, despite markedly elevated Angpt-2, suggesting a decoupling of the acute inflammatory response and chronic Angpt-2 elevation. Given chronic inflammation and hemolysis during steady state in children with SCA, chronically upregulated compensatory anti-inflammatory mechanisms (eg, free heme induction of heme oxygenase-1) may impair the endothelial and systemic inflammatory response to acute infection. It is puzzling that chronic endothelial activation does not increase parasite sequestration, suggesting a greater role for parasite-specific factors in this observation. Overall, these significant reductions in inflammatory and endovascular markers in children with SCA and SMA suggest an important modification of the typical aggressive inflammatory host response to P falciparum, which has not previously been described.
Children with SCA in this study also had increased Angpt-2 and a low l-arginine/ADMA ratio, which have been previously associated with disease severity and risk of complications, including mortality, in adults and children.20,46,47,73-75 In our cohort, lower levels of TNF-α were associated with increased risk of all-cause postdischarge mortality or readmission, whereas increased levels of Angpt-2 were associated with increase uncomplicated malaria incidence postdischarge. Children with SCA experienced both of these host responses during severe anemia with P falciparum parasitemia. Additional studies are required to further examine the long-term effects of the attenuated inflammatory response to severe anemia with P falciparum parasitemia that we note here in children with SCA on the risks of reinfection, readmission, and mortality, as this single temporal risk calculation considers only a single episode of malaria, and these children experience frequent infection.
Importantly, severe anemia commonly causes hospitalization in patients with SCA, and the etiology is often multifactorial. In this study, we defined SMA according to the Ugandan Ministry of Health guidelines, which includes any peripheral parasitemia rather than a specific parasite density threshold, as specified in the revised World Health Organization (WHO) definition. Although incidental malaria parasitemia can be seen in individuals in endemic areas, it appears likely that malaria contributed to the severe anemia in this study population. We assessed for other infection, including bacteremia, HIV infection, and stool helminth infection, and found only 1 case of bacteremia among the 22 children with HbSS, with no HIV or stool helminth infections, suggesting that other infection was not a major contribution to the anemia in these children. Importantly, all SCA-SMA biomarker and biomass associations discussed here remained significant after exclusion of all of these children and application of the stringent WHO definition of SMA (supplemental Table 4), suggesting the trends identified are not due to incidental parasitemia with concurrent severe anemia of SCA. Malnutrition can be associated with severe anemia, but the children with SCA were not chronically malnourished and had better height-for-age and weight-for-age z-scores than the children without SCA. PfHRP-2 concentrations, a measure of parasite sequestration and biomass, also correlated with inflammation, further supporting a contribution of P falciparum to the disease process in these children. Children with SCA in this study were also older than children without SCA and may have acquired greater cellular and humoral immunity to P falciparum malaria. However, the associations reported are robust after correction for age and likely reflect true differences in the molecular response to severe malaria. It is possible that the children with SCA in this cohort had milder SCA than the general population of children with SCA, as they did not have a diagnosis of SCA prior to enrollment. The physiologic differences seen between children with and without SCA were nonetheless striking and highly significant. Thus, we hypothesize that the malaria-infected host is likely balancing inflammation, parasitemia, and endovascular activation, and children with SCA may carry significant long-term risk from these maladaptive responses.
In summary, we present the first evidence that parasite sequestration and host inflammatory responses, including several proinflammatory cytokines and endovascular adhesins, are attenuated in children with SCA during severe anemia with P falciparum parasitemia, despite profoundly elevated Angpt-2 and evidence of NO dysregulation. These results give a first window into the pathophysiology of acute severe malaria in the context of SCA and provide a basis for future studies to determine the how baseline changes in inflammation and endothelial cell function in African children with SCA may affect the risk of and response to P falciparum malaria and long-term complications of SCA and malaria.
Acknowledgments
The authors thank the children and their parents who participated in this study, and the study team for their dedicated effort in treating the children and collecting these data.
This work was supported by National Institutes of Health National Institute of Neurological Disorders and Stroke and the Fogarty International Center grants R01NS055349 and D43 NS078280.
Authorship
Contribution: R.C.H. performed statistical analysis and literature review, and wrote the first draft of the manuscript; C.B. performed Cox and interaction analyses; D.D. and A.L.C. assisted with the design of the study; A.L.C. performed quantitative serological studies; R.O.O., R.N., and C.C.J. were all involved in the administration and conduct of the clinical trial upon which this study is based; C.C.J., A.L.C., R.E.W., and C.L.S. edited the manuscript; and all authors commented on and approved the final version of this manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Chandy C. John, Indiana University, 1044 W. Walnut St, Rm 402D, Indianapolis, IN 46202; e-mail: chjohn@iu.edu.
References
Author notes
For data sharing, contact the corresponding author: chjohn@iu.edu.
The full-text version of this article contains a data supplement.