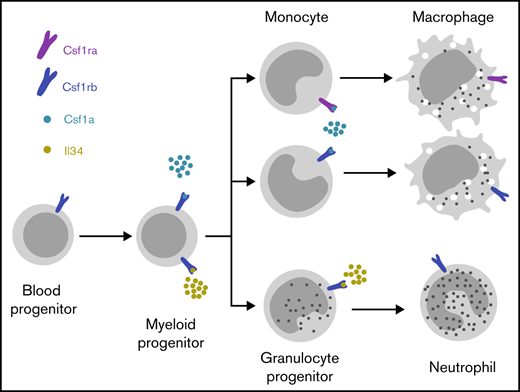
Key Points
csf1ra and csf1rb are indispensable for macrophage differentiation and, together with csf1a, regulate embryonic macrophage fates.
Il34 regulates zebrafish granulocyte development through csf1rb.
Abstract
Macrophage colony-stimulating factor receptor (M-CSFR/CSF1R) signaling is crucial for the differentiation, proliferation, and survival of myeloid cells. The CSF1R pathway is a promising therapeutic target in many human diseases, including neurological disorders and cancer. Zebrafish are commonly used for human disease modeling and preclinical therapeutic screening. Therefore, it is necessary to understand the proper function of cytokine signaling in zebrafish to reliably model human-related diseases. Here, we investigate the roles of zebrafish Csf1rs and their ligands (Csf1a, Csf1b, and Il34) in embryonic and adult myelopoiesis. The proliferative effect of exogenous Csf1a on embryonic macrophages is connected to both receptors, Csf1ra and Csf1rb, however there is no evident effect of Csf1b in zebrafish embryonic myelopoiesis. Furthermore, we uncover an unknown role of Csf1rb in zebrafish granulopoiesis. Deregulation of Csf1rb signaling leads to failure in myeloid differentiation, resulting in neutropenia throughout the whole lifespan. Surprisingly, Il34 signaling through Csf1rb seems to be of high importance as both csf1rbΔ4bp-deficient and il34Δ5bp-deficient zebrafish larvae lack granulocytes. Our single-cell RNA sequencing analysis of adult whole kidney marrow (WKM) hematopoietic cells suggests that csf1rb is expressed mainly by blood and myeloid progenitors, and the expression of csf1ra and csf1rb is nonoverlapping. We point out differentially expressed genes important in hematopoietic cell differentiation and immune response in selected WKM populations. Our findings could improve the understanding of myeloid cell function and lead to the further study of CSF1R pathway deregulation in disease, mostly in cancerogenesis.
Introduction
Hematopoiesis is a process of proliferation, differentiation, fate-commitment, and self-renewal of blood cells. It is primarily regulated by extrinsic signals such as cytokines and growth factors that bind to cell receptors and activate internal signaling pathways.1,2 One of the most prominent receptors that control the myeloid compartment is colony-stimulating factor 1 receptor (CSF1R, also known as macrophage-CSFR). In mammals, it is activated by 2 distinct ligands without obvious sequence homology (colony-stimulating factor 1 [CSF1; M-CSF] and interleukin 34 [IL-34]).3-6 However, despite the fact that both of these cytokines bind to the same receptor and can equally support cell growth and survival, they achieve this by triggering different chemokine responses.4,7 The CSF1R signaling pathway is in general critical for the proliferation, differentiation, survival, and activation of mononuclear phagocytic cells such as monocytes, macrophages, osteoclasts, or microglia in mammals,8-10 birds,11,12 and fish.13 Deregulation of the CSF1R pathway was connected to disease phenotypes14 such as osteopetrosis,9,10,15,16 brain disease,17-20 or cancer.21-24 Thus, the CSF1R signaling is of high interest as a pathway for therapeutic targeting in neurological and infectious diseases and tumorigenesis.25,26 Particularly, myeloid cells, including both neutrophils and macrophages, can act negatively in carcinogenesis. Therefore, tumor-associated macrophages (TAMs) are believed to be critical in tumor metastasis and are a good target in addition to conventional chemotherapy.27-29 It has been shown in mice that the number of TAMs can be efficiently reduced by the inhibition of CSF1R. Because of its low throughput when testing compounds, other model organisms need to be used.30 Zebrafish is a convenient model organism for human disease modeling,31-33 and the small size of zebrafish makes it advantageous for high-throughput preclinical drug screening.32,34-38 Due to the genome reduplication in teleost fish, many paralogs were generated that could possess redundant or novel biological functions.39-41 This includes both Csf1 and Csf1r, and therefore it is still needed to define the role and specificity of Csf1a, Csf1b, and Il34 toward Csf1rs (Csf1ra/b) in zebrafish myelopoiesis. So far, it seems that the function of Csf1ra and Csf1rb is only partially redundant.15 For instance, there are spatiotemporal differences in the importance of Csf1rs for microglia and HSC-derived myeloid cells development and seeding of the zebrafish brain.42-44
In this article, we focus on the roles of Csf1a, Csf1b, and Il34 cytokines in zebrafish embryonic and adult hematopoiesis, shown by ex vivo tools and single-cell RNA sequencing (scRNA-seq) of whole kidney marrows (WKM). We use a collection of zebrafish loss-of-function mutants to discern the effects of Csf1-receptor and ligand functional defects. We show that Csf1a drives the expansion of embryonic macrophages, Csf1b has no evident role in embryonic myelopoiesis, and Il34, acting through Csf1rb, is important for embryonic granulopoiesis. Finally, our observations suggest evolutionarily interesting functions of CSF1R signaling in the myelopoiesis of nonmammalian vertebrates in addition to the conventional role of CSF1 in mammalian myelopoiesis8,9,45 that should be taken into consideration when modeling human myeloid disorders in zebrafish.
Materials and methods
Animals
Zebrafish were bred, raised, and kept in ZebTEC aquatic systems (Tecniplast) according to standard procedures46 and tracked using Zebrabase.47 Zebrafish csf1-receptor mutant lines used in this study were csf1raV614M (panther),48 csf1rat36ui (further csf1raΔ5bp), csf1rbre01 (further csf1rbΔ4bp),42 and csf1raV614M;csf1rbre01 double mutants.42 The csf1r ligand mutants used were csf1ains2bp, csf1bΔ2bp, and il34Δ5bp.49 All the mutant zebrafish lines used in this study carried homozygous mutation (−/−). Transgenic reporter zebrafish lines used were Tg(mpeg1:EGFP),50 Tg(fms:GAL4;UAS:mCherry),51 Tg(mpx:EGFP),52 and Tg(pax7:GFP).53 Wild-type (WT) (AB) were used as controls. For ex vivo experiments, 6- to 12-month-old fish were used to get an optimal number of WKM cells. Animal care and experiments were approved by the Animal Care Committee of the Institute of Molecular Genetics (13/2016 and 96/2018) in compliance with national and institutional guidelines.
Multiplexed quantitative RNA fluorescence in situ hybridization
Hybridization chain reaction (HCR) v3.0 probe sets, amplifiers, and buffers were used according to the manufacturer’s protocols (Molecular Instruments).54 Probes detecting zebrafish csf1rb (XM_009295703.3), mpeg1 (NM_212737.1), and mpx (NM_001351837.1) were designed by the manufacturer. The Alexa647, Alexa546, and Alexa488 amplifiers were used.
Fluorescence imaging
Fluorescent images were acquired on Zeiss AxioZoom.V16 with Axiocam-506 mono camera. Orthogonal projections were created in ZENBlue-2.3 software. Images of HCR-stained embryos were acquired on Dragonfly-503 microscope (Andor) using Zyla-4.2 sCMOS camera. All images were processed by Fiji and Adobe Photoshop CC2021.55
scRNA-seq and transcriptomics
WKM cells were isolated as described previously,56 fractionated with Biocoll (1.077 g/mL; Merck), and counted. Between 3000 and 5000 cells were used for the preparation of chromium 3′ sequencing libraries using Chromium Single Cell 3′ Chip Kit v3.1 and sequenced with Illumina Nextseq500. The Illumina FASTQ files were used to generate filtered matrices using CellRanger (10XGenomics) with default parameters. To generate filtered matrices, data were loaded to Cellbender package57 using the following parameters: expected-cells = 5000, total-droplets-included = 15 000. Filtered matrices were then imported into R for exploration and statistical analysis using a SeuratV3 package.58 Counts were normalized according to total expression, multiplied by scale factor (10 000), and log-transformed. For cell cluster identification and visualization, gene expression values were also scaled according to highly variable genes after controlling for unwanted variation generated by sample identity. Cell clusters were identified based on UMAP of the first 20 principal components using Seurat’s method, FindClusters, with an original Louvain algorithm and resolution parameter value 0.5. Following quality control and basic clustering of each sample, we subsetted individual datasets to contain 1700 cells each and merged them. To visualize marker gene expression, Seurat’s method, Dot-Plot, was used. To merge individual datasets and to remove batch effects, SeuratV3 Integration and Label Transfer standard workflow were used.58
Other procedures and methods
The cloning of constructs for mRNA and recombinant protein expression, mRNA and protein microinjections, generation of mutant zebrafish, whole-mount in situ hybridization (ISH) of zebrafish embryos using digoxigenin-labeled antisense riboprobes, ex vivo WKM cell cultures, Sudan Black-B (SBB) staining of embryos, fluorescence-activated cell sorting (FACS) analysis, ex vivo cultures and histological staining, image processing, and statistical analysis are outlined in the supplemental Materials and Methods available in the online version of this article.
Results
Zebrafish csf1ra and csf1rb are expressed from early embryonic development and have distinct expression patterns in adults
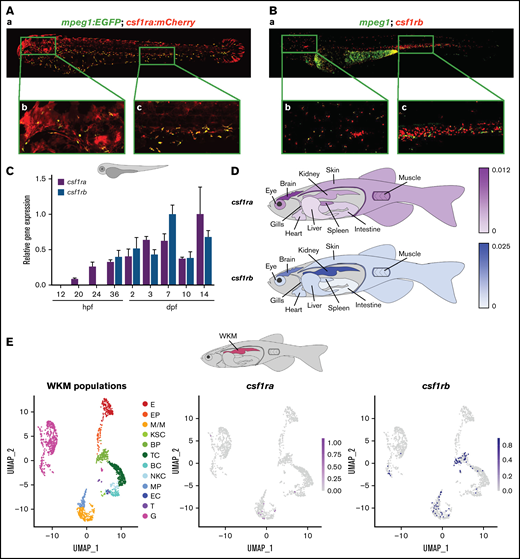
To determine whether the spatiotemporal expression pattern of zebrafish csf1ra and csf1rb overlap in embryonic development, we crossed fms:GAL4;UAS:mCherry (simplified as csf1ra:mCherry) and mpeg1:EGFP reporter lines to generate triple transgenic animals (Figure 1Aa). At 72 hours postfertilization (hpf), almost all mpeg1:EGFP+ macrophages are also csf1ra:mCherry+ (Figure 1Ab-c) (40% to 45% of fluorescent cells are double-positive: 230 ± 20 cells out of 520 ± 40 per embryo) and single csf1ra:mCherry+ cells in the skin are xanthophores,48 zebrafish pigment cells.53,59 On the contrary, by performing double fluorescent HCR using probes for mpeg1 and csf1rb at 72 hpf (Figure 1Ba), we showed that the majority of macrophages are mpeg1 single-positive with only a few mpeg1 and csf1rb double-positive cells (Figure 1Bb-c) (4% to 7% of fluorescent cells are double-positive double-positive: 30 ± 10 cells out of 550 ± 50 per embryo). In addition, we have identified a subset of csf1rb+ cells that also expressed c-myb, a marker of hematopoietic stem and progenitor cells (HSPCs) (data not shown).
csf1ra and csf1rb have distinct expression patterns in zebrafish. (A) Coexpression of csf1ra (red) and mpeg1 (green) visualized in 72 hpf Tg(fms:GAL4;UAS:mCherry);Tg(mpeg1:EGFP) triple transgenic embryos: (Aa) whole embryo, (Ab) head, (Ac) caudal hematopoietic tissue (CHT) region. (B) HCR WISH of 72 hpf embryos for csf1rb (red) and mpeg1 (green). (Ba) whole embryo, (Bb) head, (Bc) CHT region. Fluorescence images were taken on Dragonfly 503 microscope (Andor) using Zyla-4.2 sCMOS camera with magnification ×10 and processed with the Fusion software, FIJI, and Adobe Photoshop. (C) qRT-PCR analysis of pooled zebrafish embryos showing the expression dynamics of csf1ra and csf1rb in zebrafish development. Pool of 15 to 20 embryos per sample in 2 to 6 biological replicates. The expression was normalized to mob4 gene and to the time point with the highest expression (14 dpf for csf1ra and 7 dpf for csfrb). (D) qRT-PCR analysis of adult zebrafish tissues. Pool of 3 to 5 fish organs per sample in 3 to 5 biological replicates. The expression was normalized to ef1α gene. (E) scRNA-seq data showing the expression of csf1ra and csf1rb in whole kidney marrow (WKM) cell populations (pool of 2 to 3 WKMs per sample). BC, B-cells; BP, blood progenitors; E, erythroid cells; EC, endothelial cells; EP, erythroid progenitors; G, granulocytes; KSC, kidney support cells; M/M, monocytes & macrophages; MP, myeloid progenitors; NKC, NK cells; T, thrombocytes; TC, T-cells.
csf1ra and csf1rb have distinct expression patterns in zebrafish. (A) Coexpression of csf1ra (red) and mpeg1 (green) visualized in 72 hpf Tg(fms:GAL4;UAS:mCherry);Tg(mpeg1:EGFP) triple transgenic embryos: (Aa) whole embryo, (Ab) head, (Ac) caudal hematopoietic tissue (CHT) region. (B) HCR WISH of 72 hpf embryos for csf1rb (red) and mpeg1 (green). (Ba) whole embryo, (Bb) head, (Bc) CHT region. Fluorescence images were taken on Dragonfly 503 microscope (Andor) using Zyla-4.2 sCMOS camera with magnification ×10 and processed with the Fusion software, FIJI, and Adobe Photoshop. (C) qRT-PCR analysis of pooled zebrafish embryos showing the expression dynamics of csf1ra and csf1rb in zebrafish development. Pool of 15 to 20 embryos per sample in 2 to 6 biological replicates. The expression was normalized to mob4 gene and to the time point with the highest expression (14 dpf for csf1ra and 7 dpf for csfrb). (D) qRT-PCR analysis of adult zebrafish tissues. Pool of 3 to 5 fish organs per sample in 3 to 5 biological replicates. The expression was normalized to ef1α gene. (E) scRNA-seq data showing the expression of csf1ra and csf1rb in whole kidney marrow (WKM) cell populations (pool of 2 to 3 WKMs per sample). BC, B-cells; BP, blood progenitors; E, erythroid cells; EC, endothelial cells; EP, erythroid progenitors; G, granulocytes; KSC, kidney support cells; M/M, monocytes & macrophages; MP, myeloid progenitors; NKC, NK cells; T, thrombocytes; TC, T-cells.
We characterized the expression pattern of csf1ra/b during development and in adult tissues. Here, we demonstrate that csf1ra starts to be expressed at 20 hpf, whereas csf1rb expression was detected first at 36 hpf by qRT-PCR (Figure 1C). However, few csf1rb-expressing cells were observed using HCR already at 24 hpf (data not shown). The overall expression of both receptors during embryonic development gradually increases until 7 days post fertilization (dpf).
Similarly, qRT-PCR using selected adult zebrafish tissues (supplemental Figure 1) showed high expression of csf1ra in the brain, moderate expression in the spleen, muscles, eyes, kidneys, and skin, and weak expression in the remaining organs. The strongest expression of csf1rb was in adult kidney marrow and brain, whereas it was low in other organs. To summarize these results, we created representative schemas (Figure 1D).
To get a more detailed insight into the expression of csf1ra/b in adult hematopoietic tissues, we performed scRNA-seq (Figure 1E) and demonstrated that there is no overlap between csf1ra and csf1rb expression in adult WKM. Instead, the expression of csf1ra is restricted to a few cells within the population of monocytes-macrophages, whereas the csf1rb+ cells comprise blood and myeloid progenitors, monocytes-macrophages, and granulocytes (Figure 1E; supplemental Table 1). This is in agreement with our HCR expression data (Figure 1Ba-c), where we show that only a subset of csf1rb+ cells are macrophages.
csf1a drives the expansion and differentiation of zebrafish embryonic macrophages
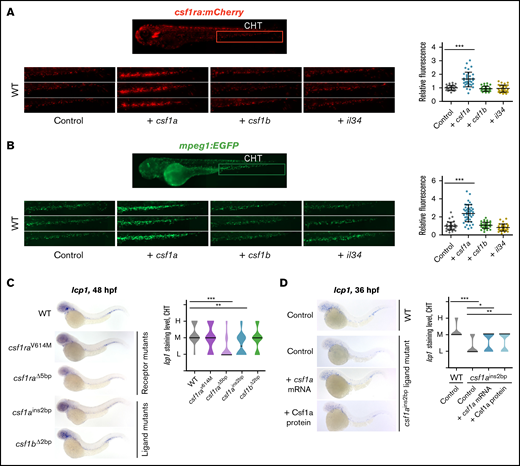
To further characterize the effects of csf1 ligands on hematopoietic cells, we in vitro transcribed and injected mRNA for csf1a, csf1b, and il34 ligands into 1-cell stage zebrafish embryos and examined their caudal hematopoietic tissue (CHT) region at 72 hpf. We noticed that the overexpression of csf1a but not of csf1b or il34 caused expansion of csf1ra:mCherry+ (Figure 2A) and mpeg1:EGFP+ macrophages (Figure 2B). These injected embryos had high expression of csf1rs and macrophage-specific markers, such as mpeg1, mfap4, and lcp1 (supplemental Figure 2A). Increased expression of lcp1 was also documented by ISH staining using lcp1 probe (supplemental Figure 2B). We also noticed that the overexpression of both csf1a/b highly increased the number of csf1ra:mCherry+ cells across the whole fish. We saw the same expansion in the xanthophore-specific pax7:EGFP transgenic line (data not shown).
csf1a, but not csf1b, and il34 drive the expansion of embryonic macrophages in vivo. (A-B) csf1a, csf1b, and il34 ligands were overexpressed by mRNA microinjection in 1-cell stage transgenic wild type (WT) embryos. Control embryos were injected with phosphate-buffered saline (PBS). Fluorescence images were acquired at 72 hpf and the area of fluorescent cells was calculated in the caudal hematopoietic tissue (CHT) (area inside of the red box) by FIJI. Results were normalized to injected controls. Scatter plots on the right represent quantification of fluorescent cells in CHT. Each dot in the scatter plot represents 1 larva. (A) Tg(fms:GAL4;UAS:mCherry); control n = 28, + csf1a n = 35, + csf1b n = 35, + il34 n = 28. (B) Tg(mpeg1:EGFP); control n = 41, + csf1a n = 45, + csf1b n = 37, + il34 n = 34. (C) WISH of 48 hpf embryos showing the expression of lcp1 in WT, 2 csf1ra mutants: csf1raV614M (panther) and csf1raΔ5bp, and in csf1ains2bp, csf1bΔ2bp ligand mutants. WT n = 15 (L = 1, M = 9, H = 5); csf1raV614M n = 15 (L = 4, M = 7, H = 4); csf1raΔ5bp n = 15 (L = 12, M = 2, H = 1); csf1ains2bp n = 14 (L = 7, M = 6, H = 1); csf1bΔ2bp n = 16 (L = 4, M = 9, H = 3). (D) lcp1 WISH of 36hpf WT embryos injected with PBS (control), and csf1ains2bp mutant embryos injected with PBS (control), csf1a mRNA or recombinant zebrafish Csf1a protein. WT, control n = 17 (L = 0, M = 14, H = 3); csf1ains2bp, control n = 45 (L = 32, M = 13, H = 0), + csf1a n = 32 (L = 15, M = 17, H = 0), + Csf1a n = 35 (L = 14, M = 21, H = 0). (C-D) Violin plot graphs show the level of lcp1 expression in the CHT region of individual embryos (L = low, M = medium, H = high) with median represented by a black line. (A-D) The level of statistical significance was determined by Mann-Whitney U test. *P < .04, **P < .007, ***P < .0001. All fluorescent images were acquired on Zeiss AxioZoom.V16 with Zeiss Axiocam 506 mono camera and ZEN Blue software. Bright field images of WISH were acquired on Zeiss AxioZoom.V16 with Zeiss Axiocam 105 color camera and processed using the Extended Depth of Focus module in the ZEN Blue software. FIJI and Adobe Photoshop were used for image processing.
csf1a, but not csf1b, and il34 drive the expansion of embryonic macrophages in vivo. (A-B) csf1a, csf1b, and il34 ligands were overexpressed by mRNA microinjection in 1-cell stage transgenic wild type (WT) embryos. Control embryos were injected with phosphate-buffered saline (PBS). Fluorescence images were acquired at 72 hpf and the area of fluorescent cells was calculated in the caudal hematopoietic tissue (CHT) (area inside of the red box) by FIJI. Results were normalized to injected controls. Scatter plots on the right represent quantification of fluorescent cells in CHT. Each dot in the scatter plot represents 1 larva. (A) Tg(fms:GAL4;UAS:mCherry); control n = 28, + csf1a n = 35, + csf1b n = 35, + il34 n = 28. (B) Tg(mpeg1:EGFP); control n = 41, + csf1a n = 45, + csf1b n = 37, + il34 n = 34. (C) WISH of 48 hpf embryos showing the expression of lcp1 in WT, 2 csf1ra mutants: csf1raV614M (panther) and csf1raΔ5bp, and in csf1ains2bp, csf1bΔ2bp ligand mutants. WT n = 15 (L = 1, M = 9, H = 5); csf1raV614M n = 15 (L = 4, M = 7, H = 4); csf1raΔ5bp n = 15 (L = 12, M = 2, H = 1); csf1ains2bp n = 14 (L = 7, M = 6, H = 1); csf1bΔ2bp n = 16 (L = 4, M = 9, H = 3). (D) lcp1 WISH of 36hpf WT embryos injected with PBS (control), and csf1ains2bp mutant embryos injected with PBS (control), csf1a mRNA or recombinant zebrafish Csf1a protein. WT, control n = 17 (L = 0, M = 14, H = 3); csf1ains2bp, control n = 45 (L = 32, M = 13, H = 0), + csf1a n = 32 (L = 15, M = 17, H = 0), + Csf1a n = 35 (L = 14, M = 21, H = 0). (C-D) Violin plot graphs show the level of lcp1 expression in the CHT region of individual embryos (L = low, M = medium, H = high) with median represented by a black line. (A-D) The level of statistical significance was determined by Mann-Whitney U test. *P < .04, **P < .007, ***P < .0001. All fluorescent images were acquired on Zeiss AxioZoom.V16 with Zeiss Axiocam 506 mono camera and ZEN Blue software. Bright field images of WISH were acquired on Zeiss AxioZoom.V16 with Zeiss Axiocam 105 color camera and processed using the Extended Depth of Focus module in the ZEN Blue software. FIJI and Adobe Photoshop were used for image processing.
Embryonic macrophage fate is impaired with the loss of csf1a signaling in zebrafish
To study impaired macrophage development upon loss of csf1ra or csf1rb, we compared lcp1 expression by ISH in CHT at 48 hpf between WT and other receptor mutants (csf1raV614M, csf1raΔ5bp, or csf1rbΔ4bp). The number of lcp1-positive cells was significantly decreased in csf1raΔ5bp mutant animals, while there was no difference in the csf1raV614M (Figure 2C), csf1rbΔ4bp, or in the csf1raV614M; csf1rbΔ4bp double-mutant fish (supplemental Figure 2C). Even though the number of lcp1+ cells was unchanged in csf1rbΔ4bp mutants, positive cells aggregated more to the rostral part of the CHT as compared with the WT (supplemental Figure 2D). In addition, we also examined mpeg1 expression in csf1raV614M as well as in csf1rbΔ4bp mutants at 48 hpf. As expected, based on published data43,44 and lcp1 expression data (Figure 2C; supplemental Figure 2C), the number of mpeg1+ macrophages in csf1raV614M CHT did not differ from those in WT (supplemental Figure 2E); however, it was significantly decreased in csf1rbΔ4bp fish (supplemental Figure 2F). To reveal the Csf1a ligand-receptor specificity, we microinjected csf1a into both of these mutants and demonstrated that ligand-overexpression-induced macrophage expansion was defective in them. Neither the number of mpeg1:EGFP+ cells in csf1raV614M (supplemental Figure 2G) nor of mpeg1+ cells in csf1rbΔ4bp mutants (supplemental Figure 2H) was changed as compared with the WT. Thus, Csf1a acts through both Csf1rs.
We examined lcp1 expression in csf1a and csf1b ligand mutants carrying frameshift mutations. ISH showed a significant decrease in the number of lcp1-expressing cells in the csf1ains2bp but not in the csf1bΔ2bp mutants (Figure 2C). Likewise, this phenotype can be rescued at 36 hpf by injection of csf1a mRNA or Csf1a proteins (supplemental Figure 3A-B) into csf1ains2bp mutant 1-cell stage embryos (Figure 2D).
Zebrafish csf1rb together with il34 regulate embryonic granulocytic fates
To test whether csf1r signaling is involved in the generation of other myeloid cell types besides macrophages, we examined the granulocytic lineage in csf1 ligand and receptor mutants. Mature granulocytes were visualized in zebrafish embryos and larvae by SBB staining, and positive cells were counted in tails (Figure 3A).
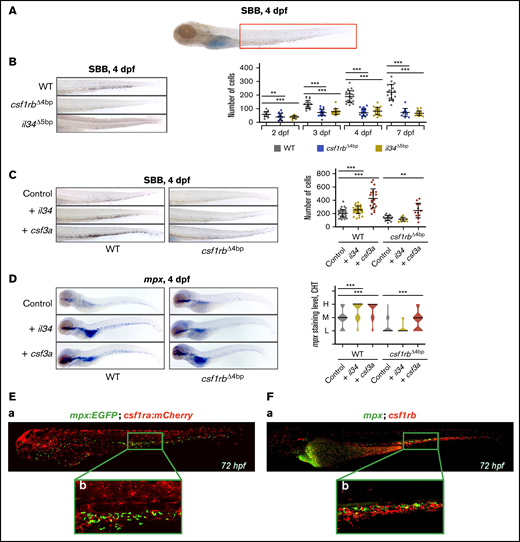
il34 binds to csf1rb and regulates the embryonic granulocytic fate. (A) SBB staining of a 4 dpf larva. The analyzed area is marked with a red rectangle. (B-C) SBB-positive cells were manually counted and the level of statistical significance was determined by Mann-Whitney U test. **P < .006, ***P < .0001. (B) SBB in WT, csf1rbΔ4bp and il34Δ5bp at 4 dpf. The graph on the right shows the number of SBB-positive cells during zebrafish embryonal and larval development, 2 to 7 dpf. 2 dpf: WT n = 21, csf1rbΔ4bp n = 24, il34Δ5bp n = 21; 3 dpf: WT n = 21, csf1rbΔ4bp n = 24, il34Δ5bp n = 18; 4 dpf: WT n = 21, csf1rbΔ4bp n = 26, il34Δ5bp n = 23; 7 dpf: WT n = 18, csf1rbΔ4bp n = 13, il34Δ5bp n = 21. (C) il34 and csf3a ligands were overexpressed by mRNA microinjection in 1-cell stage WT or csf1rbΔ4bp mutant embryos. Control embryos were injected with PBS. SBB staining was performed at 4 dpf. The graph on the right shows the number of SBB-positive cells. WT: control n = 46, + il34 n = 41, + csf3a n = 22; csf1rbΔ4bp: control n = 19, + il34 n = 18, + csf3a n = 12. (D) WISH of 4 dpf larvae showing the expression of mpx in WT or mutant csf1rbΔ4bp embryos with overexpressed il34 or csf3a ligands. Violin plots show the level of mpx expression in individual embryos (L = low, M = medium, H = high) with median represented by a black line. WT: control n = 44 (L = 11, M = 23, H = 10), + il34 n = 51 (L = 3, M = 16, H = 32), + csf3a n = 46 (L = 4, M = 11, H = 31); csf1rbΔ4bp: control n = 48 (L = 38, M = 9, H = 1), + il34 n = 40 (L = 34, M = 6, H = 0), + csf3a n = 44 (L = 19, M = 19, H = 6). The level of statistical significance was determined by Mann-Whitney U test. ***P < .0003. (E) Coexpression of csf1ra (red) and mpx (green) visualized in 72 hpf triple transgenic embryos Tg(fms:GAL4;UAS:mCherry);Tg(mpx:EGFP): (Ea) whole embryo, (Eb) caudal hematopoietic tissue (CHT) region. (F) HCR WISH of 72hpf embryos for csf1rb (red) and mpx (green). (Fa) whole embryo, (Fb) CHT region. All SBB staining and WISH bright field images were acquired on Zeiss AxioZoom.V16 with Zeiss Axiocam 105 color camera and processed using the Extended Depth of Focus module in the ZEN Blue software. FIJI and Adobe Photoshop were used for image processing. Fluorescence images were taken on Dragonfly 503 microscope (Andor) using Zyla-4.2 sCMOS camera with magnification ×10 and processed with the Fusion software, FIJI, and Adobe Photoshop.
il34 binds to csf1rb and regulates the embryonic granulocytic fate. (A) SBB staining of a 4 dpf larva. The analyzed area is marked with a red rectangle. (B-C) SBB-positive cells were manually counted and the level of statistical significance was determined by Mann-Whitney U test. **P < .006, ***P < .0001. (B) SBB in WT, csf1rbΔ4bp and il34Δ5bp at 4 dpf. The graph on the right shows the number of SBB-positive cells during zebrafish embryonal and larval development, 2 to 7 dpf. 2 dpf: WT n = 21, csf1rbΔ4bp n = 24, il34Δ5bp n = 21; 3 dpf: WT n = 21, csf1rbΔ4bp n = 24, il34Δ5bp n = 18; 4 dpf: WT n = 21, csf1rbΔ4bp n = 26, il34Δ5bp n = 23; 7 dpf: WT n = 18, csf1rbΔ4bp n = 13, il34Δ5bp n = 21. (C) il34 and csf3a ligands were overexpressed by mRNA microinjection in 1-cell stage WT or csf1rbΔ4bp mutant embryos. Control embryos were injected with PBS. SBB staining was performed at 4 dpf. The graph on the right shows the number of SBB-positive cells. WT: control n = 46, + il34 n = 41, + csf3a n = 22; csf1rbΔ4bp: control n = 19, + il34 n = 18, + csf3a n = 12. (D) WISH of 4 dpf larvae showing the expression of mpx in WT or mutant csf1rbΔ4bp embryos with overexpressed il34 or csf3a ligands. Violin plots show the level of mpx expression in individual embryos (L = low, M = medium, H = high) with median represented by a black line. WT: control n = 44 (L = 11, M = 23, H = 10), + il34 n = 51 (L = 3, M = 16, H = 32), + csf3a n = 46 (L = 4, M = 11, H = 31); csf1rbΔ4bp: control n = 48 (L = 38, M = 9, H = 1), + il34 n = 40 (L = 34, M = 6, H = 0), + csf3a n = 44 (L = 19, M = 19, H = 6). The level of statistical significance was determined by Mann-Whitney U test. ***P < .0003. (E) Coexpression of csf1ra (red) and mpx (green) visualized in 72 hpf triple transgenic embryos Tg(fms:GAL4;UAS:mCherry);Tg(mpx:EGFP): (Ea) whole embryo, (Eb) caudal hematopoietic tissue (CHT) region. (F) HCR WISH of 72hpf embryos for csf1rb (red) and mpx (green). (Fa) whole embryo, (Fb) CHT region. All SBB staining and WISH bright field images were acquired on Zeiss AxioZoom.V16 with Zeiss Axiocam 105 color camera and processed using the Extended Depth of Focus module in the ZEN Blue software. FIJI and Adobe Photoshop were used for image processing. Fluorescence images were taken on Dragonfly 503 microscope (Andor) using Zyla-4.2 sCMOS camera with magnification ×10 and processed with the Fusion software, FIJI, and Adobe Photoshop.
Strikingly, the number of SBB+ granulocytes in the tail was lower compared to the WT and failed to increase during the development in csf1rbΔ4bp as well as in il34Δ5bp mutants (Figure 3B), whereas it gradually increased in both csf1ra mutants at the same time (supplemental Figure 4A). An intermediate phenotype was documented in the receptor double mutants, in which the size of the original granulocytic pool did not change significantly with time (supplemental Figure 4A). In addition, the mutation in csf1a or csf1b ligands had no obvious effect on the number of granulocytes (data not shown). Along with these findings, the expression of mpx was also significantly downregulated in the CHT region of csf1rbΔ4bp and il34Δ5bp, but not in other receptor or ligand mutants at 4 dpf (supplemental Figure 4B; data not shown).
Further, we assessed the effects of il34 injection on granulocytic expansion in WT as well as in csf1rbΔ4bp mutants. As a positive control, we injected colony-stimulating factor 3a (csf3a, also known as gcsfa).60 As expected, the injection of csf3a mRNA led to a significant increase of SBB-positive granulocytes in either csf1rbΔ4bp mutant or WT fish at 4 dpf. Similarly, the injection of il34 mRNA into WT fish also caused an increase, however in contrast to csf3a, this il34-mediated phenotype was diminished in the csf1rbΔ4bp mutants (Figure 3C). The same effect was confirmed by mpx ISH of 4 dpf injected WT as well as csf1rbΔ4bp mutant embryos (Figure 3D). We also tested the other ligands, csf1a and csf1b, but il34 was the only one to affect granulocytic expansion (supplemental Figure 4C). In addition, we found that the overexpression of il34 led to a significant expansion of csf1rb-expressing cells at 4 dpf (data not shown). Importantly, microinjection of il34 induced granulocytic expansion also in the csf1raV614M mutant (supplemental Figure 4D). The coexpression of mpx with csf1rs in the CHT of 72 hpf embryos shows basically no overlap between csf1ra and mpx (Figure 3Ea-b) (1% to 2% of fluorescent cells are double-positive double-positive: 5 ± 3 cells out of 520 ± 40 per embryo). However, there is a small proportion of csf1rb and mpx double-positive cells (Figure 3Fa-b) (6% to 10% of fluorescent cells are double-positive: 40 ± 10 cells out of 550 ± 50 per embryo). Taken together, these results indicate that Il34 regulates embryonic granulocyte development through Csf1rb.
Zebrafish csf1rb is indispensable for definitive granulopoiesis
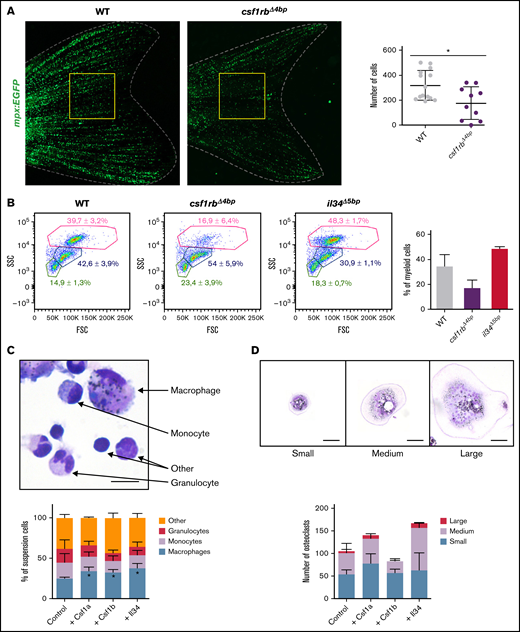
To investigate the importance of csf1rb in adult granulopoiesis, we imaged and counted the number of mpx+ cells in tail fins of 6-month-old mpx:EGFP transgenic WT and csf1rbΔ4bp mutant animals. There was a significantly reduced number of granulocytes in the periphery of csf1rbΔ4bp mutants (Figure 4A). Furthermore, we examined WKMs of csf1raV614M, csf1raΔ5bp, csf1rbΔ4bp, and il34Δ5bp animals using FACS analysis (Figure 4B), noticing a significant decrease in the number of myeloid cells in csf1rbΔ4bp mutants only (mean ± SD; WT: 39.7 ± 3.2%; csf1rbΔ4bp: 16.9 ± 6.4%; il34Δ5bp: 48.3 ± 1.7%). The other mutants were not affected (data not shown). Additionally, we prepared thin layer smears stained by May-Grünwald and Giemsa (MGG) from WKM cell suspensions (supplemental Figure 5). Most of the cells isolated from csf1rbΔ4bp mutants resembled immature, undifferentiated cells. The morphology of csf1rbΔ4bp mutant granulocytes was abnormal with a significantly decreased frequency of lobulated mature cells that were much smaller in size compared with WT cells.
csf1rb is indispensable for definitive granulopoiesis. (A) Number of neutrophils in adult Tg(mpx:EGFP) = WT and Tg(mpx:EGFP);csf1rbΔ4bp = csf1rbΔ4bp fishtails. Neutrophils were manually counted in the area of the yellow square. WT n = 14, csf1rbΔ4bp n = 10. The level of statistical significance was determined by an unpaired 2-tailed t test. *P < .04. (B) FACS analysis of WKM cell suspension from WT, csf1rbΔ4bp, and il34Δ5bp adult zebrafish. WKMs pooled from 2 fish in 3 biological replicates, and 1 representative plot is shown for each condition. The numbers in FSC/SSC plots represent the mean percentage with SD in the gates of myeloid cells (pink gate), progenitors (blue gate), and lymphoid and small progenitor cells (green gate). The percentage of WKM cells in the myeloid gate is also shown in the bar graph on the right. (C-D) Ex vivo culture of WKM cells treated with Csf1a, Csf1b, or Il34 proteins. (C) After 3 days in culture, smears of suspension cells were stained on microscopic glass slides with May-Grünwald and Giemsa (MGG), and the number of differentiated cells (monocytes, macrophages, and granulocytes) was counted. The graph on the bottom shows the mean percentage of cells with SD. The level of statistical significance was determined by an unpaired 2-tailed t test. *P < .04. The scale bar on the microscopic image is 20 µm. Results from 3 biological replicates. (D) After 3 days in culture, adherent cells on the dish were washed with PBS, stained with MGG, and the number of small, medium, and large osteoclasts was counted in 20 fields of view with a magnification ×20 objective. The graph on the bottom shows the mean percentage of cells with SD. Results from 2 biological replicates. The scale bar on the microscopic image is 50 µm. Fluorescence images were acquired on Zeiss AxioZoom.V16 with Zeiss Axiocam 506 mono camera and ZEN Blue software. ImageJ and Adobe Photoshop were used for image processing. Bright-field images of ex vivo cultures were acquired on Leica DM 2000 microscope with Zeiss Axiocam 105 color camera.
csf1rb is indispensable for definitive granulopoiesis. (A) Number of neutrophils in adult Tg(mpx:EGFP) = WT and Tg(mpx:EGFP);csf1rbΔ4bp = csf1rbΔ4bp fishtails. Neutrophils were manually counted in the area of the yellow square. WT n = 14, csf1rbΔ4bp n = 10. The level of statistical significance was determined by an unpaired 2-tailed t test. *P < .04. (B) FACS analysis of WKM cell suspension from WT, csf1rbΔ4bp, and il34Δ5bp adult zebrafish. WKMs pooled from 2 fish in 3 biological replicates, and 1 representative plot is shown for each condition. The numbers in FSC/SSC plots represent the mean percentage with SD in the gates of myeloid cells (pink gate), progenitors (blue gate), and lymphoid and small progenitor cells (green gate). The percentage of WKM cells in the myeloid gate is also shown in the bar graph on the right. (C-D) Ex vivo culture of WKM cells treated with Csf1a, Csf1b, or Il34 proteins. (C) After 3 days in culture, smears of suspension cells were stained on microscopic glass slides with May-Grünwald and Giemsa (MGG), and the number of differentiated cells (monocytes, macrophages, and granulocytes) was counted. The graph on the bottom shows the mean percentage of cells with SD. The level of statistical significance was determined by an unpaired 2-tailed t test. *P < .04. The scale bar on the microscopic image is 20 µm. Results from 3 biological replicates. (D) After 3 days in culture, adherent cells on the dish were washed with PBS, stained with MGG, and the number of small, medium, and large osteoclasts was counted in 20 fields of view with a magnification ×20 objective. The graph on the bottom shows the mean percentage of cells with SD. Results from 2 biological replicates. The scale bar on the microscopic image is 50 µm. Fluorescence images were acquired on Zeiss AxioZoom.V16 with Zeiss Axiocam 506 mono camera and ZEN Blue software. ImageJ and Adobe Photoshop were used for image processing. Bright-field images of ex vivo cultures were acquired on Leica DM 2000 microscope with Zeiss Axiocam 105 color camera.
Csf1a, Csf1b, and Il34 zebrafish proteins expand adult myeloid cells in ex vivo culture
To investigate cell-autonomous effects of Csf1 and Il34 cytokines, we performed in vitro experiments using recombinant ligand proteins. Therefore, we isolated and seeded WKM cells from WT fish, as published previously,56,60 with the addition of recombinant zebrafish Csf1a, Csf1b, or Il34 proteins. After 3 days in suspension culture, we prepared histological smears of myeloid cells for enumeration. Specifically, we counted the number of monocytes, differentiated macrophages, and granulocytes in proportion to other cells (mostly immature blood progenitors and lymphoid-like cells). In the presence of any of all 3 cytokines, suspension cells differentiated toward the myeloid lineage to mostly become mature macrophages (Figure 4C). Strikingly, mature multinucleated osteoclasts represented a major fraction of adherent cells. The addition of Csf1a or Il34 to the ex vivo culture promoted the proliferation of osteoclast progenitors and their fusion (Figure 4D).
Myelopoiesis is partially blocked in the csf1rbΔ4bp mutants
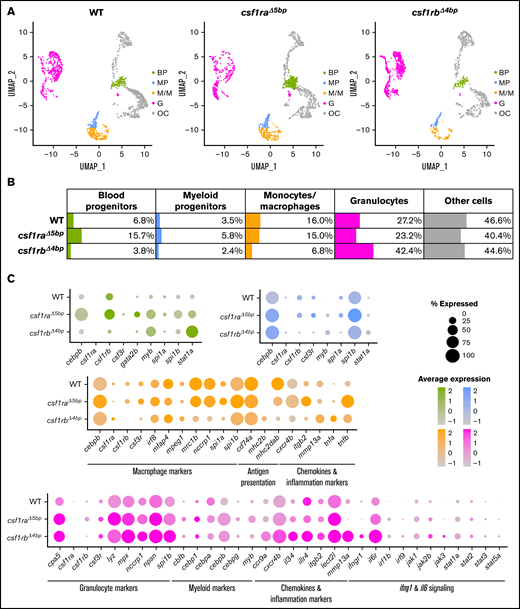
Our results thus far have shown that embryonic granulopoiesis is altered in csf1rbΔ4bp mutants. With the noted differences in the composition of the individual hematopoietic populations between WT and mutant animals, we were interested in characterizing these changes at the single-cell level. We thus used scRNA-seq to profile WKM cells isolated from 12-month-old WT, csf1raΔ5bp, and csf1rbΔ4bp mutants.
Via unsupervised clustering of single-cell transcriptomes and based upon known lineage marker genes, we named each cluster based on likely cell-type origins (Figure 5A). The percentage of cells in selected clusters of blood progenitors (BP), myeloid progenitors (MP), monocytes/macrophages (M/M), and granulocytes (G) is shown in a table (Figure 5B). We saw increased numbers of cells in BP and MP populations for csf1raΔ5bp and surprisingly G for csf1rbΔ4bp (Figure 5B; supplemental Figure 6A), whereas the number of M/M was decreased in csf1rbΔ4bp as expected.
Single-cell RNA sequencing (scRNA-seq) of adult WT, csf1raΔ5bp, and csf1rbΔ4bp WKM cells shows differentially expressed hematopoietic genes. (A-C) WKMs of 1 to 2 animals were pooled for each sample. (A) Clusters in Dim plots represent individual selected populations of WKM hematopoietic cells. The identity of each cluster is based on likely cell origin according to lineage marker gene expression. Green, blood progenitors (BP); blue, myeloid progenitors (MP); orange, monocytes/macrophages (M/M); pink, granulocytes (G); gray, other cells (OC). (B) Table representing the percentage of cells in each population for WT and respective mutants. (C) Dot plot visualization of scRNA-seq gene expression in individual populations of WKM hematopoietic cells of WT and respective mutants. The color of each dot represents the level of expression (also depicted in the histogram), and the size of the dot shows the percentage of cells expressing each individual gene.
Single-cell RNA sequencing (scRNA-seq) of adult WT, csf1raΔ5bp, and csf1rbΔ4bp WKM cells shows differentially expressed hematopoietic genes. (A-C) WKMs of 1 to 2 animals were pooled for each sample. (A) Clusters in Dim plots represent individual selected populations of WKM hematopoietic cells. The identity of each cluster is based on likely cell origin according to lineage marker gene expression. Green, blood progenitors (BP); blue, myeloid progenitors (MP); orange, monocytes/macrophages (M/M); pink, granulocytes (G); gray, other cells (OC). (B) Table representing the percentage of cells in each population for WT and respective mutants. (C) Dot plot visualization of scRNA-seq gene expression in individual populations of WKM hematopoietic cells of WT and respective mutants. The color of each dot represents the level of expression (also depicted in the histogram), and the size of the dot shows the percentage of cells expressing each individual gene.
Further, we picked out representative markers of hematopoiesis that characterize BP (green), MP (blue), M/M (orange), and G (pink) populations and created dot plot expression schemes showing their relative expression and the percent of cells expressing them (Figure 5C). We observed deregulation in most of these markers in both mutants. However, for csf1raΔ5bp mutant, the differences were more prominent in progenitors (BP and MP), and for csf1rbΔ4b, they were more prominent in differentiated myeloid cells (M/M and G). Additionally, we observed that the csf1rb gene was upregulated in the csf1raΔ5bp mutant and vice versa. After a closer look at the discrepancy between the decreased number of embryonic granulocytes and peripheral neutropenia in adult csf1rbΔ4bp fish, we noted that a high proportion of granulocyte-like cells accumulate in WKM (supplemental Figure 6A). In addition, these cells aberrantly expressed progenitor, migration, and inflammation markers (Figure 5C; supplemental Figure 6B-C).
Discussion
Differentiation, survival, and maturation of myeloid cells are tightly controlled by extrinsic factors, such as cytokines. CSF1, CSF2 (also known as granulocyte-macrophage-CSF, GM-CSF), and CSF3 belong to the most prominent ones. The critical role of CSF1 signaling on proper macrophage cell differentiation and survival was shown in mice mutants that lack CSF1R or CSF1.9,61,62 Interestingly, CSF1, CSF2, and CSF3 triple-mutant mice are not completely devoid of macrophages or granulocytes.62 This indicates that the role of these factors can be replaced by other cytokines, such as IL-6 or IL-34.4,63,64 Although CSF signaling has been historically studied mainly in mammalian and bird animal models and many transgenic and mutant lines are available, zebrafish have recently become a popular alternative model organism for modeling hematopoietic human diseases.
Here, we studied the in vivo function of zebrafish Csf1 receptor paralogs (csf1ra and csf1rb) and their ligands (csf1a, csf1b, and il34) to describe their involvement in fish myelopoiesis and to characterize their diversification.
Our findings demonstrate that Csf1 receptors and Csf1 ligands subfunctionalized during embryonic myelopoiesis. We and others show that both receptors are involved in myelopoiesis, and Csf1ra is required for pigmentation48 whereas Csf1rb was shown to be expressed and involved in HSPCs and myeloid differentiation.44
In mammals, CSF1R is expressed in erythro-myeloid progenitors (EMPs) and serves as their marker.65 Although CSF1R is not required for EMP formation,66 it is necessary for their late differentiation into myeloid lineages.67,68 In zebrafish, the role of Csf1ra/b in EMP remains unclear due to the limited number of tools available to distinguish EMPs from HSPCs.
Additionally, we show that only the Csf1a ligand is important in the development of embryonic macrophages, while both Csf1a and Csf1b are involved in pigmentation. This is consistent with previous studies demonstrating the role of Csf1a/b in adult pigment patterning.15,69
To get detailed insight into the involvement of Csf1 signaling in embryonic and adult macrophage development in zebrafish, we decided to study Csf1 ligand and receptor mutants. Zebrafish csf1ra, csf1rb, and il34 mutants have been previously described for the lack of microglia.42,49 It was shown that macrophages develop, migrate, or proliferate aberrantly in il34Δ5bp mutants43,49 and csf1raV614M;csf1rbΔ4bp double-mutant fish,70 whereas they develop normally in csf1raV614M (known as panther)42,43 and csf1rbsa1503 mutant44 embryos. The status of macrophage development in csf1rbΔ4bp single-mutant animals has remained unnoticed.
In contrast to these studies, we demonstrate that the number of embryonic macrophages in the CHT of csf1raΔ5bp and csf1rbΔ4bp mutant embryos is decreased. We suppose that the discrepancy in the number of embryonic macrophages between csf1raV614M and csf1raΔ5bp or csf1rbsa1503 and csf1rbΔ4bp could be explained by the fact that csf1raΔ5bp and csf1rbΔ4bp mutants have stronger phenotypes because they carry frameshift mutations with a premature stop codon. This has also been shown in another panther mutant with a C-terminal deletion (csf1raj4blue).48,71,72 To test the receptor-ligand specificity and in order to see the effect of csf1a overexpression, we chose the panther mutant (csf1raV614M). Even though this mutant has a weaker phenotype, it is suitable for such an experiment because it still possesses a substantial amount of embryonic macrophages that might be expanded. On the other hand, the csf1raΔ5bp animals have a very small embryonic macrophage pool that might have been difficult to expand, and therefore, an outcome of such an experiment could not be evaluated. In other experiments, we used either csf1raΔ5bp single-mutant or both (csf1raΔ5bp and panther) mutants alongside to compare their phenotypes. Surprisingly, macrophage expansion is diminished in both csf1raV614M and csf1rbΔ4bp mutants after csf1a microinjection. Therefore, we hypothesize that Csf1a drives macrophage development via either of both Csf1ra or Csf1rb receptors.
Besides the role of Csf1 signaling in macrophage differentiation, we noticed that it might also be involved in zebrafish granulocyte differentiation. Despite decades of CSF1R research, only sparse evidence exists linking CSF1R signaling to granulocyte differentiation in mammals.73 Strikingly, we show that csf1rbΔ4bp and also il34Δ5bp mutant zebrafish embryos have major defects in granulopoiesis, and the overexpression of il34 in WT animals leads to granulocyte expansion. Further, we show that the pool of embryonic granulocytes cannot be expanded in the csf1rbΔ4bp mutants by il34. This implies that Il34 plays a previously unknown role in the regulation of embryonic granulopoiesis, and it can act through Csf1rb, providing a novel Csf3r alternative pathway that is important for granulocyte differentiation. Recently, the expression of CSF1R has been detected in a subset of human fetal granulocytic progenitors and granulocytes.74
As a next step, we decided to examine adult macrophages in receptor mutant animals. In contrast to defects in embryonic macrophage myelopoiesis, we did not see any evident myeloid defects in WKM of adult csf1raΔ5bp mutants when examined histologically and by FACS. Nevertheless, the scRNA-seq analysis of csf1raΔ5bp mutants revealed that there is a slight downregulation of macrophage-specific markers, such as mfap4, mhc2dab, or mrc1b, and an upregulation of progenitor-specific markers, such as myb, cebpa, and gata2b, and the overall number of blood and myeloid progenitors is elevated as well; however, the other populations are mostly unchanged. Despite these less severe phenotypes in adult WKM cells, we hypothesize that besides Csf1ra role in the development of embryonic macrophages, it also plays a minor role during adult hematopoiesis, as previously not shown. The decreased number of monocytes/macrophages was also observed in csf1rbΔ4bp mutant using scRNA-seq. In addition, markers specific to macrophage host defense/phagosome and antigen presentation are decreased (mrc1b, nccrp1, and mhc2dab) in this mutant. These observations led us to the conclusion that Csf1rb is equally important during embryonic as well as adult macrophage development.
Additionally, csf1rbΔ4bp adult fish have a lower number of myeloid cells in WKM and also fewer mpx+ cells in the periphery. Since the majority of myeloid cells in WKM are neutrophils,75,76 we attribute the reduced number of myeloid cells to the loss of mature, physiologically normal granulocytes. However, surprisingly, our scRNA-seq data indicate that csf1rbΔ4bp mutants have more granulocytes in WKM. We explain this discrepancy by the fact that csf1rbΔ4bp granulocyte progenitors cannot fully differentiate and migrate, and progenitor-like early and also late granulocytes accumulate in WKM. This is proven by scRNA-seq marker gene expression profiling and also by FACS and histological staining of WKM cells, demonstrating that csf1rbΔ4bp fish have a high proportion of small cells with abnormal morphology and low granularity. Using scRNA-seq analysis, the predicted granulocyte population of csf1rbΔ4bp WKMs showed an overexpression of myb, cbfb, spi1b, and cebp markers, known to play essential roles in progenitor hemostasis.77-81 Further, we observed deregulated expression of chemokine and inflammation markers (il34, itgb2, mmp13a, il6r, and ifngr1) and markers connected to granulocyte migration (cxcr4b, ifngr1, and cxcl8b).82,83 Abnormal activation of the zebrafish ifngr1 signaling has been previously shown in neutropenic zebrafish.84
Next, we have shown that csf1r paralogues have functionally diverged in teleost evolution. Consistent with previous findings,44 we suggest that the function and expression of csf1 receptor paralogues is mostly nonoverlapping. At the single-cell level, using scRNA-seq analysis of adult WKM cells, we showed that there is no expression overlap between csf1ra and csf1rb and that csf1rb is highly expressed in blood and myeloid progenitors as well as in a small subset of monocytes/macrophages and granulocytes. In contrast, the csf1ra expression is restricted only to a subset of monocytes/macrophages. In correlation with these expression data, we also demonstrated that mutation in csf1ra primarily affects embryonic macrophage development, while the mutation in csf1rb is equally important during embryonic as well as adult development of both macrophages and granulocytes. Based on our scRNA-seq results, we assume that the nonoverlapping expression of Csf1ra and Csf1rb implicates their diverged roles during adult zebrafish hematopoiesis. It seems probable that Csf1rb, at least partially, takes over the function of Csf1ra in adulthood. This is also supported by the recently described role of Csf1rb in zebrafish HSPCs and adult microglia.44
Regarding ligand-receptor signaling specificities, we found that Csf1a acts via both Csf1ra and Csf1rb and that the granulocyte-related function of Il34 is executed via Csf1rb. In contrast, Csf1b has no function during embryonic myelopoiesis, and it expands xanthophores instead, together with Csf1a. Importantly, based on the fact that there is low expression of csf1rb in the skin, we assume that the Csf1-dependent xanthophore expansion is mediated via Csf1ra.
In addition, our ex vivo experiments suggest that recombinant Csf1a/b and Il34 cytokines are functional in ex vivo cultures and drive WKM-derived myeloid cell differentiation toward monocyte/macrophage and osteoclast fates. However, terminal granulocytic differentiation is not affected by Csf1r ligands.
In summary, we demonstrate that zebrafish continue to provide new biological insights relevant to disease. Using a wide range of convenient in vivo and ex vivo tools, it is possible to characterize new exciting roles of cytokines under steady-state as well as nonsteady-state conditions. Myeloid cells, including neutrophils and macrophages, are critical actors in cancerogenesis,85-87 and the CSF1 pathway is a promising target for clinical treatments.24,88 Here, we performed a detailed characterization of Csf1 signaling in zebrafish, making it suitable for preclinical disease modeling in the high-throughput discovery of new therapeutics.
Acknowledgments
The authors thank Nikol Pavlu, Tereza Hojerova, and Tereza Hingarova for animal care. They also thank Trevor Epp for editing the manuscript and Leonard Zon for providing mpeg1:EGFP and mpx:EGFP reporter fish lines. The authors acknowledge Michal Kolar for help with sc-RNA-transcriptomics (LM2018131) and the Light Microscopy Core Facility, IMG CAS, Prague, Czech Republic, supported by MEYS (LM2018129, CZ.02.1.01/0.0/0.0/18_046/0016045) for their support with the confocal imaging.
This work was supported by the Czech Science Foundation (18-18363S), LM2018130, and 68378050-KAV-NPUI to P.B. and by the ERC Advanced Grant “DanioPattern” (694289) to C.N.-V. and U.I.
Authorship
Contribution: M.H., T.M., O.M., A.P., and U.I. performed research; O.S. and P.B. designed the research; T.J.v.H. and C.N.-V. provided critical reagents for the work; and M.H., T.M., P.B., and O.S. wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Petr Bartunek, Institute of Molecular Genetics of the Czech Academy of Sciences, 142 20, Prague 4, Czech Republic; e-mail: bartunek@img.cas.cz; and Ondrej Svoboda, Institute of Molecular Genetics of the Czech Academy of Sciences, 142 20, Prague 4, Czech Republic; e-mail: osvoboda@img.cas.cz.
References
Author notes
M.H. and T.M. contributed equally to this study.
scRNA-seq data are available in ArrayExpress under accession number E-MTAB-10360. Plasmids for cytokine expression are available via Addgene: 168103 (pAc-His-zfCsf1a), 168104 (pAc-His-zfCsf1b), 168105 (pAc-His-zfIl34), and for mRNA expression: 168110 (pCS2-zfCsf1a), 168111 (pCS2-zfCsf1b), and 168112 (pCS2-zfIl34).
The full-text version of this article contains a data supplement.