TO THE EDITOR:
The adaptive humoral immune response to SARS-CoV-2 plays a critical role in establishing protective immunity after infection or vaccination.1-3 Viral clearance and immunological control seems to be affected in patients with humoral deficiency, which can present with persistent viral shedding and prolonged COVID-19.4-8 Impaired adaptive immunity related to dysfunctional or depleted B cells and hypogammaglobulinemia is a common feature in B-cell–related malignancies, such as B-cell non-Hodgkin lymphomas (B-NHLs).9-12 Because of the inability to produce a sufficient amount of SARS-CoV-2–neutralizing antibodies, those patients are at risk of prolonged COVID-197,8 and vaccination failure.13,14 Passive immunization with SARS-CoV-2–neutralizing monoclonal antibodies (nmAbs) demonstrated beneficial effects on mortality, clinical course, and viral load in immunocompetent unvaccinated patients with mild disease who were treated early,15-17 before endogenous IgG production (IgG seronegative).18 These trials did not focus on patients with immunosuppression or cancer. However, the results suggest that induction of humoral immune response several days after symptom onset marks a critical barrier for treatment efficacy. Thus, passive immunization with mAbs may be a rational treatment approach for patients who do not produce sufficient amounts of endogenous neutralizing antibodies after vaccination or infection, irrespective of symptom duration. Herein, we give a detailed description of hospitalized patients with COVID-19 who had impaired humoral immunity to underlying B-NHL and were successfully treated with nmAbs in the context of an individual healing attempt. Patients gave written informed consent for scientific evaluation. The study was approved by the local ethics committee of the University Hospital of Cologne.
From June through October 2021, 6 consecutive patients (A-F; median age, 59.5 years; range, 39-78) were enrolled in our study (Table 1; see supplemental Methods for a description of the process). All patients had known hypogammaglobulinemia, 5 had received B-cell–depleting therapy, and 4 had received regular prophylactic intravenous or subcutaneous immunoglobulin substitutions. All patients had detectable viremia and symptomatic SARS-CoV-2 infection (World Health Organization progression score, 4-6) with fever, myalgia, and malaise. Respiratory symptoms and radiologic signs of pulmonary manifestation were detectable; however, none of the patients required mechanical ventilation throughout the observation period.
Individual patient characteristics
| . | Persistent or recurrent SARS-CoV-2 infection . | Serologically defined COVID-19 vaccine failure . | ||||
|---|---|---|---|---|---|---|
| . | Patient A . | Patient B . | Patient C . | Patient D . | Patient E . | Patient F . |
| Age, y | 48 | 56 | 63 | 78 | 63 | 39 |
| Sex | Male | Male | Male | Female | Male | Female |
| Type of B-NHL | Follicular | Follicular | Follicular | Unspecified | CLL | DLBCL |
| Basal characteristics | ||||||
| B-cell depleting therapy (active and past) | O-CHOP + obinutuzumab maintenance (ongoing treatment) | R-CHOP Rituximab mono DHAP+rituximab High dose BEAM (5 m before admission) | Obinutuzumab + bendamustine (ongoing treatment) | Rituximab mono (last treatment 4 y ago) | (Venetoclax, ongoing treatment) | R-CHOP R-GemOx R-EPOCH (ongoing treatment) |
| Gammaglobulinemia, g/L (normal range, 7-16) | 2.4(IVIG)* | 3.8 | 8.3(IVIG)* | 5.4(SCIG)* | 5.4(IVIG)* | 5.7 |
| COVID-19 vaccination status (months since last dose) | Not vaccinated | Not vaccinated | One dose BNT162b2 (3) | Two doses BNT162b2 (2) | Two doses AZD1222 (4) | Two doses BNT162b2 (3) |
| SARS-CoV-2 IgG serostatus | Negative† | Negative† | 434 BAU/mL‡ | Negative† | Negative† | 8.2 BAU/mL† |
| Clinical course | ||||||
| Time from first positive PCR to nmAb treatment, d | 86 | 63 | 56 | 5 | 12 | 2 |
| Time from symptom onset to nmAb treatment, d | ∼56 | ∼63 | 13 | 11 | 13 | 2 |
| COVID-19 symptoms | Fever, dyspnea | Fever, cough | Fever, fatigue, cough | Fever, fatigue, dry cough | Fever, fatigue, dyspnea | Fever |
| WHO progression score (highest) | 6 | 5 | 5 | 5 | 4 | 4 |
| SARS-CoV-2 viremia,copies per mL | 1567 | 1336 | 1176 | 36 | 159 | 735 |
| Length of hospital stay after nmAb treatment, d | 8 | 11 | 7 | 12 | 10 | Ongoing hospitalization for treatment of lymphoma |
| . | Persistent or recurrent SARS-CoV-2 infection . | Serologically defined COVID-19 vaccine failure . | ||||
|---|---|---|---|---|---|---|
| . | Patient A . | Patient B . | Patient C . | Patient D . | Patient E . | Patient F . |
| Age, y | 48 | 56 | 63 | 78 | 63 | 39 |
| Sex | Male | Male | Male | Female | Male | Female |
| Type of B-NHL | Follicular | Follicular | Follicular | Unspecified | CLL | DLBCL |
| Basal characteristics | ||||||
| B-cell depleting therapy (active and past) | O-CHOP + obinutuzumab maintenance (ongoing treatment) | R-CHOP Rituximab mono DHAP+rituximab High dose BEAM (5 m before admission) | Obinutuzumab + bendamustine (ongoing treatment) | Rituximab mono (last treatment 4 y ago) | (Venetoclax, ongoing treatment) | R-CHOP R-GemOx R-EPOCH (ongoing treatment) |
| Gammaglobulinemia, g/L (normal range, 7-16) | 2.4(IVIG)* | 3.8 | 8.3(IVIG)* | 5.4(SCIG)* | 5.4(IVIG)* | 5.7 |
| COVID-19 vaccination status (months since last dose) | Not vaccinated | Not vaccinated | One dose BNT162b2 (3) | Two doses BNT162b2 (2) | Two doses AZD1222 (4) | Two doses BNT162b2 (3) |
| SARS-CoV-2 IgG serostatus | Negative† | Negative† | 434 BAU/mL‡ | Negative† | Negative† | 8.2 BAU/mL† |
| Clinical course | ||||||
| Time from first positive PCR to nmAb treatment, d | 86 | 63 | 56 | 5 | 12 | 2 |
| Time from symptom onset to nmAb treatment, d | ∼56 | ∼63 | 13 | 11 | 13 | 2 |
| COVID-19 symptoms | Fever, dyspnea | Fever, cough | Fever, fatigue, cough | Fever, fatigue, dry cough | Fever, fatigue, dyspnea | Fever |
| WHO progression score (highest) | 6 | 5 | 5 | 5 | 4 | 4 |
| SARS-CoV-2 viremia,copies per mL | 1567 | 1336 | 1176 | 36 | 159 | 735 |
| Length of hospital stay after nmAb treatment, d | 8 | 11 | 7 | 12 | 10 | Ongoing hospitalization for treatment of lymphoma |
N = 6.
BAU, binding antibody units; BEAM, carmustine (BCNU), etoposide, cytarabine, melphalan; CHOP, cyclophosphamide, hydroxydaunorubicin, vincristine, prednisone; CLL, chronic lymphocytic leukemia; DHAP, dexamethasone; high-dose cytarabine, cisplatin; DLBCL, diffuse large B-cell lymphoma; EPOCH etoposide, prednisone, vincristine, cyclophosphamide, hydroxydaunorubicin; GemOx, gemcitabine, oxaliplatin; O, obinutuzumab; and R, rituximab.
IVIG, intravenous immunoglobulin substitution; SCIG, subcutaneous immunoglobulin substitution.
Reference value: negative antibodies, <7.1 BAU/mL.
A live virus neutralization test (VNT) demonstrated poor neutralizing activity (100% inhibitory dilution [ID100] of 10).
Two unvaccinated patients (A and B) presented with COVID-19 persisting over more than 3 months, including constant viral shedding (the first positive polymerase chain reactions [PCRs] were 86 and 63 days, respectively, before nmAb treatment), viremia, recurrent fever episodes, and respiratory symptoms. Despite prolonged disease, both patients had nondetectable SARS-CoV-2–specific IgG. Patient A was receiving anti-CD20 maintenance therapy with obinutuzumab for follicular lymphoma. The initial detection of SARS-CoV-2 occurred during contact tracing in April 2021. Four weeks later, the patient developed symptomatic disease and presented at a secondary referral hospital. Despite treatment with anti-infective agents and dexamethasone, he showed progressive respiratory distress and was transferred to our intensive care unit for high-flow oxygen therapy where the respiratory situation stabilized during the following days. Although respiratory improvement was achieved, we observed recurrent fever episodes with persistent viral shedding and viremia so that treatment with nmAbs was initiated.
Patient B had a history of progressive follicular lymphoma and had received several regimens of immunochemotherapy, including the anti-CD20 antibodies obinutuzumab and rituximab, during the past years. In April, he was treated with high-dose chemotherapy (BEAM) and subsequent autologous stem cell transplantation. After the first positive SARS-CoV-2 PCR in August, he had recurrent fever episodes and a dry cough when he presented at the emergency room in late September. Based on a nondetectable SARS-CoV-2–specific IgG, despite prolonged infection and SARS-CoV-2 viremia, he was treated with nmAbs on the day after admission.
Patient C presented with the third episode of COVID-19, despite vaccination with 1 dose of BNT162b2 mRNA vaccine after the second episode and 3 months before the current episode. Remarkably, this patient had an IgG of 434 binding antibody units/mL upon admission, but the serum showed extremely low neutralization activity in a live SARS-CoV-2 virus neutralization test measuring a 100% inhibitory dilution of only 10. We hypothesized that the endogenous IgG antibodies were dysfunctional and had no neutralizing activity because of impaired B-cell function, and we subsequently treated the patient with nmAbs.
Three patients (D, E, and F) had serologically defined vaccination failure. Patients D and E, who had low-grade B-NHL and chronic lymphocytic lymphoma (both stable), respectively, were seronegative and presented with COVID-19 pneumonia and fever, despite having received a full course of vaccination 2 to 4 months earlier. The diagnostic approach and clinical course of patient F is of particular interest, as diagnosis required sampling the lower respiratory tract by bronchoalveolar lavage (BAL) and SARS-CoV-2 PCR from whole blood. In 2020, patient F was diagnosed with diffuse large B-cell lymphoma at the age of 38. After the initial chemotherapy resulted in a partial response, she underwent autologous stem cell transplantation in February 2021. Further disease progression led to additional treatment attempts in April and September 2021. COVID-19 vaccination was performed in May and July with BNT162b2 mRNA vaccine. In October, she was readmitted for conditioning chemotherapy and chimeric antigen receptor (CAR) T-cell immunotherapy for refractory diffuse large B-cell lymphoma. Despite full vaccination, she had an insufficient SARS-CoV-2 IgG titer (8.2 B binding antibody units/mL). Based on fever, elevated C-reactive protein levels, and progressive pulmonary ground-glass opacities visible on computed tomography, a BAL and additional diagnostics for viral and bacterial pathogens were performed (supplemental Figure 1). SARS-CoV-2 RNA was detected in the BAL (2 × 105 copies per mL) and blood (735 copies per mL) samples. Remarkably, a total of 4 oropharyngeal or nasopharyngeal swabs taken during admission including the initial screening assessment remained negative (supplemental Figure 1). No other pathogens were detected in blood or BAL samples.
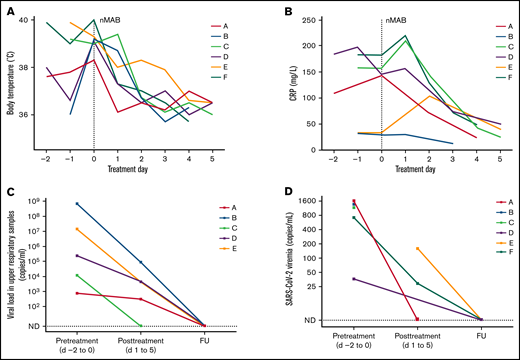
All patients were treated with 1 intravenous 1200-mg dose each of casirivimab and imdevimab. After infusion, we observed a rapid and significant clinical improvement during the following days. Fever abated within the first 48 hours, and the C-reactive protein level improved rapidly from the second day after treatment of those with marked elevation >50 mg/L (4 of 6 patients; Figure 1). Respiratory viral loads decreased from a median of 450 × 103 copies per milliliter before treatment (day −2 to 0) to 1 × 103 copies per milliliter in the posttreatment phase (days 1-5). Measurable viremia was present in all patients. In patients with data available (4 of 6), viremia had resolved in posttreatment or follow-up assessments after nmAb treatment (Figure 1). Five of 6 patients were discharged from the hospital within 8 to 12 days after treatment. Patient F received CAR T-cell immunotherapy 7 days after nmAb treatment, which led to control of COVID-19–associated symptoms and viral clearance from the blood (supplemental Figure 1). No other COVID-19–associated treatments were administered concomitantly with the nmAbs. At follow-up assessments within a period of 2 months of discharge, sustained cure of COVID-19 was confirmed in all patients, with an absence of the related symptoms. Negative SARS-CoV-2 reverse transcription-PCR (RT-PCR) of upper respiratory tract samples were available from 5 of the 6 patients at the follow-up assessment.
Evolution of maximum daily body temperature, CRP, and SARS-CoV-2 viral load, before and after treatment with nmAbs. Evolution of temperature (A) and C-reactive protein (B) in each patient from 2 days before (day −2) to 5 days after (day 5) nmAb treatment (day 0). SARS-CoV-2 RNA was determined by quantitative RT-PCR, and cycle threshold values were translated to viral concentrations based on the respective PCR core standards. Viral loads obtained from respiratory tract specimens (C) and blood (D) before and after nmAb treatment are presented cumulatively and represent samples that were taken from 2 days before until midday at the day of nmAb treatment (pre-treatment) or from days 1 to 5 after treatment (post-treatment). If several samples were taken during these time periods the median from all available assessments is shown for each patient. Patient F is not included in panel C, as only samples obtained from BAL were positive and no subsequent assessments were performed. CRP, C-reactive protein; FU, follow-up; ND, not detected.
Evolution of maximum daily body temperature, CRP, and SARS-CoV-2 viral load, before and after treatment with nmAbs. Evolution of temperature (A) and C-reactive protein (B) in each patient from 2 days before (day −2) to 5 days after (day 5) nmAb treatment (day 0). SARS-CoV-2 RNA was determined by quantitative RT-PCR, and cycle threshold values were translated to viral concentrations based on the respective PCR core standards. Viral loads obtained from respiratory tract specimens (C) and blood (D) before and after nmAb treatment are presented cumulatively and represent samples that were taken from 2 days before until midday at the day of nmAb treatment (pre-treatment) or from days 1 to 5 after treatment (post-treatment). If several samples were taken during these time periods the median from all available assessments is shown for each patient. Patient F is not included in panel C, as only samples obtained from BAL were positive and no subsequent assessments were performed. CRP, C-reactive protein; FU, follow-up; ND, not detected.
In conclusion, our series of 6 patients with COVID-19 who had B-NHL illustrates a poor humoral immune response to SARS-CoV-2 vaccination and infection that results in persistent viral shedding, viremia, and symptomatic disease. We also found that measurable SARS-CoV-2–specific IgG does not inevitably translate into equivalent neutralizing activity. The observation that patients can present with SARS-CoV-2 pneumonia and viremia while having negative RT-PCR results from nasopharyngeal swabs should be further investigated, as it may have implications for the diagnostic approach in immunocompromised patients. Our data suggest that testing for the neutralizing capacity of SARS-CoV-2–specific IgG (if detectable) and SARS-CoV-2 RT-PCR performed on whole-blood samples can be a rational addition to the diagnostic workup of patients with COVID-19 who have underlying B-NHL. Most important, we observed favorable and rapid treatment responses to nmAbs in a specific group of patients presenting with vaccination failure or prolonged or recurrent COVID-19 and SARS-CoV-2 viremia related to humoral deficiency. Thus, nmAbs may provide an attractive treatment option for treating patients with COVID-19 who are unable to mount a humoral immune response after vaccination or infection, irrespective of the duration of symptoms. Clinical studies exploiting the full potential of nmAbs for treatment and prophylaxis in these patients are warranted.
Acknowledgments: The visual abstract was created with BioRender.com.
This study was supported by funding received from the COVIM project of the NaFoUniMedCovid19 network (FKZ: 01KX2021).
Contribution: J.R. and J.J.M. initiated intensified observations and documentation of relevant cases; J.J.M. wrote the primary manuscript and prepared the figure; V.D.C., E.H. and E.K. performed the molecular and serologic diagnostics; J.J.M., J.R., P.B., C.H., E.P., J.G.B., B.B., C.L., N.J., and P.B. were involved in patient care for the described cases; R.K., G.F., F.K., M.H., and J.R. supervised data collection and interpretation; and all authors critically reviewed the primary manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Jan Rybniker, Department I for Internal Medicine, Center for Molecular Medicine Cologne, University of Cologne, Robert-Koch-Strasse 21, 50931 Cologne, Germany; e-mail: jan.rybniker@uk-koeln.de.
References
Author notes
The data used and analyzed will be provided by the corresponding author (jan.rybniker@uk-koeln.de) on reasonable request and in agreement with protection of data privacy.
The full-text version of this article contains a data supplement.