Key Points
Patients with SCNSL have poor prognosis.
ASCT for large B-cell lymphoma patients with secondary central nervous system is associated with prolonged survival.
Abstract
Secondary central nervous system large B-cell lymphoma (SCNSL) is rare, with a generally poor prognosis. There is limited data about the role of autologous stem cell transplantation (ASCT) in these high-risk patients. We explored in this study treatment outcomes and prognostic factors for patients with SCNSL who underwent ASCT. We included all consecutive patients who underwent ASCT at our institution. Primary endpoints were progression-free survival (PFS) and overall survival (OS). One-hundred two patients were identified. Median age at transplant was 56 (range, 21-71) years. With a median follow-up of 56 (range, 1-256) months, the median PFS and OS were 40 and 88 months, respectively. The 4-year PFS and OS were 48% and 57%, respectively. In univariate analysis, complete remission (CR) at transplant, prior lines of therapy (≤2), normal lactate dehydrogenase, and parenchymal involvement were significantly associated with improved PFS. For OS, only CR at transplant and ≤2 prior lines of therapy were associated with improved survival. On multivariable analysis for PFS, CR at transplant (hazard ratio [HR], 0.278; 95% CI, 0.153-0.506; P ≤ .0001) and ≤2 prior lines of therapy (HR, 0.485; 95% CI, 0.274-0.859; P = .0131) were significantly associated with superior PFS. Similarly, CR at transplant (HR, 0.352; 95% CI, 0.186-0.663; P = .0013) and ≤2 prior lines of therapy (HR, 0.476; 95% CI, 0.257-0.882; P = .0183) were associated with improved survival. In the largest single-center study, our findings indicate that ASCT is associated with durable responses and prolonged survival in patients with SCNSL. Patients in CR at transplant and those who received ≤2 lines of therapy have particularly excellent outcomes.
Introduction
Non-Hodgkin lymphoma (NHL) consists of a heterogeneous group of malignant lymphoproliferative disorders with an estimated 74 200 new cases in 2019,1 of which diffuse large B-cell lymphoma (DLBCL) is the most common subtype, accounting for ∼32% of cases.2 It is estimated ∼2% to 10% of patients with NHLs may develop secondary central nervous system lymphoma (SCNSL), the majority of which are derived from aggressive B-cell NHLs in the relapsed setting.3-6 The overall prognosis of SCNSL is extraordinarily poor despite remarkable advances in treatment, with median survival of 2 to 5 months across several studies.7,8
There remains an unmet need to standardize treatment of patients with SCNSL. Given the rarity of the disease, the associated morbidity, and the overall poor prognosis, patients with SCNSL are, unfortunately, frequently excluded from clinical trials, including studies exploring novel agents. Autologous stem cell transplantation (ASCT) with high-dose chemotherapy (HDC) remains the standard of care treatment for patients with relapsed chemosensitive DLBCL.9,10 HDC and ASCT have been used anecdotally for patients with SCNSL who are otherwise deemed eligible for transplant, with promising durable remissions achieved.11,12 However, most of these were small observational studies and included heterogeneous patient populations with a variety of lymphoma subtypes.
We aim with this report to present data from a large single-center study evaluating the prognostic factors and treatment outcomes of HDC/ASCT in a homogenous patient population, all of which have large B-cell lymphoma with secondary central nervous system (CNS) involvement.
Methods
Study design and patients
All consecutive patients with histologically confirmed large B-cell lymphoma and with CNS involvement who underwent ASCT between September 1990 and March 2019 were identified. We included in this retrospective analysis all adult patients who were 18 years or older. Patients with primary CNS lymphoma were excluded from this analysis. SCNSL was defined as large B-cell lymphoma involving the brain parenchyma, leptomeninges, cerebrospinal fluid, and/or eyes, either at initial presentation or at time of relapse. Patients with neurolymphomatosis or spinal cord involvement were also included. If tissue biopsy or cytology was not available, diagnosis of SCNSL was concluded by imaging. Primary endpoints were progression-free survival (PFS) and overall survival (OS). Secondary endpoints included cumulative incidence of relapse (CIR), non-relapse mortality (NRM), and exploring prognostic factors for both PFS and OS. The study was approved by the Institutional Review Board of The University of Texas MD Anderson Cancer Center and was conducted in accordance with the Declaration of Helsinki.
Statistics
Descriptive statistics were used to characterize the patient cohort. PFS was measured from the date of transplant to date of disease progression or death, and patients who were alive without disease progression at last follow-up were censored. OS was calculated from date transplant to last known vital status, and patients were censored if they were alive at last follow-up. PFS and OS rates were computed using Kaplan-Meier estimates and compared using the log-rank test with statistical significance value of P < .05. CIR and NRM were estimated using the competing risks method. Univariate and multivariate analyses were performed using Cox proportional hazards regression models to assess for significant predictive risk factors for PFS and OS. We used a cutoff P value of <.1 to include univariate risk factors in multivariate analyses. All other statistical tests used a significance level of 5%. All statistical analyses were performed using SAS 9.4 for Windows (SAS Institute Inc., Cary, NC).
Results
Patient characteristics
A total of 102 patients with SCNSL who underwent HDC/ASCT were identified during the study period. All patients had a confirmed diagnosis with aggressive large B-cell lymphoma, 91% (n = 93) of which were de novo DLBCL, 4% each (n = 4) were transformed large B-cell and discordant/composite lymphomas, and 1 patient had lymphoma not otherwise specified. The median age at transplant was 56 (range, 21-71) years, with male predominance. Of all study patients, 24 had CNS involvement at time of initial diagnosis, 75 had CNS involvement upon progression/relapse, and for 3 patients, the timing could not be determined from available records. Of the 24 patients who had CNS involvement at initial diagnosis, 12 patients received upfront consolidation with ASCT while in first remission and 12 received ASCT upon relapse (7 had systemic disease alone without CNS involvement at time of relapse). Of the 92 patients with CNS involvement prior to last treatment before transplant, 26% (n = 24) had isolated CNS relapse and 74% (n = 68) had concurrent systemic disease and CNS involvement. Leptomeningeal disease (LMD) with/without parenchymal disease was the most frequent site of CNS involvement (45%), followed by parenchymal disease (30%). Median number of prior lines of therapies was 2 (range, 1-6). Data about frontline systemic therapy was available for 99 patients, of which 67% received cyclophosphamide, doxorubicin, vincristine, and prednisone–based and 20% received etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin–based regimens. The majority of patients (n = 58) received high-dose methotrexate-based therapy prior to transplant. A variety of conditioning regimens were used during the study period, at the discretion of the treating physician, with carmustine, etoposide, cytarabine, and melphalan (BEAM) with/without rituximab being the most frequently used regimen (53%), followed by thiotepa-based (24.5%) and gemcitabine, busulfan, and melphalan–based (17.5%) regimens. The other 5% (n = 5) of patients received other, less commonly used regimens. Baseline patient, disease, and transplant characteristics are outlined in Table 1.
Baseline patient, disease, and transplant characteristics
| Patient characteristics . | Frequency (%) n = 102* . |
|---|---|
| Age | |
| <60 y | 63 (62) |
| ≥60 y | 39 (38) |
| Gender | |
| Female | 40 (39) |
| Male | 62 (61) |
| KPS at transplant* | |
| ≥90 | 69 (69) |
| 70-80 | 31 (31) |
| Stage at diagnosis* | |
| Stage 1/2 | 22 (22) |
| Stage 3/4 | 78 (78) |
| IPI at transplant | |
| >2 | 34 (33) |
| ≤2 | 68 (67) |
| LDH at transplant* | |
| High | 40 (40) |
| Normal | 61 (60) |
| CNS involvement site | |
| Leptomeningeal | 35 (34) |
| Parenchymal | 31 (30) |
| Leptomeningeal and parenchymal | 11 (11) |
| CSF only | 8 (8) |
| Other† | 17 (17) |
| Extent of disease prior to transplant‡ | |
| CNS alone | 24 (24) |
| CNS and systemic | 68 (69) |
| Systemic alone | 7 (7) |
| Prior lines of therapy | |
| ≤2 | 68 (67) |
| >2 | 34 (33) |
| Time from diagnosis to transplant | |
| >18 mo | 49 (48) |
| ≤18 mo | 53 (52) |
| Disease status at transplant | |
| CR/CRu | 76 (75) |
| Not remission | 26 (25) |
| CNS directed therapy before transplant* | |
| MTX-based regimen | 28 (30) |
| ARA-C–based regimen | 21 (23) |
| MTX- and ARA-C–based regimen | 30 (32) |
| Intrathecal alone | 14 (15) |
| Conditioning regimen§ | |
| BEAM +/− rituximab | 54 (53) |
| Thiotepa-based | 25 (24) |
| GBM-based | 18 (18) |
| Other | 5 (5) |
| Patient characteristics . | Frequency (%) n = 102* . |
|---|---|
| Age | |
| <60 y | 63 (62) |
| ≥60 y | 39 (38) |
| Gender | |
| Female | 40 (39) |
| Male | 62 (61) |
| KPS at transplant* | |
| ≥90 | 69 (69) |
| 70-80 | 31 (31) |
| Stage at diagnosis* | |
| Stage 1/2 | 22 (22) |
| Stage 3/4 | 78 (78) |
| IPI at transplant | |
| >2 | 34 (33) |
| ≤2 | 68 (67) |
| LDH at transplant* | |
| High | 40 (40) |
| Normal | 61 (60) |
| CNS involvement site | |
| Leptomeningeal | 35 (34) |
| Parenchymal | 31 (30) |
| Leptomeningeal and parenchymal | 11 (11) |
| CSF only | 8 (8) |
| Other† | 17 (17) |
| Extent of disease prior to transplant‡ | |
| CNS alone | 24 (24) |
| CNS and systemic | 68 (69) |
| Systemic alone | 7 (7) |
| Prior lines of therapy | |
| ≤2 | 68 (67) |
| >2 | 34 (33) |
| Time from diagnosis to transplant | |
| >18 mo | 49 (48) |
| ≤18 mo | 53 (52) |
| Disease status at transplant | |
| CR/CRu | 76 (75) |
| Not remission | 26 (25) |
| CNS directed therapy before transplant* | |
| MTX-based regimen | 28 (30) |
| ARA-C–based regimen | 21 (23) |
| MTX- and ARA-C–based regimen | 30 (32) |
| Intrathecal alone | 14 (15) |
| Conditioning regimen§ | |
| BEAM +/− rituximab | 54 (53) |
| Thiotepa-based | 25 (24) |
| GBM-based | 18 (18) |
| Other | 5 (5) |
ARA-C, cytarabine; CR, complete response; CRu, complete response uncertain; CSF, cerebrospinal fluid; GBM, gemcitabine, busulphan, melphalan; IPI, international prognostic index; KPS, Karnofsky performance scale; MTX, methotrexate.
Numbers do not add up for a total of 102 in some subgroups because of missing information, unless otherwise specified.
Included patients with isolated neurolymphomatosis, intramedullary spinal dissemination, and others not otherwise classified such as epidural with nerve root or spine involvement.
Disease involvement prior to salvage therapy for relapsed patients and at presentation for patients who underwent upfront consolidation with transplant.
BEAM and GBM-based conditioning were given as previously described (references 18,–20). Thiotepa-based regimens included BCNU/thiotepa plus or minus rituximab (n = 17) and thiotepa/busulfan/cyclophosphamide plus or minus rituximab (n = 8). Five patients received “other” less commonly used regimens.
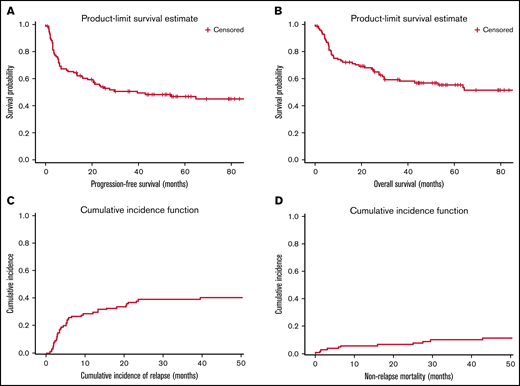
Efficacy
With a median follow-up of 56 (range, 1-256) months, the median PFS and OS of all study patients were 40 and 88 months, respectively (Figure 1). The 1-year, 2-year, and 4-year PFS rates were 65%, 54%, and 48%, respectively. The respective 1-year, 2-year, and 4-year OS rates were 72%, 68%, and 57%, respectively.
Survival, relapse and mortality rates for all study patients. (A) PFS. (B) OS. (C) CIR. (D) NRM.
Survival, relapse and mortality rates for all study patients. (A) PFS. (B) OS. (C) CIR. (D) NRM.
In univariate analysis (Table 2), disease status (reference, CR) at transplant (HR for PFS, 4.883; 95% CI, 2.806-8.499 [P < .001]; HR for OS, 3.971; 95% CI, 2.215-7.718 [P < .0001]) and >2 prior lines of therapy (HR for PFS, 2.849; 95% CI, 1.663-4.882 [P = .0001]; HR for OS, 3.045; 95% CI, 1.713-5.411 [P = .0001]) were significantly associated with worse PFS and OS. Additionally, parenchymal brain involvement (reference, LMD) was associated with favorable PFS (HR, 0.495; 95% CI, 0.245-0.998; P = .0493), and high LDH at transplant was associated with inferior PFS (HR, 2.08; 95% CI, 1.216-3.557; P = .0075). There was a trend for inferior OS with high LDH (HR, 1.709; 95% CI, 0.968-3.017; P = .0646).
Univariable analysis for PFS and OS
| . | . | PFS . | OS . | Reference . | ||
|---|---|---|---|---|---|---|
| Variable . | . | HR (95% CI) . | P value . | HR (95% CI) . | P value . | |
| Age | Age ≥60 y | 1.247 (0.729-2.133) | .4206 | 1.327 (0.753-2.338) | .3282 | Age <60 y |
| Gender | Female | 0.983 (0.546-1.649) | .8527 | 0.919 (0.508-1.661) | .7798 | Male |
| KPS | KPS 70-80 | 1.366 (0.779-2.397) | .2765 | 1.421 (0.789-2.56) | .242 | KPS 90-100 |
| Stage | 3-4 | 0.957 (0.504-1.819) | .8938 | 0.786 (0.408-1.512) | .4705 | Stage 1-2 |
| LDH | High LDH | 1.105 (0.584-2.09) | .7585 | 1.179 (0.6-2.316) | .6331 | Normal LDH |
| IPI at transplant | IPI at SCT ≥2 | 1.432 (0.774-2.649) | .2528 | 1.093 (0.558-2.14) | .7963 | IPI at SCT <2 |
| LDH at transplant | High LDH | 2.08 (1.216-3.557) | .0075 | 1.709 (0.968-3.017) | .0646 | Normal LDH |
| CNS involvement site | Parenchymal | 0.495 (0.245-0.998) | .0493 | 0.529 (0.252-1.108) | .0915 | LMD |
| Leptomeningeal and parenchymal | 0.913 (0.392-2.131) | .8341 | 0.95 (0.401-2.252) | .9078 | ||
| CSF Only | 0.762 (0.263-2.208) | .6162 | 0.958 (0.326-2.816) | .9385 | ||
| Others* | 0.635 (0.293-1.378) | .2511 | 0.55 (0.231-1.306) | .1752 | ||
| Extent of disease prior to transplant | CNS and Systemic | 1.677 (0.813-3.458) | .1619 | 1.512 (0.724-3.156) | .2707 | Isolated CNS relapse |
| Systemic alone | 1.115 (0.302-4.122) | .8703 | 1.197 (0.323-4.429) | .7878 | ||
| Prior lines of therapy | >2 | 2.849 (1.663-4.882) | .0001 | 3.045 (1.713-5.411) | .0001 | Prior lines of therapy ≤2 |
| Time from diagnosis to transplant | ≤18 mo | 0.714 (0.418-1.218) | .2158 | 0.613 (0.347-1.084) | .0926 | >18 mo |
| Disease status at transplant | Not in remission | 4.883 (2.806-8.499) | <.0001 | 3.971 (2.215-7.118) | <.001 | CR/CRu |
| CNS-directed therapy | Cytarabine-based | 0.983 (0.385-2.515) | .9722 | 1.203 (0.413-3.505) | .7343 | Intrathecal chemotherapy alone |
| Methotrexate-based | 1.037 (0.418-2.572) | .9375 | 1.241 (0.437-3.529) | .6852 | ||
| Methotrexate/Cytarabine-based | 1.238 (0.512-2.994) | .6363 | 1.556 (0.567-4.270) | .3909 | ||
| Conditioning | Thiotepa-based | 1.495 (0.-2.83) | .2166 | 1.131 (0.5-2.317) | .7367 | BEAM |
| GBM-based | 1.508 (0.7-3.217) | .2876 | 1.411 (0.6-3.139) | .3983 | ||
| . | . | PFS . | OS . | Reference . | ||
|---|---|---|---|---|---|---|
| Variable . | . | HR (95% CI) . | P value . | HR (95% CI) . | P value . | |
| Age | Age ≥60 y | 1.247 (0.729-2.133) | .4206 | 1.327 (0.753-2.338) | .3282 | Age <60 y |
| Gender | Female | 0.983 (0.546-1.649) | .8527 | 0.919 (0.508-1.661) | .7798 | Male |
| KPS | KPS 70-80 | 1.366 (0.779-2.397) | .2765 | 1.421 (0.789-2.56) | .242 | KPS 90-100 |
| Stage | 3-4 | 0.957 (0.504-1.819) | .8938 | 0.786 (0.408-1.512) | .4705 | Stage 1-2 |
| LDH | High LDH | 1.105 (0.584-2.09) | .7585 | 1.179 (0.6-2.316) | .6331 | Normal LDH |
| IPI at transplant | IPI at SCT ≥2 | 1.432 (0.774-2.649) | .2528 | 1.093 (0.558-2.14) | .7963 | IPI at SCT <2 |
| LDH at transplant | High LDH | 2.08 (1.216-3.557) | .0075 | 1.709 (0.968-3.017) | .0646 | Normal LDH |
| CNS involvement site | Parenchymal | 0.495 (0.245-0.998) | .0493 | 0.529 (0.252-1.108) | .0915 | LMD |
| Leptomeningeal and parenchymal | 0.913 (0.392-2.131) | .8341 | 0.95 (0.401-2.252) | .9078 | ||
| CSF Only | 0.762 (0.263-2.208) | .6162 | 0.958 (0.326-2.816) | .9385 | ||
| Others* | 0.635 (0.293-1.378) | .2511 | 0.55 (0.231-1.306) | .1752 | ||
| Extent of disease prior to transplant | CNS and Systemic | 1.677 (0.813-3.458) | .1619 | 1.512 (0.724-3.156) | .2707 | Isolated CNS relapse |
| Systemic alone | 1.115 (0.302-4.122) | .8703 | 1.197 (0.323-4.429) | .7878 | ||
| Prior lines of therapy | >2 | 2.849 (1.663-4.882) | .0001 | 3.045 (1.713-5.411) | .0001 | Prior lines of therapy ≤2 |
| Time from diagnosis to transplant | ≤18 mo | 0.714 (0.418-1.218) | .2158 | 0.613 (0.347-1.084) | .0926 | >18 mo |
| Disease status at transplant | Not in remission | 4.883 (2.806-8.499) | <.0001 | 3.971 (2.215-7.118) | <.001 | CR/CRu |
| CNS-directed therapy | Cytarabine-based | 0.983 (0.385-2.515) | .9722 | 1.203 (0.413-3.505) | .7343 | Intrathecal chemotherapy alone |
| Methotrexate-based | 1.037 (0.418-2.572) | .9375 | 1.241 (0.437-3.529) | .6852 | ||
| Methotrexate/Cytarabine-based | 1.238 (0.512-2.994) | .6363 | 1.556 (0.567-4.270) | .3909 | ||
| Conditioning | Thiotepa-based | 1.495 (0.-2.83) | .2166 | 1.131 (0.5-2.317) | .7367 | BEAM |
| GBM-based | 1.508 (0.7-3.217) | .2876 | 1.411 (0.6-3.139) | .3983 | ||
ARA-C, cytarabine; CRu, complete response uncertain; CSF, cerebrospinal fluid; GBM, gemcitabine, busulphan, melphalan; HR, hazard ratio; KPS, Karnofsky performance scale; LDH, lactate dehydrogenase.
Included patients with isolated neurolymphomatosis, intramedullary spinal dissemination, and others not otherwise classified such as epidural with nerve root or spine involvement.
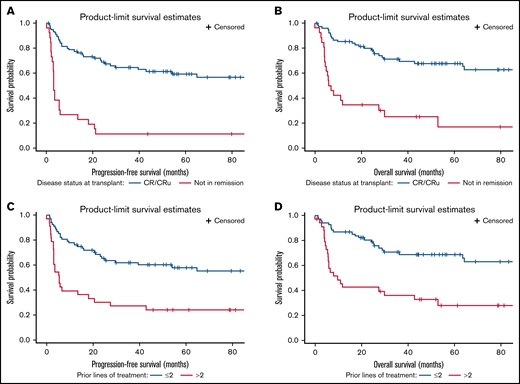
In multivariable analysis (Table 3), only being in remission at transplant (HR, 0.268; 95% CI, 0.142-0.506; P < .0001) and ≤2 prior lines of therapy (HR, 0.483; 95% CI, 0.266-0.876; P = .0165) remained significant predictors for improved PFS. Achieving complete remission (HR, 0.344; 95% CI, 0.175-0.677; P = .002) and ≤2 prior lines of therapy (HR, 0.475; 95% CI, 0.252-0.896; P = .0214) were also significant predictors for improved OS. The 4-year PFS and OS for patients transplanted while in complete remission were 61% and 68%, respectively, compared with 12% and 25%, respectively, for those with residual disease at transplant. (Figure 2) The 4-year PFS and OS for patients who received ≤2 prior lines of therapy were 60% and 69%, respectively, compared with 24% and 33%, respectively, for those who received >2 prior lines of therapy (Figure 2).
Multivariable analysis for PFS and OS
| . | . | PFS . | OS . | Reference . | ||
|---|---|---|---|---|---|---|
| Variable . | . | HR (95% CI) . | P value . | HR (95% CI) . | P value . | |
| LDH at transplant | High LDH | 1.559 (0.887-2.739) | .1229 | 1.305 (0.725-2.348) | .3745 | Normal LDH |
| CNS involvement site | Leptomeningeal | 1.659 (0.781-3.522) | .1878 | 1.789 (0.801-3.998) | .156 | Parenchymal |
| Leptomeningeal and Parenchymal | 1.702 (0.653-4.433) | .2762 | 1.488 (0.553-4.003) | .4312 | ||
| CSF Only | 2.143 (0.67-6.849) | .1986 | 2.676 (0.806-8.882) | .1078 | ||
| Others* | 1.019 (0.396-2.622) | .9697 | 0.939 (0.331-2.667) | .9064 | ||
| Prior lines of therapy | ≤2 | 0.483 (0.266-0.876) | .0165 | 0.475 (0.252 - 0.896) | .0214 | Prior lines of therapy >2 |
| Time from diagnosis to transplant | ≤18 mo | 0.643 (0.354-1.167) | .1462 | >18 mo | ||
| Disease status at transplant | CR/ Cru | 0.268 (0.142-0.506) | <.0001 | 0.344 (0.175-0.677) | .002 | Not in remission |
| . | . | PFS . | OS . | Reference . | ||
|---|---|---|---|---|---|---|
| Variable . | . | HR (95% CI) . | P value . | HR (95% CI) . | P value . | |
| LDH at transplant | High LDH | 1.559 (0.887-2.739) | .1229 | 1.305 (0.725-2.348) | .3745 | Normal LDH |
| CNS involvement site | Leptomeningeal | 1.659 (0.781-3.522) | .1878 | 1.789 (0.801-3.998) | .156 | Parenchymal |
| Leptomeningeal and Parenchymal | 1.702 (0.653-4.433) | .2762 | 1.488 (0.553-4.003) | .4312 | ||
| CSF Only | 2.143 (0.67-6.849) | .1986 | 2.676 (0.806-8.882) | .1078 | ||
| Others* | 1.019 (0.396-2.622) | .9697 | 0.939 (0.331-2.667) | .9064 | ||
| Prior lines of therapy | ≤2 | 0.483 (0.266-0.876) | .0165 | 0.475 (0.252 - 0.896) | .0214 | Prior lines of therapy >2 |
| Time from diagnosis to transplant | ≤18 mo | 0.643 (0.354-1.167) | .1462 | >18 mo | ||
| Disease status at transplant | CR/ Cru | 0.268 (0.142-0.506) | <.0001 | 0.344 (0.175-0.677) | .002 | Not in remission |
CRu, complete response uncertain; CSF, cerebospinal fluid.
Included patients with isolated neurolymphomatosis, intramedullary spinal dissemination, and others not otherwise classified such as epidural with nerve root or spine involvement.
Comparison of survival outcomes by disease status at transplant and prior lines of therapy. (A-B) PFS and OS by disease status. (C-D) PFS and OS by prior lines of therapy. CR/CRu, complete response/complete response uncertain.
Comparison of survival outcomes by disease status at transplant and prior lines of therapy. (A-B) PFS and OS by disease status. (C-D) PFS and OS by prior lines of therapy. CR/CRu, complete response/complete response uncertain.
Relapse and deaths
During the study period, 42 (41%) patients progressed, and 49 (48%) patients died. The 1-, 2-, and 4-year CIRs were 26%, 39%, and 40%, respectively, and the respective NRM rates were 6%, 7%, and 11% (Figure 1). The majority (90%) of relapses occurred within 2 years of transplant, and none progressed after 5 years, but 1 patient who had disease relapse at 65 months from transplant. Twenty-nine (69%) patients had systemic disease at relapse with (n = 5) or without (n = 24) CNS relapse. Isolated CNS relapse occurred in 31% (n = 13) of patients. Seventy-one percent of deceased patients (n = 35) died of relapsed disease. Common causes of NRM were second malignancy (n = 4), infections (n = 4), cardiac (n = 2), 1 each for pulmonary toxicity and hepatic veno-occlusive disease, and 2 patients had no documented cause of death.
Subgroup analyses
Disease status and prior lines of therapy.
Disease status at transplant and prior lines of therapy were strongly predictive for survival in multivariable analysis. Hence, we explored outcomes stratifying patients by these 2 variables. The 2-year and 4-year PFS/OS for patients who transplanted in remission and received ≤2 lines of prior therapy were 68%/82% and 62%/71%, respectively, compared with 2-year and 4-year PFS/OS of 0%/21% and 0%/14%, respectively, for those who had residual disease at transplant and received >2 prior lines of therapy. The other 2 groups, patients who had >2 prior lines but were in remission at transplant and those who received ≤2 prior lines of therapy but had residual disease at transplant, have relatively a favorable intermediate prognosis (supplemental Figure).
Long-term outcomes.
We explored long-term outcomes for a subgroup of patients who were alive at 5 years after transplant (n = 31). With a median follow up of 8.9 (range, 5-21.4) years, both median PFS and OS were not reached, and the 8-year PFS and OS rates were 82% and 83%, respectively.
Isolated CNS relapse.
Twenty-four patients had isolated CNS relapse prior to salvage therapy and transplant. All patients received CNS-directed salvage treatment; a combination of high-dose MTX plus cytarabine-based therapy was the most common regimen (n = 14), followed by high-dose MTX-based (n = 7) and high-dose cytarabine (n = 3). Eleven patients received radiation therapy prior to transplant, of which in 9 included whole-brain radiation therapy, and 1 each received ocular and γ knife radiation. Ten patients received BEAM plus or minus rituximab, 9 patients received thiotepa-based conditioning, and 5 patients received other, less commonly used regimens. The PFS and OS of these patients were not statistically significant different compared with patients with concurrent CNS and systemic disease. Seven of the 24 patients relapsed after transplant; all had CNS involvement at time progression (3 received prior whole-brain radiation therapy, and 4 received thiotepa-based conditioning).
CNS involvement for patients with relapsed/refractory lymphoma.
Eighty patients, at a median age of 57 (range, 21-71) years, underwent ASCT for relapsed/refractory disease with evidence of CNS lymphoma at time of progression. Overall, the results were comparable to all study patients with 4 year-PFS and OS of 47% and 58%, respectively. In univariate analysis, disease status (reference, CR) at transplant (HR for PFS, 5.331; 95% CI, 2.889-9.835 [P < .001]; HR for OS, 4.149; 95% CI, 2.157-7.980 [P < .0001]) and >2 prior lines of therapy (HR for PFS, 2.339; 95% CI, 1.290-4.242 [P = .005]; HR for OS, 2.648; 95% CI, 1.387-5.057 [P = .0032]) were significantly associated with worse PFS and OS. Additionally, parenchymal brain involvement (reference, LMD) was associated with favorable PFS (HR, 0.432; 95% CI, 0.202-0.921; P = .0298), and high LDH at transplant was associated with inferior PFS (HR, 1.954; 95% CI, 1.076-3.548; P = .0278), and age ≥60 years was associated with worse OS (HR, 1.952; 95% CI, 1.038-3.671; P = .038). Patients with active disease at transplant and who received >2 prior lines of therapy have again dismal prognosis, with 4-year PFS and OS of 0% and 20%, respectively.
Discussion
Patients with SCNSL generally have a poor prognosis. In the largest single institutional study to explore the outcomes of secondary CNS involvement by large B-cell lymphoma, our findings show HDC/ASCT is associated with durable remissions and improved survival. Patients achieving complete remission prior to transplant and/or receiving ≤2 lines of prior lines of therapy have an excellent prognosis. Patients who transplanted with active disease and received >2 lines of therapy (n = 19) had very poor prognosis, with 4-year OS of 14%.
There remains no consensus-driven standardized guidelines for treatment of SCNSL, particularly in the relapsed setting. Although treatment in the context of a clinical trial setting is best for these high-risk patients, the rarity of this disease entity and the associated morbidity at presentation have excluded them from most therapeutic clinical trials. Hence, treatment and outcome data are largely derived from observational studies, most of which were small case series or registry data, which included heterogeneous patient population with multiple lymphoma subtypes. We report durable outcomes in patients with SCNSL, all of which had large B-cell lymphoma and received an intensified consolidation therapy with HDC/ASCT. Our findings are consistent with others,13 indicating HDC/ASCT should be a preferred option for transplant-eligible patients with SCNSL. In the large study by the Center for International Blood and Marrow Transplant Research,13 authors compared transplant outcomes of 151 patients with SCNSL (83 patients with DLBCL) to 4688 patients with no CNS involvement and found no statistically significant differences in PFS and OS between the 2 groups. Without transplant, patients with SCNSL, particularly in the relapsed setting, have had historically dismal outcomes even with intensified non-transplant regimens, and cure is an exception for these high-risk patients.7,14,15 For instance, of 173 patients with SCNSL treated with intensive regimens in a multicenter international study,7 the overall median OS was 7.5 months, but patients who underwent HDC/ASCT (n = 25) had notable improved median OS of 61 months, with 2-year OS of 65%. A more recent smaller (n = 79) but prospective phase 2 study confirmed the same results for improved outcomes for patients who underwent HDC/ASCT (n = 37).16
Several potential prognostic factors were explored to identify susceptible groups that might benefit the most from HDC/ASCT (Table 2). Consistent with other studies,7,13 we have showed achieving complete remission is a strong predictor for improved survival. In one report,7 achieving complete remission was associated with improved outcomes for a subgroup of patients with isolated CNS relapse (n = 38) regardless of proceeding with transplant; however, only 13 patients underwent transplant in this cohort. In contrast, Ferreri et al reported significantly improved survival with HDC/ASCT (n = 20) in a homogenous group of 38 patients who achieved complete remission after induction therapy.17 Another strong predictor of outcomes in our cohort was the number of prior lines of therapy. Similar to our experience in patients without CNS lymphoma,18 patients who received >2 prior lines of therapy had inferior outcomes. Realizing the limitations of cross-trial comparisons, the 4-year OS of 34% for patients who received >2 prior lines of therapy was comparable to our previously published non-SCNSL report (n = 93) with 5-year OS of 30%.18 In a subgroup analysis combining disease status and number of prior lines of therapy, we were able to identify a subgroup of ultra-high-risk patients (those with active disease and received >2 prior lines of therapy) who were unlikely to benefit from HDC/ASCT. Alternative options should be sought for these patients, including novel therapeutics and a consideration for allogeneic stem cell transplantation.
Several limitations inherent to retrospective studies are to be acknowledged. First, we included patients who were treated in over 2 decades. However, given the rarity of SCNSL, collecting data over a prolonged period becomes essential to understanding the natural history and prognostic factors of these high-risk patients. We were indeed able to show that patients who survive beyond 5 years are potentially cured by HDC/ASCT; median PFS and OS are not reached yet with a median follow-up of 8.9 years. Second, we did not include in our analysis data on some of the baseline risk factors at diagnosis (eg, IPI and LDH) as this information was missing in many patients. Pertinent risk factors at relapse/prior to transplant were available for most patients, and these are thought to provide more meaningful prognostic value on whom may or may not benefit from HDC/ASCT. Third, we did not analyze outcomes by cell of origin or genetic rearrangements because their use was limited during the study period. Finally, this was a noncontrolled single-arm study with a potential for selection bias for a fit group of patients who were otherwise eligible for transplant. Nevertheless, our findings are of great importance, showing excellent long-term outcomes associated with HDC/ASCT compared to published historical controls. Recognizing the difficulty of conducting randomized clinical trials for this rare entity, we hope large collaborative groups can at minimum initiate prospective controlled observational studies to gather treatment and outcome data, in addition to baseline disease and patient characteristics, which would help us better understand the nature of this disease and perhaps risk-stratify and standardize treatments for these high-risk patients.
In conclusion, in a large homogenous cohort of patients with large B-cell lymphoma with CNS involvement, our findings indicate that consolidation with HDC/ASCT is highly effective, leading to durable remissions and is potentially curative. Results are particularly encouraging for patients who achieve complete remission prior to transplant and/or those who are less heavily pretreated.
Authorship
Contribution: S.A.S. conceived and designed the research and performed statistical analysis; S.A. and S.A.S. analyzed and interpreted data and wrote the manuscript; S.A., C.H., I.K., Sairah Ahmed, A.A., N.F., J.J., J.T., J.L.R., F.M., G.R., M.D., J.S.I., R.S., J.W., S.P.I., B.D., P.A., U.R.P., M.H.Q., C.R.F., E.S., R.E.C., Y.N., and S.A.S. critically reviewed and edited the manuscript for important intellectual content; and all the authors contributed to the acquisition and interpretation of data and approved the final manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Samer A. Srour, Department of Stem Cell Transplantation and Cellular Therapy, The University of Texas MD Anderson Cancer Center, 1515 Holcombe Blvd, Houston, TX 77030; e-mail: ssrour@mdanderson.org.
References
Author notes
For data sharing, contact the corresponding author: ssrour@mdanderson.org.
The full-text version of this article contains a data supplement.