Key Points
The induction of an activated profile in NK cells by NKTR-255 correlates with an effective enhancement of the anti-MM effector functions.
NKTR-255 increases NK-mediated ADCC against MM cells and partially overcomes daratumumab-induced NK cell depletion in vivo.
Abstract
Multiple myeloma (MM) is characterized by an immunosuppressive microenvironment that enables tumor development. One of the mechanisms of immune evasion used by MM cells is the inhibition of natural killer (NK) cell effector functions; thus, the restoration of NK cell antitumor activity represents a key goal to increase tumor cell recognition, avoid tumor escape and potentially enhancing the effect of other drugs. In this study, we evaluated the ability of the investigational medicine NKTR-255, an IL-15 receptor agonist, to engage the IL-15 pathway and stimulate NK cells against MM cells. We observed that incubation with NKTR-255 was able to tilt the balance toward an activated phenotype in NK cells isolated from peripheral blood mononuclear cells of patients with MM, with increased expression of activating receptors on the surface of treated NK cells. This resulted in an enhanced degranulation, cytokine release, and anti-tumor cytotoxicity when the NK cells were exposed to both MM cell lines and primary MM cells. We further evaluated the in vivo effect of NKTR-255 in fully humanized immunocompetent mice subcutaneously engrafted with H929 MM cells. Compared with placebo, weekly injection of the mice with NKTR-255 increased the number of circulating NK cells in peripheral blood and delayed tumor growth. Finally, we observed that combination of NKTR-255 with the anti-CD38 antibody, daratumumab, was effective against MM cells in vitro and in vivo. Taken together, our data suggest a significant impact of NKTR-255 in inducing NK cell function against MM cells with important translational implications.
Introduction
Multiple myeloma (MM) is a highly heterogeneous disease that resides in a complex ecosystem composed of immune, endothelial, and stromal cells.1-4 The introduction of agents targeting MM cells in their microenvironment, such as immunomodulatory drugs, has markedly improved the survival of patients with MM.5 In MM, however, the efficacy of immune-based approaches is hampered by an immunosuppressive bone marrow (BM) milieu,6-8 which includes defective antigen processing and presentation by dendritic cells,9,10 increased numbers of myeloid-derived suppressor cells with immunosuppressive activity,11,12 and/or promotion of plasma cell survival through IL-6 and VEGF release by macrophages.3 Moreover, decline in total number of natural killer (NK) and natural killer T (NKT) cells has been observed in advanced stages of the disease along with impairment of their effector functions.13-19
During the past few years, different immunotherapeutic strategies have been designed to increase tumor cell recognition, avoid tumor escape, and enhance efficacy of anti-cancer agents by harnessing the mechanisms of a patient’s own immune system. Among these strategies, cytokines represent a potential therapeutic resource due to their capability to control the proliferation of the different immune subsets and increase anti-tumor cytotoxicity.20
Interleukin-15 (IL-15) is structurally and functionally related to interleukin-2 (IL-2); both share the β- and γ-components of their receptors21 and promote the proliferation and cytolytic properties of NK and T cells, enhancing innate and adaptive immunity.22-25 Even when the receptor complexes act through similar transduction cascades, their activation results in different functional activities in lymphocytes.26 Recombinant human IL-15 (rhIL-15) has the advantage of preferentially acting on effector cells compared with regulatory T cells,27,28 and seems to have a better safety profile than IL-2, among other distinctions between the 2 cytokines.23,29 The rhIL-15–primed NK cells from patients with MM displayed enhanced ex vivo functional responses against autologous MM cells30,31; and rhIL-15 delayed senescence of memory CD8+ T cells, improving the effectiveness of adoptive T cell therapy in a MM murine model.32
The safety and potential to control tumor proliferation by administration of rhIL-15 as a single agent have been tested in different phase-I clinical trials in human.33,34 However, the triggering of cytokine release and macrophage activation syndromes as well as the instability and early renal clearance of rhIL-15 has limited its clinical use.34,35 Here we have evaluated the efficacy of NKTR-255,36,37 an investigational IL-15 receptor agonist. NKTR-255 is a polymer-conjugated rhIL-15 that, through optimal in vivo engagement of the IL-15Rα/IL-2Rβγ receptor complex, may enhance formation of long-term immunological memory and lead to restored activity against MM cells. NKTR-255 displays an extended in vivo half-life with reduced renal clearance, sustained signaling, and prolonged pharmacodynamic effect.36-38 In studies with rodent and non-human primates, a single dose of NKTR-255 enabled a sustained activation of the IL-15 receptor pathway, specifically in NK cells, but also in memory CD8+ T cells, maintaining and enhancing their proliferation and effector functions over time.37-40
In the present study, we show that treatment of NK cells with NKTR-255 resulted in increased degranulation and release of pro-inflammatory cytokines after tumor exposure, leading to improved NK-mediated killing of MM cells. NKTR-255 synergized with the FDA-approved CD38-directed cytolytic antibody, daratumumab, ex vivo and in vivo in a humanized mouse model of MM, providing support for its clinical use as a novel immunotherapeutic agent for treatment of myeloma.
Materials and methods
Cells
NCI-H929 (Cat.#CRL-9068) and U266B1 (Cat.#TIB-196) were purchased from American Type Culture Collection (Manassas, VA), and KMS12BM (Cat.#JCRB0429) and KMS26 (Cat.#JCRB1187) were purchased from the Japanese Collection of Research Bioresources Cell Bank (Osaka, Japan). Cell lines were cultured in Roswell Park Memorial Institute 1640 (RPMI 1640; Gibco [Gaithersburg, MD], Cat.#10379144) supplemented with 10% fetal bovine serum (Gibco, Cat.#10437010), 2 mmol/L l-glutamine (Gibco, Cat.#35050061), 100 mg/mL streptomycin (Gibco, Cat.#10378016) and 100 U/mL penicillin (Gibco, Cat.#10378016). Effector cells were obtained from blood aspirate of patients with MM treated at Dana-Farber Cancer Institute (DFCI); samples from healthy donors were obtained through the Kraft Family Blood Donor Center at DCFI and Brigham and Women’s Hospital. Human NK cells were isolated using a commercial kit (Miltenyi Biotec, Bergisch Gladbach, Germany, Cat.#130-092-657) through negative selection with immunomagnetic columns (Miltenyi Biotec, Cat.#130-042-401) following the manufacturer’s instructions. NK cells were isolated immediately prior the assays and after expansion and activation of total peripheral blood mononuclear cells (PBMCs).
NK immunophenotyping assays
Total PBMCs were collected and cultured at 0.5 to 1 × 106 cells/mL with NKTR-255 at a concentration of 1000 ng/mL. NK cells were recognized and gated as CD3-CD56+ and expression of the different markers was assessed at baseline and after treatment, taking the median fluorescence intensity as reference. For this assay the following combination of fluorochrome conjugated anti-human monoclonal antibodies was employed: anti-CD3-PE/Cy7 (Biolegend, Cat.#300420), anti-CD56-BV786 (BD Biosciences, Cat.#564058), anti-NKG2A-APC (R&D Systems [Minneapolis, MN] Cat.#FAB1059A-100), anti-NKG2C-AlexaFluor488 (R&D Systems, Cat.#FAB138G-025), anti-NKG2D-BV421 (Biolegend, Cat.#320821), anti-NKp30-PE (Biolegend, Cat.#325207), anti-NKp46-BV421 (Biolegend, Cat.#331914), anti-DNAM1-APC (Biolegend, Cat.#338311), anti-LAG3-FITC (Invitrogen [Waltham, MA], Cat.#11-2239-42), anti-TRAIL-PE (Biolegend, Cat.#308206), anti-TIGIT-FITC (Invitrogen, Cat.#11-9500-42) and anti-KIR2D-PE (Biolegend, Cat.#312606). LIVE/DEAD fixable dead cell stain (ThermoFisher Scientific [Waltham, MA], Cat.#L34966) was included to label and discard dead cells. Analysis was performed using a BD LSRFortessa X-20 (BD Biosciences) cytometer and FlowJo v9 (FlowJo LLC, Ashland, OR) software.
Cytotoxicity assay
Direct NK cytotoxicity and antibody-dependent cellular cytotoxicity (ADCC) against MM cells were evaluated through a standard flow cytometry approach previously described.41 Briefly, myeloma target cells previously labeled with CellTrace Violet (ThermoFisher Scientific, Cat.#C34557) were cocultured with NK cells at different target-to-effector (T:E) ratios for 4 hours. The percentage of dead target cells was estimated by flow cytometry using the fixable viability dye eFluor 780 (ThermoFisher Scientific, Cat.#501129035). Killing efficiency was obtained after correcting by background, considered as the spontaneous lysis of target cells not exposed to NK cells. The same approach was applied to assess the ADCC of NK cells upon treatment of MM cells with commercially obtained daratumumab (Darzalex®). For these experiments, the CellTrace Violet-labeled target cells were incubated for 30 minutes with daratumumab at the concentrations indicated and washed to remove excess antibody before co-incubation with NK cells. All experiments were performed in triplicate per biological sample and condition.
Degranulation assay
Briefly, NK cells previously stimulated with NKTR-255 or rhIL-15 were collected and cultured for 4 hours alone or with CTV-stained MM cells at a T:E ratio of 1:1 in a 96-well U-bottom plate with 200 μL/well of culture medium without cytokines and 5 μL/well of anti-CD107a-PE (BD Biosciences, Cat.#555801) or mouse IgG1-PE isotype control (BD Biosciences, Cat.#555749). After 1 hour of incubation at 37°C, 5 μL/well of a monensin solution produced by mixing 2 μL of GolgiStop (BD Biosciences, Cat.#BDB554724) and 75 μL of culture medium was added. Following the 4 hours of incubation, cells were washed and stained in FACS buffer with anti-CD3-PerCP/Cy5.5 (Biolegend, Cat.#317336) and anti-CD56-BV786 (BD Biosciences, Cat.#564058), followed by flow cytometry evaluation as mentioned above.
Cytokine release assay
NK cells were incubated for 16 hours in a 96-well U-bottom plate in triplicate, alone or in the presence of myeloma cells at T:E ratio of 1:2, and the levels of IFN-γ and TNF-α in the supernatant were measured using commercial ELISA kits (Invitrogen, Cat.#EHIFNG and Cat.#BMS2034, respectively) according to the manufacturer’s instructions.
Ex vivo assays
To assess the effect of NKTR-255 on the NK cell compartment and MM cells in an autologous setting, BM samples from patients newly diagnosed with MM were collected under sterile conditions and processed within 24 hours of extraction to minimize loss of tumor cellularity. Whole BM samples were diluted with IMDM (Gibco, Cat.#12440053), supplemented with 20% (vol/vol) autologous serum, 2% HEPES (Gibco, Cat.#11560496), 1% ZellShield antimicrobial solution (Labclinics, Barcelona, Spain, Cat.#13-0050), and 1% l-glutamine (Lonza, Hopkinton, MA, Cat.#BE17-605E) to a final volume of 60 μL per well. For this experiment, 96-well plates were previously prepared using an Echo 550 Liquid Handler (LabCyte, Sunnyvale, CA) containing 4 increasing NKTR-255 doses. Whole BM samples were maintained in culture for 120 hours at the pre-specified concentrations of NKTR-255, including control wells without the compound. Drug effect on BM MM and NK cell populations was measured ex vivo using an automated flow cytometry platform that enables the counting of absolute numbers of live cells as described previously.42 Based on these absolute counts, the T:E ratio was estimated at baseline. NKG2D and CD69 expressions were analyzed as markers for NK cell activation. To identify live plasma cells and NK cells, the following combination of antibodies was employed: annexin V-FITC (Immunostep, Salamanca, Spain, Cat.#ANXVF-200T), anti-CD138-PE (Cytognos, Salamanca, Spain, Cat.#CYT-138PE4), anti-CD69-PerCP (Biolegend, Cat.#310928), anti-CD3-Pacific Blue (BD Biosciences Cat.#558117), anti-CD45-Pacific Orange (Invitrogen, Cat.#MHCD4530), anti-CD38-APC (Biolegend, Cat.#356606), and anti-CD56-APC/Cy7 (Biolegend, Cat.#318332).
In vivo studies
All animal experiments were ethically conducted complying with the U.S. Public Health Service Policy on Human Care and Use of Laboratory Animals. The experiments were carried out in a female NOD/Shi-scid/IL-2Rγnull immunodeficient mouse strain (NCG) at the facilities of TransCure bioServices (TCS) SAS (Archamps, France). Mice were humanized using hematopoietic stem cells (CD34+) isolated from human cord blood following TCS’s proprietary humanization protocol. Only mice with a humanization rate (hCD45/total CD45) above 25% were used. The hu-NCG mice used in this experiment were enhanced for myeloid cell populations by receiving (1 week before tumor engraftment) a boost based on the transient expression of the recombinant human cytokines IL-15, IL-3, IL-4, and GM-CSF after hydrodynamic liver injection. Seven days after hydrodynamic liver injection of the recombinant human cytokines IL-15, IL-3, IL-4, and GM-CSF, mice were engrafted with 5 × 106 NCI-H929 MM cells subcutaneously. Mice were randomized to receive vehicle, NKTR-255 0.3 mg/kg weekly, daratumumab 5 mg/kg weekly, or NKTR-255 0.3 mg/kg weekly + daratumumab 5 mg/kg weekly, when the average tumor volume reached 50 mm3. Day 0 was defined as the first day of treatment. All procedures described in this study have been reviewed and approved by the local ethics committee (Comité d’Éthique Local pour l’Expérimentation Animale – GenevoisCELEAG).
Readouts from the in vivo study
Mice were monitored daily for unexpected signs of distress. Body weight was monitored thrice weekly, to check for bodyweight loss > 25%. The tumor volume was monitored using a caliper and the formula: 1/2 length × width2. Mice were sacrificed after 2 weeks of treatment for phenotypic analysis. Flow cytometry was performed on cells isolated from blood and analyses performed on an Attune NxT Flow Cytometer (Life Technologies, Carlsbad, CA). Blood (100 μL) was collected from the retro-orbital sinus in EDTA-coated tubes (Microvette 100 MCVT100-EDTA; Sarstedt, Cat.#20.1278.100). Red blood cells were lysed with High Yield Lyse buffer (Life Technologies, Cat.#HYL250) at RT for 15 min. Samples were incubated with human FcR blocking reagent (Miltenyi Biotec, Cat.#130-059-901), stained with surface antibodies and viability dye for 30 min at 4°C prior to washing with flow cytometry staining buffer (Invitrogen, Cat.#00-4222) before analysis.
Statistical analysis
Data were analyzed using GraphPad Prism 9 (GraphPad Software, San Diego, CA) and comparisons were performed using a two-sample t-test or an ANOVA test for multiple comparisons. In the case of unequal variance between groups a non-parametric test was employed. Results for quantitative variables are presented as mean ± standard deviation depicted as error bars in the figures. A two-sided value of P < .05 was considered significant for the experiments performed.
Results
NKTR-255 enhances antitumor responses of human NK cells against MM target cells
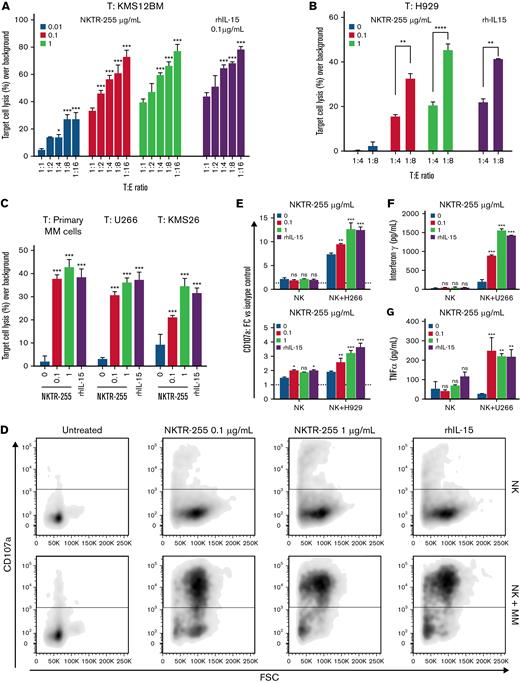
With the role of rhIL-15 in regulating NK cell proliferation and activity,43,44 we first assessed the ability of NKTR-255 to augment NK cytotoxic function and induce effector cell-dependent killing of target MM cell lines. We observed a significant dose and T:E ratio-dependent increase in cytotoxicity following stimulation of purified CD56+CD3- NK cells from healthy donors (Figure 1A) or patients with myeloma (Figure 1B-C) with either NKTR-255 or rhIL-15 used as positive control in a panel of MM cell lines. The efficacy of NKTR-255 to induce activation of myeloma patient-derived NK cells was also confirmed against primary MM cells from a patient with myeloma (Figure 1C).
NKTR-255 enhances effector functions of NK cells and increases their responsiveness to MM cell exposure, displaying higher cytotoxicity against MM target cells. PBMCs from healthy donors or patients with MM were treated for 7 days with and without different doses of NKTR-255 or 0.1 μg/mL rhIL-15 (as positive control) and NK cells were subsequently isolated by negative selection using immunomagnetic columns prior to every assay. MM cell lines were labeled with CTV stain and co-incubated with the effector cells in a 4-hour in vitro cytotoxicity assay. The percentage of target cell lysis was assessed by flow cytometry and corrected by background lysis. (A) Dose and T:E ratio-dependency was tested using NK cells from healthy donors as effector cells and KMS12BM MM cell line as target cells. (B) Dose and T:E ratio-dependency was tested using NK cells from a patient with myeloma as effector cells and H929 MM cell line as target cells. (C) NK cells isolated from patients with myeloma were treated with different concentrations of NKTR-255 or rhIL-15 and tested against either primary MM cells isolated from BM aspirate of a patient newly diagnosed with MM or U266 and KMS26 MM cell lines as target cells. The percentage of target cell killing over background (spontaneous lysis of target cells not exposed to NK cells) is shown and results are depicted as mean ± standard deviation (SD). (D-E) CD107a cell surface expression was used as a surrogate for quantifying cellular degranulation of NK cells. CD107a expression on NK cells incubated with different doses of NKTR-255 or rhIL-15 was measured at rest and after target MM cell exposure by flow cytometry. (F and G) IFN-γ (F) and TNFα (G) released by NK cells isolated from patients with MM and incubated with different doses of NKTR-255 or rhIL-15 were measured by ELISA assay before and after the co-incubation with different MM cell lines. Results are expressed as concentration (pg/mL) of cytokine in the culture supernatant and compared for statistical analysis with non-treated NK cells. Data shown represent mean ± SD of triplicates for each condition in the same experiment. ∗P < .05, ∗∗P < .01, ∗∗∗P < .001, nsP ≥ .05. BM, bone marrow; MM, multiple myeloma; NK, natural killer; ns, not significant; PBMCs, peripheral blood mononuclear cells; SD, standard deviation; T, target; T:E, target-to-effector.
NKTR-255 enhances effector functions of NK cells and increases their responsiveness to MM cell exposure, displaying higher cytotoxicity against MM target cells. PBMCs from healthy donors or patients with MM were treated for 7 days with and without different doses of NKTR-255 or 0.1 μg/mL rhIL-15 (as positive control) and NK cells were subsequently isolated by negative selection using immunomagnetic columns prior to every assay. MM cell lines were labeled with CTV stain and co-incubated with the effector cells in a 4-hour in vitro cytotoxicity assay. The percentage of target cell lysis was assessed by flow cytometry and corrected by background lysis. (A) Dose and T:E ratio-dependency was tested using NK cells from healthy donors as effector cells and KMS12BM MM cell line as target cells. (B) Dose and T:E ratio-dependency was tested using NK cells from a patient with myeloma as effector cells and H929 MM cell line as target cells. (C) NK cells isolated from patients with myeloma were treated with different concentrations of NKTR-255 or rhIL-15 and tested against either primary MM cells isolated from BM aspirate of a patient newly diagnosed with MM or U266 and KMS26 MM cell lines as target cells. The percentage of target cell killing over background (spontaneous lysis of target cells not exposed to NK cells) is shown and results are depicted as mean ± standard deviation (SD). (D-E) CD107a cell surface expression was used as a surrogate for quantifying cellular degranulation of NK cells. CD107a expression on NK cells incubated with different doses of NKTR-255 or rhIL-15 was measured at rest and after target MM cell exposure by flow cytometry. (F and G) IFN-γ (F) and TNFα (G) released by NK cells isolated from patients with MM and incubated with different doses of NKTR-255 or rhIL-15 were measured by ELISA assay before and after the co-incubation with different MM cell lines. Results are expressed as concentration (pg/mL) of cytokine in the culture supernatant and compared for statistical analysis with non-treated NK cells. Data shown represent mean ± SD of triplicates for each condition in the same experiment. ∗P < .05, ∗∗P < .01, ∗∗∗P < .001, nsP ≥ .05. BM, bone marrow; MM, multiple myeloma; NK, natural killer; ns, not significant; PBMCs, peripheral blood mononuclear cells; SD, standard deviation; T, target; T:E, target-to-effector.
The perforin/granzyme (granule exocytosis) pathway is a major mechanism used by NK cells to kill targets. To assess for degranulation and cytokine production, purified NK cells from patients with MM were cultured for 7 days with or without rhIL-15 or NKTR-255. Cells were then washed and triggered with target tumor cells, after which degranulation (measured as CD107a surface expression) and cytokine production (IFN-γ and TNFα) were assessed. NKTR-255 or IL-15 treatment robustly enhanced degranulation (Figure 1D-E) and enhanced release of IFN-γ and TNFα (Figure 1F-G) by NK cells in response to myeloma target cells in a dose-dependent manner. Importantly, we did not observe any direct effect of NKTR-255 or rhIL-15 on growth and viability of MM cells (supplemental Figure 1A-B).
Altogether, these results suggest that NKTR-255 enhances NK cell effector functions against myeloma cells.
NKTR-255 increases ex vivo expression of NK activating receptors and adhesion molecules
NK cell function depends on the integration and balance of multiple inhibitory and activation signals received through a broad repertoire of receptors, adhesion molecules, and co-stimulatory signals.45,46
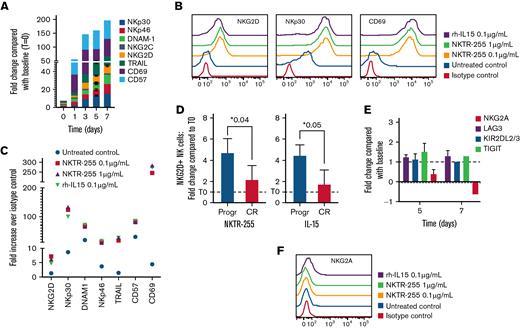
We therefore evaluated the impact of NKTR-255 on the expression of a panel of surface proteins on NK cells purified from patients with MM, including markers of activation and exhaustion, and receptors involved in tumor cell recognition and induction of cell death. A significant increase of the activating receptors NKG2D, DNAM-1, NKp30, and NKp46 required for tumor cell recognition and killing was observed over time on the surface of myeloma NK cells following stimulation with NKTR-255 or rhIL-15, but not in the untreated NK cells (Figure 2A-C). We also looked at the expression of alternate mediators of NK cell target killing and observed a significant increase of the death receptor ligand TRAIL which has been shown to regulate the signaling downstream of the IL-15 receptor leading to enhanced NK cell-mediated killing (Figure 2A-C).31,47 Moreover, the expression of the activation markers CD57 and CD69, which have been shown to sustain NK cell cytotoxicity, was also significantly enhanced after treatment (Figure 2A-C).48-51
NK cells treated with NKTR-255 are characterized by increased expression of stimulatory receptors and activation markers. (A) NK cells purified from patients with MM were continuously cultured with NKTR-255 1 μg/mL for up to 7 days and the expression pattern of indicated surface markers was tracked by flow cytometry at different time points. Graphics show the fold-change of MFI compared with the MFI at day 0 (before treatment with NKTR-255) for each time point. (B-C) PBMCs were collected from patients with MM samples and cultured with and without NKTR255 or rhIL-15. After 5 days, PBMCs were stained with CD3 and CD56 antibodies to gate NK cell population as well as with indicated surface proteins. The expression profile of these markers was measured by flow cytometry. Representative histograms of the isotype control and the 4 culture conditions are shown (B). Fold change increase over isotype control in untreated and treated samples is shown in the graph (C). (D) PBMCs from patients with MM with progressive disease (n = 3) and complete response (n = 6) were treated with 1 μg/mL of NKTR-255 or 100 ng/ml of rh-IL15 for 5 days. PBMCs were then stained with CD3 and CD56 antibodies to gate NK cell population and NKG2D. Results are expressed as mean ± standard deviation of fold-change in absolute count after 5 days. (E) NK cells purified from patients with MM were continuously cultured with NKTR-255 1 μg/mL for up to 7 days and the expression pattern of indicated surface markers was tracked by flow cytometry at different time points. Graphics show the fold-change of MFI compared with the MFI at day 0 (before treatment with NKTR-255) for each time point. (F) PBMCs were collected from peripheral blood of patients with MM and culture with and without NKTR255 or rhIL-15. After 5 days, PBMCs were stained with CD3 and CD56 antibodies, to gate NK cell population, as well as with NKG2A antibody or isotype control. Representative histograms of the isotype control and the 4 culture conditions are shown in the graph. ∗P < .05. CR, complete response; MFI, median fluorescence intensity; MM, multiple myeloma; ns, not significant; NK, natural killer; PBMCs, peripheral blood mononuclear cells; Progr, progression; SD, standard deviation.
NK cells treated with NKTR-255 are characterized by increased expression of stimulatory receptors and activation markers. (A) NK cells purified from patients with MM were continuously cultured with NKTR-255 1 μg/mL for up to 7 days and the expression pattern of indicated surface markers was tracked by flow cytometry at different time points. Graphics show the fold-change of MFI compared with the MFI at day 0 (before treatment with NKTR-255) for each time point. (B-C) PBMCs were collected from patients with MM samples and cultured with and without NKTR255 or rhIL-15. After 5 days, PBMCs were stained with CD3 and CD56 antibodies to gate NK cell population as well as with indicated surface proteins. The expression profile of these markers was measured by flow cytometry. Representative histograms of the isotype control and the 4 culture conditions are shown (B). Fold change increase over isotype control in untreated and treated samples is shown in the graph (C). (D) PBMCs from patients with MM with progressive disease (n = 3) and complete response (n = 6) were treated with 1 μg/mL of NKTR-255 or 100 ng/ml of rh-IL15 for 5 days. PBMCs were then stained with CD3 and CD56 antibodies to gate NK cell population and NKG2D. Results are expressed as mean ± standard deviation of fold-change in absolute count after 5 days. (E) NK cells purified from patients with MM were continuously cultured with NKTR-255 1 μg/mL for up to 7 days and the expression pattern of indicated surface markers was tracked by flow cytometry at different time points. Graphics show the fold-change of MFI compared with the MFI at day 0 (before treatment with NKTR-255) for each time point. (F) PBMCs were collected from peripheral blood of patients with MM and culture with and without NKTR255 or rhIL-15. After 5 days, PBMCs were stained with CD3 and CD56 antibodies, to gate NK cell population, as well as with NKG2A antibody or isotype control. Representative histograms of the isotype control and the 4 culture conditions are shown in the graph. ∗P < .05. CR, complete response; MFI, median fluorescence intensity; MM, multiple myeloma; ns, not significant; NK, natural killer; PBMCs, peripheral blood mononuclear cells; Progr, progression; SD, standard deviation.
Importantly, a significant expansion of NKG2D-positive NK cells in PBMCs from 9 patients with MM was observed after treatment with NKTR-255 (or rhIL-15), which was more pronounced in patients with progressive or refractory disease compared with those patients who had achieved a disease response (Figure 2D). Conversely, the surface expression of receptors with an inhibitory role such as NKG2A and KIR2DL2/3 was only marginally impacted by NKTR-255 (Figure 2E-F).
Augmenting NK cell number and functions is effective against MM cells in the context of their BM milieu
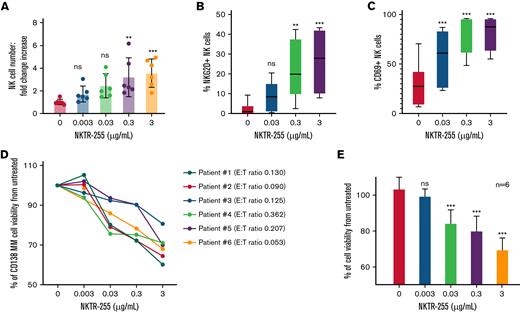
For a more accurate assessment of the effect of NKTR-255 on NK cell activity and MM cell survival in an autologous setting in the presence of the BM milieu, we cultured whole BM samples from 6 treatment-naïve patients newly diagnosed with MM with and without 4 different 10-fold increasing doses of NKTR-255 for 5 days. Using an automated flow cytometry assay, we measured the absolute numbers of CD138+ MM cells and NK cells at baseline and after 5 days of incubation in the treated and untreated samples, detecting a dose-dependent increase in the total number of BM NK cells following stimulation with NKTR-255 (Figure 3A; supplemental Figure 2A). We also assessed the expression of NKG2D and CD69 on NK cells as markers of NK activation and observed an increased number of NKG2D- and CD69-positive NK cells compared with untreated cells (Figure 3B-C; supplemental Figure 2B-C). Importantly, NKTR-255 treatment caused a significant reduction of viability of CD138+ MM cells compared with untreated samples in a dose-dependent manner (Figure 3D-E).
NKTR-255 induces proliferation and activation of BM-derived NK cells, reducing viability of autologous MM cells. (A) Whole BM samples (n = 6) from patients newly diagnosed with MM without prior treatment were incubated for 120 hours with and without 4 increasing doses of NKTR-255. The effect of the compound on the NK (effector) and MM (target) cell populations in whole BM was assessed through an automated flow cytometry platform that allows for absolute cell counting. Based on this data, T:E ratio for each sample was calculated at baseline and it was not modified since the whole BM sample was employed unaltered for this assay. Absolute numbers of NK cells within whole BM samples were measured at baseline and after 120 hours of incubation and results are presented as mean ± standard deviation fold increase in NK cell absolute number in the 6 patients according to concentration level of NKTR-255. (B-C) CD69 and NKG2D expression was measured as a marker of NK cell activation. Percentages of NK cells expressing NKG2D (B) and CD69 (C) over total NK cell population after 120 hours of incubation at different NKTR-255 concentrations are shown as mean and range (minimum to maximum). (D-E) Percentage of live CD138+ MM cells in BM samples of each patient (D) was calculated at baseline and after 120 hours of culture with and without different doses of NKTR-255. Mean of CD138+ cell viability compared with untreated is shown (E). ∗∗P < .01, ∗∗∗P < .001, nsP ≥ .05. BM, bone marrow; E:T, effector-to-target; ns, not significant; NK, natural killer; MM, multiple myeloma; SD, standard deviation.
NKTR-255 induces proliferation and activation of BM-derived NK cells, reducing viability of autologous MM cells. (A) Whole BM samples (n = 6) from patients newly diagnosed with MM without prior treatment were incubated for 120 hours with and without 4 increasing doses of NKTR-255. The effect of the compound on the NK (effector) and MM (target) cell populations in whole BM was assessed through an automated flow cytometry platform that allows for absolute cell counting. Based on this data, T:E ratio for each sample was calculated at baseline and it was not modified since the whole BM sample was employed unaltered for this assay. Absolute numbers of NK cells within whole BM samples were measured at baseline and after 120 hours of incubation and results are presented as mean ± standard deviation fold increase in NK cell absolute number in the 6 patients according to concentration level of NKTR-255. (B-C) CD69 and NKG2D expression was measured as a marker of NK cell activation. Percentages of NK cells expressing NKG2D (B) and CD69 (C) over total NK cell population after 120 hours of incubation at different NKTR-255 concentrations are shown as mean and range (minimum to maximum). (D-E) Percentage of live CD138+ MM cells in BM samples of each patient (D) was calculated at baseline and after 120 hours of culture with and without different doses of NKTR-255. Mean of CD138+ cell viability compared with untreated is shown (E). ∗∗P < .01, ∗∗∗P < .001, nsP ≥ .05. BM, bone marrow; E:T, effector-to-target; ns, not significant; NK, natural killer; MM, multiple myeloma; SD, standard deviation.
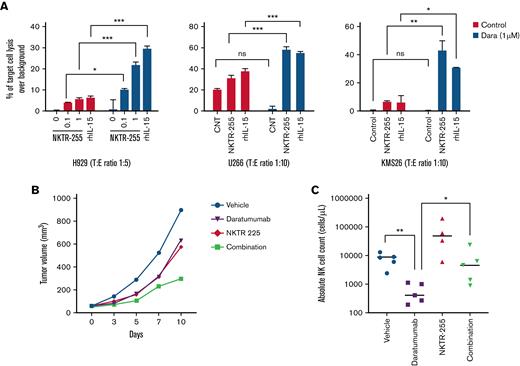
NKTR-255 enhances in vitro ADCC of NK cells and synergizes with daratumumab to reduce MM growth in a humanized MM mouse model
Daratumumab and its use in combination with other agents is becoming a new standard of care for the treatment of MM.52 Daratumumab mediates its anti-MM activity by binding to the CD38 that is highly expressed on MM cells, causing ADCC. To determine whether NKTR-255 was also able to enhance the NK cell-mediated ADCC against MM cells, we performed standard 4-hour ADCC assays using NK cells from patients with MM as effector cells and MM cells pre-treated with daratumumab as target cells. This led to an increased MM cell killing as compared with control conditions using MM cells that had not been pre-incubated with daratumumab, suggesting an important NK cell role mediated through ADCC. Daratumumab-mediated ADCC against MM cells was limited in the presence of NK cells not previously activated with NKTR-255 (Figure 4A); however, ADCC was significantly augmented when NK cells were stimulated with NKTR-255. Enhanced NK cell-dependent ADCC was confirmed in the presence of rhIL-15 as a positive control as well.
NKTR-255 enhances in vitro ADCC of NK cells and synergizes with daratumumab to reduce MM growth in a humanized MM mouse model. (A) NK cells isolated from peripheral blood mononuclear cells of patients with MM treated for 14 days with NKTR-255 at indicated doses (or rhIL-15 as positive control) were employed to test ADCC. MM cell lines were labeled with CTV stain, pre-treated with or without daratumumab, and co-incubated with the effector cells in a 4-hour in vitro cytotoxicity assay. The percentage of target cell killing over background (spontaneous lysis of target cells not exposed to NK cells) is shown and results are depicted as mean ± standard deviation. (B-C) A fully humanized mouse model engrafted with NCI-H929 MM tumor cell line was used to study the efficacy of NKTR-255, daratumumab, or their combination on tumor growth and NK cell proliferation. Humanized mice were boosted with the human recombinant cytokines IL-15, IL-3, IL-4 and GM-CSF. One week after boost, mice were engrafted with 5 × 106 NCI-H929 cells subcutaneously. When tumors reached an average volume of 50 mm3 these mice were randomized into vehicle, NKTR-255 (0.3 mg/kg weekly), daratumumab (5 mg/kg weekly), or NKTR-255 (0.3 mg/kg weekly) + daratumumab (5 mg/kg weekly). Tumor volume (B) was monitored 3 times a week using a caliper. Blood samples were collected at the end of the experiment for immunophenotyping, and the absolute count of NK cells was assessed per cohort of treatment (C). N = 5 mice/group. ∗P < .05, ∗∗P < .01, ∗∗∗P < .001, nsP ≥ .05. CNT, control; MM, multiple myeloma; ns, not significant; NK, natural killer; SD, standard deviation.
NKTR-255 enhances in vitro ADCC of NK cells and synergizes with daratumumab to reduce MM growth in a humanized MM mouse model. (A) NK cells isolated from peripheral blood mononuclear cells of patients with MM treated for 14 days with NKTR-255 at indicated doses (or rhIL-15 as positive control) were employed to test ADCC. MM cell lines were labeled with CTV stain, pre-treated with or without daratumumab, and co-incubated with the effector cells in a 4-hour in vitro cytotoxicity assay. The percentage of target cell killing over background (spontaneous lysis of target cells not exposed to NK cells) is shown and results are depicted as mean ± standard deviation. (B-C) A fully humanized mouse model engrafted with NCI-H929 MM tumor cell line was used to study the efficacy of NKTR-255, daratumumab, or their combination on tumor growth and NK cell proliferation. Humanized mice were boosted with the human recombinant cytokines IL-15, IL-3, IL-4 and GM-CSF. One week after boost, mice were engrafted with 5 × 106 NCI-H929 cells subcutaneously. When tumors reached an average volume of 50 mm3 these mice were randomized into vehicle, NKTR-255 (0.3 mg/kg weekly), daratumumab (5 mg/kg weekly), or NKTR-255 (0.3 mg/kg weekly) + daratumumab (5 mg/kg weekly). Tumor volume (B) was monitored 3 times a week using a caliper. Blood samples were collected at the end of the experiment for immunophenotyping, and the absolute count of NK cells was assessed per cohort of treatment (C). N = 5 mice/group. ∗P < .05, ∗∗P < .01, ∗∗∗P < .001, nsP ≥ .05. CNT, control; MM, multiple myeloma; ns, not significant; NK, natural killer; SD, standard deviation.
Finally, we assessed the efficacy of NKTR-255 alone and in combination with daratumumab against MM cell growth in the context of the immune milieu in a humanized immunocompetent mouse model engrafted first with human immune cells and subsequently with the H929 MM cell line. While monotherapy with NKTR-255 or daratumumab treatment delayed tumor growth (35.4% and 29.6%, respectively) as compared with the control group, the combination regimen was additive (66.4%) in inhibiting tumor growth in this model (Figure 4B), without causing significant increase in toxicity in mice (supplemental Figure 3). Importantly, NKTR-255 treatment was associated with increased absolute count of NK cells in the peripheral blood, and the combination therapy significantly prevented the NK cell depletion observed with daratumumab alone (P = .032) (Figure 4C).
Discussion
Cytokine activation of NK cells to promote antitumor responses has been explored in many clinical settings.30,53-55 In this context, rhIL-15 represents an example of a cytokine with promising antitumor activity via enhancement of both NK and memory T cell functions. However, with the issues observed with utilization of rhIL-15 in a clinical setting, including the need to modify the route and schedule of administration due to cytokine release and macrophage activation syndromes taking place within hours after an intravenous bolus administration, and potential constraints with the instability and early renal clearance of rhIL-15,34,35 there is a need to provide and develop rhIL-15 agonists with a similar biological response but improved pharmacokinetics and tolerability (among other beneficial properties).
patients with MM are characterized by an immunosuppressive microenvironment that allows immune escape and continued myeloma cell growth. In addition to humoral immunodeficiency affecting B-cell subsets and the adaptive immune system dysfunction, a significant impairment of the innate immunity impacting the functional state of NK cells has been reported. Here we investigate the potential of NKTR-255, a novel polymer-conjugated rhIL-15, to engage the IL-15 pathway and overcome the inhibitory status observed in NK cells from patients with MM.36,37 NKTR-255 displays an extended in vivo half-life with reduced renal clearance, sustained signaling, and prolonged pharmacodynamics when compared with rhIL-15, thus providing an attractive alternative for its clinical evaluation in MM.36,38,40
Similarly to induction with rhIL-15, treatment with NKTR-255 resulted in a significant increase in the expression of activating receptors with a key role in antitumor cytotoxicity (such as NKG2D or DNAM-1)8,16,56-59 on NK cells from patients with MM after ex vivo treatment with NKTR-255. Interestingly, the upregulation of NKG2D expression was more pronounced in patients with myeloma with progressive disease compared with those who had achieved disease response.13,60,61 These findings correlated with improved degranulation, effector cytokine production, and increased direct cytotoxicity in purified NK cells in response to primary MM cells or MM cell lines in a dose-dependent manner after incubation with NKTR-255.
The experiments employing whole BM samples are of particular interest since they were collected from patients newly diagnosed with MM who had not received any prior anti-MM therapy. Thus, the effect of NKTR-255 against survival of MM cells was assessed in a microenvironment favorable to MM cell growth where the activity of NK cells should be impaired. Even though it is not possible to ascertain the precise mechanism leading to the inhibition of MM cell viability and the role played by other immune cell populations, we can hypothesize that the dose-dependent expansion and activation of NK cells observed after treatment with NKTR-255 may have played a role on this outcome. Since we did not observe a direct cytotoxicity when different MM cell lines were cultured in vitro with the compound, it is feasible to postulate that among other immune cells, the NK cell expansion and improved function induced by NKTR-255 is contributing to more effective control of MM tumor growth, raising a potential scope for synergism with other anti-MM therapies such as monoclonal antibodies.
ADCC represents one of the major mechanisms underlying the clinical efficacy of therapeutic monoclonal antibodies. Enhancement of the activation status in the immune cell subsets involved as effectors in the ADCC process may improve the anti-tumor efficacy of antibody-based therapies, as observed with rhIL-15 therapy.44,62,63 Here we show that NKTR-255 enhances ADCC, increasing MM cell killing by NK cells when target cells are pre-treated with daratumumab.
Importantly, in vivo studies using a fully humanized MM mouse model suggested superiority of the combination regimen compared with single agents in controlling MM growth. NK cells express CD38 at the highest level among other normal hematological lineage cells. In fact, daratumumab depletes patient NK cells within the first week after treatment,64,65 potentially affecting its efficacy to target MM cells66 and increasing the risk of infections.67,68 Nevertheless, in our in vivo model, NKTR-255 partially rescued the NK cell depletion observed after treatment with daratumumab, providing a potential rationale for combination. Of note, the majority of surviving cells in the combination group were CD38−/low NK cells (data not shown).
In conclusion, this study supports NKTR-255 as a potent inducer of expansion and anti-tumor functions of NK cells, providing rationale for its further evaluation as a novel immunotherapeutic approach in MM, alone or in combination with monoclonal antibodies or potentially with other immunomodulatory drugs.
Acknowledgments
The authors thank Sanika Derebail for the technical support; John Daley and Suzan Lazo (Flow Cytometry Core Facility, Dana-Farber Cancer Institute) for their help and support with the flow cytometry analysis.
Funding support for this article was provided by the NIH grant P01-155258-07 (K.C.A., N.C.M., and M.F.); the Department of Veterans Affairs Merit Review Award 1 I01BX001584 (N.C.M.); Fundación Española de Hematología y Hemoterapia (R.A.F.); a NIH/NCI R01 (CA207237-05) and a Paula and Rodger Riney Foundation Grant (K.C.A.); an NIH/NCI SPORE grant (P50-CA100707-18) (N.C.M. and K.C.A.); and research support from Nektar Therapeutics.
Authorship
Contribution: R.A.F. and M.F. were responsible for the conception, design and execution of the study; J.M.-L., D.P., T.M., K.C.A., and N.C.M. provided study materials, technical resources and/or patient samples; R.A.F., J.E.M., L.P.-L., Y.X., Y.Y., S.M., D.P., and T.M. performed the experiments; R.A.F., T.M., R.P., W.W.O., N.C.M., and M.F. were involved with data analysis and interpretation; R.A.F., T.M., K.C.A., W.W.O., N.C.M., and M.F. wrote, reviewed, and edited the manuscript; and all authors approved the final manuscript.
Conflict-of-interest disclosure: T.M. and W.W.O. are employees of Nektar Therapeutics. K.C.A. serves on advisory boards to Celgene, Takeda, BMS, and Gilead and holds equity ownership in Oncopep and Acetylon. N.C.M. serves on advisory boards/consultant to Takeda, BMS, Janssen, Amgen, AbbVie, Oncopep, Karyopharm, Adaptive Biotechnology, and Novartis, and holds equity ownership in Oncopep. The remaining authors declare no competing financial interests.
Correspondence: Mariateresa Fulciniti, 440 Brookline Ave, Mayer Building, 5th floor, room M-551, Boston, MA 02215; e-mail: nikhil_munshi@dfci.harvard.edu; and Nikhil Munshi, 440 Brookline Ave, Mayer Building, 2nd floor, Boston, MA 02215; e-mail: mariateresa_fulciniti@dfci.harvard.edu.
References
Author notes
Data sharing requests should be sent to: Mariateresa Fulciniti (mariateresa_fulciniti@dfci.harvard.edu).
The full-text version of this article contains a data supplement.