Key Points
Excess IL-18 and complete or partial perforin deficiency cooperate to drive clonal CD8 T-cell expansion.
Concurrent terminal exhaustion and effector programs define CD8 T-cell hyperactivation.
Abstract
Hemophagocytic lymphohistiocytosis (HLH) and macrophage activation syndrome (MAS) are life-threatening hyperinflammatory syndromes. Familial HLH is caused by genetic impairment of granule-mediated cytotoxicity (eg, perforin deficiency). MAS is linked to excess activity of the inflammasome-activated cytokine interleukin-18 (IL-18). Though individually tolerated, mice with dual susceptibility (Prf1⁻/⁻Il18tg; DS) succumb to spontaneous, lethal hyperinflammation. We hypothesized that understanding how these susceptibility factors synergize would uncover key pathomechanisms in the activation, function, and persistence of hyperactivated CD8 T cells. In IL-18 transgenic (Il18tg) mice, IL-18 effects on CD8 T cells drove MAS after a viral (lymphocytic choriomeningitis virus), but not innate (toll like receptor 9), trigger. In vitro, CD8 T cells also required T-cell receptor (TCR) stimulation to fully respond to IL-18. IL-18 induced but perforin deficiency impaired immunoregulatory restimulation-induced cell death (RICD). Paralleling hyperinflammation, DS mice displayed massive postthymic oligoclonal CD8 T-cell hyperactivation in their spleens, livers, and bone marrow as early as 3 weeks. These cells increased proliferation and interferon gamma production, which contrasted with increased expression of receptors and transcription factors associated with exhaustion. Broad-spectrum antibiotics and antiretrovirals failed to ameliorate the disease. Attempting to genetically “fix” TCR antigen-specificity instead demonstrated the persistence of spontaneous HLH and hyperactivation, chiefly on T cells that had evaded TCR fixation. Thus, drivers of HLH may preferentially act on CD8 T cells: IL-18 amplifies activation and demand for RICD, whereas perforin supplies critical immunoregulation. Together, these factors promote a terminal CD8 T-cell activation state, combining features of exhaustion and effector function. Therefore, susceptibility to hyperinflammation may converge on a unique, unrelenting, and antigen-dependent state of CD8 T-cell hyperactivation.
Introduction
The description of a fulminant syndrome of fever, pancytopenia, coagulopathy, central nervous system inflammation, hepatitis, and hepatosplenomegaly associated with reticuloendothelial histiocytosis and prominent hemophagocytosis dates back at least 70 years.2,3 With intensive study, the current classification of this diverse syndrome, dubbed hemophagocytic lymphohistiocytosis (HLH), relies primarily on the context in which it occurs (familial, primary, or infection-, malignancy-, rheumatic-, or immunotherapy-associated).4 However, accumulating evidence suggests multiple mechanisms drive susceptibility and disease activity between contexts and even individual patients. Thus, a more mechanistic framework may improve the categorization and management of patients with HLH.5
Familial HLH is a mechanistic category of HLH, wherein biallelic genetic defects in granule-mediated cytotoxicity cause clinical hyperinflammation.6-9 HLH in the context of rheumatic diseases is usually called macrophage activation syndrome (MAS). MAS is best studied in systemic juvenile idiopathic arthritis (SJIA), in which the 2 exist on a spectrum of severity.10 Interleukin-18 (IL-18) is an inflammasome-activated cytokine and a valuable biomarker of MAS susceptibility. IL-18 that is unbound by its abundant soluble inhibitor, IL-18BP, is called free IL-18.11,12 SJIA-related and monogenic forms of MAS11,13-19 are tightly associated with very high circulating levels of total IL-18 and detectable free IL-18. Neither genetic familial HLH lesions nor free IL-18 are sufficient to cause HLH in mice or humans; both require triggers such as infection8,20-24 to convert susceptibility into immunopathology.9,25,26 Monoallelic defects in cytotoxicity genes are common, but they are enriched up to fourfold in patients with SJIA-MAS.27,28
Recent experimental and clinical observations suggest a convergence on T-cell hyperactivation and pathogenic interferon gamma (IFN-γ). Patients with HLH showed a preponderance of activated CD8 T cells in the peripheral blood29,30 and affected tissues.31-33 In mice infected with typically benign lymphocytic choriomeningitis virus (LCMV), both excess IL-18 and perforin deficiency led to an HLH-like phenotype,25,34 and CD8 T cells are essential for disease in Prf1⁻/⁻ mice.35 We recently showed that mice with perforin deficiency and transgenic expression of IL-18, so-called dual susceptibility (DS) mice, develop spontaneous, lethal, IFN-γ–dependent hyperinflammation.25 This strongly supports the hypothesis that cytotoxic defects and excess IL-18 contribute to, but are not individually sufficient for, hyperinflammation. Herein, we explore this hypothesis and identify specific and nonredundant effects of these HLH susceptibility factors on (possibly self-) antigen-specific CD8 T-cell activation.
Methods
Mouse lines
Mice were housed in specific pathogen-free conditions under a protocol approved by the University of Pittsburgh Institutional Animal Care and Use Committee. All animals originated from Jackson Laboratories except: Il18tg mice were a gift from Tomoaki Hoshino (Kurume University),36 mice bearing the Il18r1f/f allele were a gift from G. Trinchieri (National Cancer Institute),25 and mice bearing the E8iCreERT2/GFP allele were a gift from D. Vignali (University of Pittsburgh).37
Tissue harvesting
Spleens, lymph nodes, bone marrows, and thymuses were processed in RPMI supplemented with 10% fetal bovine serum (FBS) (R10) and filtered through 100uM strainers. Livers were perfused with 10mL of cold phosphate-buffered saline, then processed in R10, supplemented with 0.5 units per mL Collagenase D and 20 μg/mL DNase, processed using GentleMACS C Tubes, and purified using a 40% Percoll gradient. Tissues with visible red blood cell contamination underwent ammonium–chloride–potassium lysis. Single-cell suspensions were prepared in phosphate-buffered saline + 2% FBS for flow cytometry or flow sorting.
Flow cytometry and analysis
Peptide and phorbol myristate acetate/ionomycin stimulations were performed as per the description by Burn et al.38 Flow cytometric data were collected on a 5 laser Cytek Aurora Spectral Cytometer and analyzed using FlowJo version 10.9.0 (Treestar). T-distributed stochastic neighbor embedding analysis was carried out using the EXCYT2 program39 on Matlab R2022a. Glycoprotein (GP) tetramer25,34-41 (GP33) was obtained from the National Institutes of Health tetramer core facility.
In vivo neutralization/depletion
Adult mice were administered with 500 μg per mouse of either anti-CD4 (GK1.5), anti-CD8 (YTS169.4), or control (LTF-2) antibody intraperitoneally on day 1, and 250 μg per mouse every third day, for 4 consecutive treatments.
In vivo tamoxifen and LCMV Armstrong treatment
Mice were injected intraperitoneally with 1 mg tamoxifen for 5 consecutive days and rested for 2 days before infection intraperitoneally with 2 × 105 plaque-forming units of LCMV Armstrong. Mice were administered 1 mg of tamoxifen 4 and 7 days after infection and assessed on day 9.
Antibiotic treatment
Adult mice were administered with broad-spectrum antibiotics ad libitum in drinking water for 5 weeks. Antibiotic water supplemented with saccharin contained metronidazole (0.5 mg/mL), vancomycin (0.5 mg/mL), neomycin (1 mg/mL), and ampicillin (1 mg/mL). Control mice received only saccharin-supplemented water.
Antiretroviral and etoposide treatment
For antiretroviral treatment (ART), adult mice were administered 200 μL of combined tenofovir (12.5 mg/mL) and emtricitabine (7.5 mg/mL) daily for 3 weeks via oral gavage, as per that described by Lima-Junior et al.40 Mice were collected 1 day after the last ART treatment. Etoposide was administered to adult mice at 30 mg/kgintraperitoneally twice weekly for 4 doses, and mice were evaluated 1 week after the last treatment, as per that described by Koh et al.41
In vitro assays
Whole splenocytes from P14 or Prf1⁻/⁻P14 mice (bearing a transgenic TCR recognizing the LCMV GP25,34-42 peptide) were processed as described and then cultured for 24 hours with cognate GP25,34-42 peptide (1 μg/mL) in complete R10 media (RPMI supplemented with 10% FBS, 1 × penicillin-streptomycin, 2 mM L-glutamine, 1 mM sodium pyruvate, 10 mM nonessential amino acids, 50 μM 2-mercaptoethanol, and 12 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid), supplemented with 50 U/mL IL-2. Peptide was removed, and cells were resuspended in complete media with 50 U/mL IL-2 (± 50 ng/mL IL-18) and allowed to rest for 3 days. Every other day thereafter, live cells were cultured in fresh, complete media with 25 U/mL IL-2 (± 50 ng/mL IL-18). At 9-11 days after activation, cells were reactivated via anti-CD3 (3 μg/mL coated plate) and anti-CD28 (5 μg/mL) in complete media (25 U/mL IL-2 ±50 ng/mL IL-18) and assessed for activation markers and cytokine production via flow cytometry.
RNA sequencing and TCR-seq
Messenger RNA purification and fragmentation, complementary DNA synthesis, and target amplification were performed with Smart-Seq v4 ultralow input RNA kit with Nextera XT. Pooled complementary DNA libraries were sequenced on the Illumina NextSeq500, mapped to Mus_musculus_ensembl_v80 reference sequence, gene track Mus_musculus_ensembl_v86, and quantified using CLC Main Workbench software (V22, Qiagen). Reads were converted to transcripts per million (TPM), and a 0.5 TPM offset was added to all transcripts. Batch correction between in-house data and publicly available data was completed using ComBat-seq,42 which uses a negative binomial regression to model and adjust for batch effects. Heat maps were generated using Morpheus analysis software.43 Principal component analysis plots were generated from ComBat-adjusted TPM tables using the R package pcaExplorer.44 Differential gene expression between ComBat-adjusted data sets was determined using the BioJupies online analysis suite.45
T-cell receptor (TCR) sequencing (TCR-seq) was performed on bulk splenic CD8 T cells using the TakaraBio SMARTer Mouse TCR a/b Profiling Kit. Libraries were sequenced, mapped, and quantified as decribed earlier. TCR-seq analysis and visualizations were performed using the R packages Immunarch (V0.6.8),46 Mixcr,47 VDJtools,48 and packcircles (Bubble Plots), using the University of Pittsburgh’s Center for Research Computing.
Results
IL-18 preferentially drives CD8 T-cell hyperactivation and immunopathology in LCMV-triggered hyperinflammation
Although both perforin deficiency and excess IL-18 promote hyperinflammation during acute LCMV (Armstrong) infection, viral clearance is normal in mice with excess IL-18 (Il18tg).25,34 Prior work demonstrated that the development of LCMV-induced MAS in Il18tg mice required Il18r1 expression by T cells but suggested a specific effect on CD8 T cells.25,34 To test this, we temporally and selectively deleted Il18r1 in CD8 T cells using E8iERT2-Cre-GFPIl18r1flox/flox mice (Il18r1Δ8).37 By itself, tamoxifen treatment substantially (but incompletely) reduced the expression of IL-18R1 on blood CD8 T cells but not CD4 T cells (∼70% in control mice to ∼20% in Cre+ mice; supplemental Figure 1A,B). Upon LCMVa infection, Cre+ mice were protected from mortality, severe weight loss, and anemia (Figure 1A,B; supplemental Figure 1C).
Loss of Il18r1 on CD8 T cells improves LCMV-induced hyperinflammation. (A) Weight loss throughout tamoxifen treatment and LCMV Armstrong infection (2e5 plaque-forming units per mouse). Il18tg and Il18tg;Il18r1Δ8 mice were compared by mixed effects modeling because only 63% of Il18tg mice survived until experimental end point. (# indicates that mice succumbed 7-8 days after infection). No other mice succumbed to infection. (B) Anemia in surviving mice 10 days after infection. (C) Percentage of splenic CD8 T cells producing IFN-γ by intracellular flow cytometry after a 6 hour GP33 stimulation 10 days after infection. (D) Percentage of splenic CD8 and CD4 T cells expressing IL-18R1. (E) Percentage of splenic CD8 and CD4 T cells expressing PD-1 in mice directly ex vivo 10 days after infection. (A-E) Combined data from >3 experiments. P values in panels B and E are calculated using one-way analysis of variance (ANOVA) with Tukey post hoc test of comparisons between Il18tg, Il18r1Δ8 and all other groups. Significance for comparisons between Il18tg and Il18tg;Il18r1Δ8 groups are shown for adjusted P value < .05. PD-1, programmed cell death protein-1.
Loss of Il18r1 on CD8 T cells improves LCMV-induced hyperinflammation. (A) Weight loss throughout tamoxifen treatment and LCMV Armstrong infection (2e5 plaque-forming units per mouse). Il18tg and Il18tg;Il18r1Δ8 mice were compared by mixed effects modeling because only 63% of Il18tg mice survived until experimental end point. (# indicates that mice succumbed 7-8 days after infection). No other mice succumbed to infection. (B) Anemia in surviving mice 10 days after infection. (C) Percentage of splenic CD8 T cells producing IFN-γ by intracellular flow cytometry after a 6 hour GP33 stimulation 10 days after infection. (D) Percentage of splenic CD8 and CD4 T cells expressing IL-18R1. (E) Percentage of splenic CD8 and CD4 T cells expressing PD-1 in mice directly ex vivo 10 days after infection. (A-E) Combined data from >3 experiments. P values in panels B and E are calculated using one-way analysis of variance (ANOVA) with Tukey post hoc test of comparisons between Il18tg, Il18r1Δ8 and all other groups. Significance for comparisons between Il18tg and Il18tg;Il18r1Δ8 groups are shown for adjusted P value < .05. PD-1, programmed cell death protein-1.
Nearly all CD8 T cells expressed IL-18r1 in infected control (Cre⁻) mice, whereas there was more “leak-through” IL-18R1 expression on CD8 T cells from Il18tg;Il18r1Δ8 mice than those without excess IL-18. Likewise, CD8 T cells from Il18tg;Il18r1Δ8 mice produced IFN-γ comparably with those from Il18r1-sufficient Il18tg controls but showed less activation as measured by programmed cell death protein-1 (PD-1) expression (Figure 1C-E). These findings suggest cell-intrinsic activation, survival, and/or proliferative advantages conferred by IL-18 on CD8 T cells during infection. In addition to more leak-through IL-18R1 on CD8 T cells, Il18tg;Il18r1Δ8 mice also showed more CD4 T-cell activation (Figure 1C,E), suggesting that IL-18 responsive CD8 T cells compete for cytokines/growth factors and/or suppress CD4 T-cell responses. This apparent compensation by CD4 T cells may help explain the persistence of serum IFN-γ elevation in Il18tg;Il18r1Δ8 mice (supplemental Figure 1D).
Minimal effects of IL-18 on CD8 T cells in the absence of TCR stimulation
IL-18 is known to amplify disease in the toll like receptor 9 (TLR9)-driven model of MAS, which typically does not require T cells.11,49 Nevertheless, cytokines such as IL-12 can provoke effector responses from activated and memory T cells in the absence of TCR stimulation.50,51 Thus, we predicted that mice specifically lacking Il18r1 in all T cells might be protected from increased inflammation in the setting of systemic TLR9 agonism, which profoundly induces systemic IL-12 production.52 However, Il18tg mice lacking Il18r1 on all T cells (Il18tg;Il18r1ΔT) were not protected from increased weight loss, splenomegaly, or thrombocytopenia (supplemental Figure 2A-C). Interestingly, T-cell responses to IL-18 made a significant contribution to increased systemic IFN-γ levels, suggesting IL-18 mediated more severe MAS in this model via early effects of IFN-γ and/or factors other than IFN-γ.
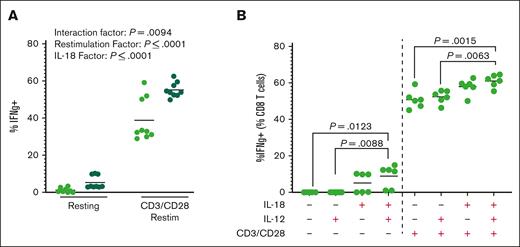
To further assess the effects of IL-18 with and without TCR stimulation, we cultured CD8 T cells with IL-18 during in vitro TCR restimulation. Consistent with the role of IL-18 as an amplifier of IFN-γ production, we found that maximal IFN-γ production required the interaction of CD3/CD28 and IL-18 (Figure 2A). In CD8 T cells restimulated in vitro (up to 2 weeks after initial TCR stimulation), we found that neither IL-12 alone nor IL-18 plus IL-12 substantially affected IFN-γ production by CD8 T cells in the absence of TCR restimulation (Figure 2B). Together, these data suggest that the effects of long-term IL-18 on T cells may require active or recent TCR signaling.
CD8 T cells require TCR stimulus in the presence of IL-18 for hyperactivation. (A) Percentage of CD8 T cells producing IFN-γ after reactivation with or without TCR restimulation for 24 hours in the presence (dark green) or absence of 50 ng/mL recombinant murine IL-18. Each point represents an average of 3 wells in an experiment, 9 experiments shown. Analysis by two-way ANOVA. (B) Percentage of CD8 T cells producing IFN-γ after reactivation with or without TCR restimulation for 24 hours in the presence or absence of 50 ng/mL recombinant murine IL-18 and/or 10 ng/mL recombinant murine IL-12. Graph shows representative data from 2 independent experiments; each dot is a technical replicate. P value is based on one-way ANOVA with Tukey post hoc test, comparing cytokine addition within resting and CD3/CD28 groups separately.
CD8 T cells require TCR stimulus in the presence of IL-18 for hyperactivation. (A) Percentage of CD8 T cells producing IFN-γ after reactivation with or without TCR restimulation for 24 hours in the presence (dark green) or absence of 50 ng/mL recombinant murine IL-18. Each point represents an average of 3 wells in an experiment, 9 experiments shown. Analysis by two-way ANOVA. (B) Percentage of CD8 T cells producing IFN-γ after reactivation with or without TCR restimulation for 24 hours in the presence or absence of 50 ng/mL recombinant murine IL-18 and/or 10 ng/mL recombinant murine IL-12. Graph shows representative data from 2 independent experiments; each dot is a technical replicate. P value is based on one-way ANOVA with Tukey post hoc test, comparing cytokine addition within resting and CD3/CD28 groups separately.
CD8 T-cell hyperactivation in DS mice
Despite the apparent need in CD8 T cells for recent TCR stimulation to respond fully to IL-18, most patients with MAS lack a known antigenic MAS trigger. We previously showed that mice with DS, that is both excess IL-18 and perforin deficiency (Prf1+/−;Il18tg or Prf1⁻/⁻;Il18tg, hereafter referred to as DSHET/DSKO or collectively DS), developed spontaneous hyperinflammation that was lethal in DSKO mice.25 Lymphohistiocytosis in human HLH/MAS consists primarily of histiocytes and activated CD8 T cells and can occur in the peripheral blood,29,53 liver,32 and other organs.33 We performed a flow cytometric survey of multiple organs in DS mice, focusing on lymphocytes as potential sources of pathogenic cytokines such as IFN-γ.25 We found that DSKO mice have more CD8 T cells in reticuloendothelial organs (bone marrow, liver, and spleen; Figure 3A; supplemental Figure 3A). Those CD8 T cells showed dramatically increased PD-1 and IL-18R1 expression, indicative of activation, and were observed as early as 3 weeks of age in DS mice (supplemental Figure 3D). CD8 T-cell numbers and activation states were comparable between DS and control (particularly Il18tg control) mice in the mesenteric and inguinal lymph nodes and thymus (Figure 3B).
DuDS mice have more activated CD8 T cells with a distinct immunological profile. (A) Total number of splenic CD8 and CD4 T cells. (B) Percentage of CD8 T cells homeostatically expressing activation markers IL-18r1 and PD-1 in various organs. Graph shows combined data from >3 experiments; each data point represents a mouse. P value based on one-way ANOVA with Tukey post hoc test of comparisons between Il18tg to DSKO. ∗∗∗∗P < .0001; ∗∗P < .001; ∗P < .05. (C) Unbiased K means clustering t-distributed stochastic neighbor embedding projection of splenic T cells (7649 cells per mouse) from 3 mice of each genotype with representation of each cluster within each genotype. (D) Heat map of relative geometric mean fluorescent intensity (MFI) of selected markers. ILN, inguinal lmph node; MLN, mesenteric lymph node; WT, wild-type.
DuDS mice have more activated CD8 T cells with a distinct immunological profile. (A) Total number of splenic CD8 and CD4 T cells. (B) Percentage of CD8 T cells homeostatically expressing activation markers IL-18r1 and PD-1 in various organs. Graph shows combined data from >3 experiments; each data point represents a mouse. P value based on one-way ANOVA with Tukey post hoc test of comparisons between Il18tg to DSKO. ∗∗∗∗P < .0001; ∗∗P < .001; ∗P < .05. (C) Unbiased K means clustering t-distributed stochastic neighbor embedding projection of splenic T cells (7649 cells per mouse) from 3 mice of each genotype with representation of each cluster within each genotype. (D) Heat map of relative geometric mean fluorescent intensity (MFI) of selected markers. ILN, inguinal lmph node; MLN, mesenteric lymph node; WT, wild-type.
Though far less numerous, CD4 T cells in DS mice showed slightly greater PD-1 and IL-18R1 expression (supplemental Figure 4A). As previously demonstrated,25 CD8 (but not CD4) T cells from DS mice produced more IFN-γ after ex vivo phorbol myristate acetate/ionomycin stimulation (supplemental Figure 4B,C). Because Foxp3+ regulatory CD4 T cells are reduced in the Prf1⁻/⁻-LCMV model,54 we hypothesized that regulatory CD4 T-cell deficiency might contribute to immunopathology in DS mice but observed only a small decrease largely restricted to the bone marrow (supplemental Figure 4D). Like in patients with SJIA,55 natural killer (NK) cells were less abundant in DS mice than in controls (supplemental Figure 4E). Flow cytometry analysis using unsupervised T-stochastic neighbor embedding clustering of splenic T cells identified 2 clusters of CD8 T cells specific to DS mice. Both clusters showed high expression of IL-18R1 and PD-1 and low expression of CD62L. Combined with coexpression of Tim-3, Lag-3, and PD-1 in the CD8 T cells in these clusters (Figure 3C-D; supplemental Figure 3B), these data suggest a CD8 T-cell effector/exhaustion state56-60 most prominent in DSKO mice. Of these, 1 cluster was distinguished by low CD44 expression and was more prominent in DSKO than in DSHET mice, making up nearly 65% of DSKO splenic CD8 T cells (supplemental Figure 3C). This CD44/CD62L double-negative CD8 T-cell population may be comparable with terminally differentiated CD8 T cells induced in vitro with repeated TCR stimulation and intact phosphoinositide 3-kinase.61 The combination of expression of multiple inhibitory receptors and retained effector function (possibly including CD44 downregulation) suggests a state of CD8 T-cell hyperactivation.
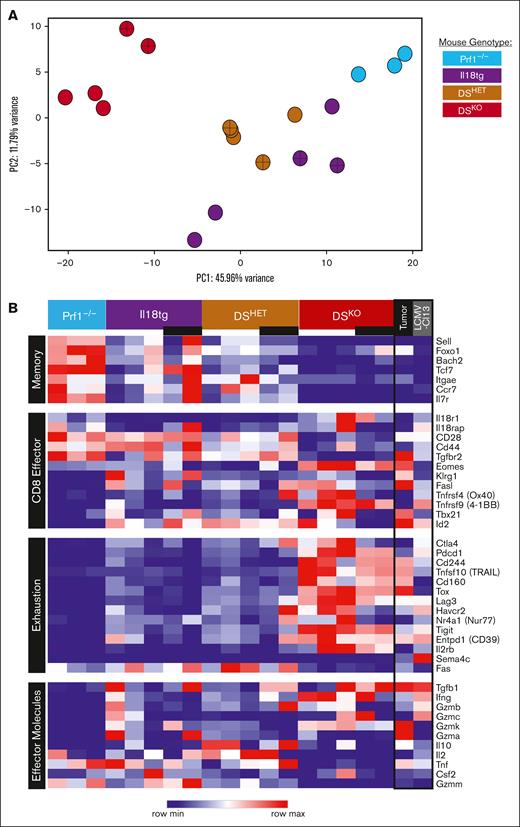
Next, we performed bulk RNA sequencing of both CD44+ and CD44lo/⁻/CD62L⁻ CD8 T-cell populations and found that these populations were transcriptionally similar within genotypes (although CD44 was downregulated) but that DSKO CD8 T cells were transcriptionally distinct from control genotypes (Figure 4A). CD44+ CD8 T cells from Prf1⁻/⁻ mice had a central-memory phenotype typical of an unstimulated mouse, with high expression of progenitor/memory markers t-cell factor (Tcf) 7 and Il7r and high expression of Sell (encoding CD62L; Figure 4B). Cytokine receptors Il12rb1, Il15r, Il2rb, and Il2rg were increased in DS CD8 T cells, consistent with an effector phenotype (supplemental Table 1). CD8 T cells from DS mice showed evidence of terminal differentiation, with high expression of effector transcripts, such as Eomes, Tgfb1, IFN-γ, Gzmk, and Il10 and low expression of Il7r and Tcf7 (TCF-1).63-65 Despite the absence of experimental antigen, the most differentially upregulated transcripts in DS CD8 T cells were exhaustion-associated inhibitory receptors such as Pdcd1 (PD-1), Havcr2 (Tim-3), Ctla4, Entpd1 (CD39), and Tigit (Figure 4B). These cells also expressed Tox, a key transcription factor driving the exhaustion program.66-68
DSKO CD8 T cells have a unique, terminally differentiated transcriptional profile with effector and exhaustion features. (A) Principal component analysis (PCA) from bulk CD8 T-cell RNA sequencing of >3 adult mice from each genotype under homeostatic conditions. (B) Heat map comparison of selected genes of homeostatic CD8 T cells and terminal TEXs from tumor and chronic infection (LCMV-Cl13).62 Crosses within data point (A) and black bars under genotype (B) indicate CD44LoCD62L⁻ CD8 T cells, otherwise CD44+ CD8 T cells. Rows are z-score normalized.
DSKO CD8 T cells have a unique, terminally differentiated transcriptional profile with effector and exhaustion features. (A) Principal component analysis (PCA) from bulk CD8 T-cell RNA sequencing of >3 adult mice from each genotype under homeostatic conditions. (B) Heat map comparison of selected genes of homeostatic CD8 T cells and terminal TEXs from tumor and chronic infection (LCMV-Cl13).62 Crosses within data point (A) and black bars under genotype (B) indicate CD44LoCD62L⁻ CD8 T cells, otherwise CD44+ CD8 T cells. Rows are z-score normalized.
Given the transcriptional overlap with CD8 T-cell exhaustion, we analyzed these data sets alongside published transcriptomes of terminally exhausted CD8 T cells (TEXs; PD-1+Tim-3+) and CD8 T-cell exhaustion precursors (PD-1IntTim-3-).62 CD8 T cells from DS mice clustered separately from both populations, but the trajectory of change from CD8 T-cell exhaustion precursor and TEX paralleled that of CD8 T cells from Prf1⁻/⁻to DSKO (supplemental Figure 5A-C). Differentially expressed genes between DSKO and published TEX included adhesion/chemotaxis (eg, Nek7, Tgfbr2, Ccr5, and PECAM1) and T-cell hyperactivation (eg, Il18r1, Sh2d1a, and Ikzf2) genes (Supplemental Figure 5D; supplemental Table 2). Thus, the transcriptomes of CD8 T cells from DS mice (both CD44+ and CD44lo/⁻/CD62L⁻ populations) include features of both terminal effector differentiation and exhaustion.
Effects of HLH susceptibility factors on CD8 T-cell RICD and proliferation promote hyperinflammation
We previously reported that IFN-γ neutralization led to an amelioration of hyperinflammation in DSKO mice.25 Despite the profound CD8 T-cell expansion/activation, less impressive CD4 T-cell activation, and near absence of NK1.1+ innate lymphoid cells in DSKO mice (supplemental Figure 4E), antibody-mediated depletion of either T-cell subset was not sufficient to rescue hyperinflammation (supplemental Figure 6A-B). CD8 depletion had no effect on the hyperinflammatory phenotype, yet it substantially increased the activation state and IFN-γ production of CD4 T cells. Depletion of CD4 T cells affected neither hyperinflammatory features nor CD8 T-cell activation (Supplemental Figure 6C). These data together suggest temporal depletion of CD8 T cells is insufficient to blunt the long-term effects of IFN-γ overproduction by lymphocytes while also highlighting the compensatory capacity of CD4 T-cell activation in DS mice.
Etoposide is a cornerstone of the regimen used to dampen hyperinflammation and prepare patients with familial HLH for hematopoietic stem cell transplant.69 It also selectively ablates dividing, pathogenic CD8 T cells and ameliorates hyperinflammation in Prf1⁻/⁻ mice infected with LCMV.70 We hypothesized that a similar etoposide treatment regimen might ameliorate HLH in DS mice, but we observed no appreciable improvement in splenomegaly, anemia, or hyperactivated CD8 T cells in DS mice after 2 weeks’ treatment (supplemental Figure 7A-C).
Impaired restimulation-induced cell death (RICD) has been implicated in the pathogenesis of X-linked lymphoproliferative syndrome type 1, a type of primary HLH due to SH2D1A deficiency and usually triggered by Epstein-Barr virus infection.71 Perforin may also help mediate activation-induced apoptotic cell death, specifically at early time points before Fas-mediated killing.72,73 To determine whether perforin deficiency and IL-18 conferred CD8 T-cell resistance to cell death, we tested the effects of these 2 factors (individually and in concert) in an in vitro RICD system. We found that the addition of IL-18 significantly increased RICD (Figure 5A). Consistent with prior results,72,74 perforin deficiency caused a slight decrease in RICD that persisted even in the presence of IL-18. This reinforces the notion that, when present, perforin prevents hyperinflammation by killing both target (eg, dendritic) cells35 and other cytotoxic cells (fratricide).75 IL-18 has also been linked to increased CD8 T-cell proliferation,76,77 and we observed greater 5-ethynyl-2’-deoxyuridine (EdU) incorporation in DS than in wild-type or Prf1⁻/⁻ CD8 T cells (Figure 5B). Il18tg CD8 T-cell EdU incorporation was comparable with that in DS mice, but such mice had far less profound CD8 T-cell activation (Figure 3). Thus, inflammatory/proliferative stimuli such as excess IL-18 may increase demand for immunoregulatory RICD, a demand that goes unmet when cytotoxicity is (even partially) impaired.
IL-18 and perforin deficiency alter CD8 T-cell RICD and proliferation. (A) RICD in CD8 T cells in the presence (purple) or absence (blue) of 50 ng/uL IL-18 after a 24-hour restimulation with CD3/CD28. Each data point represents the average of technical triplicates within 1 experiment. P values are determined using two-way ANOVA. (B) Percent and representative graphs of in vivo EdU incorporation in CD8 T cells 19h after systemic administration. Data from >3 separate experiments; each data point represents a single mouse. P values calculated by one-way ANOVA with Tukey multiple comparisons test, evaluating all comparisons with DSKO group. Only adjusted P < .05 is shown.
IL-18 and perforin deficiency alter CD8 T-cell RICD and proliferation. (A) RICD in CD8 T cells in the presence (purple) or absence (blue) of 50 ng/uL IL-18 after a 24-hour restimulation with CD3/CD28. Each data point represents the average of technical triplicates within 1 experiment. P values are determined using two-way ANOVA. (B) Percent and representative graphs of in vivo EdU incorporation in CD8 T cells 19h after systemic administration. Data from >3 separate experiments; each data point represents a single mouse. P values calculated by one-way ANOVA with Tukey multiple comparisons test, evaluating all comparisons with DSKO group. Only adjusted P < .05 is shown.
Noncommensal antigens may drive CD8 T-cell hyperactivation in DS mice
Given that CD8 T cells in DS mice showed increased proliferation, expression of exhaustion markers, and TCR-dependent IL-18 responses (in vitro), we sought to understand the nature of the TCR stimuli driving CD8 T-cell activation. We found a very similar proportion of CD8 T cells were hyperactivated in 2-3–week–old DS pups as in adult DS mice (supplemental Figure 3D), suggesting that the antigen(s) and immunologic circumstances needed to promote hyperactivation were present continuously.
To determine whether the CD8 T cells were responding to a common antigen, we performed TCR sequence analysis of all splenic CD8 T cells from DS and control mice. We found the expected broad distribution of clonotypes in control mice, but a very high proportion (up to half) of the sequenced clonotypes belonged to the most prominent clones in DSKO CD8 T cells. This oligoclonality correlated with the degree of genetic susceptibility to hyperinflammation, as it was present to a lesser extent in DSHET and Il18tg mice (Figure 6A,B). TCR clones of public antigen-specific TCRs present in available databases were equally rare in DS and control mice (data not shown). Likewise, there were no shared clonotypes between the hyperexpanded clones of individual DS mice (Figure 6C). No substantive differences in variable region gene usage were found between DS mice and controls (supplemental Figure 8A,B). Together, these analyses suggest that CD8 T-cell expansion in DS mice is oligoclonal but not TCR-restricted.
IL-18 and perforin deficiency drive oligoclonal CD8 T-cell hyperexpansion unaffected by broad-spectrum antibiotic treatment. (A) A bubble plot represents the proportion of mapped TCRs attributable to individual clonotypes within Prf1⁻/⁻, Il18tg, DSHET, and DSKO mice. (B) Sums of the proportion of mapped TCRs attributable to clonotypes that individually occupy a large (0.1% to 1%) or hyperexpanded (1% to 100%) proportion of all mapped TCRs. (C) Representative plot showing that the top 50 hyperexpanded clonotypes in a single DSKO mouse account for ∼50% of all mapped TCRs in that mouse, but the same clonotypes account for a negligible proportion of all mapped TCRs in all other sequenced mice. (D) Splenomegaly, anemia, and thrombocytopenia in DSKO mice after >30 days of antibiotic treatment with metronidazole, neomycin, ampicillin, and vancomycin. The dotted line represents the average platelet count for WT mice. (E) CD8 T-cell activation and ex vivo stimulated IFN-γ production in DSKO mice after treatment. All graphs are combined from 2 experiments; each point represents an individual mouse.
IL-18 and perforin deficiency drive oligoclonal CD8 T-cell hyperexpansion unaffected by broad-spectrum antibiotic treatment. (A) A bubble plot represents the proportion of mapped TCRs attributable to individual clonotypes within Prf1⁻/⁻, Il18tg, DSHET, and DSKO mice. (B) Sums of the proportion of mapped TCRs attributable to clonotypes that individually occupy a large (0.1% to 1%) or hyperexpanded (1% to 100%) proportion of all mapped TCRs. (C) Representative plot showing that the top 50 hyperexpanded clonotypes in a single DSKO mouse account for ∼50% of all mapped TCRs in that mouse, but the same clonotypes account for a negligible proportion of all mapped TCRs in all other sequenced mice. (D) Splenomegaly, anemia, and thrombocytopenia in DSKO mice after >30 days of antibiotic treatment with metronidazole, neomycin, ampicillin, and vancomycin. The dotted line represents the average platelet count for WT mice. (E) CD8 T-cell activation and ex vivo stimulated IFN-γ production in DSKO mice after treatment. All graphs are combined from 2 experiments; each point represents an individual mouse.
Though DS mice do not develop mucosal immunopathology,25 1 potential source of antigen driving T-cell activation and expansion in DS mice could be commensal microbes. We hypothesized that broad-spectrum antibiotics would decrease bacterial antigen load and diminish CD8 T-cell activation and immunopathology. Aggressive decolonization with continuous, ad libitum ingestion of ampicillin, vancomycin, metronidazole, and neomycin did not appreciably alter CD8 T-cell activation or clinical features of hyperinflammation in DS mice (Figure 6D,E). We hypothesized that endogenous retroviruses (ERVs) might be driving T-cell activation in DS mice as they do in multiple sclerosis78 and some primary HIV infections.79 Treatment of adult DS mice with the ART drugs emtricitabine and tenofovir daily for 3 weeks40 did not significantly alter CD8 T-cell hyperactivation or clinical features of hyperinflammation (supplemental Figure 7D-F). Thus, neither commensal bacteria nor (ART-susceptible) ERVs appear necessary for driving spontaneous activation of CD8 T cells in DS mice.
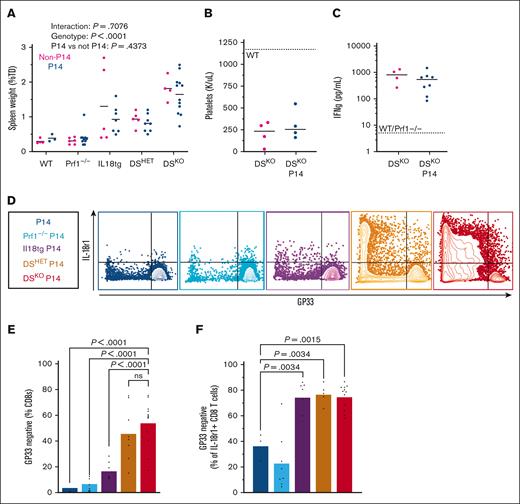
We wondered whether we could prevent hyperinflammation and CD8 T-cell hyperactivation in DS mice by genetically limiting T-cell encounters with cognate antigen. To test this, we bred DS mice to express the P14 transgene, generating DS-P14 mice. The P14 transgene encodes a dominant TCR (by allelic exclusion) that recognizes an immunodominant peptide of the LCMV glycoprotein, GP.25,34-41 Despite this attempt to fix their TCRs, DSKO-P14 mice developed spontaneous immunopathology nearly identical to control DSKO mice, including splenomegaly, thrombocytopenia, and systemic IFN-γ (Figure 7A-C). In addition, like control DSKO mice, DSKO-P14 mice developed (almost exclusively) CD8 T-cell hyperactivation. However, most CD8 T cells in DS-P14 mice were GP33 tetramer–negative (GP33TET-negative; Figure 7D-F). This enrichment for GP33TET-negative CD8 T cells was highly concentrated in the most activated (IL-18R1+) cells. These breakthrough CD8 T cells produced IFN-γ at a significantly higher rate than GP33TET-positive cells in DS-P14 mice (supplemental Figure 9A).
DS mice bearing the P14 transgene succumb to hyperinflammation but develop breakthrough GP33TET-negative CD8 T-cell activation. (A-C) Splenomegaly, thrombocytopenia, and systemic IFN-γ in DSKO, DSKO-P14, and control mice. (D) Representative flow cytometry plots of splenic CD8 T cells. (E) Proportion of splenic CD8 T cells that bind GP33TET. (F) Proportion of the activated (IL-18R1+) CD8 T-cell pool occupied by GP33TET-negative cells. Graphs show combined data from 3+ experiments, each point represents an individual mouse. P values determined by two-way ANOVA (A) or one-way ANOVA with Tukey multiple comparisons test of pairwise comparisons against WT-P14 group.62
DS mice bearing the P14 transgene succumb to hyperinflammation but develop breakthrough GP33TET-negative CD8 T-cell activation. (A-C) Splenomegaly, thrombocytopenia, and systemic IFN-γ in DSKO, DSKO-P14, and control mice. (D) Representative flow cytometry plots of splenic CD8 T cells. (E) Proportion of splenic CD8 T cells that bind GP33TET. (F) Proportion of the activated (IL-18R1+) CD8 T-cell pool occupied by GP33TET-negative cells. Graphs show combined data from 3+ experiments, each point represents an individual mouse. P values determined by two-way ANOVA (A) or one-way ANOVA with Tukey multiple comparisons test of pairwise comparisons against WT-P14 group.62
The P14 TCR includes the TCRVβ8.1 chain,80 and as expected, GP33TET-negative CD8 T cells expressed TCRVβ chains other than TCRVβ8.1. Because we surmised that breakthrough CD8 T cells were responding to a peptide other than GP,25,34-41 we predicted that the few GP33TET-positive T cells in DSKO-P14 mice would also express another TCR. As predicted, the GP33TET-positive cells from DS-P14 mice showed increased coexpression of TCRVβ8.1 and other TCRVβ chains (supplemental Figure 9B,C). Thus, although all T cells in DS-P14 mice werw deficient in perforin and exposed to the same high levels of IL-18, those bearing the GP33-specific TCR (typically the vast majority) were specifically excluded from becoming hyperactivated. Simultaneously, CD8 T cells bearing other TCRs and, thereby, capable of encountering cognate peptides come to dominate the pool of hyperactivated T cells. These data underscore the necessity of antigen stimulation for IL-18 and perforin deficiency to exert their synergistic effects on CD8 T-cell hyperactivation.
Discussion
HLH is a recognizable clinical syndrome of systemic inflammation, the proper management of which requires prompt identification of its life-threatening immunophysiology and simultaneous consideration of its diverse contributors.81 Though widely described in diverse contexts, its contributors are incompletely enumerated and poorly understood. How these contributors converge on this phenotype also remains unclear and a source of clinical confusion. Nonetheless, IFN-γ seems to play a prominent role. Our findings suggest antigen-specific CD8 T-cell hyperactivation may be a necessary upstream event and possibly the convergent mechanism in the pathogenesis of HLH. Circumstantial observations that oligoclonal CD8 T cells dominate the peripheral blood,29,53 and liver,31,32 and nodular parenchymal lesions33 in diverse patients with HLH reinforces the translational validity of these murine results.
The predilection for CD8 (over CD4) T-cell activation in our model remains incompletely explained. The greater expression of perforin by CD8 T cells82,83 and its cell-intrinsic role in terminating immune synapses (ISs)35,84,85 are likely major contributors. Beyond perforin, the observed tropism for CD8 activation could be due to the more potent effects of IL-18 on CD8 T cells, although receptor expression appears comparable (supplemental Figure 1). LCMV-infected Il18tg mice display normal cytotoxicity, yet CD8 T cells remain critical drivers of hyperinflammation25 (Figure 1). Other possible factors include competition for nutrients or growth factors,54 formation of less stable ISs, slower proliferation,86 and/or active suppression of CD4 responses by IL-18responsive CD8 T cells.87
Neutralization of IFN-γ helped decrease anemia and weight loss but did not affect CD8 T-cell activation25 Further exploring T-cell–directed therapies for HLH in DSKO mice, we found that HLH and CD8 T-cell activation were remarkably recalcitrant. At least 5 factors may contribute to this resistance: (1) non–T cells may be critical contributors, although we observed a dramatic reduction of NK1.1+ cells and B cells in DS mice (supplemental Figure 4E; data not shown). (2) Morbidity in these mice arises spontaneously but, we suspect, follows many weeks’ accumulation of immunopathology. Antibody-based depletion of neither CD4 nor CD8 T cells was sufficient to ameliorate immunopathology, but longer-term depletion is not possible because of the development of neutralizing antibodies.88 Likewise, etoposide (chosen for its CD8-specificity in the Prf1⁻/⁻-LCMV model70) was ineffective in treating spontaneous HLH in DS mice, but longer use would likely compound HLH phenotypes due to bone marrow suppression.89 (3) Antibody-based depletion may not be complete90 (supplemental Figure 6). (4) We found that CD4 T cells compensated by increasing their activation status and producing more IFN-γ when CD8 T cells lacked Il18r1 or were depleted (Figure 1D,E; supplemental Figure 6B). (5) Hyperactivated CD8 T cells have shown remarkable persistence despite (attempted) IL-18 receptor deletion, broad-spectrum antibiotics, antiretrovirals, etoposide, and attempts at TCR fixation.
Hyperactivated CD8 T cells were seen specifically in reticuloendothelial organs: sites of both homeostatic and HLH-associated hemophagocytosis.91 These may be sites where CD8 T cells encounter antigen and exert local effects such as producing IFN-γ,92 an established driver of hemophagocytosis. Phenotypically, the protein and transcriptional signatures of CD8 T cells in DS mice contained both terminal effector and exhaustion features. Though typically considered immunosuppressive, TEXs are not intrinsically anti-inflammatory. Terminal TEXs require continuous antigen stimulation and can still produce IFN-γ until the latest stages of exhaustion.93 Features of exhaustion in CD8 T cells of DS mice may indicate progression of naïve cells through an IL-18–amplified effector state and toward exhaustion.94,95 The inflammatory effects of IL-18 on exhausting and exhausted T cells are being actively explored in cancer immunotherapy.96-98 Terminal TEXs do not express Il18r1,99 yet it is highly expressed (at transcript and protein levels) on hyperactivated CD8 T cells in DS mice (Figures 3 and 4B). As a population, these hyperactivated CD8 T cells could represent a pathogenic transitional T effector–like phenotype.100
Mechanistically, DS mice may reveal the nature of immune defects likely to promote CD8 T-cell hyperactivation and HLH. IL-18 appears to be a potent and possibly CD8-tropic amplifier of cytokine production, proliferation, and cytotoxicity. In addition to inducing IL-18 binding protein via IFN-γ, our data suggest activation induced cell death/RICD are also important mechanisms regulating IL-18 effects. Thus, IL-18 (like LCMV, Epstein-Barr virus, or other HLH stimuli) may stress cytotoxic function and, thus, increase sensitivity to common, partial defects like Prf1 haploinsufficiency.101 These stimuli, without sufficient cytotoxic capacity, can drive proliferation, persistence, cytokine production, and potentially inflammatory cell death (eg, necroptosis) or exhaustion.
TCR stimulation appears to be critical to both susceptibility factors. In vitro, IL-18 became relatively inactive on T cells after just a few days without TCR stimulation. In vivo, IL-18 amplified both antigen-specific (LCMV) and innate (TLR9) models of HLH.25 Spontaneous HLH in DS mice gave rise to an oligoclonal expansion of hyperactivated CD8 T cells, but different DS mice did not share CD8 TCR clonotypes. The introduction of the P14 TCR transgene created an in vivo competition between the abundant GP33-specific CD8 T cells and the rare CD8 T cells who escaped allelic exclusion. Cells expressing non-P14 TCRs were at a clear advantage, with IL-18 amplifying and perforin deficiency failing to meet increased demand for regulation. Notably, Prf1⁻/⁻ CD19 chimeric antigen receptor T cells may expand even after elimination of target antigen when wild-type CD19 chimeric antigen receptor T cells have been contracted,102 suggesting an important cell-intrinsic or fratricidal role for Prf1.
The precise nature of TCR stimulation in the DS mice remains an enigma reminiscent of CD8 T virtual memory.103 Classic autoimmunity seems unlikely, because DS mice did not appear to develop autoimmune immunopathology, had disturbed splenic germinal centers,25 and had few CD19+ B cells. Our data cannot entirely rule out responses to self-antigens, commensal or ERV antigens, or even selection for above-normal peptide-independent TCR stimulation are possible. Rather than continuous/cumulative expansion of the same clones, DS mice may experience rolling waves of immunization, hyperactivation, and ultimately exhaustion: a hypothesis that would fit our data in 2-3–week–old pups as well as the absence of overlapping hyperexpanded clonotypes between DS mice.
Experimental combinations of hyperinflammatory contributors have illuminated HLH pathogenesis, promoted clinical test development, and identified both specific and general nodes of therapeutic intervention. For example, both LCMV and IL-18, when combined with perforin deficiency, drive lethal HLH. Perforin deficiency does not enhance TLR9-MAS.25 IL-18 amplifies both LCMV and TLR9, but Il18tg mice (like patients with SJIA) appear to have less aggressive/recalcitrant disease. The clinical correlates of these scenarios are well-documented: patients with FHL require hematopoietic transplants to fully recover,4 infections trigger SJIA-MAS in a sizable minority104; and both patients with MAS and severe infection may be enriched for partial genetic defects in cytotoxicity.9,105,106
Overall, our data reinforce the primacy of CD8 T cells and IFN-γ; identify an effector/exhaustion state arising in spontaneous CD8 T-cell activation; and reveal synergy between CD8 TCR stimulation, signal amplification (eg, IL-18 and LCMV), and impaired immunoregulation. Blocking convergent CD8 T-cell effector functions (eg, with steroid and cytokine, particularly IFN-γ, blockade) may be important temporizing measures, but our data reinforce the ultimate need to identify and minimize long-term drivers of T-cell hyperactivation by targeting type 1 inflammatory stimuli (eg, IL-18), restoring T-cell regulation (eg, cytotoxicity), and clearing relevant antigens. Thus, we believe they support a novel framework for the classification of HLH/MAS based on multiple mechanistic contributors. Rethinking HLH in this way may improve its classification, support novel diagnostic strategies, and encourage personalized, multipronged treatment strategies to improve outcomes in this often-fatal syndrome.
Acknowledgments
The authors thank Eline Luning Prak for assistance in TCR-seq analysis and Dario Vignali for sharing the E8iCreERT2/GFP mice.
Authorship
Contribution: E.L. and S.W.C. planned and performed experiments and wrote the manuscript; J.V., V.D., and A.S.-W. assisted in experiments and data collection; and E.L., S.W.C., and L.P.K. analyzed data.
Conflict-of-interest disclosure: S.W.C. is a site principal investigator for clinical trials run by Novartis and provides consultancy for Simcha Therapeutics, Apollo Therapeutics, and Sobi. The remaining authors declare no competing financial interests.
Correspondence: Scott W. Canna, Rheumatology & Immune Dysregulation, Children's Hospital of Philadelphia, 3615 Civic Ctr Blvd, Philadelphia, PA 19104; e-mail: cannas@chop.edu.
References
Author notes
The full-text version of this article contains a data supplement.