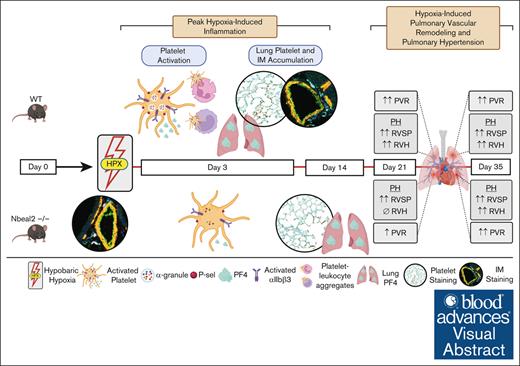
Key Points
Hypoxia-induced platelet activation is not prevented in Nbeal2−/− mice.
This mutation did not prevent hypoxia-induced increase in lung PF4, platelets, or the development of chronic hypoxic PH.
Visual Abstract
Inflammation drives the initiation and progression of pulmonary hypertension (PH). Platelets, increasingly recognized as immune cells, are activated and increased in the lungs of patients with PH. Platelet activation leads to the release of α-granule chemokines, many of which are implicated in PH. We hypothesized that hypoxia-induced secretion of platelet α-granule–stored proteins and PH would be prevented in Neurobeachin-like 2 knockout (Nbeal2−/−) α-granule–deficient mice. Wild-type (WT) and Nbeal2−/− mice were maintained in normoxia or exposed to 10% hypobaric hypoxia for 3, 14, 21, or 35 days. We observed macrothrombocytopenia, increased circulating neutrophils and monocytes, and increased lung interstitial macrophages (IMs) in Nbeal2−/− mice at baseline. Hypoxia-induced platelet activation was attenuated, and hypoxia-induced increase in lung platelet factor 4 (PF4) and platelets was delayed in Nbeal2−/− mice compared with in WT mice. Finally, although pulmonary vascular remodeling (PVR) and PH were attenuated at day 21, Nbeal2−/− mice were not protected against hypoxia-induced PVR and PH at day 35. Although this mutation also affected circulating monocytes, neutrophils, and lung IMs, all of which are critical in the development of experimental PH, we gained further support for the role of platelets and α-granule proteins, such as PF4, in PH progression and pathogenesis and made several observations that expand our understanding of α-granule–deficient mice in chronic hypoxia.
Introduction
Pulmonary hypertension (PH) is a progressive inflammatory disease characterized by increased pulmonary vascular resistance and remodeling that results in right ventricular (RV) pressure overload and hypertrophy.1-3 Clinically, PH is defined by elevated mean pulmonary artery systolic pressure of ≥20 mm Hg and pulmonary vascular resistance of ≥2.0 Wood units.4 Despite advancements in available therapies, survival rates remain poor, with a median survival rate between 5 and 10 years from the time of diagnosis.1,2,5,6 Inflammation is a key driver in the initiation and progression of PH. Increased circulating and pulmonary proinflammatory chemokines and infiltration of immune cells, such as macrophages, occur in preclinical models and patients with PH.3,7-13 Several groups have shown that systemic and local inflammation occurs before vasculopathic changes and the development of experimental PH.1,2,12-18
Platelets are increasingly recognized as immune cells in addition to their roles in hemostasis and thrombosis.19-24 Thrombotic lesions are a common pathological finding in PH, and we recently showed that platelets are increased in the lungs of patients with PH.1,14,25,26 Platelets are activated and circulating platelet-derived proteins are increased in patients with PH.27-29 Our group and others have demonstrated that platelets are activated and increased in the lungs of rodents with experimental PH.14,28-34 Platelets contain lysosomes, dense (δ) granules, and alpha (α) granules. Over 300 proteins are stored in platelet α granules, many of which are increased in the lungs and circulation of patients with PH.20,35,36 One of these proteins, platelet factor 4 (PF4), binds with high affinity to glycosaminoglycans and contributes directly to vascular remodeling by inducing myofibroblast differentiation and collagen synthesis as well as promoting monocyte activation, recruitment, and differentiation of macrophages toward a proinflammatory phenotype.20,35,37-44
The neurobeachin-like 2 (Nbeal2) gene is required for α-granule maturation and the packaging of α-granule proteins.45-47Nbeal2−/− platelets have a disorganized and diffuse distribution of α-granule proteins that are secreted abnormally.35,47 Loss of function in this gene results in macrothrombocytopenia and impaired platelet adhesion, thrombus formation, and wound healing.35,45,48 However, Nbeal2−/− mice have no signs of spontaneous bleeding and have normal platelet lifespans.45,48,49 In addition to platelets, aberrant α-granule formation secondary to Nbeal2 gene mutation results in decreased granularity and impaired functions of neutrophils, monocytes, T cells, and mast cells.50-54 These α-granule–deficient mice have been used to demonstrate the importance of α-granule–stored proteins in several inflammatory-mediated disease processes. These studies reveal that host defense to viral and bacterial infection is decreased in Nbeal2−/− mice, yet they are protected from thrombo-inflammation in ischemic stroke and dermal injury models.42,45,49,53 Given increasing support for platelets and α-granule proteins such as PF4 in the pathogenesis of PH, we used Nbeal2−/− (α-granule–deficient) mice to test our hypothesis that hypoxia-induced secretion of platelet α-granule–stored proteins would be prevented in Nbeal2−/− mice and therefore these mice would be protected against the development of hypoxia-induced pulmonary vascular remodeling (PVR) and PH.
Material and methods
Mouse model
C57Bl/6 wild-type (WT) mice were obtained from The Jackson Laboratories (Bar Harbor, ME) and Nbeal2−/− mice (on C57Bl/6 background) were obtained from Jorge Di Paola (Washington University School of Medicine, Department of Pediatrics, St. Louis, MO). C57BL/6 mice were bred in Denver, and male and female offspring were weaned into sea-level acclimatization chambers (barometric pressure of 760 mm Hg) and evaluated at 6 to 8 weeks of age. Control groups remained in the sea-level chambers, and experimental groups were placed into hypobaric (barometric pressure of 380 mm Hg; FiO2 = 10%) hypoxic chambers for 3, 14, 21, or 35 days. All studies were approved by the institutional animal care and use committee at the University of Colorado, Denver, Anschutz Medical Campus.
Preparation of blood, platelets, and plasma
Mice were anesthetized with 1% to 2% isoflurane, and blood was obtained via terminal closed-chest cardiac puncture of the right ventricle as previously described.14,55 Complete blood counts were obtained from heparin-anticoagulated blood using the hematologic Heska HT5 analyzer (Loveland, CO). Platelet-rich plasma was obtained by centrifugation of heparin-anticoagulated whole blood at 100g for 16 minutes. Platelet-rich plasma was supplemented with prostaglandin I2 (PGI2; 1 μg/mL, Cayman Chemical) and apyrase (0.02 U/mL, Sigma-Aldrich) to avoid platelet preactivation, and incubated at room temperature (RT) for 3 minutes before centrifugation at 2000g for 2 minutes to obtain platelet-poor plasma (PPP) and platelet pellets. Platelet pellets were resuspended in 100 μL of modified Tyrode buffer, pH 7.3 (NaCl, 129 mM; KCl, 2.9 mM; MgCl2, 1 mM; NaH2PO4, 0.34 mM; NaHCO3, 12 mM; and glucose, 5 mM). Platelet counts were obtained using the hematologic Heska HT5 analyzer (Loveland, CO). Platelets were pelleted at 2000g for 2 minutes, and the supernatant was discarded. Platelet pellets and plasma samples were stored at −80°C until use.
PF4 western blot
Platelet pellets (10 ×106 platelets per pellet) were obtained as described above and frozen at −80°C. Platelets were lysed and separated by gel electrophoresis as previously described.56 Membranes were incubated with rabbit anti-mouse PF4 primary antibody (PF4; Abclonal, A3694; 1:2500) overnight at 4°C followed by donkey anti-rabbit horseradish peroxidase (HRP)–conjugated secondary antibody (Santa Cruz, 2313; 1:10 000). Membranes were developed using SuperSignal Femto Chemiluminescent substrates (ThermoFisher, Waltham, MA). Protein bands were quantified by densitometry using Image Laboratory Software (Bio-Rad).
Assessment of platelet activation
Acid citrate dextrose–anticoagulated whole blood was obtained as described above, and washed platelets were obtained as previously described.56,57 Washed platelets were diluted (1 × 106 platelets) in 100 μL of warm Tyrode buffer containing 1 mM CaCl2. Platelets were incubated for 10 minutes at RT in the dark in the presence of the appropriate antibodies (supplemental Table 1). The assay was quenched at 10 minutes by diluting the blood 1:5 using RT 1% paraformaldehyde (PFA) in Tyrode buffer. Specific monoclonal antibody (mAb) binding was expressed as a percentage of positive cells in the target gate (supplemental Figure 1).
Assessment of PLAs
Acid citrate dextrose–anticoagulated whole blood was immediately diluted 1:10 in M199 media containing 100 U/mL heparin for platelet-leukocyte aggregation studies. For the characterization of platelet-leukocyte aggregates (PLAs), 300 μL of diluted, citrated, blood was incubated for 15 minutes at 37°C in the dark in the presence of the appropriate antibodies (supplemental Table 1). The assay was quenched using 1.5 mL FACS red cell lysis buffer (BD Biosciences) and fixed for 15 minutes in the dark at RT. Samples were centrifuged at 800g for 5 minutes, the supernatant was discarded, and fixed cells were resuspended in 600 μL FACS red cell lysis buffer (BD Biosciences). Specific mAb binding was expressed as a percentage of positive cells in the target gate (supplemental Figure 2).
PF4 ELISA
PF4 was measured in PPP, platelet lysates (250 000 platelets per well), and lung homogenates using the mouse PF4 enzyme-linked immunosorbent assay (ELISA) kit (Abcam, Cambridge, MA). PPP and platelets were obtained as described above. Lungs were collected and homogenized as previously described.56 Homogenized samples were incubated for 30 minutes on ice, then centrifuged at 10 000g for 5 minutes, and stored at −80°C.
Evaluation of lung platelets
Lungs were flushed with 10 mL of phosphate-buffered saline and inflated for paraffin embedding and immunohistochemistry (IHC) was performed for CD41 as previously described.14,55-57 IHC staining and quantification were performed on lung sections using anti-mouse CD41 (Genetex, GTX113758; 1:200) and Dako EnVision + Dual Link System anti-rabbit HRP 3,3′-diaminobenzidine (DAB+ chromagen) detecting system (Agilent, Carpinteria, CA). Whole-lung scans were collected using the Leica Aperio VERSA brightfield scope (40× objective, 0.5-micron resolution). Fifteen random images per slide were captured, excluding lung fields containing large vessels or airways, and distal lung platelets were assessed by quantifying CD41+ pixels per high-powered field (×20). CD41+ pixels were quantified in whole-lung sections using a random, nonbiased approach using ImageScope, version 12.4.3.500 (Leica Biosystems Imaging, Inc, Deer Park, IL) pixel quantification software as previously described.1 An investigator blinded to the experimental groups performed the analysis.
Multiplex imaging of lung tissue
Lungs were collected as described above. We collaborated with the human immune monitoring shared resource at the University of Colorado School of Medicine and performed multispectral imaging using the Akoya Vectra Polaris instrument. Tissue sections were stained consecutively with specific primary antibodies according to standard protocols provided by Akoya and performed routinely by the human immune monitoring shared resource followed by HRP-conjugated secondary antibody polymer, and HRP-reactive OPAL fluorescent reagents. Platelet and PF4 imaging: slides were stained with anti-mouse CD41 (Genetex, GTX113758, 1:100), CD45 (CST, Clone No. D3F8Q, 60257S, 1:100), CD68 (CST, Clone No. E307V, 97778S, 1:1000), PF4 (Abclonal, A3694, 1:200), and DAPI (4’,6-diamidino-2-phenylindole). Images were captured using the 3I Marianas Spinning Disk Confocal attached to a Zeiss Axio Observer 7 microscope (100× objection, 0.1-micron resolution. Images were processed using ImageJ 1.54f open-source software (Wayne Rasband and contributors, National Institute of Health). Interstitial macrophage (IM) imaging: slides were stained with anti-mouse α-smooth muscle actin (α-SMA; Cell Signaling Technology [CST], clone no. D4k9N; 1:400), CD31 (CST, clone no. D8V9E/PECAM-1, 77699S; 1:100), CD45 (CST, clone no. D3F8Q, 60257S; 1:200), and CD68 (CST, clone no. E307V, 97778S; 1:1000), and counterstained with DAPI. Whole-slide scans were collected using the Vectra Polaris spatial imaging scope (40× objective, 0.5-micron resolution). Representative distal lung and pulmonary vessel images were captured using Phenochart 1.1.0 open-source software (Akoya Biosciences, Hopkinton, MA).
In situ hybridization for PF4 expression
RNAScope detection of Pf4 messenger RNA (mRNA) was used to perform in situ hybridization according to the manufacturer’s protocol (Advanced Cell Diagnostics, Hayward, CA), as previously described.58 Probes directed against Pf4 mRNA were applied at +40°C in the following order: target probes, preamplifier, amplifier, and label probe for 10 minutes. After each hybridization step, slides were washed 2 times at RT. mRNA detection was followed by immunofluorescent staining for nucleated hematopoietic cells and platelets (CD45+ and CD41+). Slides were blocked in 5% bovine serum albumin buffer for 1 hour at RT, incubated with primary antibodies, anti-mouse CD41 (Genetex, GTX113758; 1:100) and CD45 (CST, clone no. D3F8Q, 60257S; 1:200) overnight at 4°C, stained with appropriate fluorescently labeled secondaries antibodies and counterstained with DAPI at 1:20 000 (BioLegend, 422801). Staining was visualized using an OlympusVS120 (Evident, Waltham, MA) whole-slide scanner using 40× objective 0.5-micron resolution. Representative distal lung and pulmonary vessel images were captured using QuPath 0.5.1 open-source software.59
Assessment of lung IMs
Mice were retro-orbitally injected with 100 μL of CD45 in phosphate-buffered saline 5 minutes before lung collection to label intravascular leukocyte populations as previously described.14 Briefly, the lungs were flushed to dislodge the majority of circulating leukocytes, then collected and digested in 1 mL of Hanks balanced salt solution with no calcium or magnesium (Sigma), Liberase thermolysin medium (Sigma, 0.4 mg/mL), and DNase I (Sigma, 100 U/mL). Lungs were processed to obtain a single-cell suspension, as previously described.60 Cells were then incubated for 30 minutes in the dark in the presence of the appropriate antibodies (supplemental Table 1). Specific mAb binding was expressed as total cells in the target gate and absolute counts were calculated using 123 counting beads (Invitrogen, Waltham, MA). IM subtypes were differentiated as previously described by Gibbings et al,61 bearing in mind, there are other accepted segregation strategies for segregating lung IM subsets (supplemental Figure 5).62-64
Evaluation of pulmonary vessel density and MWT
Mouse lungs were flushed and inflated for paraffin embedding, and IHC was performed for CD41, as previously described55 IHC staining and quantification of muscularized vessels were performed on lung sections using anti-mouse α-SMA (Sigma, A2547, 1:1200) and Dako Envision anti-rabbit detecting system (Statlab). Whole-slide scans were collected using the Leica Aperio VERSA brightfield scope (40× objective, 0.5-micron resolution). Ten random images per slide were captured, excluding lung fields containing large vessels or airways. Muscularization of small vessels was assessed by counting the number of vessels of <50 μm staining positive for α-SMA per high-power field (×10). Medial wall thickness (MWT) was calculated for α-SMA–stained vessels of 50 to 200 μm located adjacent to airways; ×10 vessels were included per mouse. The width of the medial wall was measured in 4 perpendicular locations, and an average was calculated. The external diameter was measured in 2 locations, this was then averaged and divided by 2 to calculate the average radius. The MWT was expressed as the average width of the medial wall/average radius. An investigator blinded to the experimental groups performed the analysis.
Hemodynamic measurements and evaluation of RVH
PH was assessed by RV puncture via closed chest to obtain RV systolic pressures (RVSPs) and the Fulton Index for RV hypertrophy at 21 and 35 days. RVSPs were obtained from averaging 3-second waveforms and analyzed using AcqKnowledge, version 3.9.1-100M (BIOPAC Systems, Inc, Goleta, CA). Hearts were removed and dissected to isolate the free wall of the RV from the left ventricle (LV) and septum (S). The ratio of RV weight over LV + S weight (RV/LV + S), or the Fulton Index, was used as an index of RV hypertrophy (RVH) resulting from PH. An investigator blinded to the experimental group performed the analysis.
Open chest solid state catheterization for RV function analysis
Normoxic and hypoxic WT and Nbeal2−/− mice underwent terminal open chest RV function measurements with a 1.2F, FTE-1212B-4018 pressure-volume catheter (Transonic Systems Inc, Ithaca, NY) inserted by direct cardiac puncture, as previously described.65 Briefly, anesthesia induction was achieved with inhaled isoflurane (4%-5%), a tracheal tube was inserted and connected to an Anesthesia Workstation, and a plane was maintained at 1.0% to 2.5% isoflurane in 100% oxygen. The heart was exposed by bilateral thoracotomy, and the pericardium was resected. Steady-state hemodynamics are collected with short pauses in ventilation (up to 10 seconds) or high-frequency oscillatory ventilation to eliminate ventilator artifacts from the pressure-volume recordings obtained via a pressure-volume catheter. Occlusions of the inferior vena cava were performed by applying pressure to the inferior vena cava (up to 10 seconds) through the abdominal opening. Data were recorded continuously using LabScribe2 (iWorx, Dover, NH) and analyzed offline.
Statistical analysis
Data were analyzed using Prism (GraphPad Software, La Jolla, CA) by Student t test or 2-way analysis of variance with Tukey post hoc analysis where appropriate. Data are expressed as mean ± standard error of the mean; significance is defined as P ≤.05. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−.
Results
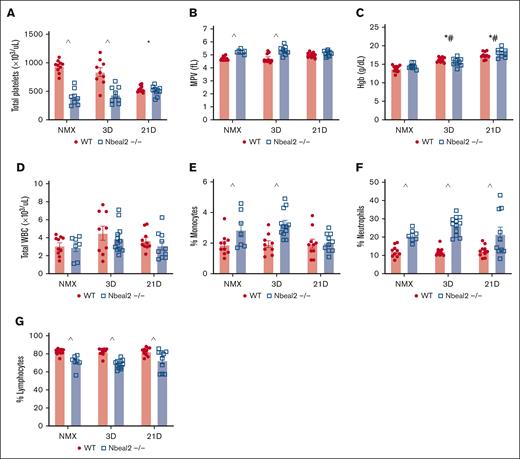
Hypoxia-exposed WT mice developed thrombocytopenia, whereas in Nbeal2−/− mice, platelet count did not further decrease
Thrombocytopenia is reported in up to 20% of patients with idiopathic pulmonary arterial hypertension and independently predicts mortality.66 In agreement with prior studies, we found that Nbeal2−/− mice have a ∼50% reduction in platelet count at baseline (Figure 1A).45,47,67 Hypoxia-exposed WT mice developed thrombocytopenia, resulting in similar platelet numbers between genotypes at day 21 (Figure 1A). Hemoglobin was similar between WT and Nbeal2−/− mice and increased similarly with hypoxia (Figure 1C). There was no difference in total white blood cell count between WT and Nbeal2−/− at baseline (Figure 1D). However, we observed significant differences in baseline leukocyte subpopulations, with an increased percentage of neutrophils and monocytes in Nbeal2−/− mice. In hypoxic Nbeal2−/− mice, the percentage of monocytes decreased at day 21 compared with day 3, while total white blood cells or the percentage of neutrophils or lymphocytes did not change (Figure 1D-G).53,54
Hypoxia-exposed WT mice developed thrombocytopenia, whereas in Nbeal2−/− mice, platelet count did not further decrease. (A) Platelet counts were decreased in Nbeal2−/− compared with WT mice at baseline and after 3 days of hypoxia. At day 21, platelet counts decreased in hypoxic WT mice. (B) MPV was decreased in hypoxic Nbeal2−/− compared with WT mice at baseline and after 3 days of hypoxia. (C) Hemoglobin (Hgb) increased in hypoxic WT and Nbeal2−/− mice at days 3 and 21. (D) There was no difference in total white blood cell (WBC) count between WT and Nbeal2−/− at baseline or with hypoxia. (E-G) In Nbeal2−/− mice at baseline and at day 3, the percentages of circulating neutrophils and monocytes were increased, and the percentage of circulating lymphocytes was decreased compared with WT mice. In Nbeal2−/− mice at day 21, the percentage of circulating monocytes decreased, whereas the percentage of circulating neutrophils remained elevated and circulating lymphocytes remained decreased compared with WT mice. Statistics: P ≤.05 by 2-way analysis of variance (ANOVA) with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. MPV, mean platelet volume; NMX, normoxia.
Hypoxia-exposed WT mice developed thrombocytopenia, whereas in Nbeal2−/− mice, platelet count did not further decrease. (A) Platelet counts were decreased in Nbeal2−/− compared with WT mice at baseline and after 3 days of hypoxia. At day 21, platelet counts decreased in hypoxic WT mice. (B) MPV was decreased in hypoxic Nbeal2−/− compared with WT mice at baseline and after 3 days of hypoxia. (C) Hemoglobin (Hgb) increased in hypoxic WT and Nbeal2−/− mice at days 3 and 21. (D) There was no difference in total white blood cell (WBC) count between WT and Nbeal2−/− at baseline or with hypoxia. (E-G) In Nbeal2−/− mice at baseline and at day 3, the percentages of circulating neutrophils and monocytes were increased, and the percentage of circulating lymphocytes was decreased compared with WT mice. In Nbeal2−/− mice at day 21, the percentage of circulating monocytes decreased, whereas the percentage of circulating neutrophils remained elevated and circulating lymphocytes remained decreased compared with WT mice. Statistics: P ≤.05 by 2-way analysis of variance (ANOVA) with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. MPV, mean platelet volume; NMX, normoxia.
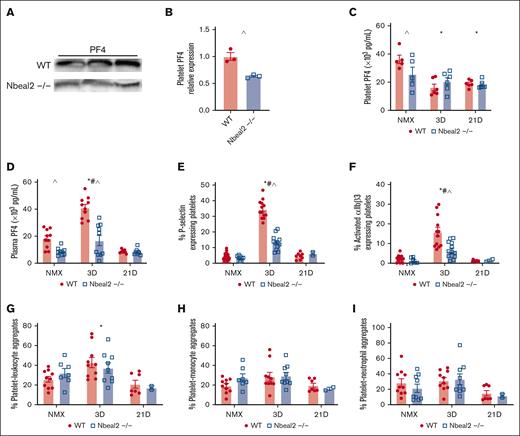
Platelet activation increased in both WT and Nbeal2−/− hypoxic mice
We, and others, have demonstrated that hypoxia causes platelet activation.14,19,24 We assessed hypoxia-induced platelet activation by quantifying plasma PF4, platelet P-selectin expression, activation of the αIIBβ3 integrin, and formation of PLAs, including platelet-monocyte aggregates (PMAs), and platelet-neutrophil aggregates (PNAs). Nbeal2−/− mice have ∼30% to 40% less platelet PF4 than WT mice at baseline (Figure 2A-C). Platelet PF4 decreased in hypoxic WT mice at day 3 and 21 (Figure 2C). In WT hypoxic mice, we observed peak platelet activation at day 3, evidenced by increased plasma PF4, increased expression of platelet P-selectin, αIIBβ3 activation, and increased PLAs (Figure 2D-G). There was no change in PMAs or PNAs in hypoxic WT mice (Figure 2H-I). In Nbeal2−/− mice, hypoxia-induced increase in the platelet α-granule–stored proteins, PF4, and P-selectin, and activation of αIIBβ3 was attenuated, and we observed no change in PLAs, PMAs, or PNAs (Figure 2B-I).
Platelet activation increased in WT and Nbeal2−/− hypoxic mice. (A) Representative western blot of platelet lysates for PF4. (B) Quantification of western blot demonstrates platelet PF4 is reduced in Nbeal2−/− mice compared with WT mice at baseline. (C) Platelet PF4 levels are reduced in Nbeal2−/− mice compared with WT mice at baseline. Platelet PF4 is decreased in hypoxic WT mice at days 3 and 21 but not Nbeal2−/− mice. (D) Plasma PF4 was decreased at baseline in Nbeal2−/− compared with WT mice. Plasma PF4 increased in hypoxic WT and Nbeal2−/− at day 3. Hypoxia-induced increase in plasma PF4 was attenuated in Nbeal2−/− mice. (E) The percentage of P-selectin expressing platelets increased in hypoxic WT and Nbeal2−/− mice at day 3. Hypoxia-induced increase in P-selectin expressing platelets was attenuated in Nbeal2−/− compared with WT mice. (F) The percentage of activation of αIIBβ3 (JONA) expressing platelets increased in hypoxic WT and Nbeal2−/− mice at day 3. The hypoxia-induced increase in the percentage of platelets expressing activated αIIBβ3 (JONA) was attenuated in Nbeal2−/− compared with WT mice. (G-I) PLAs are increased in hypoxic WT mice at day 3 but not Nbeal2−/− mice. PMAs and PNAs are similar between WT and Nbeal2−/− mice at baseline and unchanged in hypoxia. Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. NMX, normoxia.
Platelet activation increased in WT and Nbeal2−/− hypoxic mice. (A) Representative western blot of platelet lysates for PF4. (B) Quantification of western blot demonstrates platelet PF4 is reduced in Nbeal2−/− mice compared with WT mice at baseline. (C) Platelet PF4 levels are reduced in Nbeal2−/− mice compared with WT mice at baseline. Platelet PF4 is decreased in hypoxic WT mice at days 3 and 21 but not Nbeal2−/− mice. (D) Plasma PF4 was decreased at baseline in Nbeal2−/− compared with WT mice. Plasma PF4 increased in hypoxic WT and Nbeal2−/− at day 3. Hypoxia-induced increase in plasma PF4 was attenuated in Nbeal2−/− mice. (E) The percentage of P-selectin expressing platelets increased in hypoxic WT and Nbeal2−/− mice at day 3. Hypoxia-induced increase in P-selectin expressing platelets was attenuated in Nbeal2−/− compared with WT mice. (F) The percentage of activation of αIIBβ3 (JONA) expressing platelets increased in hypoxic WT and Nbeal2−/− mice at day 3. The hypoxia-induced increase in the percentage of platelets expressing activated αIIBβ3 (JONA) was attenuated in Nbeal2−/− compared with WT mice. (G-I) PLAs are increased in hypoxic WT mice at day 3 but not Nbeal2−/− mice. PMAs and PNAs are similar between WT and Nbeal2−/− mice at baseline and unchanged in hypoxia. Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. NMX, normoxia.
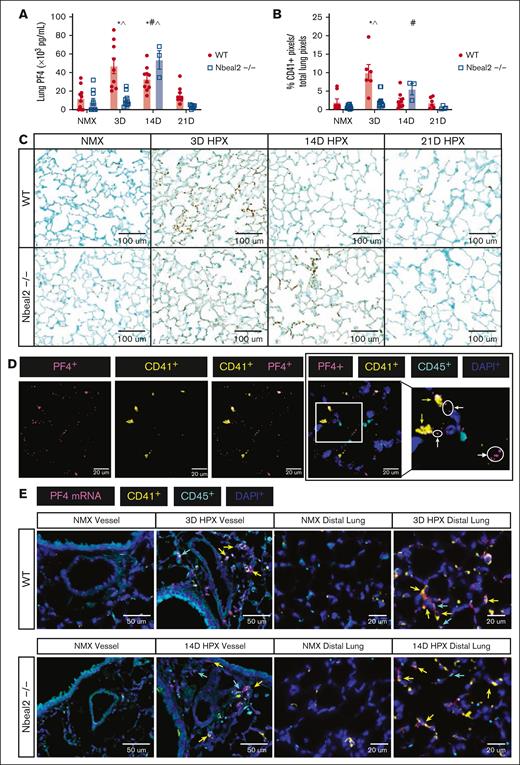
Hypoxia-induced increase in lung PF4 and accumulation of lung platelets was delayed in Nbeal2−/− mice
We previously demonstrated that platelets are increased in the lungs of hypoxic WT mice and patients with end-stage PH.14 Platelets store the majority of PF4, and we and others have shown that platelet-depleted mice have greatly diminished levels of PF4.14,42,68 To determine whether α-granule deficiency prevents hypoxia-induced increase in lung PF4 and platelets, we performed ELISA for PF4 and IHC for the platelet-specific protein CD41. Lung PF4 and lung platelet accumulation did not differ by genotype at baseline (Figure 3A-C). We observed peak hypoxia-induced increase in lung PF4 and lung platelets at day 3 in WT mice, and day 14 in Nbeal2−/− mice (Figure 3A-C). Lung PF4 protein accumulated both extracellularly and within platelets (Figure 3D; supplemental Figure 3). We assessed Pf4 mRNA expression in the pulmonary vessels and distal lung by performing RNA Scope. Qualitatively, we observed peak hypoxia-induced Pf4 mRNA expression at day 3 in hypoxic WT mice and day 14 in Nbeal2−/− mice (Figure 4E; supplemental Figure 4). Pf4 mRNA was expressed in megakaryocytes, as well as leukocytes and other cells throughout the lung.
Hypoxia-induced increase in lung PF4 and accumulation of lung platelets was delayed in Nbeal2−/− mice. (A) Lung PF4 increased in hypoxic WT mice at days 3 and 14. Lung PF4 increased in hypoxic Nbeal2−/− mice at day 14. Hypoxia-induced increase in lung PF4 was decreased at day 3 and increased at day 14 in Nbeal2−/− compared with WT mice. (B) Platelets accumulated in the distal lung of hypoxic WT mice at day 3, which resolved by day 21. Accumulation of distal lung platelets was not observed in hypoxic Nbeal2−/− mice. (C) Whole-slide scans were obtained using the Leica Aperio VERSA brightfield scope (×40 objective, 0.5-micron resolution). Representative images of CD41 staining in the distal lung (original magnification ×20; scale bars, 100 μm. As previously described, CD41+ pixels were quantified in whole-lung sections using a random, nonbiased approach using ImageScope, version 12.4.3.500 (Leica Biosystems Imaging, Inc, Deer Park, IL) pixel quantification software.1 (D) Lung sections were stained with anti-PF4, anti-CD41, anti-CD45, and DAPI. Whole-slide scans were obtained using the 3I Marianas Confocal Microscope (×100 objective, 0.1-micron resolution). Representative images of intracellular and extracellular PF4 were captured. Original magnification ×100; scale bars, 50 μm. White arrows indicate extracellular PF4 (magenta), and yellow arrows indicate intracellular PF4 within platelets (yellow). (E) Lung sections were probed with a PF4 mRNA primer and anti-CD41, anti-CD45, and DAPI antibodies. Whole-slide scans were obtained using (×40 objective, 0.5-micron resolution). Representative images of pulmonary vessels and the distal lung were captured. Representative images of peak Pf4 mRNA expression within megakaryocytes and leukocytes in the pulmonary vessels and distal lung of WT (day 3) and Nbeal2−/− (day 14) vs their baseline, as well as other unidentified cells (original magnification ×40; scale bars, 50 μm). Yellow arrows indicate PF4 mRNA (magenta) within megakaryocytes (yellow), and blue arrows indicate Pf4 mRNA within leukocytes (cyan). Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. HPX, hypoxia; NMX, normoxia.
Hypoxia-induced increase in lung PF4 and accumulation of lung platelets was delayed in Nbeal2−/− mice. (A) Lung PF4 increased in hypoxic WT mice at days 3 and 14. Lung PF4 increased in hypoxic Nbeal2−/− mice at day 14. Hypoxia-induced increase in lung PF4 was decreased at day 3 and increased at day 14 in Nbeal2−/− compared with WT mice. (B) Platelets accumulated in the distal lung of hypoxic WT mice at day 3, which resolved by day 21. Accumulation of distal lung platelets was not observed in hypoxic Nbeal2−/− mice. (C) Whole-slide scans were obtained using the Leica Aperio VERSA brightfield scope (×40 objective, 0.5-micron resolution). Representative images of CD41 staining in the distal lung (original magnification ×20; scale bars, 100 μm. As previously described, CD41+ pixels were quantified in whole-lung sections using a random, nonbiased approach using ImageScope, version 12.4.3.500 (Leica Biosystems Imaging, Inc, Deer Park, IL) pixel quantification software.1 (D) Lung sections were stained with anti-PF4, anti-CD41, anti-CD45, and DAPI. Whole-slide scans were obtained using the 3I Marianas Confocal Microscope (×100 objective, 0.1-micron resolution). Representative images of intracellular and extracellular PF4 were captured. Original magnification ×100; scale bars, 50 μm. White arrows indicate extracellular PF4 (magenta), and yellow arrows indicate intracellular PF4 within platelets (yellow). (E) Lung sections were probed with a PF4 mRNA primer and anti-CD41, anti-CD45, and DAPI antibodies. Whole-slide scans were obtained using (×40 objective, 0.5-micron resolution). Representative images of pulmonary vessels and the distal lung were captured. Representative images of peak Pf4 mRNA expression within megakaryocytes and leukocytes in the pulmonary vessels and distal lung of WT (day 3) and Nbeal2−/− (day 14) vs their baseline, as well as other unidentified cells (original magnification ×40; scale bars, 50 μm). Yellow arrows indicate PF4 mRNA (magenta) within megakaryocytes (yellow), and blue arrows indicate Pf4 mRNA within leukocytes (cyan). Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. HPX, hypoxia; NMX, normoxia.
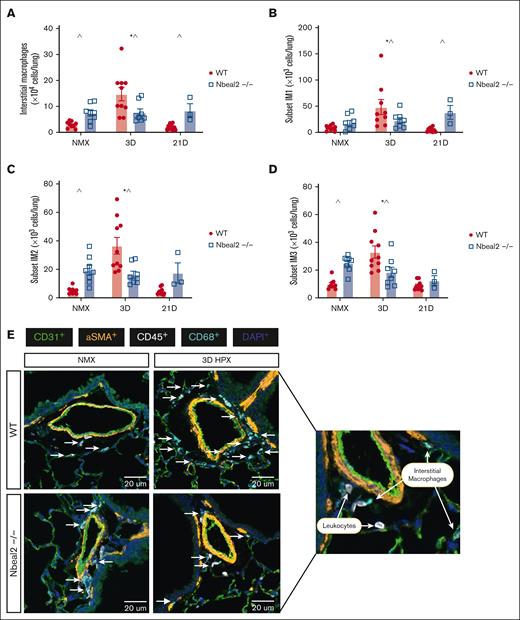
Lung IMs were increased at baseline and hypoxia-induced increase in lung IMs was prevented in Nbeal2−/− mice. (A) Lung IMs were increased in Nbeal2−/− mice at baseline compared with WT mice. (A-D) At day 3, total IMs and IM1, IM2, and IM3 subsets increased in WT but not Nbeal2−/− mice. At day 21, total lung IMs and IM1s were increased in Nbeal2−/− compared with WT mice. (E) Lung sections were stained with anti-CD31, anti-α-SMA, anti-CD68, anti-PF4, anti-CD41, and anti-CD45, and DAPI. Whole-slide scans were obtained using the Vectra Polaris spatial imaging scope (×40 objective, 0.5-micron resolution). Representative images of pulmonary vessels were captured. Representative image of IMs in WT and Nbeal2−/− mice (original magnification ×40; scale bars, 50 μm). Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. HPX, hypoxia; NMX, normoxia.
Lung IMs were increased at baseline and hypoxia-induced increase in lung IMs was prevented in Nbeal2−/− mice. (A) Lung IMs were increased in Nbeal2−/− mice at baseline compared with WT mice. (A-D) At day 3, total IMs and IM1, IM2, and IM3 subsets increased in WT but not Nbeal2−/− mice. At day 21, total lung IMs and IM1s were increased in Nbeal2−/− compared with WT mice. (E) Lung sections were stained with anti-CD31, anti-α-SMA, anti-CD68, anti-PF4, anti-CD41, and anti-CD45, and DAPI. Whole-slide scans were obtained using the Vectra Polaris spatial imaging scope (×40 objective, 0.5-micron resolution). Representative images of pulmonary vessels were captured. Representative image of IMs in WT and Nbeal2−/− mice (original magnification ×40; scale bars, 50 μm). Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. HPX, hypoxia; NMX, normoxia.
In Nbeal2−/−mice, lung IMs were increased at baseline, and the hypoxia-induced increase in lung IMs was prevented
Lung IM accumulation in the perivascular/adventitial space is a prominent driver of PH.7,14 Hypoxia-induced lung IM accumulation occurs early in WT mice, peaking at day 3 to 4 and waning by day 14.69 We found that total IMs and IM2 and IM3 subsets were increased in Nbeal2−/− mice at baseline compared with WT mice (Figure 4A,C-D). Consistent with prior reports, in hypoxic WT mice, total lung IMs increased at day 3, returning to baseline by day 21 (Figure 4A).11,69 IM1, IM2, and IM3 subsets increased in hypoxic WT mice at day 3 and returned to baseline by day 21 (Figure 4B-D). We previously reported that platelet depletion prevents hypoxia-induced increase in lung IMs.14 We wondered whether α-granule deficiency would prevent hypoxia-induced pulmonary IM expansion. Hypoxia-induced increase in total IMs and IM subtypes was not observed in Nbeal2−/− mice (Figure 4A-D). Using immunofluorescent staining, we visualized hypoxia-induced lung IM accumulation at day 3 in the perivascular space of airway-adjacent vessels in WT but not Nbeal2−/− mice (Figure 4E).
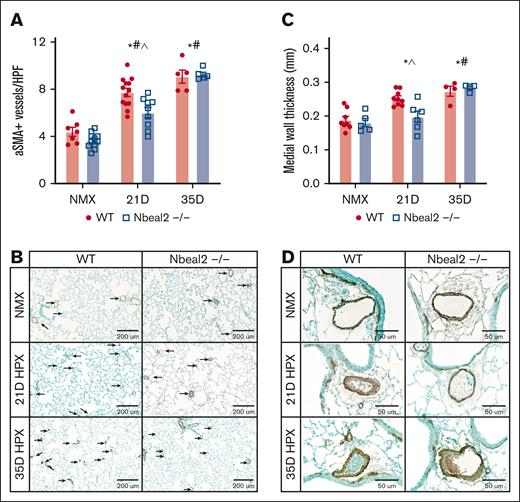
Hypoxia-induced PVR was delayed in Nbeal2−/− mice compared with WT mice
In experimental models and patients with PH, remodeling of the pulmonary arteries is a pathologic feature and contributes to increased pulmonary artery resistance.1,3,70,71 We examined PVR in WT and α-granule–deficient Nbeal2−/− mice by quantifying the number of α-SMA–positive vessels of <50 μm and MWT of vessels between 50 and 200 μm at day 21 and 35. In hypoxic WT mice, the number of α-SMA–positive vessels of <50 μm was increased at days 21 and 35. Hypoxia-induced increase in the number of α-SMA–positive vessels of <50 μm was attenuated in Nbeal2−/− mice compared with WT mice at day 21, but similar to WT mice at day 35 (Figure 5A-B). MWT increased in hypoxic WT mice at days 21 and 35. MWT was unchanged in hypoxic Nbeal2−/− mice at day 21, but at day 35, MWT increased similarly to that in WT mice (Figure 5C-D).
Hypoxia-induced PVR was delayed in Nbeal2−/− mice compared with WT mice. Whole-slide scans were obtained using the Leica Aperio VERSA brightfield scope (×40 objective, 0.5-micron resolution). (A) At day 21, hypoxia-induced muscularization of vessels of <50 μm was attenuated in Nbeal2−/− compared with WT mice. At day 35, hypoxia-induced muscularization of vessels of <50 μm was similar between WT and Nbeal2−/− mice. (B) Representative α-SMA staining in the distal lung (original magnification ×10; scale bars, 200 μm). (C) At day 21, the hypoxia-induced increase in MWT was prevented in Nbeal2−/− mice. At day 35, the hypoxia-induced increase in MWT was similar between WT and Nbeal2−/− mice. (D) Representative vessel images (original magnification ×40; scale bars, 50 μm). Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. HPF, high-powered field; HPX, hypoxia; NMX, normoxia.
Hypoxia-induced PVR was delayed in Nbeal2−/− mice compared with WT mice. Whole-slide scans were obtained using the Leica Aperio VERSA brightfield scope (×40 objective, 0.5-micron resolution). (A) At day 21, hypoxia-induced muscularization of vessels of <50 μm was attenuated in Nbeal2−/− compared with WT mice. At day 35, hypoxia-induced muscularization of vessels of <50 μm was similar between WT and Nbeal2−/− mice. (B) Representative α-SMA staining in the distal lung (original magnification ×10; scale bars, 200 μm). (C) At day 21, the hypoxia-induced increase in MWT was prevented in Nbeal2−/− mice. At day 35, the hypoxia-induced increase in MWT was similar between WT and Nbeal2−/− mice. (D) Representative vessel images (original magnification ×40; scale bars, 50 μm). Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. HPF, high-powered field; HPX, hypoxia; NMX, normoxia.
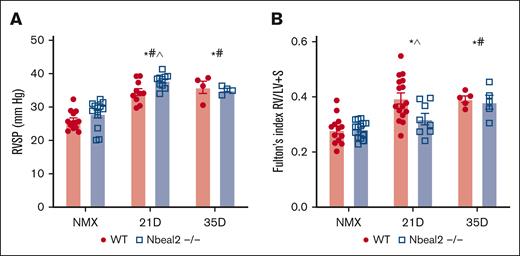
Hypoxia-induced increase in RVSPs was similar in WT and Nbeal2−/− mice, but hypoxia-induced RVH was delayed in Nbeal2−/− mice
An elevation in pulmonary artery pressure defines PH. RVH develops as a consequence of increased RV pressure, which may lead to right-sided heart failure.1,2,5 We evaluated hypoxic PH by measuring RVSPs and RVH using the Fulton Index (ratio weight of the right ventricle-to-LV + S) at days 21 and 35. We found that RVSP increased similarly in hypoxic WT and Nbeal2−/− mice at days 21 and 35 (Figure 6A). Hypoxic WT mice developed RVH at days 21 and 35 (Figure 6B). Nbeal2−/− mice developed RVH at day 35 (Figure 6B). We considered that the lack of RVH after 21 days of hypoxia could be maladaptive. We conducted invasive hemodynamic studies at this time point, revealing that Nbeal2−/− had normal RV function (supplemental Figure 6).
Hypoxia-induced increase in RVSPs was similar in WT and Nbeal2−/− mice; hypoxia-induced RVH was delayed in Nbeal2−/− mice. (A) RVSP increased in hypoxic WT and Nbeal2−/− mice at days 21 and 35. At day 21, the hypoxia-induced elevation in RVSP was increased in Nbeal2−/− compared with WT mice. (B) At day 21, hypoxia-induced RVH was prevented in Nbeal2−/− mice. At day 35, hypoxia-induced RVH was similar between WT and Nbeal2−/− mice. Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. NMX, normoxia.
Hypoxia-induced increase in RVSPs was similar in WT and Nbeal2−/− mice; hypoxia-induced RVH was delayed in Nbeal2−/− mice. (A) RVSP increased in hypoxic WT and Nbeal2−/− mice at days 21 and 35. At day 21, the hypoxia-induced elevation in RVSP was increased in Nbeal2−/− compared with WT mice. (B) At day 21, hypoxia-induced RVH was prevented in Nbeal2−/− mice. At day 35, hypoxia-induced RVH was similar between WT and Nbeal2−/− mice. Statistics: P ≤.05 by 2-way ANOVA with Tukey post hoc analysis. Comparisons: (∗) WT compared with WT baseline, (#) Nbeal2−/− compared with Nbeal2−/− baseline, (^) WT compared with Nbeal2−/−. NMX, normoxia.
Discussion
Circulating platelets are activated, and thrombosis is commonly observed in the lungs of patients with PH.25-29,72,73 Platelet activation leads to the release of α-granule–stored chemokines, many of which are implicated in the pathogenesis of PH. α-Granule formation is impaired in Nbeal2−/− mice, resulting in impaired storage of proteins within α-granules and impaired release of these proteins.45,47,67 We hypothesized that hypoxia-induced secretion of platelet α-granule–stored proteins would be prevented in Nbeal2−/− mice; therefore, these mice would be protected against the development of hypoxia-induced PVR and PH. We were surprised to find that although attenuated compared with WT mice, hypoxia-induced release of platelet α-granule–stored proteins, PF4 and P-selectin, still occurred in Nbeal2−/− mice. We observed that hypoxia-induced increase in lung PF4 and accumulation of lung platelets was delayed in Nbeal2−/− mice. Finally, although PVR and PH at day 21 were attenuated, Nbeal2−/− mice were not protected against hypoxia-induced PVR or PH at day 35.
In short-term infectious models, platelet surface expression and release of α-granule proteins are prevented or significantly reduced in Nbeal2−/− mice.45,47,54,67 Similar to others, we found that the α-granule protein, platelet PF4, is decreased in Nbeal2−/− compared with WT mice. Our first major finding is that the release of platelet α-granule–stored proteins, PF4 and P-selectin, and activation of αIIBβ3 is attenuated in hypoxic Nbeal2−/− compared with WT mice. To our knowledge, this is the first report of the effects of chronic hypoxia on the release of platelet α-granule–stored proteins and platelet activation in Nbeal2−/− mice. Circulating PLAs are increased in patients with PH.27 Hypoxia-induced increase in PLAs was prevented in Nbeal2−/− mice. We speculate that this finding, in addition to the attenuation of platelet activation and increase in proinflammatory α-granule proteins, such as PF4, may contribute to the delayed PH phenotype in Nbeal2−/− mice.
The Nbeal2 gene is required for α-granule maturation and the packaging of α-granule proteins in megakaryocytes, platelets, and other α-granule–containing immune cells.35,47,50,52-54 Similar to recent publications, we found that this mutation also affected circulating immune cells.45,47,53,54 We observed that the percentage of circulating neutrophils and monocytes was increased at baseline in Nbeal2−/− compared with WT mice and that the percentage of monocytes decreased in Nbeal2−/− mice at day 21 compared with baseline and day 3. Although the relevance of these findings is unclear, it is well described that neutrophils and monocytes are critical in the development of experimental PH. As such, our results may be affected by aberrant α-granule formation in neutrophils and monocytes in Nbeal2−/− mice. Neutrophils participate in early inflammation in hypoxic PH and exacerbate PVR and hemodynamic dysfunction.74,75 Similarly, blood-borne monocytes are recruited to the lungs during early inflammation and differentiate into pulmonary IMs.76 Accumulation of pulmonary IMs is associated with muscularization of small pulmonary arteries and increased disease severity.71,76 The impact of this mutation on leukocyte populations at baseline and in hypoxia provides new insight into the phenotype of these mice, which is necessary to understand when investigating the role of α-granule proteins in disease pathogenesis.
Hypoxia-induced increase in lung PF4 and lung platelet accumulation was delayed yet persisted in Nbeal2−/− mice. PF4 is 1 of the most abundant platelet α-granule–stored chemokines and is increased in patients with PH.77 We were interested in determining the source of lung PF4 because it has recently been reported that PF4 may be expressed by mast cells, monocytes, macrophages, and T cells, particularly in inflammatory conditions.38,44,78 Qualitatively, we observed peak hypoxia-induced Pf4 mRNA expression at day 3 in WT mice and day 14 in Nbeal2−/− mice and found that several cell types within the lung express Pf4 mRNA, including megakaryocytes, leukocytes, and others. Although we observed expression of Pf4 mRNA in several lung cell types, we found that lung PF4 protein expression was either extracellular or restricted to platelets and megakaryocytes. We speculate that the increase in lung PF4 in hypoxic WT and Nbeal2−/− mice is secondary to the accumulation of lung platelets and/or release after platelet activation, which may occur in the circulation or within the lung. The contribution of resident lung megakaryocytes to the increase in lung PF4 and the accumulation of lung platelets is unclear and will be explored further in future studies.
In addition to examining lung platelets and PF4, we examined lung IM accumulation. The accumulation of pulmonary IMs, which undergo reprogramming to drive inflammation and resolution, is critical to the progression of PVR and inflammation in PH.7,10,79-81 We previously demonstrated that platelet depletion prevents hypoxia-induced increase in pulmonary IMs.14,20,37,81 Interestingly, at baseline, total lung IMs and IM2 and IM3 subsets were increased in Nbeal2−/− mice compared with WT mice, although hypoxia did not increase IM accumulation as seen in WT mice. It is unclear whether the observed difference in baseline lung IM number is a direct result of the Nbeal2 mutation in macrophages or an indirect effect of this mutation within platelets, neutrophils, or monocytes, or secondary to changes in the bone marrow affecting myeloid precursors. We speculate that changes in myeloid precursors may drive the increase in lung IMs. This is supported by prior studies revealing that the bone marrow is altered in Nbeal2−/− mice and our observation that circulating monocytes are increased at baseline in Nbeal2−/− mice. Furthermore, although we measured the number of lung IMs, we recognize that further differentiation of these macrophages may reveal alterations in phenotype or dysregulation of proinflammatory and/or proresolution macrophage functions depending on genotype and/or hypoxia exposure.82,83 Because pulmonary IMs undergo a shift to a proresolving phenotype in chronically hypoxic WT mice, it is possible that this switch does not occur in these mice, contributing to the development of PH.69,84,85
Our final series of observations demonstrated that the Nbeal2−/− mice were not protected against late PH. This observation was consistent with the abovementioned observation of platelet activation and increased lung PF4 and platelets in these mice. PF4 is an important modulator of vascular smooth muscle cell injury, increasing both local inflammation and cell proliferation. Proliferation and muscularization of pulmonary artery small- and medium-sized vessels leads to medial wall thickening, increased pulmonary resistance, and RVH.1,86,87 We were particularly intrigued by the time course of PH, with attenuation of hypoxia-induced PH at 21 days in Nbeal2−/− mice but eventually development of PH similar to WT mice. This observation supports the premise that the disease response in these mice might be attenuated acutely in disease processes, as reported in models of ischemic stroke and dermal injury, but in this chronic model of PH, disease progression continued in these mice. Overall, these data indicate that these mice were not protected against PH and provide further evidence to support a role for platelets and PF4 in the development of experimental PVR and PH.
In summary, this study provides new insight into the Nbeal2−/− mouse strain’s phenotype and highlights several novel findings. Our main observations were that the defect in α-granule maturation and packaging of α-granule proteins in Nbeal2−/− mice attenuated hypoxia-induced release of platelet α-granule–stored proteins, PF4 and P-selectin, and activation of αIIBβ3 and delayed lung platelet accumulation. Although platelet activation was attenuated, and lung platelet accumulation and PVR and RVH were delayed, ultimately, Nbeal2−/− mice were not protected from the development of chronic hypoxic PVR and PH. This mutation affected not only platelet numbers but also circulating monocytes and neutrophils, as well as lung IMs. Monocytes, neutrophils, and lung IMs are critical in the development of experimental PH. Collectively, although our results may be affected by quantitative and qualitative changes in cells other than platelets, we gained further support for the role of platelets and α-granule proteins, such as PF4, in PH progression and pathogenesis and made several observations that expand our understanding of α-granule–deficient mice in chronic hypoxia.
Acknowledgments
The authors thank the Human Immune Monitoring Shared Resource Core (RRID: SCR_021985), Neuro Technology Center Advanced Light Microscopy Resource Core, and the Cancer Center Flow Cytometry Shared Resource Core (RRID: SCR_022035) for their expert assistance in immunofluorescent imaging and flow cytometry–generated research.
Grant support for this study was received from the American Heart Association Career Development Award (23CDA1045594 [C.V.L.]), the International Society of Hemostasis and Thrombosis Fundamental Research Career Development Award (F.D.), NIH National Heart, Lung, and Blood Institute (1P01HL152961-01 [K.S.], 1R35HL139726 [E.S.N.], and 1P01HL152961-01 [C.D.]). This work was supported by the University of Colorado Human Immunology and Immunotherapy Initiative, the University of Colorado NeuroTechnology Center (supported, in part, by Rocky Mountain Neurological Disorders core grant [P30NS048154], and by a Diabetes Research Center grant [P30DK116073]), and the University of Colorado Cancer Center (P30CA046934).
Authorship
Contribution: J.N.P., C.D., and E.S.N. conceptualized the study; J.N.P., C.D., E.S.N. F.D., and D.I. were responsible for study methodology; J.D.P., M.J., D.S, and C.V.L. were responsible for formal analysis; J.D.P., M.J., C.V.L., and C.D performed investigation; J.D.P. and D.I. provided resources; J.N.P., M.J., C.V.L., E.D. C.D., and E.S.N. were responsible for data curation; J.D.P. prepared the original manuscript draft; J.D.P., C.V.L., C.S., D.I., C.D., E.S.N, and K.S. reviewed and edited the manuscript; C.D., E.S.N., and K.S. supervised the study; C.D. and E.S.N. were responsible for project administration; C.D., E.S.N., and K.S. were responsible for funding acquisition; and all authors have read and agreed to the published version of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Cassidy Delaney, Department of Pediatrics, Section of Neonatology, University of Colorado Anschutz Medical Campus, 13243 E 23rd Ave, Aurora, CO 80045; email: cassidy.delaney@childrenscolorado.org.
References
Author notes
Original data are available on request from the corresponding author, Cassidy Delaney (cassidy.delaney@childrenscolorado.org).
The full-text version of this article contains a data supplement.