Abstract
The success of allogeneic stem cell transplantation has demonstrated the potential for immunotherapy to treat acute myeloid leukemia (AML). Although alternative T-cell-based immunotherapies have shown efficacy, they also pose the risk of on-target off-leukemia hematotoxicity. So far, adoptive autologous or allogeneic chimeric antigen receptor (CAR) T/natural killer cell therapy is almost exclusively employed as a bridge-to-transplant strategy in the context of clinical trials. For now, clinical trials predominantly target lineage-restricted antigens, but emerging approaches focus on leukemia-associated/specific intracellular target antigens, including dual and split targeting strategies. Adapter CAR T cells and T-cell-recruiting bispecific antibodies offer transient exposure with enhanced safety and multitargeting potential against antigen-escape variants. However, these have yet to demonstrate sustained responses and should be used earlier to treat low leukemia burden, preferably if measurable residual disease is present. To address immune dysregulation and enhance T-cell fitness, novel CAR T and bispecific designs, along with combinatorial strategies, might prove essential. Furthermore, genetic associations with inflammatory bone marrow signatures suggest the need for tailored platforms in defined AML subtypes. The eagerly anticipated results of trials investigating magrolimab, an anti-CD47 antibody targeting the “do not eat me” signal in p53-mutated AML, should shed further light on the potential of these evolving immunotherapeutic approaches.
Learning Objectives
Learn about the different categories of AML antigens for immunotherapeutic targeting and recognize their relevance for the unique challenges confronted in AML
Review the current landscape of immunotherapy platforms for AML in adults
Understand the barriers to the successful development of AML immunotherapy
CLINICAL CASE
A 64-year-old male patient with European Leukemia Net (ELN) intermediate risk, fms like tyrosine kinase 3 (FLT3) and nucleophosmin 1 (NPM1) gene wildtype (wt), de novo acute myeloid leukemia (AML) received “7 + 3” intensive induction therapy and achieved complete remission. The patient received 1 cycle of high-dose cytarabine consolidation therapy. A measurable residual disease (MRD) assessment by multiparameter flow cytometry revealed persistent disease at 0.2%. The patient was evaluated as eligible for transplantation from 2 suitable haploidentical children. The patient received a Reduced-intensity conditioning (RIC) regimen, followed by a haploidentical stem cell transplant (T-cell replete/post-transplantation cyclophosphamide [PTCy]), and went into MRD-negative complete remission with full donor chimerism. Unfortunately, the patient relapsed 6 months later and was enrolled in a phase 1 clinical trial of autologous CD33-redirected chimeric antigen receptor (CAR) T-cell immunotherapy with a backup graft from the initial stem cell donor. He successfully underwent T-cell apheresis and manufacturing, and entered complete response with incomplete cell recovery (CRi) 30 days after CD33 CAR T-cell infusion with antecedent lymphodepletion.
Introduction
Patients with relapsed or refractory (R/R) AML have poor prognoses, and their treatment remains challenging. In the majority of patients, allogeneic hematopoietic stem cell transplantation (allo-SCT) represents the only curative approach, albeit with limited long-term benefit (<15% 3-year overall survival [OS]).1,2 Even in patients in first remission, the risk of relapse after allo-SCT is markedly increased in those with detectable MRD prior to allo-SCT (eg, 2-year cumulative incidence of relapse 41% in MRD-positive vs 20% in MRD-negative patients), although reported results vary.1,3-5 Importantly, disease biology is associated with MRD status and significantly impacts outcome after allo-SCT: for example, patients with an adverse genetic risk profile, such as TP53 mutation/deletion, do poorly after allo-SCT (2-year OS: 28%).6 Hence, for patients with an increased risk of relapse, either determined by genetics upon initial diagnosis or MRD positivity throughout the course of the disease, as demonstrated by our clinical case, there is a high medical need for further treatment strategies prior to or after allo-SCT. In addition, despite the feasibility of allo-SCT in an increasing number of older patients, the majority of patients with AML are ineligible for allo-SCT (of an estimated 20 380 new cases of AML in 2020, only 3373 patients (16%) received a stem cell transplant).7 Importantly, non-Hispanic Black patients have the lowest allo-SCT rate compared to Hispanic patients and non-Hispanic White patients highlighting disparaties in access to allo-SCT.8
Alternative nontransplant immunotherapy approaches are therefore needed and aim to elicit immune responses (often T-cell mediated) against AML cells. This encompasses, on the one hand, therapies that either reactivate (immune checkpoint inhibition) or transfer (donor lymphocyte infusions [DLI] after allo-SCT) existing immune responses. On the other hand, many novel immunotherapeutic approaches, such as T-cell-engaging antibodies or CAR T cells, are often directed against self-antigen targets that are not immunogenic per se due to their expression and/or function in healthy tissues. Furthermore, and in contrast to the successful immunotherapy platforms implemented in B-cell precursor acute lymphoblastic leukemia (eg, antibody–drug conjugates [ADCs], T-cell bispecifics, and CAR T cells), translation to the myeloid setting is further challenged by an immune-dysregulated tumor microenvironment (TME) consisting of dysfunctional T cells enriched with regulatory T cells, accumulated myeloid-derived suppressor cells (MDSCs), immunosuppressive cytokines (eg, transforming growth factor β [TGF-β], interleukin [IL]-10), soluble inhibitory factors (eg, soluble Fas ligand [sFasL], soluble tumor necrosis factor related apoptosis inducing ligand [sTRAIL], and AML cells expressing indoleamine 2,3-dioxygenase, inhibitory checkpoint molecules, as well as a downregulated antigen-presentation machinery.9 Considering the biologic, phenotypic, and genetic heterogeneity of AML in adults and AML's impact on the leukemia microenvironment, including T-cell fitness, it is likely that various immunotherapy platforms need to be developed to improve outcomes.10,11 Here, we review the various emerging immunotherapy platforms and summarize some of the early clinical trial data. Current challenges in development and optimal time points for application will be addressed.
Target antigens
In stark contrast to B-cell malignancies, in which the B-lineage-restricted antigens CD19, CD20, and CD22 have successfully been targeted by various immunotherapy platforms, the ideal target antigen for AML is still to be identified. Such an antigen would be strongly expressed not only on every AML cell in each individual AML case, but also on leukemic stem cells (LSCs) with self-renewal capacities. In addition, healthy tissues would not express the antigen, thus avoiding on-target off-leukemia toxicity. Finally, ideal targets would also be involved in leukemogenesis, and downregulation of the target antigen would lead to a survival disadvantage for the individual cell.
Target antigens usually derive from one of the following categories (Table 1, adapted from Daver et al12 ):
lineage-restricted antigens: antigens confined to the myeloid lineage
leukemia-associated antigens: mostly non-lineage-specific antigens frequently overexpressed on AMLs cells relative to healthy tissue
leukemia-specific antigens: neoantigens resulting from leukemia-specific mutations, usually expressed intracellularly and presented in the context of HLA molecules.
Currently, the majority of immunotherapy approaches in AML target myeloid-lineage antigens (mainly CD33, CD123, and CLL-1), which are hardly ideal targets.13 As AML derives from myeloid progenitor cells, many potential target antigens for immunotherapy are also expressed on hematopoietic stem cells, imparting the risk of long-lasting or even permanent myelosuppression. This leads to the more common application of CAR T-cell therapy to patients who are also candidates for subsequent allo-SCT. Combinatorial targeting is one way to circumvent toxicity against vital tissues, but can increase the risk of immune escape (when used in an AND-gate strategy). Logic-gating strategies make use of differential expression patterns of several targets by employing, for example, AND, OR, or NOT gates for CAR T-cell activation,14 and first-in-human clinical trials using such strategies started enrollment in 2023. Other platforms, including T-cell- recruiting bispecifics and adapter CAR T cells, offer the possibility to switch cytotoxicity on and off and hence hold the promise to mitigate ongoing myelotoxicity. However, these platforms still face the challenge that the majority of myeloid lineage antigens are internalized, contributing to an “antigen sink” effect, which might ultimately impair their efficacy. Finally, the role of the targeted myeloid antigens in inducing immune-related adverse events, particularly cytokine release syndrome (CRS), is not fully understood. Their potential contribution to the occurrence and severity of CRS might necessitate the extensive step-up dosing regimens used in the early clinical trials using bispecifics.
Hence, there is still a great medical need to identify novel AML-specific target antigens, or, alternatively, adapt targeting approaches so that flexible multitargeting can overcome target antigen–related toxicities and, at the same time, counteract immune escape. Multiomic approaches15,16 have identified novel AML-associated surface antigens, and clinical trials targeting such target candidates have recently been initiated. Moreover, a plethora of possible novel target antigens will be derived from intracellular proteins that are presented in the context of defined human leukocyte antigen (HLA)-molecules. These include leukemia-specific neoepitopes of recurrent mutations in genes such as NPM1 or TP53, which could be targeted by T-cell receptor (TCR)-mimic bispecifics or TCR-transgenic T cells.17
Antibody–drug conjugates, antibodies, and bispecific antibodies
The only immunotherapeutic agent approved for AML thus far is gemtuzumab ozogamicin (GO), an ADC administered in combination with daunorubicin and cytarabine for the treatment of newly diagnosed CD33-positive AML, with most significant activity in AML patients in the ELN-favorable risk group. However, its efficacy as a monotherapy in the R/R setting is limited (only the FDA approved its use in R/R). In addition to GO, ADCs directed against CD33 and CD123 and carrying alternative payloads are under investigation (eg, IMGN632 [pivekimab sunirine], a CD123-targeting ADC with a DNA-alkylating payload). In patients with blastic plasmacytoid dendritic cell neoplasia, tagraxofusp—a fusion molecule of IL-3, which targets CD123, with a truncated form of diphteria toxin—was recently approved. Several clinical trials are currently testing the safety, tolerability, and efficacy of tagraxofusp and related ADCs in combination with standard chemotherapy or hypomethylating agents (HMAs) in de novo and in relapsed AML patients.18 Evolving data suggest that combination strategies are especially advantageous for patients eligible for allo-SCT, as they are associated with a greater likelihood of achieving MRD negativity and, consequently, a reduced risk of relapse after allo-SCT. Such approaches could have been advantageous in the context of the case we have presented in this article.
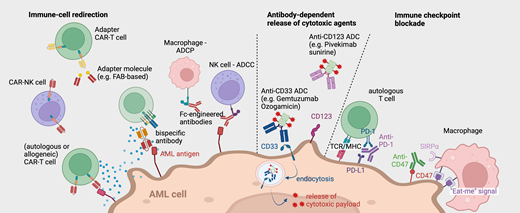
Mode of action of the different immunotherapy platforms in AML. BsAb, bispecific antibody.
Mode of action of the different immunotherapy platforms in AML. BsAb, bispecific antibody.
Other monoclonal antibodies include naked antibodies directed against CD33 (eg, lintuzumab), CD123 (eg, talacotuzumab), and CD70 (eg, cusatuzumab). Cusatuzumab was administered in conjunction with azacitidine to patients with newly diagnosed AML ineligible for intensive chemotherapy. Nineteen of 38 patients achieved an objective response and 14/38 a complete response with a median duration of response of 4.5 months and median OS of 11.5 months.19 In contrast to monoclonal antibodies, bispecific antibodies are recombinant proteins that recruit T cells through CD3 engagement and bind to target antigens on tumor cells, typically with higher affinity. Many different formats of bispecific antibodies have been developed, such as bispecific T-cell engagers (BiTEs), half-life-extended BiTEs, dual-affinity retargeting (DART) proteins, tandem diabodies, DuoBody, affinity-tailored adaptors for T cells, and tetravalent bispecific antibodies.12 Despite differences in pharmacokinetics due to molecule size, it remains unclear to what extent the different formats impact efficacy and toxicity. Validated by GO, CD33 is one of the antigens most commonly targeted by the bispecifics. Sixty patients with highly advanced AML (48/60 had >4 prior lines of treatment, 27/60 had prior allo-HSCT) were treated with a CD33-directed BiTE (AMG 330, continuous intravenous infusion [CIV]) in a ramp-up approach together with dexamethasone prophylaxis in order to manage cytokine release syndrome (CRS). The complete response (CR)/CRi rate was 17% with doses ≥120 µg/day and the median duration of response was 58.5 days. Responders were more likely to have higher AMG 330 exposure and lower baseline leukemic burden, with no correlation between CD33 expression on AML blasts and response.20 Instead of a continuous application, AMG 673, a half-life-extended CD33-BiTE construct (fusion of the N terminus of a single-chain IgG Fc region), is given as a short-term infusion (1 h) twice weekly. A reduction in blasts was observed in 16/38 patients, however none of the responses were sustainable. Neither of these trials nor those of related drugs (eg, JNJ-67571244, AMG 427) were able to achieve the projected exposure level within a reasonable timeframe, partly due to integration of multiple dose steps and trial interruptions to mitigate immune-related toxicity as well as hematotoxicity.
Flotetuzumab is the most advanced investigational bispecific antibody directed against CD123. This DART molecule is given as continuous infusion and was tested in a phase 1/2 trial in R/R AML. Notably, within the refractory/early relapse patient cohort, flotetzumab achieved an objective response rate (ORR) of 30%, including a CR rate of 24% and MRD negativity in 21%. This was correlated to higher density of CD123 receptors on AML blasts, higher inflammatory chemokine signature score, and higher interferon gamma signaling gene expression score compared to patients with late AML relapse.21 Other bispecific constructs of different design targeting various antigens (eg, FLT3, CLL-1, CD70) are being tested in early clinical trials (Table 2). In addition, bifunctional and trifunctional natural killer (NK)-cell engagers are currently in the early stages of clinical development. Preliminary data from a phase 1/2 trial involving patients with R/R AML showed promising results for SAR’579, a novel CD123 targeting and coengaging NKp46 and CD16a trifunctional NK-cell engager. That trial enrolled 23 patients across 6 dose levels and demonstrated an excellent safety profile.22 Across the various clinical trials using bispecifics, reasonable safety data have been obtained for integrating prophylactic anti-inflammatory drugs (eg, tocilizumab, corticosteroids) and step-up dosing. Combinatorial approaches might improve the efficacy of bispecifics by modulating TME and boosting T-cell function.23 The strategies that are currently used in this as well as other disease entities include the following: application of 2 bispecifics, one of which provides a positive costimulatory molecule (thereby mimicking second-generation CAR T); small-molecule modulators of T-cell function (eg, bruton tyrosine kinase [BTK] inhibitors, immunomodulatory drugs [IMiDs], B-cell lymphoma 2 [bcl-2] inhibitors); conditional bispecifics that are only active within the TME24 ; combination with AML-approved drugs (eg, HMAs); integration into the conditioning regimen prior to and in conjunction with DLI after allo-SCT. Importantly, bispecifics appear to work best at an earlier line of therapy in a low disease burden/MRD+ disease setting. In the future, it will be necessary to assess the optimal clinical context for bispecifics. This could involve their use as a transitional strategy to eradicate MRD before allo-SCT, as illustrated in our case, or potentially in conjunction with DLIs to preempt an impending relapse after allo-SCT. Another potential application is as a standalone treatment approach following cytoreductive treatment to eliminate refractory disease.
Results of early clinical trials on bispecific antibodies for AML
| Clinical trial no. . | Target . | Construct design . | Dosing . | Safety . | Efficacy (CR/CRi) . | No. patients treated to date . | Enrollment stage . |
|---|---|---|---|---|---|---|---|
| NCT02520427 | CD33 | BiTE | 0.5-720 µg/day; 0-3 dose steps; 14-28 days CIV | CRS 67% (≥G3 13%) | 7/42 | 96 | Terminated |
| NCT03224819 | CD33 | HLE-BiTE | 0.05-72 µg per dose, 2 IV infusions in 14 days | CRS 50% (≥G3 13%) | 1/27 | 46 | Terminated |
| NCT03144245 | CD33 | TandAb | 0.5-300 µg/day; 14 days CIV; 28-day cycle | CRS NA (≥G3 0%) | 2/35 | 53 | Completed |
| NCT02152956 | CD123 | DART | RP2D: 500 ng/kg/day; 7 dose steps; 28 days CIV; then 4 days/week | CRS 50% (≥G3 7%) | 8/30 | 246 | Terminated |
| NCT02715011 | CD123 | DuoBody | 0.6-6 µg/kg Q2W IV; 0.15-4.8 µg/kg twice weekly IV; 2.4-4.8 µg/kg twice weekly SC; 0-4 dose steps | CRS 44% (≥G3 15%) | 0/62 | 62 | Completed |
| NCT02730312/ NCT05285813 | CD123 | XmAb | 1.7 µg/kg IV; 4 dose steps on days 1, 3, 5, and 8 followed by weekly administration | CRS 44% (≥G3 15%) | 5/51 | 106 | Dose finding completed/ phase 2 initiated |
| NCT05086315 | CD123 | Trifunctional NK cell engager | 10-3000 µg/kg/dose in cycle 1; 100-3000 µg/kg QW for the rest of induction cycles | CRS 9% (≥G3 n.r.), IRR 43% | 3/23 | 23 | Recruiting |
| NCT03038230 | CLL1 | Biclonics IgG format | 0.675-240 mg weekly after initial ramp-up dosing; 3-4 dose steps | CRS 36% (≥G3 9%) | 0/58 | 62 | Active, not recruiting |
| Clinical trial no. . | Target . | Construct design . | Dosing . | Safety . | Efficacy (CR/CRi) . | No. patients treated to date . | Enrollment stage . |
|---|---|---|---|---|---|---|---|
| NCT02520427 | CD33 | BiTE | 0.5-720 µg/day; 0-3 dose steps; 14-28 days CIV | CRS 67% (≥G3 13%) | 7/42 | 96 | Terminated |
| NCT03224819 | CD33 | HLE-BiTE | 0.05-72 µg per dose, 2 IV infusions in 14 days | CRS 50% (≥G3 13%) | 1/27 | 46 | Terminated |
| NCT03144245 | CD33 | TandAb | 0.5-300 µg/day; 14 days CIV; 28-day cycle | CRS NA (≥G3 0%) | 2/35 | 53 | Completed |
| NCT02152956 | CD123 | DART | RP2D: 500 ng/kg/day; 7 dose steps; 28 days CIV; then 4 days/week | CRS 50% (≥G3 7%) | 8/30 | 246 | Terminated |
| NCT02715011 | CD123 | DuoBody | 0.6-6 µg/kg Q2W IV; 0.15-4.8 µg/kg twice weekly IV; 2.4-4.8 µg/kg twice weekly SC; 0-4 dose steps | CRS 44% (≥G3 15%) | 0/62 | 62 | Completed |
| NCT02730312/ NCT05285813 | CD123 | XmAb | 1.7 µg/kg IV; 4 dose steps on days 1, 3, 5, and 8 followed by weekly administration | CRS 44% (≥G3 15%) | 5/51 | 106 | Dose finding completed/ phase 2 initiated |
| NCT05086315 | CD123 | Trifunctional NK cell engager | 10-3000 µg/kg/dose in cycle 1; 100-3000 µg/kg QW for the rest of induction cycles | CRS 9% (≥G3 n.r.), IRR 43% | 3/23 | 23 | Recruiting |
| NCT03038230 | CLL1 | Biclonics IgG format | 0.675-240 mg weekly after initial ramp-up dosing; 3-4 dose steps | CRS 36% (≥G3 9%) | 0/58 | 62 | Active, not recruiting |
IV, intravenous; IRR, infusion-related reaction; NA, not applicable; QW, once weekly; Q2W, once every 2 weeks; SC, subcutaneous injection; TandAb, tandem diabody.
Adoptive cellular therapy
Similar to bispecifics, the antigens most commonly targeted by CAR T cells in trials are CD33, CD123, and CLL-1 (Table 3).25 In contrast to the common target antigen denominators, there is great variability in cell source, CAR T-cell constructs, and study design between these clinical trials. Some of the variables include cell source (autologous versus allogeneic), heterogenous lymphodepletion regimens, cell number, manufacturing method (traditional viral vectors versus rapid manufacturing), and the modifications made to products to enhance killing and/or safety (Table 3).
For example, PRGN-3006 CAR T cells are manufactured in a decentralized and rapid process via electroporation of the Sleeping Beauty plasmid (from apheresis to patient infusion within 2 days); these cells target CD33 and have membrane-bound IL-15 incorporated into the product to enhance in vivo expansion.26 The completed phase 1 dose escalation data for PRGN-3006 CAR T cells were presented at the American Society of Hematology (ASH) annual meeting 2022. Lymphodepletion was mandatory, as expansion was significantly lower, and 0% efficacy was observed in the nonlymphodepletion cohort. In AML patients treated in the lymphodepletion cohort (n = 11), the product was very well tolerated (1 G3 CRS), and 27% of patients showed a response, although no patient with myelodysplastic syndrome (MDS) responded (0/3). However, responses were nondurable outside of a bridge to allo-SCT concept. The trial has since moved to dose expansion at 1 × 106 cells/kg and notably now incorporates a second infusion of CAR T cells in an attempt to augment efficacy.
UCART123 cells are a non-HLA-matched allogeneic product targeting CD123 that have been additionally modified to prevent graft-versus-host disease via TCRαβ elimination as well as CD52 knockout to allow for intensified lymphodepletion by alemtuzumab.27 Optimal kinetics were observed in the cohort of patients treated with fludarabine, cyclophosphamide, and alemtuzumab; this cohort is the current one in the ongoing in clinical study. Cytokine release syndrome was observed in all patients; notably, events of severity ≥G3 occurred in 24% of patients (4/17), including 2 G5 events. (G stands for grade.) Immune effector cell–associated neurotoxicity syndrome (ICANS) was rare, a finding consistent across AML CAR T studies. Early evaluation of efficacy showed responses in several patients, albeit most notably in a heavily pretreated AML patient (including prior allo-SCT) who achieved MRD-negative CR beyond 12 months without DLI/second allo-SCT at the time of data reporting. That study is also evaluating multiple dosing strategies.
To date, anti-CLL1 CAR T-cell data have been primarily collected from pediatric populations with MRD-negative responses, although the degree of hematopoietic recovery is unclear and challenges with durability exist.28,29 So far, the only adult anti-CLL1 CAR T data was from a cohort of 10 patients with a reported CR/CRi rate of 70%, although all patients but 1 were transplanted very early in the course of therapy (days 18-34). Cytokine release syndrome occurred in 100% of these patients (60% G3).30 Perhaps the most exciting results to date concern the CLL1-CD33 compound CAR, which were most recently updated at the European Hematology Association (EHA) annual meeting 2020.31 Notably, 7/9 patients achieved MRD-negative remission, with 1/2 nonresponding patients being CLL1-negative. These data support that aggressive “OR gating” strategies might be optimal for efficacy in order to cover the majority of AML blasts/LSCs and prevent antigen escape. Notably, allo-SCT appears to be required in the setting of CLL1 CAR T, secondary to a lack of neutrophil recovery.
Saar Gill and colleagues proposed a novel approach in which they initiated the treatment sequence by utilizing a CD33 clustered regularly interspaced short palindromic repeats (CRISPR)-Cas-knockout allogeneic stem cell graft followed by CD33-directed CAR T cells.32 More recently, the first-in-human trial demonstrated successful engraftment of CD33-deleted stem cells and tolerance to post-allo-SCT GO without incidence of cytopenia.33
Given the lack of a single tumor-specific antigen in AML, the development of CAR T-cell therapies that could be agnostic to phenotypic targets are of considerable interest. CYAD-01 is an autologous CAR T-cell product expressing NKG2D, the critical killing receptor of NK cells, and targets 1 of 8 ligands that are overexpressed on nearly 100% of blasts (ie, MICA/MICB and the ULBP family). In the completed phase 1 dose escalation study, 25% (3/12) of AML patients had objective responses, although these were nondurable with the exception of 1 patient bridged to allo-SCT.34 Additional efforts to enhance efficacy with CYAD-01 have been the incorporation of lymphodepletion chemotherapy as well as a second-generation product (CYAD-02) to abrogate potential fratricide in vivo via CAR T-cell upregulation of NKG2D ligands. However, a key factor in the limited efficacy with this approach is that LSCs have been shown to lack NKG2D ligands, which raises the potential for combinatorial therapy with agents that can upregulate NKG2D ligands on LSCs (eg, PARP1 inhibitors).35
As critical as T-cell dysfunction is to the impaired efficacy of CAR T cells, universal or adapter CAR T programs might be optimal in overcoming these challenges, as recently shown.36 These programs might also have therapeutic advantages by helping prevent antigen escape while ameliorating CRS/ICANS and/or on-target toxicity. Notably, we now have supportive clinical data in the recent presentation of a universal CAR T cell with a CD28 costimulatory domain (UniCAR T) and a CD123 targeting module (TM).37 Although clinical activity was modest (3/14; 2 CRi, 1 MRD-negative conversion), a G2 ICANS was reversed with TM interruption, and a reexpansion of CAR T cells was noted with re-initiation of TM. There will remain clinical obstacles with adapter programs because multiple factors require optimization, undoubtedly a challenge in the clinical investigation of AML.
Optimal timing of adoptive cellular therapy in the context of the clinical case
One paramount question in the field is how the impact of disease burden and past therapies impact the outcomes of CAR T and other cellular therapies. Given that overall efficacy, and particularly durability of response, has been low, with overall enriched responses in patients with lower blast burdens, intervention at the time of MRD positivity and prior to overt relapse is potentially critical. Notably, the T-cell fitness is also likely enhanced in MRD-positive settings, which is most relevant to autologous products. As with our patient, who ultimately relapsed after allo-SCT, the future paradigm is likely intervening at the time of MRD positivity, with the goal of improving the outcome of transplantation. Furthermore, applying T-cell based immunotherapy platforms, such as bispecific antibodies and CAR T cells, earlier in treatment lines with low disease burden should enhance their safety profiles and result in lower rates of immune-related adverse events such as CRS or macrophage activation syndrome.
Checkpoint blockade
Immune checkpoint inhibitors have been investigated in AML with the intent of harnessing components of the immune microenvironment to generate an immune response against AML.12 Single-agent immune-checkpoint-inhibitor therapies have demonstrated almost no activity in patients with R/R AML who have not undergone allo-SCT. Liu et al conducted a multicenter, randomized phase 2 study to assess the efficacy of nivolumab (every 2 weeks for up to 46 doses) as a maintenance therapy for patients with AML with CR/CRi who were not candidates for allo-SCT.38 A total of 79 patients were randomized. The primary endpoint of progression free survival (PFS) was not met, with median PFS of 13.2 versus 10.9 months (P = 0.38) and 2-year PFS rates of 30.3% versus 30% in the nivolumab and observation arms, respectively. Similarly, median OS and 2 year OS were not improved. The grade 3 adverse event rate was significantly higher in the nivolumab arm.
However, Zeidner et al conducted a phase 2 single-arm study of high-dose cytarabine followed by pembrolizumab in R/R AML, demonstrating a CR + CRi rate of 38% and median OS of 11.3 months, with patients with primary refractory/first salvage having further improved OS compared to later salvage patients (13.2 vs 11.3 months).39 The authors considered this an encouraging response rate in this R/R AML population.
Further encouragement can be taken from the results of giving the anti-CTLA4 antibody ipilimumab to patients with hematologic malignancies relapsing following allo-SCT, including 12 with AML and 2 with MDS. Among these 14 patients, CR was achieved in 5 (36%), including 4 of 5 with extramedullary disease, with all responses being observed at the higher ipilimumab dose of 10 mg/kg. Grade 3/4 immune-related adverse events (irAEs) were noted in about 20% of patients and were reported to be generally manageable.40
Hypomethylating agents upregulate genes in the interferon-gamma pathway, increase the expression of HLA class I antigens, and activate viral defense pathways, thereby promoting antitumor immune signaling; data suggest they increase the expression of PD-1 and PD-L1 on T cells via methylation.41 The anti-PD-1 antibody nivolumab was combined with azacitidine in a single-arm phase 2 study (n = 70) with an ORR of 33% (CR + CRi 22%) in a heavily pretreated population.42 A higher response rate was noted in patients with higher CD3 and CD8 T-cell levels in the blood and bone marrow prior to therapy, suggesting that to be a useful and simple biomarker for selecting patients more likely to benefit from anti-PD-1-centered approaches in future trials.43 In a subsequent phase 2 study, 129 AML patients ≥65 years old and ineligible for intensive chemotherapy were randomized (1:1) to azacitidine with durvalumab or azacitidine alone with no significant difference in CR rate (17.2% vs 21.5%) or OS (13.0 vs 14.4 months).44 Based on these data, interest in further developing HMAs with PD-1-, PD-L1-, or CTLA-4-targeted interventional trials has waned in recent years.
Novel AML- and MDS-specific checkpoints, such as TIM3 antibodies, in combination with azacitidine have entered clinical development. Although the CR rate in newly diagnosed AML was only 25% (CR + CRi: 30%) in the initial single-arm phase 1B trial, the durability of response in patients with ELN adverse-risk mutations (TP53/RUNX1/ASXL1) was encouraging (median duration of response [DOR] 12.6 months).45 In a subsequent randomized phase 2 study of azacitidine with sabatolimab or with placebo in newly diagnosed higher-risk myelodysplastic syndrome (MDS), the primary endpoint of improved CR rates and/or PFS was not reached.46 Sabatolimab is currently being investigated in an ongoing frontline randomized phase 3 trial in higher-risk MDS, and the data are eagerly awaited.
The cell-surface molecule CD47 functions as a phagocyte immune checkpoint, impeding phagocytosis and serving as a “don't eat me” signal.47 CD47 is overexpressed in most cancer cells, especially in AML and MDS, which is associated with adverse prognosis in AML.48 Azacitidine upregulates prophagocytic signals on AML cells and was combined with magrolimab (a first-in-class humanized monoclonal antibody against CD47) in a recently completed phase 1B study in patients with previously untreated AML or higher-risk MDS. A total of 87 patients were treated in the AML cohort, with an unplanned enrichment of patients with TP53 mutations (TP53m; n = 72); the unplanned enrichment was initiated because azacitidine and magrolimab showed encouraging signs of early efficacy in TP53m patients, emerging data show the limited benefit of adding venetoclax in TP53m frontline AML, and an extremely high but unmet need exits for effective therapies in this population.49 The combination of azacitidine and magrolimab was well tolerated. The most significant adverse event was on-target anemia, seen predominantly in the first 7-10 days, postulated to be due to the removal of CD47 antibody-coated sensescent red blood cells by the reticuloendothelial system. The median hemoglobin drop from baseline to first postdose magrolimab assessment was −0.9 g/dL (range: −3.6 to 2.5 g/dL); however, it is important to note that 5%-6% of patients could have a >2.0 g/dL early drop in hemoglobin, and close monitoring and transfusion to try to maintain hemoglobin at >8.5-9.0 g/dL during the first 7-10 days of therapy is strongly recommended. The ORR and CR rates in the 15 TP53wt patients were 47% and 33%, respectively, and, interestingly, these were similar (47% and 32%) in the 72 TP53m patients. The median DOR in TP53m patients was about 8 months and median OS was around 11 months. These CR, CR + CRi rates, and median OS intervals appear favorable compared to those recently published with azacitidine and venetoclax50 and to those in a retrospective observational study of outcomes in frontline TP53m AML patients. Badar et al. looked at patients treated with 6 different frontline induction regimens in 291 patients across 8 US academic centers between 2012 and 2021.51 The outcomes were unfortunately dismal, with an overall CR + CRi rate of 29%, ranging from 17% to 41%, and with HMA + venetoclax showing the highest CR + CRi rate, 41%. The median OS ranged from 2.0 to 9.5 months, with allo-SCT being the only factor associated with improved OS in a multivariate analysis. By analyzing survival outcomes for TP53m AML over time, the authors observed a maintained but modest response to induction therapy and poor OS that has not improved in the era of novel therapies. The combination of azacitidine and magrolimab might be a step forward, but the phase 1B results require confirmation in a randomized study. Interestingly, in that study, the median OS was 18.2 months in the 15 TP53wt patients, suggesting magrolimab is also active in TP53wt patients. Another single-arm study combining azacitidine with venetoclax and magrolimab was initiated based on observed preclinical synergy of this triplet beyond either doublet in both TP53m and TP53wt patient-derived xenograft (PDX) models.52 The ongoing phase 1B study has demonstrated encouraging response rates; CR + CRi rates were about 60% in TP53m and about 80% in TP53wt, and median OS was not reached in either population.53 Based on these results, 2 phase 3 trials are currently enrolling: one with azacitidine with magrolimab in patients with newly diagnosed TP53-mutant AML (ENHANCE-2; NCT04778397) and another with azacitidine and venetoclax with magrolimab in patients with newly diagnosed AML who are ineligible for intensive chemotherapy (ENHANCE-3; NCT05079230). These results are eagerly awaited.
Conclusions
Adoptive cellular therapy: bridge to transplant and beyond
Allogeneic stem cell transplantation has proven effective against AML, harnessing the graft-versus-leukemia effect to eliminate residual disease. Similarly, CAR T-cell therapy has shown promise, as exemplified by the induction of bone marrow aplasia, indicating highly potent antileukemic activity. The most recent developments appear to recapitulate these responses utilizing allogeneic T cell or NK cell sources. Current challenges revolve around identifying suitable target antigens to elevate these T and NK cell–based platforms beyond a bridge-to-transplant strategy. Besides the obvious on-target off-leukemia toxicity of the healthy hematopoietic compartment and possible other vital tissues, targeting myeloid lineage antigens might contribute to antigen sink, aggravation of CRS and possibly, through continuous antigen exposure, T-cell exhaustion. Given the interindividual and intraindividual heterogeneity of genotypes and phenotypes in AML, antigen-escape variants appear likely. Future strategies to combat AML need to consider multiple targeting, ideally based on the phenotypic profile of the AML of each individual patient.
Bispecifics: moving to earlier treatment lines in low disease burden, preferentially MRD-positive
T-cell-recruiting bispecific antibodies offer the considerable advantages of being off-the-shelf products with increased safety profiles as a result of on–off switchable cytotoxicity. However, despite that and their success against B-cell malignancies, bispecifics have so far failed to deliver sustainable responses in AML. The bispecific antibody platform relies on endogenous T-cell fitness, and hence the interpretation of clinical results must take into account that clinical trials were conducted on patients with advanced AML and high disease burdens after several prior treatment lines. Hence, not surprisingly, and in line with poor outcomes after allo-SCT upon relapse, responses so far have been limited. We propose to promote this therapeutic platform to a first-line therapy in genetically high-risk patients, preferentially in MRD-positive patients, in a clinical scenario involving a less immunosuppressive leukemic microenvironment, better T-cell fitness, and favorable effector:target ratio. In line with this approach are current clinical trials utilizing targeting or adapter molecules in conjunction with universal CAR T cells or DLI to combine the safety and flexibility of bispecific antibodies with a functional adoptive cell therapy.
Tailoring immunotherapy to AML phenotype, AML genotype, and bone marrow inflammatory signature: combination strategies to overcome adversity
Finally, learning the lessons from allo-SCT, we need to particularly adopt combinatorial approaches to improve T-cell function and counteract immune dysregulation. Early clinical trials utilizing additional bispecifics that provide positive costimulation (eg, CD20 x CD3 plus a CD28 x CD3 or 4-IBB x CD3) are currently recruiting participants with B-cell malignancies; other approaches might encompass sequential or in-parallel application of small molecules such as tyrosine kinase inhibitors (TKI), venetoclax–azacitidine, IMiDs, or BTK inhibitors. Recent evidence demonstrates the association between genetics and the inflammatory signature of the bone marrow,54 supporting the notion that defined immunotherapy platforms might only be successful in certain AML subtypes.55 We eagerly await the results of randomized clinical trials (currently recruiting) in TP53-mutated AML that will utilize the anti-CD47 antibody magrolimab; this approach might be better suited against AML subtypes less susceptible to T cell–based immunotherapy. Ideally, applied immunotherapy strategies integrate synthetic and natural immunity leading to innate but, importantly, adaptive immunity.56 In the future, immunotherapy platforms should be applied early in a treatment line; should be based on AML genotype and phenotype, and the inflammatory signature of the bone marrow; and should guide the use of single or combinatorial approaches.
Conflict-of-interest disclosure
Marion Subklewe: research funding from Amgen, Bristol Myers Squibb, Gilead, Janssen, Miltenyi Biotec, MorphoSys, Novartis, Roche, Seagen, and Takeda; travel support from Celgene, Gilead, Pfizer, and Takeda; consultant/scientific advisory board member at Autolus, AvenCell, CanCell Therapeutics, CDR-Life, Genmab US, Ichnos Sciences, Incyte Biosciences, Interius BioTherapeutics, Janssen, Millennium Pharmaceuticals, Miltenyi Biomedicine, Molecular Partners, Nektar Therapeutics, Novartis, Pfizer, Ridgeline Discovery, and Takeda; speakers' bureau at Amgen, AstraZeneca, Bristol Myers Squibb/Celgene, Gilead, GSK, Janssen, Novartis, Octapharma, Pfizer, Roche, Springer Healthcare, and Takeda.
Veit Bücklein: research funding from Bristol Myers Squibb, Celgene, Kite/Gilead, Novartis, and Takeda; honoraria from Amgen, Bristol Myers Squibb, Kite/Gilead, Novartis, Pfizer, and Roche; travel support from Amgen, Pfizer, and Pierre Fabré; consultant to Amgen, Kite/Gilead, Pfizer, and Priothera.
David Sallman: research funding from Aprea; advisory board member at AbbVie, Agios, Aprea, Bristol Myers Squibb, Intellia, Kite, Novartis, Shattuck Labs, and Syndax; consultant to Magenta, Novartis, and Takeda; speakers' bureau at Bristol Myers Squibb and Incyte.
Naval Daver: research funding from AbbVie, Amgen, Astellas, Bristol Myers Squibb, Fate Therapeutics, Genentech, Gilead, Glycomimetics, Hanmi, ImmunoGen, Kite, Novimmune, Pfizer, Servier, Trillium, and Trovagene; consultant to AbbVie, Agios, Amgen, Arog, Astellas, Bristol Myers Squibb, Celgene, Daichii– Sankyo, Genentech, Gilead, ImmunoGen, Jazz, Kite, Novartis, Pfizer, Servier, Shattuck Labs, Stemline/Menarini, Syndax, and Trillium.
Off-label drug use
Marion Subklewe: nothing to disclose.
Veit Bücklein: nothing to disclose.
David Sallman: nothing to disclose.
Naval Daver: nothing to disclose.