Abstract
The granulocyte-macrophage colony-stimulating factor (GM-CSF) analog E21R binds to the GM-CSF receptor complex with low affinity and acts as a competitive antagonist. In addition, it has been reported to be a potent direct activator of apoptosis in primary human acute myeloid leukemia (AML) cells. We have confirmed the ability of E21R to neutralize the biologic effects of GM-CSF and investigated its activity on primary AML blasts. We find that it failed to induce cell death in blast cells from 23 separate AML cases treated in vitro at concentrations of E21R up to 30 μg/mL. Significant cell death resulted in all cases after incubation with cytosine arabinoside. The lack of effect of E21R on AML blasts was unlikely to be due to an absence of functional GM-CSF receptors because 13 cases demonstrated an increase in cell number with the addition of exogenous GM-CSF. These results do not support the use of E21R for the treatment of AML. (Blood. 2004;103: 3230-3232)
Introduction
Granulocyte-macrophage colony-stimulating factor (GM-CSF) promotes the survival, proliferation, and activation of cells ranging from myeloid progenitors to terminally differentiated neutrophils.1 The effects of GM-CSF are mediated via high-affinity binding to heterodimeric cell surface receptors consisting of a GM-CSF–specific α chain and a β chain that is shared with the receptors for interleukin-3 (IL-3) and IL-5.2,3 Binding of GM-CSF to the α chain alone is of low affinity.4 The glutamate residue at position 21 of GM-CSF is involved in GM-CSF binding to both chains of the αβ complex; its substitution by arginine (GM-CSF E21R) results in a molecule capable of binding with low affinity to the α chain but with no high-affinity binding to the αβ complex.5 E21R functions as a competitive antagonist of GM-CSF binding to the αβ complex and can neutralize its biologic effects.6
In addition to its neutralizing activity, E21R is reported to directly induce apoptosis in cells expressing high-affinity GM-CSF receptors even in the absence of exogenous GM-CSF. Cells reported to undergo apoptosis include normal hemopoietic progenitors,7 primary acute myeloid leukemia (AML) blasts,7 eosinophils,8 and the Jurkat T-cell line engineered to express the high-affinity GM-CSF receptor.9 This potent proapoptotic effect was shown to be an active process requiring protein kinase activity, the synthesis of new proteins,7 and the membrane proximal portion of the intracytoplasmic domain of the common β chain to be present.9 Caspase inhibitors were shown to block the effects of E21R, whereas overexpression of Bcl-2 was ineffective.9 E21R has also been shown to have biologic effects against the survival and proliferation of juvenile myelomonocytic leukemic (JMML) cells in vitro10 and in vivo11 and has anti–chronic myelomonocytic leukemia (anti-CMML) activity demonstrated by inhibition of spontaneous colony formation in vitro.12
Functional GM-CSF receptors are found on the blast cells of most patients with AML,13-15 and exogenously added recombinant GM-CSF can stimulate proliferation of AML cells.16-21 There is evidence that autocrine production of GM-CSF may play a part in leukemia cell proliferation.22,23 The profound proapoptotic effects of E21R in all 21 cases of primary AML previously reported raised the possibility that E21R may be very useful in the treatment of AML and prompted us to investigate its effects on AML cell survival and proliferation.
Study design
Blasts were isolated from the peripheral blood or bone marrow of patients with newly diagnosed AML by standard Ficoll-Hypaque separation. Informed consent was obtained according to the Joint University College London/University College London Hospitals Ethics Committee guidelines. Samples were either used fresh or after recovery from liquid nitrogen. A total of 21 of 23 samples were thawed and incubated in RPMI/10% fetal calf serum (FCS) at 37°C/5% CO2 and underwent a further Ficoll separation to remove dead cells where necessary. Samples were incubated with varying concentrations of E21R (BresaGen, British Biotech, Oxford, United Kingdom), GM-CSF (Hoechst-Behringwerke, Marburg, Germany) (or both), or cytosine arabinoside (Sigma, St Louis, MO) at a cell density of 0.5 × 106/mL to 1 × 106/mL in RPMI/10% FCS for up to 72 hours. Cell proliferation was measured using an MTS (3-(4,5 dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl-2-(4-sulfophenyl)-2H-tetrazolium) assay (CellTiter 96, Promega, Madison, WI). The assay was performed according to manufacturer's instructions, and the results are expressed as a percentage of the absorbance reading obtained with cells incubated in RPMI/10% FCS alone. Apoptosis was measured by annexin V binding as previously described.24
Results and discussion
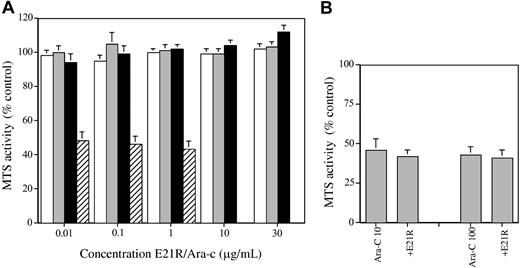
Increasing concentrations of E21R were incubated with primary blast cells from a total of 23 patients with AML. After 48 hours, surviving cells were quantified using an MTS-based assay. Figure 1 shows that there was no reduction in cell number at 48 hours at any concentration of E21R up to a maximum of 30 μg/mL. Flow cytometric assays for annexin V binding carried out at 48 hours in a subset of 10 patients also failed to show an increase in apoptosis in the presence of E21R (control, 45% ± 6% annexin-positive; E21R 10 μg/mL, 46% ± 6% positive).
Effects of E21R and cytosine arabinoside. (A) Effect of E21R or cytosine arabinoside (Ara-c) on the survival of AML blasts. Primary AML blasts were incubated with increasing concentrations of E21R for the indicated times or with increasing concentrations of cytosine arabinoside for 48 hours. Samples were assayed in duplicate by the MTS technique for viable cell number, and the data are presented as the mean ± SEM activity as a percentage of the result obtained with cells incubated without additives. For E21R, n = 15 at 24 hours (□), 23 at 48 hours (▦), and 14 at 72 hours (▪). For Ara-c, n = 21 at 48 hours (▨). (B) Effect of E21R combined with Ara-c on the survival of AML blasts. Primary AML blasts were incubated with Ara-c alone (at 10 ng/mL or 100 ng/mL) or in combination with E21R (10 μg/mL) for 48 hours and cell survival measured by MTS assay (mean ± SEM of 21 samples).
Effects of E21R and cytosine arabinoside. (A) Effect of E21R or cytosine arabinoside (Ara-c) on the survival of AML blasts. Primary AML blasts were incubated with increasing concentrations of E21R for the indicated times or with increasing concentrations of cytosine arabinoside for 48 hours. Samples were assayed in duplicate by the MTS technique for viable cell number, and the data are presented as the mean ± SEM activity as a percentage of the result obtained with cells incubated without additives. For E21R, n = 15 at 24 hours (□), 23 at 48 hours (▦), and 14 at 72 hours (▪). For Ara-c, n = 21 at 48 hours (▨). (B) Effect of E21R combined with Ara-c on the survival of AML blasts. Primary AML blasts were incubated with Ara-c alone (at 10 ng/mL or 100 ng/mL) or in combination with E21R (10 μg/mL) for 48 hours and cell survival measured by MTS assay (mean ± SEM of 21 samples).
Because of these unexpected findings, prolonged incubation for 72 hours was carried out in 14 patient samples. Again, no decrease in cell survival was detected. In addition to its lack of effect on primary AML blasts, E21R failed to induce apoptosis in HL-60 and U937 cell lines, both of which express high-affinity GM-CSF receptors.25 E21R did not affect the survival of TF-1 cells cultured in erythropoietin (Figure 2), MO7e cells cultured in stem cell factor (SCF) or thrombopoietin (data not shown), or primary human CD34+-derived cells cultured in G-CSF (Figure 2).
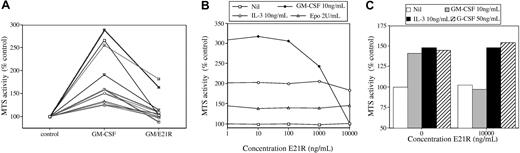
Neutralization of GM-CSF by E21R. (A) Response of AML blasts to exogenous GM-CSF and its neutralization by E21R. Primary AML blasts from 13 individuals showing a response to exogenous GM-CSF. Cells were incubated in medium plus GM-CSF at 10 ng/mL for 48 to 72 hours either without or with an at least 1000-fold excess of E21R (10 to 30 μg/mL). MTS assay was carried out, and the results are expressed as a percentage of the activity in control cells incubated in medium alone. (B) Effect of E21R in neutralizing the proliferative response of TF-1 cells to GM-CSF. TF-1 cells were washed and placed in medium alone, GM-CSF (10 ng/mL), IL-3 (10 ng/mL), or erythropoietin (2 U/mL) with the indicated concentrations of E21R for 48 hours. MTS assay was carried out, and the results are expressed as a percentage of the activity in control cells incubated in medium alone. (C) Effect of E21R in neutralizing the proliferative response of normal myeloid cells to GM-CSF. Primary CD34+ cells were placed in medium with SCF, IL-3, and G-CSF for 7 days to induce myeloid differentiation. After washing, cells were incubated for 48 hours in medium alone or with GM-CSF (10 ng/mL), IL-3 (10 ng/mL), or G-CSF (50 ng/mL) without or with E21R (10 μg/mL). MTS assay was carried out, and the results are expressed as a percentage of the activity in control cells incubated in medium alone.
Neutralization of GM-CSF by E21R. (A) Response of AML blasts to exogenous GM-CSF and its neutralization by E21R. Primary AML blasts from 13 individuals showing a response to exogenous GM-CSF. Cells were incubated in medium plus GM-CSF at 10 ng/mL for 48 to 72 hours either without or with an at least 1000-fold excess of E21R (10 to 30 μg/mL). MTS assay was carried out, and the results are expressed as a percentage of the activity in control cells incubated in medium alone. (B) Effect of E21R in neutralizing the proliferative response of TF-1 cells to GM-CSF. TF-1 cells were washed and placed in medium alone, GM-CSF (10 ng/mL), IL-3 (10 ng/mL), or erythropoietin (2 U/mL) with the indicated concentrations of E21R for 48 hours. MTS assay was carried out, and the results are expressed as a percentage of the activity in control cells incubated in medium alone. (C) Effect of E21R in neutralizing the proliferative response of normal myeloid cells to GM-CSF. Primary CD34+ cells were placed in medium with SCF, IL-3, and G-CSF for 7 days to induce myeloid differentiation. After washing, cells were incubated for 48 hours in medium alone or with GM-CSF (10 ng/mL), IL-3 (10 ng/mL), or G-CSF (50 ng/mL) without or with E21R (10 μg/mL). MTS assay was carried out, and the results are expressed as a percentage of the activity in control cells incubated in medium alone.
To confirm that the assay systems were effective at detecting cell death, we incubated AML blasts with cytosine arabinoside. This effectively reduced viable cell numbers by 48 hours as detected by the MTS assay to 48% ± 5% (range, 9% to 91%) of control (Figure 1; n = 21) and led to a significant increase in the number of annexin-binding cells from 45% ± 6% to 76% ± 6% (n = 10). The absence of any effect of E21R could be due to a lack of GM-CSF receptors on AML blasts. However, previous publications have shown that blast cells from most AML patients express the GM-CSF receptor,13-15 and we have previously demonstrated the presence of GM-CSF receptors on AML blasts in all 3 patients investigated using 125I–GM-CSF with a mean of 107 ± 28 high-affinity binding sites (dissociation constant [KD], 64 ± 10 pM) and 2684 ± 1511 low-affinity sites (KD, 5.6 ± 3.7 nM) per cell (A.K. and D.C.L., unpublished data, July 1993). We found that 13 of 23 patients studied here had a clear response to increasing concentrations of GM-CSF with a significant increase in MTS activity at 48 to 72 hours (Figure 2). These data are in keeping with those published by others showing GM-CSF responsiveness in primary AML blasts.16-21 In addition to investigating the effects of E21R as a single agent, we have combined it with cytosine arabinoside and did not find any evidence of synergy (Figure 1).
To confirm that the E21R used was biologically active, we used it as a competitive antagonist of GM-CSF. Figure 2 shows that E21R neutralizes the effect of human GM-CSF in stimulating the survival and proliferation of the factor-dependent cell line TF-1 and of myeloid cells in a primary CD34+ cell–derived liquid culture. In addition, Figure 2 shows that in primary AML blasts from patients who showed a response to GM-CSF in the MTS assay, this could be neutralized by the addition of an excess of E21R.
In summary, we found that the GM-CSF antagonist E21R does not have antileukemic activity in primary AML blasts obtained from 23 patients. This lack of effect was unlikely to be due to an absence of high-affinity GM-CSF receptors, because 13 of 23 cases had a clear survival response to exogenous GM-CSF. The reason for the difference between our findings and those of Iversen et al is not clear. We used concentrations of E21R up to 30-fold higher than those previously shown to induce death of more than 95% of AML blasts from 21 patients.7 These blasts had been selected using an anti–GM-CSF receptor α antibody and magnetic beads and represented 50% to 75% of the total blast population. It is possible that this process could influence cell survival. However, Iversen et al7 also found that unselected AML blasts underwent apoptosis in response to E21R and that the proportion of cells apoptotic at 48 hours correlated completely with the number of cells expressing GM-CSF receptor α (ranging up to 75%). We did not find any reduction in viable cells using either the MTS technique or annexin V binding in response to E21R, whereas clear effects of cytosine arabinoside were detected. The E21R used in our experiments was biologically active in neutralizing the effects of exogenous GM-CSF. In conclusion, these studies do not support the use of E21R for the treatment of AML.
Supported by British Biotech (I.J.), the Kay Kendall Leukaemia Fund (V.L.G.), and the Medical Research Council, United Kingdom (A.K.).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
Prepublished online as Blood First Edition Paper, December 30, 2003; DOI 10.1182/blood-2003-06-1985.