Abstract
The chimeric anti-CD20 monoclonal antibody (mAb), rituximab, is an established part of the management of many non-Hodgkin lymphomas. The in vivo action of rituximab remains elusive, and this partially reflects a lack of highly specific reagents to detect rituximab binding at the cell surface. Here we report a new high-affinity mAb (MB2A4) with fine specificity for the idiotype of rituximab. It is able to detect rituximab in vitro, in the presence of high levels of human immunoglobulin G (IgG), in the serum of patients receiving rituximab therapy, and, surprisingly, when rituximab is bound to CD20 on the cell surface. We propose that the anti–idiotype (Id) binds to rituximab molecules bound univalently at the cell surface, facilitated by the relatively high off-rate of rituximab. This reagent provides new insights into the binding of rituximab at the cell surface and demonstrates a mode of binding that could be exploited for the surface detection of other mAbs with clinical and biologic applications.
Introduction
The chimeric anti-CD20 monoclonal antibody (mAb), rituximab, has now been used in more than 300 000 patients and has become integrated into the treatment of many B-cell lymphomas.1-5 The use of rituximab is likely to increase even further with the recent observations that it may be of clinical benefit to patients with autoimmune diseases.5-8 Remarkably, given its widespread use, we still have a relatively poor understanding of the nature of rituximab binding to B cells and how this mAb actually mediates its therapeutic effects in vivo.3,9-15
There are currently few well-characterized, highly specific reagents to detect rituximab. Previous reagents have been polyclonal and directed either to the rituximab idiotype (Id)16,17 or the remaining mouse regions in rituximab.18 Clearly, these may suffer from batch variation, lack of specificity, and/or cross-reactivity to normal human immunoglobulin G (IgG). For this reason, we raised a highly specific mAb MB2A4 directed to the rituximab Id and have used this tool to probe the nature of rituximab binding to B-cell tumors.
Study design
Cell lines and antibodies
Cells were obtained from the European collection of cell cultures (ECACC) and maintained in supplemented RPMI. mAbs and their fragments were produced using standard methods.19
Generation of anti-Id mAbs
Anti-Id mAbs were generated by immunizing Lou rats with purified rituximab F(ab′)2 fragments and fusing their splenocytes with NS-1 cells to generate hybridomas. The hybridomas were screened by enzyme-linked immunosorbent assay (ELISA) and assessed for binding to rituximab-coated plates.
ELISA for rituximab
To detect rituximab in patient serum, MB2A4 was coated onto ELISA plates, blocked, and washed. Subsequently, appropriately diluted rituximab-containing samples were added to the plate for 90 minutes, washed, and incubated with anti–human Fc–horseradish peroxidase (Hu Fc-HRP) mAb (SB2H2) for 60 minutes, before further washing and addition of the ELISA substrate.
Flow cytometry for the detection of cell-bound rituximab
To detect rituximab bound to the surface of CD20+ cells, standard flow cytometry techniques were used.20 Briefly, rituximab was coated to CD20+ cells for 15 minutes at room temperature (RT), and the cells were washed twice and incubated with fluorescein isothiocyanate (FITC)–labeled MB2A4. Labeled cells were assessed using a FACScan flow cytometer with CellQuest software (BD Biosciences, San Jose, CA).
Results and discussion
Anti-rituximab idiotype (MB2A4) detects rituximab in sera
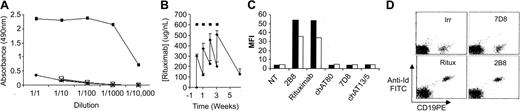
Following immunization of Lou rats and subsequent fusions, a number of mAbs were raised that detected rituximab by ELISA. MB2A4 was chosen for further study, as it failed to bind to normal IgG in serum or another chimeric anti-CD20 mAb, chAT80 (Figure 1A). Importantly, MB2A4 did bind to 2B8, the nonchimeric parent mAb of rituximab (data not shown) and thus appears specific for the idiotype region of rituximab. Next, we addressed whether MB2A4 bound rituximab in patients' sera. The linear region of the standard curve occurred at less than 100 ng/mL, so patients' sera was first diluted accordingly. Figure 1B illustrates the variation in rituximab concentration over time for a representative patient during and following weekly administration of rituximab and demonstrates that MB2A4 is able to detect serum rituximab down to 5 to 10 ng/mL.
Ability of MB2A4 to detect rituximab in sera and on the surface of cells. (A) MB2A4 was coated to the surface of ELISA plates before blocking nonspecific binding sites and adding various dilutions (1:1-1:10 000) of either pure serum (♦), or serum spiked with 100 μg/mL rituximab (▪), chAT13/5 (X), or chAT80 (□). The amount of human Fc captured to the plate was then detected using anti–human Fc coupled to HRP. (B) The amount of rituximab present in a patients' serum was detected over a period of time during which they had received rituximab. After collection of serum, a number of dilutions of the sample were made and then measured in an ELISA, such as detailed in panel A, along with a standard curve of known rituximab concentrations. Subsequently, reference to the standard curve allowed calculation of the serum rituximab level in the original sample. ▪ indicates when a 375 mg/m2 dose of rituximab was given. Error bars represent the standard error of the mean for duplicate points. MFI indicates mean fluorescence intensity. (C) Following binding of various mAbs (10 μg/mL) to EHRB (□) or Raji cells (▪) for 15 minutes at RT, cells were washed twice in phosphate-buffered saline/bovine serum albumin/Azide before staining with FITC-labeled mAb MB2A4 (10 μg/mL) for 15 minutes at RT, washing, and detection by flow cytometry. (D) Rituximab (Ritux), 2B8, or 7D8 (10 μg/mL) was added to whole blood for 15 minutes at RT along with CD19-phycoerythrin before washing and staining with FITC-labeled mAb MB2A4 as in panel C.
Ability of MB2A4 to detect rituximab in sera and on the surface of cells. (A) MB2A4 was coated to the surface of ELISA plates before blocking nonspecific binding sites and adding various dilutions (1:1-1:10 000) of either pure serum (♦), or serum spiked with 100 μg/mL rituximab (▪), chAT13/5 (X), or chAT80 (□). The amount of human Fc captured to the plate was then detected using anti–human Fc coupled to HRP. (B) The amount of rituximab present in a patients' serum was detected over a period of time during which they had received rituximab. After collection of serum, a number of dilutions of the sample were made and then measured in an ELISA, such as detailed in panel A, along with a standard curve of known rituximab concentrations. Subsequently, reference to the standard curve allowed calculation of the serum rituximab level in the original sample. ▪ indicates when a 375 mg/m2 dose of rituximab was given. Error bars represent the standard error of the mean for duplicate points. MFI indicates mean fluorescence intensity. (C) Following binding of various mAbs (10 μg/mL) to EHRB (□) or Raji cells (▪) for 15 minutes at RT, cells were washed twice in phosphate-buffered saline/bovine serum albumin/Azide before staining with FITC-labeled mAb MB2A4 (10 μg/mL) for 15 minutes at RT, washing, and detection by flow cytometry. (D) Rituximab (Ritux), 2B8, or 7D8 (10 μg/mL) was added to whole blood for 15 minutes at RT along with CD19-phycoerythrin before washing and staining with FITC-labeled mAb MB2A4 as in panel C.
MB2A4 binds rituximab on the surface of B cells
We then assessed whether MB2A4 could detect rituximab bound to CD20 on the surface of B-cell tumor lines, using FITC-labeled MB2A4. To our initial surprise, these experiments indicated that MB2A4 bound to rituximab-coated cells (Figure 1C-D). Binding was specific for the idiotypic region of rituximab and not other chimeric or fully human anti-CD20 mAbs, or chimeric mAbs against other B-cell markers (Figure 1C), and was dependent upon the amount of idiotype-containing mAbs (rituximab or 2B8) bound to the cells (data not shown). Further specificity was demonstrated when rituximab was incubated with whole blood where the anti-Id detected only the CD19+ B cells (Figure 1D).
MB2A4 binds to rituximab through univalent binding
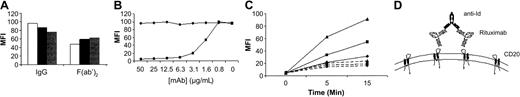
Because the specificity of anti-Id mAbs is highly dependent on the epitope structure of the complementarity determining regions (CDRs), it is expected that binding the target antigen will block binding of the anti-Id. Therefore, we wanted to determine the mechanism through which MB2A4 was binding to rituximab-coated cells. The first possibility addressed was whether rituximab was binding to the cells via Fcγ receptor IIb (FcγRIIb), thus allowing the idiotype regions to be free for the anti-Id to bind. To examine this, we performed experiments using high concentrations of normal human plasma that would be expected to out-compete rituximab binding to FcγRIIb and also by directly blocking the FcγRIIb with mAb (AT10). In addition, we used rituximab F(ab′)2 fragments incapable of binding through FcγR. These experiments (Figure 2A) show that MB2A4 binding to rituximab on the surface of lymphoma cells occurred whether the Fc-FcR interaction was blocked or absent. To probe the binding mechanism further, we next sought to determine whether rituximab binding to CD20 could be blocked by the addition of MB2A4 prior to rituximab binding the cell surface. These experiments (Figure 2B) show clearly that MB2A4 but not another anti-Id mAb (specific for another anti-CD20) was able to block rituximab binding to CD20 on cells in a dose-dependent manner. These data indicate that while rituximab cannot bind to CD20 in the presence of high levels of the anti-Id, once rituximab is bound to CD20, the anti-Id may bind to rituximab. From these experiments, we conclude that the sites for anti-Id and CD20 binding are indeed the same, confirming our suggestion that binding of antigen via the mAb CDRs in the V-regions blocks access of the anti-Id mAb. With these results, we continued to investigate an explanation for the simultaneous binding of antigen and anti-Id mAb at the cell surface. The simplest explanation is that one Fab arm of rituximab is bound to CD20, while the second Fab arm is bound by the anti-Id. This would require rituximab to exhibit a reasonably high off-rate, allowing one Fab arm to be released from CD20 binding. Recently, we assessed the off-rate of various anti-CD20 mAb fragments and showed that rituximab has a relatively fast off-rate in comparison with other anti-CD20 mAbs.21 Furthermore, off-rate, unlike on-rate is highly sensitive to differences in temperature.22,23 Therefore, we addressed whether the binding of MB2A4 to rituximab-coated cells was temperature sensitive. These experiments detailed in Figure 2C clearly show the temperature dependence of MB2A4 binding, indicating that an increase in temperature (and hence off-rate) greatly facilitates the binding of the anti-Id. Presumably, within this model, MB2A4 carries out a bridging and stabilizing function, binding 2 separate Fab-bound rituximab molecules, thereby preventing rapid dissociation from the cell surface (Figure 2D). Interestingly, and in support of this model, we could detect no binding of MB2A4 to the Fab′ fragment of rituximab (data not shown). Support for this model was also found when a second, low off-rate anti-CD20 mAb (and its associated anti-Id mAb) was assessed and shown to be much more difficult to detect on the surface of B cells (Figure 2C).
Mechanism of binding of anti-Id to rituximab bound to CD20 on the surface of lymphoma cells. (A) Raji cells were preincubated in normal media (NT, open bar), 33% plasma (solid bar), or 50 μg/mL anti-FcγRII mAb AT10 (gray bar) prior to incubation with rituximab IgG or F(ab′)2, followed by washing and incubation with FITC-labeled mAb MB2A4 as in Figure 1C. (B) FITC-labeled rituximab was added to Raji cells in the presence of varying concentrations of either MB2A4 (▪) or SAB1.3 (♦). Bound FITC-mAb was detected as before. (C) Raji cells were coated with rituximab (solid lines) or 7D8 (dashed lines) at 10 μg/mL for 15 minutes at RT and washed, and then MB2A4-FITC or SAB1.3-FITC was added to the cells for 5 or 15 minutes at 4°C (♦), 20°C (▪), or 37°C (▴) before washing and detection of FITC-mAb binding. (D) Schematic of MB2A4 binding to rituximab bound to the surface of CD20+ cells.
Mechanism of binding of anti-Id to rituximab bound to CD20 on the surface of lymphoma cells. (A) Raji cells were preincubated in normal media (NT, open bar), 33% plasma (solid bar), or 50 μg/mL anti-FcγRII mAb AT10 (gray bar) prior to incubation with rituximab IgG or F(ab′)2, followed by washing and incubation with FITC-labeled mAb MB2A4 as in Figure 1C. (B) FITC-labeled rituximab was added to Raji cells in the presence of varying concentrations of either MB2A4 (▪) or SAB1.3 (♦). Bound FITC-mAb was detected as before. (C) Raji cells were coated with rituximab (solid lines) or 7D8 (dashed lines) at 10 μg/mL for 15 minutes at RT and washed, and then MB2A4-FITC or SAB1.3-FITC was added to the cells for 5 or 15 minutes at 4°C (♦), 20°C (▪), or 37°C (▴) before washing and detection of FITC-mAb binding. (D) Schematic of MB2A4 binding to rituximab bound to the surface of CD20+ cells.
In summary, we have developed a new high-affinity anti-Id mAb (MB2A4) that is equally capable of binding rituximab in the serum of patients or when bound to CD20 at the cell surface. This reagent represents a significant advance in our ability to accurately probe the pharmacokinetics, tissue distribution, and modulation of rituximab in vivo. We believe that this novel mode of mAb cell surface detection provides new insights into the mode of rituximab binding to B cells and this type of approach could be exploited for the detection of other mAbs with clinical and biologic applications.
Prepublished online as Blood First Edition Paper, June 22, 2004; DOI 10.1182/blood-2004-05-1733.
Supported by Tenovus of Cardiff, the Leukemia Research Fund, and Cancer Research UK.
M.S.C. and M.B.B. contributed equally to this work.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
The authors would like to acknowledge the assistance and input of Prof Jan van de Winkel and Carin EG Havenith (Genmab) and the members of the CD20 team both in Southampton and Utrecht.