Abstract
Leukemic blasts from patients with acute myeloid leukemia (AML) frequently express high levels of the interleukin-3 receptor α chain (IL-3Rα). In the present study, we have explored the sensitivity of primary leukemic blasts obtained from 34 patients with AML to a diphtheria toxin (DT) composed of the catalytic and translocation domains of DT (DT388) fused to IL-3 (DT388IL-3) and to DT388 fused to a variant IL-3 with increased binding affinity (DT388IL-3[K116W]). On a molar basis, DT388IL-3[K116W] was significantly more active than DT388IL-3 in mediating leukemic cell killing. The rate of cell killing induced by the 2 DT/IL-3 fusion proteins was significantly correlated with the level of IL-3Rα/IL-3Rβ expressed on leukemic blasts. These observations support a potential use of DT388IL-3[K116W] in the treatment of refractory AMLs and provide a simple biochemical parameter for the selection of eligible patients. (Blood. 2005;106:2527-2529)
Introduction
Acute myeloid leukemia (AML) encompasses a group of heterogeneous disorders characterized by the expansion of malignant clones of hematopoietic cells blocked at various stages of differentiation. Several lines of evidence suggest that multiple molecular mechanisms are involved in the pathogenesis of AMLs.1 For example, chromosome translocations resulting in fusion genes that generate new proteins have been shown to play a major role in the genesis of leukemias.2 However, for the development of an overt leukemia condition, additional mutations and phenotypic abnormalities are required. Among them, a key role is played by mutations or alterations in the level of expression of membrane receptors involved in the control of the proliferation of hemopoietic cells, such as fms-related tyrosine kinase 3 (flt3), cellular-receptor tyrosine kinase (c-kit), and interleukin-3 receptor (IL-3R).
Flt3 is mutated and/or overexpressed in 30% to 40% of AMLs,3 whereas IL-3R is overexpressed in about 45% of AMLs.4 Furthermore, the analysis of an AML subpopulation enriched in leukemic stem cells showed that these cells express elevated levels of the IL-3R α chain (IL-3Rα), whereas in the normal counterpart low levels of IL-3Rα have been observed, thus indicating that IL-3Rα is a unique marker of leukemic stem cells.5 According to these findings, it has been suggested that IL-3R may represent an important target for the development of new antileukemic drugs.6
A genetically engineered fusion toxin composed of the first 388 amino acid residues of diphtheria toxin (DT) with a His-Met (H-M) linker is fused to human IL-3.7 This IL-3 DT was found to be toxic toward leukemic blasts8 and in vivo studies in Cynomolgus monkeys have shown that it is relatively well tolerated up to 100 μg/kg.9,10 These results contrast with the limited tolerance to the granulocyte macrophage-colony-stimulating factor (GMCSF) DT fusion protein (DT388GMCSF), which produced liver injury above 4 μg/kg to 7 μg/kg in monkeys and patients with AML.11,12 Preclinical studies with rodent-cell-directed DT fusion proteins (DT388mGMCSF and DT388mIL-3) showed Kupffer-cell-mediated liver injury for the GMCSF DT fusion protein but not the IL-3 DT fusion protein, consistent with the presence of GMCSFR but not IL-3R on the target cells.13 Based on these findings, a phase 1 clinical trial has recently been initiated with DT388IL-3 for the treatment of patients with AML. To date, 6 patients with AML have been treated with DT388IL-3 without evidence of severe toxicities or liver damage.14
Finally, with the aim of improving antileukemic activity, diphtheria toxin has been fused to IL-3 variants. Based on mutagenesis studies by several laboratories, substitution of a large, bulky hydrophobic residue (tryptophan) at IL-3 position 116 enhanced the hydrophobic interaction with IL-3Rα. We prepared the modified DT388IL-3[K116W], and the resulting fusion protein exhibited an enhanced binding to the IL-3R and more potent cytotoxicity toward leukemic cell lines.15
Using primary blasts from patients with AML in vitro, we compared in the present study the antileukemic cytotoxic activity of DT388IL-3 and that of DT388IL-3[K116W]. We found that the variant IL-3 fusion protein exhibited a more potent antileukemic activity than the wild-type (WT) IL-3 fusion protein.
Effect of increasing concentrations of DT388IL-3 and DT388IL-3[K116W] on the induction of apoptosis in primary AML leukemic blasts. (A) Leukemic cells from one representative case were grown in vitro for 24 hours in the presence of either DT388IL-3 or DT388IL-3[K116W] and then the percentage of apoptotic cells was determined by the annexin V binding assay. ▪ indicates DT388IL-3; ▴, DT388IL-3[K116W]. (B) Percentage of apoptotic cells observed on the leukemic blasts of 25 patients with AML grown for 24 hours in the presence of either DT388IL-3 or DT388IL-3[K116W]. ▪ indicates DT388IL-3; ▴, DT388IL-3[K116W]. (C, D) Correlation between the percentage of apoptotic cells induced by a 24-hour incubation in the presence of either DT388IL-3 or DT388IL-3[K116W] and the level of IL-3Rα (C) or IL-3Rβ (D). □ indicates DT388IL-3; ▴,DT388IL-3[K116W].
Effect of increasing concentrations of DT388IL-3 and DT388IL-3[K116W] on the induction of apoptosis in primary AML leukemic blasts. (A) Leukemic cells from one representative case were grown in vitro for 24 hours in the presence of either DT388IL-3 or DT388IL-3[K116W] and then the percentage of apoptotic cells was determined by the annexin V binding assay. ▪ indicates DT388IL-3; ▴, DT388IL-3[K116W]. (B) Percentage of apoptotic cells observed on the leukemic blasts of 25 patients with AML grown for 24 hours in the presence of either DT388IL-3 or DT388IL-3[K116W]. ▪ indicates DT388IL-3; ▴, DT388IL-3[K116W]. (C, D) Correlation between the percentage of apoptotic cells induced by a 24-hour incubation in the presence of either DT388IL-3 or DT388IL-3[K116W] and the level of IL-3Rα (C) or IL-3Rβ (D). □ indicates DT388IL-3; ▴,DT388IL-3[K116W].
Study design
Cells
Fresh leukemic blasts from 34 patients with AML, obtained after informed consent, were isolated from either bone marrow or peripheral blood by Ficoll-Hypaque density gradient centrifugation and were immediately processed. All patients were consecutively diagnosed at the Division of Hematology, Department of Cellular Biotechnologies and Hematology of the University “La Sapienza,” Rome, Italy. Leukemias were classified by morphologic criteria according to the French-American-British (FAB) classification and samples contained more than 70% leukemic blasts. Approval for these studies was obtained from the institutional review board of the Istituto Superiore di Sanità, Rome, Italy. Informed consent was obtained in accordance with the Declaration of Helsinki.
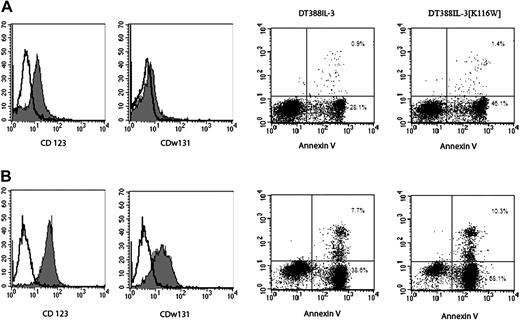
Flow cytometry analysis. Left panels show analysis of IL-3Rα and IL-3Rβ expression; right panels, of annexin V-propidium iodide (PI) staining. Data from one representative patient with low IL-3R expression (A) and one with high IL-3R expression (B) are shown. Numbers in the scatterplots indicate the percentage of annexin V-positive cells in that quadrant.
Flow cytometry analysis. Left panels show analysis of IL-3Rα and IL-3Rβ expression; right panels, of annexin V-propidium iodide (PI) staining. Data from one representative patient with low IL-3R expression (A) and one with high IL-3R expression (B) are shown. Numbers in the scatterplots indicate the percentage of annexin V-positive cells in that quadrant.
Preparation of DT388IL-3
Hematopoietic growth factor receptor expression
Phycoerythrin (PE)-labeled anti-IL-3Rα chain (CD123) monoclonal antibody clones 7G3 and 9G5 were purchased from Pharmingen (San Diego, CA). PE-labeled anti-IL-3Rβ chain (CDw131) monoclonal antibody was obtained from Santa Cruz Biotechnology (Santa Cruz, CA). Negative controls were represented by isotype-matched PE-labeled mouse immunoglobulins. Cells were incubated with optimal concentrations of the antibodies and, after 2 washings in cold phosphate-buffered saline (PBS), were analyzed for fluorescence by flow cytometry (FACSCAN; Becton Dickinson, San Diego, CA) by Cell Quest Software (Becton Dickinson).
In vitro incubation of leukemic blasts with IL-3 diphtheria fusion proteins and study of apoptosis
Leukemic cells were grown in vitro in freshly prepared Iscoves-modified minimal essential medium (IMDM; Gibco, New York, NY) containing 10% fetal calf serum (Gibco) at a final concentration of 5 × 105 cells/mL either in the absence (control) or in the presence of either DT388IL-3 or DT388IL-3[K116W] at various concentrations (10-11 M-10-7 M). At various days of culture, one cell aliquot was removed and assayed by FACS analysis for the presence of apoptotic cells using the annexin V binding assay.
Results and discussion
Newly diagnosed patients with AML were investigated for their in vitro sensitivity to the DT fused to the wild-type IL-3 (DT388IL-3) and to the DT fused to variant IL-3 (DT388IL-3[K116W]). Leukemic blasts from 2 M0, 4 M1, 8 M2, 2 M3, 10 M4, and 8 M5 were incubated in vitro for 24 to 48 hours either in the absence (control) or in the presence of increasing concentrations (from 1 × 10-11 to 1 × 10-7 M) of either DT388IL-3 or DT388IL-3[K116W] and the percentage of apoptotic cells was measured by the annexin V-propidium iodide (PI) labeling. A representative experiment reported in Figure 1A clearly indicated that maximal cytotoxicity was observed using the 1 × 10-7 M concentration of the 2 toxins and that DT388IL-3[K116W] was more active than DT388IL-3 in killing AML blasts.
We therefore tested the effects of the 2 fusion proteins at 1 × 10-7 M on AML cells. The results of this analysis showed that after 24 hours of incubation, DT388IL-3[K116W] was more active than DT388IL-3 in inducing apoptosis of leukemic blasts (percentage of apoptotic cells equal to 52.4 ± 4.5, versus 27.1 ± 3.5; P < .001; Figure 1B).
It is of interest to note that the rate of cell killing induced by these diphtheria IL-3 fusion proteins varied among the different AML cases tested. In order to understand the origin of this heterogeneity, we investigated whether the rate of blast-cell killing induced by the IL-3 diphtheria fusion proteins was dependent on the levels of IL-3R expression on leukemic cells. For this purpose, the expression of IL-3Rα (CD123) and IL-3Rβ (CDw131) was analyzed by flow cytometry using specific monoclonal antibodies conjugated with phycoerythrin. This analysis showed that the levels of IL-3Rα and IL-3Rβ expressed on AML cells are heterogeneous and that they clearly correlated with the percentage of apoptotic cells induced by the 2 IL-3 diphtheria fusion proteins (Figure 1C-D).
AML cases expressing very low or low levels of IL-3Rα/IL-3Rβ showed only a very low or low rate of cell killing after 24 to 48 hours of incubation with the 2 IL-3 diphtheria fusion proteins (data not shown); AML cases expressing moderate IL-3Rα/IL-3Rβ levels (see representative example in Figure 2, top panels) exhibited a moderate rate of cell killing after 24 hours of incubation with DT388IL-3 or DT388IL-3[K116W]; AML cases exhibiting high IL-3Rα and IL-3Rβ levels (see representative case in Figure 2, bottom panels) showed a high rate of cell killing after incubation with IL-3 diphtheria fusion proteins, particularly with DT388IL-3[K116W].
Based on these observations, we conclude that the levels of IL-3Rα/IL-3Rβ on the surface of AML blasts represent a major determinant in their sensitivity to IL-3 diphtheria toxins.
The results of the present study clearly indicate that DT388IL-3[K116W] exerts a potent in vitro antileukemic activity. The cytotoxic activity of this IL-3/diphtheria toxin is particularly pronounced in samples from patients with AML expressing high IL-3Rα/IL-3Rβ levels; such patients are known to have a poor prognosis and a high rate of relapse.4 These patients could thus represent the natural candidates for an antileukemic therapy based on the use of DT388IL-3[K116].
In summary, our data suggest that the variant DT388IL-3[K116W] is a promising new agent for patients with chemotherapy-refractory AML. Because of the persistently poor prognosis of a large proportion of patients with AML, the further preclinical development of this agent appears warranted.
Prepublished online as Blood First Edition Paper, May 31, 2005; DOI 10.1182/blood-2005-02-0540.
Supported by grants from the Italian Stem Cell Research Program, Ministry of Health, Rome, Italy, and by 1% Progetto Finalizzato 4AC/F, Italian Ministry of Health (U.T.) and by grants from the National Institutes of Health (R01CA76 178 and R01CA90 263) and the Leukemia and Lymphoma Society (A.E.F.).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

![Figure 1. Effect of increasing concentrations of DT388IL-3 and DT388IL-3[K116W] on the induction of apoptosis in primary AML leukemic blasts. (A) Leukemic cells from one representative case were grown in vitro for 24 hours in the presence of either DT388IL-3 or DT388IL-3[K116W] and then the percentage of apoptotic cells was determined by the annexin V binding assay. ▪ indicates DT388IL-3; ▴, DT388IL-3[K116W]. (B) Percentage of apoptotic cells observed on the leukemic blasts of 25 patients with AML grown for 24 hours in the presence of either DT388IL-3 or DT388IL-3[K116W]. ▪ indicates DT388IL-3; ▴, DT388IL-3[K116W]. (C, D) Correlation between the percentage of apoptotic cells induced by a 24-hour incubation in the presence of either DT388IL-3 or DT388IL-3[K116W] and the level of IL-3Rα (C) or IL-3Rβ (D). □ indicates DT388IL-3; ▴,DT388IL-3[K116W].](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/106/7/10.1182_blood-2005-02-0540/6/m_zh80190584610001.jpeg?Expires=1767870926&Signature=iMPXmgWFNjIf1eNpsTG5Q0Rb8J20x2mOuR-RbFIP-a84t--jgxLQWNW6Jxkldp5tlx3JRnr-y7VYBbuAr0gLIzqx118hsEggpqnuAha0q0vw4r~aROe2wEcRSKsAO7GDVCOtTx1Fa0431-ve9GXqNUctM6ULZzSU5WTqDpqimiJWpSytpkKpqUma8C1DtPcGZG7a8cFNgv4tUWn7mcmiAJ~OaD-wr~rMVK8Ulz7KPC-RaDdYXoKPq7CZG3FpUbxmrn-9C3Vv0BpyiMp88bhA-9-qyYjQqNVm-RNGO~e1id~j2QiqHSCxZTkXcq2cbNYIrY5m4Fl6Qd-y-DoptYfiIQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)