In a recent issue of Blood, Mathas et al1 suggested elevated BCL3 expression to be functionally important in classical Hodgkin lymphoma (cHL) and peripheral T-cell lymphoma (PTCL). The authors reported strong BCL3 protein expression in the vast majority of cHLs and a subset of PTCLs.1 These results corroborated similar immunohistochemical findings by Canoz et al.2
Mathas et al1 reported chromosomal gains of the BCL3 locus in chromosome band 19q13 as a potential cause of BCL3 upregulation in 3 of 6 cHL cell lines and 8 of 37 PTCLs. Here, we provide evidence that not only chromosomal gains but also translocations affecting the BCL3 locus are recurrent in cHL and PTCL.
A BCL3 break-apart fluorescence in situ hybridization (FISH) assay using differentially labeled bacterial artificial chromosome (BAC) clones was applied to a series of 20 cHLs, and BCL3 breakpoints were detected in 2 cases. In one cHL, in which cytogenetic analysis suggested a t(14;19),3 an IGH-BCL3 juxtaposition was proven by FISH (Figure 1A-B). This translocation is a recurrent event in B-cell chronic lymphatic leukemia4 (B-CLL), although there was no evidence for a concomitant or previous B-CLL in this case. As the immunoglobulin genes are downregulated in cHL,5 the pathogenetic significance of an IGH-BCL3 translocation remains uncertain. Unfortunately, no material was available for immunohistochemistry. In the second cHL, none of the IG loci were translocated to BCL3. Additionally, 3 (15%) of 20 cHLs showed gains of the BCL3 locus in the Hodgkin/Reed-Sternberg (HRS) cells exceeding the ploidy level (defined as median number of FISH signals for 12 different genomic loci; data not shown). Thus, the frequency of BCL3 gains in our series of primary cHLs was lower than that reported in cHL cell lines.1 However, our analyses did not consider the absolute number but the number of BCL3 copies relative to the ploidy level. Applying this approach to 4 cHL cell lines (HDLM-2, KM-H2, L428, and L1236), no gains of the BCL3 locus were detected, though the absolute numbers of signals were the same as those reported.1 These findings were corroborated by array-comparative genomic hybridization (CGH) analyses that failed to detect significant gains of BCL3 in these 4 cell lines (data not shown).
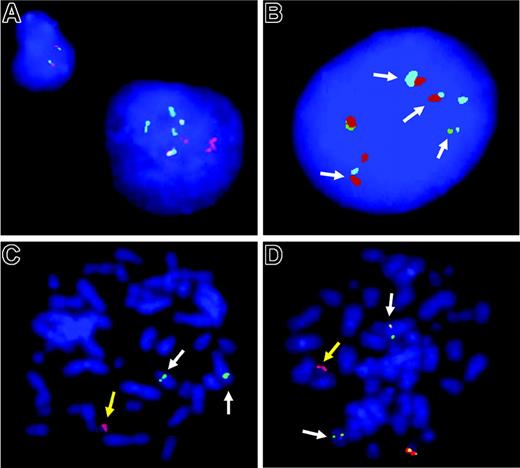
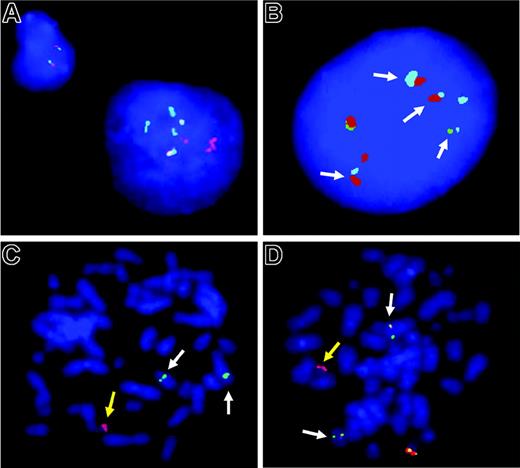
FISH analysis. (A-B) Interphase FISH analyses in cHL with a IGH-BCL3 fusion. (A) The large Hodgkin cell nucleus on the right shows multiple splits of BCL3 break-apart probe, whereas the small cell on the left shows the normal signal pattern for 2 intact BCL3 loci (ie, 2 colocalized signals). (B) Multiple colocalizations of IGH-telomeric (green) and IGH-centromeric (red) probes with a BCL3 spanning probe (pale blue; arrows) confirming IGH-BCL3 juxtaposition in an HRS nucleus of the same case. (C-D) Metaphase FISH analyses in PTCL. (C) Split of the TCRAD break-apart probe indicating a TCRAD breakpoint and concomitant loss of the normal TCRAD allele. The yellow arrow points to the chromosome containing a TCRAD centromeric signal (red), whereas white arrows point to marker chromosomes containing TCRAD telomeric signals (green). (D) Extra red signal for the BCL3 telomeric probe (yellow arrow) and residual red signals colocalizing with the green BCL3 centromeric signal (white arrows), indicating a chromosomal breakpoint slightly telomeric to the BCL3 gene. The yellow and white arrows in panels C and D correspond to the same marker chromosomes, respectively. Images were acquired using a 63 ×/1.40 numeric aperture oil objective in a Zeiss Axioskop2 fluorescence microscope (Zeiss, Göttingen, Germany) equipped with the appropriate filter sets (AHF, Tübingen, Germany) and documented using the ISIS imaging system (MetaSystems, Altlussheim, Germany).
FISH analysis. (A-B) Interphase FISH analyses in cHL with a IGH-BCL3 fusion. (A) The large Hodgkin cell nucleus on the right shows multiple splits of BCL3 break-apart probe, whereas the small cell on the left shows the normal signal pattern for 2 intact BCL3 loci (ie, 2 colocalized signals). (B) Multiple colocalizations of IGH-telomeric (green) and IGH-centromeric (red) probes with a BCL3 spanning probe (pale blue; arrows) confirming IGH-BCL3 juxtaposition in an HRS nucleus of the same case. (C-D) Metaphase FISH analyses in PTCL. (C) Split of the TCRAD break-apart probe indicating a TCRAD breakpoint and concomitant loss of the normal TCRAD allele. The yellow arrow points to the chromosome containing a TCRAD centromeric signal (red), whereas white arrows point to marker chromosomes containing TCRAD telomeric signals (green). (D) Extra red signal for the BCL3 telomeric probe (yellow arrow) and residual red signals colocalizing with the green BCL3 centromeric signal (white arrows), indicating a chromosomal breakpoint slightly telomeric to the BCL3 gene. The yellow and white arrows in panels C and D correspond to the same marker chromosomes, respectively. Images were acquired using a 63 ×/1.40 numeric aperture oil objective in a Zeiss Axioskop2 fluorescence microscope (Zeiss, Göttingen, Germany) equipped with the appropriate filter sets (AHF, Tübingen, Germany) and documented using the ISIS imaging system (MetaSystems, Altlussheim, Germany).
We additionally studied 3 PTCLs (2 unspecified and 1 angioimmunoblastic T-cell lymphoma) with a cytogenetically proven t(14;19)(q11;q13) with FISH break-apart probes for the TCRAD locus in 14q11 and BCL3 (Figure 1C-D). In all 3 cases, chromosomal breaks affecting both loci were detected. Colocalization of TCRAD and BCL3 was confirmed by 3-color assays, suggesting TCRAD-driven BCL3 activation as a possible novel oncogenic mechanism in T-cell neoplasms. Nuclear BCL3 protein expression was shown by immunohistochemistry in 2 PTCLs with available material (not shown). According to published cytogenetic data in PTCL, the estimated frequency of TCRAD breaks and TCRAD-BCL3 fusions is approximately 5% to 10% and 2%, respectively.6
Our results show for the first time that the spectrum of lymphatic neoplasias with BCL3 rearrangements goes beyond B-CLL and also includes cHL and PTCL. These findings are in line with the published immunohistochemical data on BCL3 expression1,2 and highlight the importance of this gene in B- and T-cell lymphomagenesis.
Chromosomal rearrangements involving BCL3 support its pathogenetic role in classical Hodgkin and peripheral T-cell lymphoma
We recently described high expression of the putative protooncogene Bcl-3 in Hodgkin/Reed-Sternberg (HRS) cells of classical Hodgkin lymphoma (cHL) and a proportion of peripheral T-cell lymphomas (PTCLs), including anaplastic large-cell lymphomas (ALCLs).1 In cHL and ALCL cell lines, Bcl-3 contributes via induction of NF-κB-p50 homodimer activity to the constitutive NF-κB activity and might be involved in cell-cycle and apoptosis regulation. Bcl-3 was originally discovered by molecular characterization of the t(14;19)(q32.3;q13.2) translocation in B-cell chronic lymphocytic leukemia (B-CLL).2 In this issue of Blood, Martin-Subero et al describe chromosomal rearrangements in cHL and PTCL that involve the BCL3 locus. These data provide interesting additional information concerning the possible underlying molecular mechanism of Bcl-3 upregulation in these lymphomas. Apart from a BCL3 fluorescence in situ hybridization (FISH) analysis for the detection of rearrangements involving the BCL3 locus, the authors investigated the number of BCL3 copies in HRS cells. Whereas we assessed in our analysis of HRS cell lines the absolute number of BCL3 copies per cell, Martin-Subero et al analyzed BCL3 copy number gains relative to the ploidy level. This analysis demonstrated that, in addition to BCL3 gains resulting from copy number gains of the entire chromosome (eg, in polyploid cells), intrachromosomal rearrangements of the BCL3 locus in HRS cells occur. In our opinion, all of these mechanisms could explain the finding of frequent Bcl-3 overexpression in cHL and PTCL. However, the results of Martin-Subero et al additionally point to a selection for chromosomal alterations involving the BCL3 locus that might lead to activation and overexpression of BCL3. These data further underline the importance of the BCL3 gene for the pathogenesis of cHL and PTCL.
Correspondence: Stephan Mathas, Max-Delbrück-Center for Molecular Medicine, FG Dörken, Robert-Rössle-Str 10, Berlin, Germany; e-mail: smathas@mdc-berlin.de.
The authors thank Claudia Becher and Dorit Schuster for their excellent technical assistance.
Supported by grants from the Deutsche Krebshilfe (70-3173-Tr3/B1), the Fund for Scientific Research of Flanders (FWO-Vlaanderen, grant no. G.0338.01), and a Collaborative Experimental Scholarship for Central & Eastern Europe from the Federation of European Biochemical Societies (FEBS). M.G. receives a PhD fellowship from the President of the Polish Academy of Sciences.
J.I.M.-S. designed and performed research, analyzed data, and wrote the paper; I.W. and C.B. provided tumor samples and performed cytogenetic analyses; J.-M.P. provided tumor samples for immunohistochemistry; W.K. provided tumor samples, performed research, and was involved in histopathologic review; J.H. and M.G. performed research and analyzed data; and R.S. designed research, analyzed data, and wrote the paper.