Activation of natural killer-like T (NKT) cells with the CD1d ligand α-galactosylceramide enhances T-dependent humoral immune responses against coadministered T-dependent Ag. At present, there is little information on the mechanisms involved other than a dependence on CD1d expression by antigen-presenting cells and/or development of the NKT subset. We therefore tested the hypothesis that direct presentation of α-GC by B cells was required for NKT-enhanced Ab responses against T-dependent Ag. We reconstituted B cell–deficient μMT mice with B cells from C57Bl/6 donors or CD1d−/− donors before immunization with NP-KLH alone or NP-KLH mixed with α-GC. We made the surprising observation that B-cell expression of CD1d is absolutely required for the NKT-enhanced Ab response. Our data show that the mechanism by which NKT cells enhance humoral immune responses involves interaction with CD1d-expressing B cells.
Introduction
CD1d is expressed by antigen-presenting cells (APCs) and presents glycolipids, including α-galactosylceramide (α-GC) to CD1d-restricted natural killer–like T (NKT) cells.1,–3 NKT cells stimulate Th1 and Th2 responses in vivo and are thus implicated in asthma, infectious disease, cancer, and autoimmunity.4,,,–8 A newly appreciated aspect of NKT function is their potential to regulate, enhance, and sustain humoral immune responses against foreign Ags, including those expressed by parasites, bacteria, and viruses.9,,,,,,,,,–19
Despite clear demonstrations that NKT cells impact humoral immunity, the mechanisms are not defined. We therefore tested the hypothesis that CD1d glycolipid presentation by B cells in vivo was required for NKT-enhanced humoral immunity. After reconstitution of B cell–deficient μMT mice with CD1d+/+ or CD1d−/− B cells and subsequent immunization, we observed that NKT-enhanced antibody (Ab) responses were deficient without CD1d expression by B cells. Our data introduce an important concept in humoral immunity: the direct interaction of B cells and NKT cells to facilitate enhanced Ab responses.
Methods
Mice
C57Bl/6 mice were from the National Cancer Institute (Bethesda, MD) and μMT mice from the Jackson Laboratory (Bar Harbor, ME). CD1d−/− mice have been described previously.20 All procedures were approved by the Institutional Animal Care and Use Committee at University of Oklahoma Health Sciences Center and performed on female mice between 6 and 10 weeks of age.
Antibodies and fluorochromes
Anti-CD1d, -TCRβ, and isotype control mAbs were from BD Biosciences (San Jose, CA), anti-B220 and -CD19 mAbs from Biolegend (San Diego, CA), anti-CD4, -Thy1.2, and 2.4G2 mAbs from BioXpres (West Lebanon, NH), CD1d/α-GC tetramers from the National Institute of Allergy and Infectious Diseases (NIAID) Tetramer Facility (Emory University, Atlanta, GA), and carboxyfluorescein diacetate succinimidyl ester (CFSE) from Invitrogen (Carlsbad, CA).
Cells and adoptive transfers
Spleens and thymi were subjected to mechanical disruption. Splenic erythrocytes were removed by hypotonic lysis. NKT and T cells were depleted by Lo-Tox complement-mediated lysis of anti-CD4/Thy1.2-labeled splenocytes (Cedarlane, Burlington, NC). Magnetic sorting was performed according to manufacturer's instructions using anti-fluorescein isothiocyanate microbeads in conjunction with a fluorescein isothiocyanate-anti-CD19 mAb (Miltenyi Biotech, Auburn, CA). A BD FACScalibur was used to confirm cell purity. Where indicated, cells were labeled with CFSE (9 μM in phosphate-buffered saline [PBS]/0.1% w/v bovine serum albumin) for 10 minutes at 37°C. This was followed by incubation for 5 minutes at 4°C and washing with PBS. Para-orbital injection was used to adoptively transfer 2 × 107 cells into recipient mice.
Immunization and sera collection
Recipient mice (μMT) were immunized intraperitoneally 24 hours after adoptive transfer with either 20 μg NP-KLH (Biosearch Technologies, Novato, CA) or 20 μg NP-KLH plus 4 μg α-GC Axxora (San Diego, CA) or NP-KLH adsorbed to Alum Imject (Pierce, Rockford, IL) in sterile PBS. The mice were boosted with NP-KLH on day 28 and sera were collected on day 33.
Cytokine assay
Mice were immunized intraperitoneally with 100 μL sterile PBS containing 0.05% w/v polysorbate-20 vehicle or vehicle plus 4 μg α-GC. Sera were collected at 2, 6, and 22 hours after immunization, and interleukin-4 (IL-4) and interferon-γ (IFN-γ) concentration was measured using a Bio-Plex instrument (Bio-Rad, Hercules, CA).
Enzyme-linked immunosorbent assay
Endpoint anti-NP IgG1, IgG2b, IgG2c, and IgM titers in serum were measured as described previously.18
Results and discussion
NKT cells develop in μMT mice
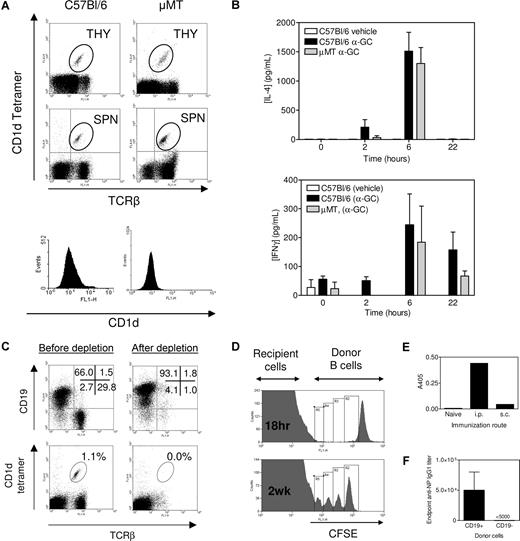
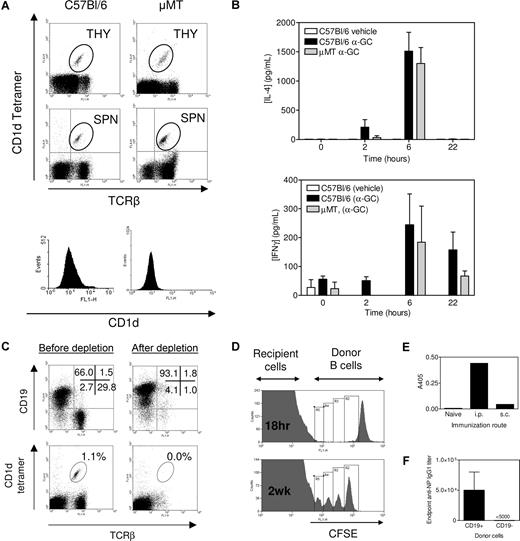
NKT cells were present in the thymus of μMT and C57Bl/6 mice at a similar frequency (Figure 1A). NKT cells were present at a higher frequency in spleens from μMT mice, but the total number of splenic NKT cells was similar between the 2 strains. Therefore, NKT thymic development and subsequent export to the periphery were intact in μMT mice. We immunized C57Bl/6 and μMT mice with α-GC and then measured IL-4 and IFN-γ production (Figure 1B). The C57Bl/6 and μMT mice produced equivalent amounts of IL-4 and IFN-γ in response to α-GC immunization. Bezbradica et al observed enhanced IL-4 and IFN-γ production in μMT mice after α-GC treatment.21 A larger dose of α-GC and higher concentration of vehicle in that study may have led to higher dendritic cell (DC) activation and in turn NKT activation in the absence of a buffering effect by B cells. Our results, however, are in broad agreement with that of Bezbradica et al21 in that NKT cells are functional in μMT mice.
μMT mice as a tool for examining NKT-enhanced Ab production. (A) Thymocytes (THY) and splenocytes (SPN) were assessed for TCRβ+, CD1d tetramer+ cells (dot plots) and CD1d+ splenocytes (histograms). Data are representative of several (> 6) determinations. (B) Mice were immunized as described in “Immunization and sera collection,” and sera were collected at times indicated before Bio-Plex analysis. Data show serum IL-4 and IFNγ concentration (mean ± SEM, n = 2 for vehicle, n = 5 for C57Bl/6 and n = 2 for μMT). Samples were analyzed in duplicate. No response was elicited with α-GC in CD1d−/− mice (not shown). Data are representative of 2 independent experiments. (C) Complement-mediated lysis of Thy1.2- and CD4-labeled cells depletes T and NKT cells resulting in a B cell–enriched preparation. Data are representative of several (> 6) determinations. (D) CFSE-labeled C57Bl/6 B cells were adoptively transferred to μMT mice. Cells were detected in recipient spleen after 18 hours (top panel) and 2 weeks after immunization with NP-KLH/Alum (bottom panel). (E) B cells were transferred into μMT recipients, which were then untreated (naive) or immunized 24 hours later with NP-KLH/Alum by the intraperitoneal or subcutaneous route. Sera were obtained after 2 weeks and anti-NP IgG1 detected by enzyme-linked immunosorbent assay. Data show A405 at a 1 of 1000 dilution of serum. (F) After T/NKT-depletion, CD19+ cells and CD19− (non-B/non-T cells) were resolved by magnetic sorting. C57Bl/6 mice were adoptively transferred with 2 × 107 CD19+ cells or 106 CD19− cells before immunization with NP-KLH/α-GC. The graph shows that the resulting Ab response was the result of the CD19+ fraction. The CD19− cellular fraction was determined by flow cytometry to be approximately 50% granulocytes and 50% DCs (not shown). Data in panels D-F show the results from single experiments.
μMT mice as a tool for examining NKT-enhanced Ab production. (A) Thymocytes (THY) and splenocytes (SPN) were assessed for TCRβ+, CD1d tetramer+ cells (dot plots) and CD1d+ splenocytes (histograms). Data are representative of several (> 6) determinations. (B) Mice were immunized as described in “Immunization and sera collection,” and sera were collected at times indicated before Bio-Plex analysis. Data show serum IL-4 and IFNγ concentration (mean ± SEM, n = 2 for vehicle, n = 5 for C57Bl/6 and n = 2 for μMT). Samples were analyzed in duplicate. No response was elicited with α-GC in CD1d−/− mice (not shown). Data are representative of 2 independent experiments. (C) Complement-mediated lysis of Thy1.2- and CD4-labeled cells depletes T and NKT cells resulting in a B cell–enriched preparation. Data are representative of several (> 6) determinations. (D) CFSE-labeled C57Bl/6 B cells were adoptively transferred to μMT mice. Cells were detected in recipient spleen after 18 hours (top panel) and 2 weeks after immunization with NP-KLH/Alum (bottom panel). (E) B cells were transferred into μMT recipients, which were then untreated (naive) or immunized 24 hours later with NP-KLH/Alum by the intraperitoneal or subcutaneous route. Sera were obtained after 2 weeks and anti-NP IgG1 detected by enzyme-linked immunosorbent assay. Data show A405 at a 1 of 1000 dilution of serum. (F) After T/NKT-depletion, CD19+ cells and CD19− (non-B/non-T cells) were resolved by magnetic sorting. C57Bl/6 mice were adoptively transferred with 2 × 107 CD19+ cells or 106 CD19− cells before immunization with NP-KLH/α-GC. The graph shows that the resulting Ab response was the result of the CD19+ fraction. The CD19− cellular fraction was determined by flow cytometry to be approximately 50% granulocytes and 50% DCs (not shown). Data in panels D-F show the results from single experiments.
Adoptive transfer of T- and NKT-depleted splenocytes
The CD4- and Thy 1.2-mediated T-cell depletion method also depleted NKT cells from splenocytes as noted by a loss of TCR+/CD1d-tetramer+ cells (Figure 1C). This resulted in the isolation of a highly enriched CD19+ B-cell preparation. The number of DCs (0.5%) in the postdepletion sample was calculated to increase the recipient DC pool by no more than 1.0% (not shown). We then adoptively transferred CFSE-labeled B cells from C57Bl/6 donors into μMT recipients and monitored their splenic homing (Figure 1D). Transferred B cells homed preferentially to spleen compared with lymph node or bone marrow (not shown) and splenic B cells proliferated (Figure 1D) and led to Ab production following an intraperitoneal route of immunization (Figure 1E). After T/NKT-depletion, we resolved CD19+ cells and CD19− non-B/non-T cells by magnetic sorting and showed that only the CD19+ cells led to Ab production after immunization (Figure 1F).
CD1d expression by B cells is required for NKT-enhanced Ab responses
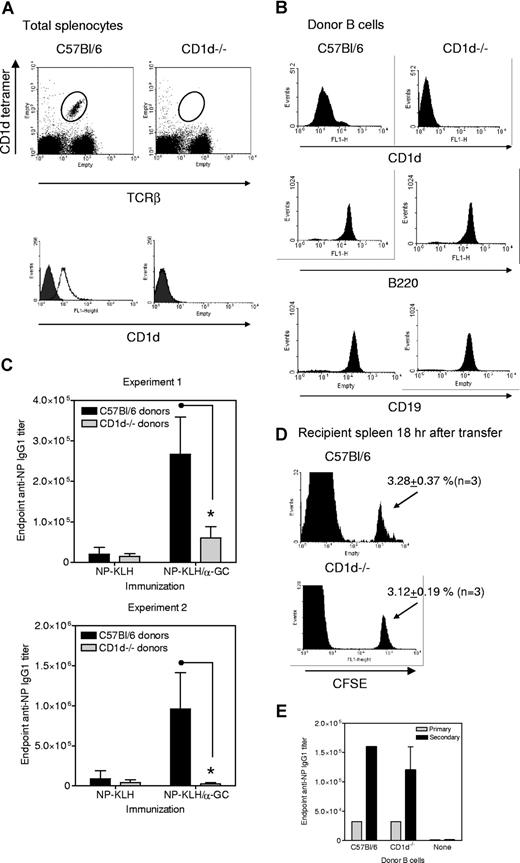
Splenocytes from C57Bl/6 and CD1d−/− donors were assessed for CD1d expression before (Figure 2A) and after (Figure 2B) T/NKT depletion and B cell enrichment confirmed. In the presence of equivalent B220 and CD19 expression, only B cells from C57Bl/6 donors expressed CD1d (Figure 2B).
CD1d expression by B cells is required for NKT-enhanced Ab recall responses. (A) Splenocytes from C57Bl/6 and CD1d−/− mice were assessed for TCRβ+, CD1d tetramer+ cells (dot plots) and CD1d+ (histograms). Empty histograms show anti-CD1d and filled show isotype control mAb. Data are representative of several (> 20) determinations. (B) Shows CD1d status of the T- and NKT-depleted B220+ cells used for adoptive transfer. Data are representative of several (> 12) determinations. (C) B cells from C57Bl/6 and CD1d−/− donors were adoptively transferred to μMT recipients before immunization with NP-KLH or NP-KLH plus α-GC 24 hours after transfer. All groups were boosted with NP-KLH on day 28 and sera collected on day 33. Each mouse was immunized and boosted with 10 μg (experiment 1) and 20 μg (experiment 2) NP-KLH. Data show endpoint IgG1 titer (mean ± SD) for 3 mice per group from 2 independent experiments. * Significant differences in titer between mice receiving cells from C57Bl/6 and CD1d−/− donors. (D) CFSE-labeled B cells from C57Bl/6 and CD1d−/− donors were adoptively transferred into μMT mice. After 18 hours, splenocytes were prepared and assessed for CFSE+ cells. Data are representative of 3 independent experiments. (E) Experiment was performed as in panel C, except that mice were immunized with NP-KLH/Alum on day 0, bled (primary bleed), and then boosted with NP-KLH on day 21 and bled again on day 26 (secondary bleed). Results from a single experiment are shown in panel E.
CD1d expression by B cells is required for NKT-enhanced Ab recall responses. (A) Splenocytes from C57Bl/6 and CD1d−/− mice were assessed for TCRβ+, CD1d tetramer+ cells (dot plots) and CD1d+ (histograms). Empty histograms show anti-CD1d and filled show isotype control mAb. Data are representative of several (> 20) determinations. (B) Shows CD1d status of the T- and NKT-depleted B220+ cells used for adoptive transfer. Data are representative of several (> 12) determinations. (C) B cells from C57Bl/6 and CD1d−/− donors were adoptively transferred to μMT recipients before immunization with NP-KLH or NP-KLH plus α-GC 24 hours after transfer. All groups were boosted with NP-KLH on day 28 and sera collected on day 33. Each mouse was immunized and boosted with 10 μg (experiment 1) and 20 μg (experiment 2) NP-KLH. Data show endpoint IgG1 titer (mean ± SD) for 3 mice per group from 2 independent experiments. * Significant differences in titer between mice receiving cells from C57Bl/6 and CD1d−/− donors. (D) CFSE-labeled B cells from C57Bl/6 and CD1d−/− donors were adoptively transferred into μMT mice. After 18 hours, splenocytes were prepared and assessed for CFSE+ cells. Data are representative of 3 independent experiments. (E) Experiment was performed as in panel C, except that mice were immunized with NP-KLH/Alum on day 0, bled (primary bleed), and then boosted with NP-KLH on day 21 and bled again on day 26 (secondary bleed). Results from a single experiment are shown in panel E.
C57Bl/6 and CD1d−/− donor B cells were adoptively transferred into μMT mice, which were then immunized intraperitoneally with NP-KLH, or NP-KLH/α-GC and then boosted with NP-KLH (Figure 2C). The μMT mice adoptively transferred with B cells from C57Bl/6 donors had minimal Ab titers in response to NP-KLH but enhanced Ab responses when immunized with NP-KLH/α-GC. This shows that an NKT-enhanced Ab response can be reconstituted in μMT mice. In μMT mice receiving CD1d−/− B cells, α-GC did not lead to enhanced anti-NP Ab titers. To confirm that lack of CD1d on B cells did not simply skew the Ab subclasses produced, we analyzed the same sera for anti-NP IgG2b and IgG2c. The IgG2c subclass was not induced under any condition. IgG2b titers approximately 2.5% of the magnitude of the IgG1 titer were induced, and the pattern matched that of the IgG1 titers. IgM titers also showed the same pattern as IgG1 (not shown). The α-GC NKT-enhanced Ab response was not observed despite CD1d expression by DCs in vivo (not shown). This shows that CD1d+ B cells are required for NKT-enhanced Ab responses.
We confirmed that the different Ab responses in μMT mice reconstituted with C57Bl/6 and CD1d−/− donor B cells were not the result of differential splenic homing. The frequency of CFSE-labeled C57Bl/6 and CD1d−/− donor B cells observed in the recipient spleens 18 hours after transfer was identical (Figure 2D). Control groups of μMT mice that had not received donor B cells were immunized with NP-KLH, NP-KLH/α-GC, α-GC, or NP-KLH/Alum. Neither of these agents stimulated an Ab response in the absence of B cells (not shown). Comparable in vivo function of transferred C57Bl/6 and CD1d−/− B cells was confirmed by immunization of μMT recipients with NP-KLH/Alum (Figure 2E).
We have uncovered an important mechanism by which CD1d-restricted NKT cells enhance humoral immune responses. CD1d+ B cells were required for NKT-enhanced Ab responses to T-dependent Ag. This shows that B cell presentation of CD1d ligand to NKT cells is a crucial interaction, perhaps resulting in additional contact-dependent B-cell help. The degree to which B-cell CD1d was required for the α-GC effect was surprising. Our prediction was that B-cell CD1d Ag presentation would make a measurable but partial contribution to NKT-enhanced Ab responses. However, our data do not preclude a role for CD1d Ag presentation by other APCs, including DCs, because α-GC induces DC maturation in vivo and leads to enhanced Th and CTL responses.22,23 Arguably, CD1d+ DCs could stimulate NKT cells to increase Th priming and contribute to NKT-enhanced Ab production. Indeed, our previous work suggested α-GC-induced Th priming contributed to NKT-enhanced Ab responses.18 We therefore expect that CD1d+ DCs are involved during the earliest phases of NKT-enhanced Ab production (Th priming) and that CD1d+ B cells play a major role in stimulating NKT cells to provide direct B-cell help.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Dr Mark Exley (Harvard Medical School) for providing CD1d−/− mice, the NIAID tetramer facility (Emory University, Atlanta, GA) for supplying the CD1d tetramers, Dr Carol Webb (Oklahoma Medical Research Foundation) for critical review of the manuscript, and Hemangi B. Shah (University of Oklahoma Health Sciences Center) for helpful discussions.
This work was supported by the American Lung Association (Research Grant RG-21 019-N) (M.L.L.) and the National Institutes of Health (Grant 5P20RR015564-07) from the National Center for Research Resources.
National Institutes of Health
Authorship
Contributions: G.A.L, T.S.D., and M.L.L. performed experiments. G.A.L. and M.L.L. designed experiments and interpreted results; and G.A.L. and M.L.L. wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Mark L. Lang, Department of Microbiology and Immunology, BMSB1035, University of Oklahoma Health Sciences Center, 940 Stanton L. Young Blvd, Oklahoma City, OK 73104; e-mail: mark-lang@ouhsc.edu.