Abstract
In adaptation of risk-directed combined chemotherapies, the initial remission rate in treatment of childhood acute lymphoblastic leukemia (ALL) has exceeded 95%. Hematological relapse during maintenance therapy, in which methotrexate (MTX) and thiopurine are applied, is the major cause of treatment failure. A retrospective study was performed to evaluate the role of pharmacogenomic effects in the treatment of children with ALL in the southern Chinese population. A total of 105 Taiwanese children with ALL, who received combined chemotherapy of different intensities based on risk-directed Taiwan Pediatric Oncology Group (TPOG)-ALL-93 protocols between Oct. 1993 to Dec. 2001, were recorded in long-term follow-up (6.5 to 13.7 years) for events (hematological relapse or death) occurrence (Figure 1). Seventeen genetic polymorphisms in 13 pharmacogenomic targets that implicated in MTX/thiopurine metabolism were analyzed by PCR-based restriction length polymorphism (RFLP) or sequence-specific oligonucleotide (SSO) probe hybridization. Pharmacogenomic polymorphisms were correlated with long-term event-free survival (EFS) of patients, with confounding effects adjusted by multivariate regression. Homozygosity of the 2677–3435 G-C allele in the multi-drug resistance gene (MDR-1, ABCB1) was highly associated with a significant reduction in long-term EFS in those patients treated with the standard risk protocol (TPOG-ALL-93-SR) (Figure 2). In the 36 patients receiving TPOG-ALL-93-SR treatment protocol, 6 out of 12 (50%) subjects carried homozygotic MDR1 2677–3435 G-C/G-C genotype suffered hematological relapse in 2 years, compared to 21 of 24 (88%) the non-homozygotic subjects remained event-free after 5 years (hazard ratio: 6.8, p=0.01). Among patients treated with the a high risk protocol (TPOG-ALL-93-HR) due to the presence of myeloid markers on the leukemic cells or manifested central nervous system leukemia, the thymidylate synthase (TYMS) enhancer 28-bp repeats 3R3R, and the glutathione-S-transferase M1 (GSTM1) null genotypes were associated with inferior clinical outcomes (p=0.029 and 0.058, respectively). Moreover, for patients with T-cell ALL that received the very high risk protocol (TPOG-ALL-97-VHR), the methionine synthase reductase (MTRR) 66AA genotype correlated with a superior prognosis compared to the AG or GG genotypes. These findings indicated independent pharmacogenomic determinants could be identified in subsets of Taiwanese children with ALL and correlated to the treatment outcome. In conclusion, we propose the pharmacogenomic determinants disclosed in the context of TPOG-ALL-93 protocols could be used to refine protocols for the treatment of pediatric ALL patients.
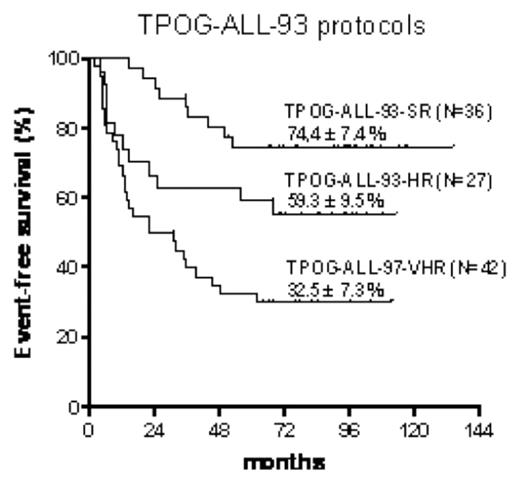
Kaplan-Meier plot depicting event-free survival of the ALL patients. The estimate of five-year event-free-survival (and the standard error) is illustrated for 105 patients received TPOG-ALL-93 protocols.
Kaplan-Meier plot depicting event-free survival of the ALL patients. The estimate of five-year event-free-survival (and the standard error) is illustrated for 105 patients received TPOG-ALL-93 protocols.
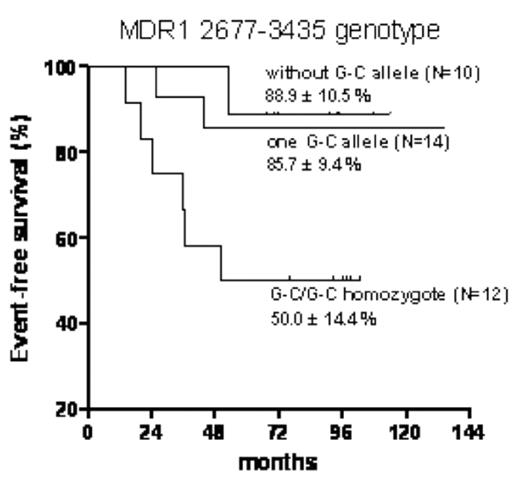
MDR1 2677–3435 G-C/G-C genotype identified a subset with poor prognosis in the 36 ALL patients received TPOG-ALL-93-SR treatment protocol.
MDR1 2677–3435 G-C/G-C genotype identified a subset with poor prognosis in the 36 ALL patients received TPOG-ALL-93-SR treatment protocol.
Disclosures: No relevant conflicts of interest to declare.
Author notes
Corresponding author