Abstract
The pivotal role of tumor-associated macrophages (TAMs) in tumor progression is now well established. TAMs have been shown to influence multiple steps in tumor development including the growth, survival, invasion, and metastasis of tumor cells as well as angiogenesis and lymphangiogenesis in tumors. The molecular circuits that polarize TAMs toward such a protumoral phenotype are now the focus of intense investigation. The transcription factor, nuclear factor–κB (NF-κB), is a master regulator of many cellular processes and been shown to regulate various pathways that impact on the function of TAMs. Much evidence for this has come from the use of elegant transgenic murine tumor models in which modification of single components of the NF-κB signaling pathway has been shown to regulate the pro-tumor repertoire of TAMs. Here, we outline this evidence and attempt to reconcile the various views that have emerged recently over the exact role of NF-κB in this phenomenon.
Introduction
Solid tumors consist of both malignant cells and a number of nonmalignant stromal cell types, including endothelial cells, fibroblasts, and various cells derived from the bone marrow. Complex interactions occur between these within the tumor microenvironment and impact on tumor growth, progression, metastasis, and angiogenesis.1 There is a marked myeloid cell infiltrate in most tumors, and activation of these is now known to play a key role in tumor progression.2,3 A cell that has achieved considerable prominence recently is the tumor-associated macrophage (TAM). These are recruited into tumors as monocytes from the bloodstream by the release of chemoattractants, such as CCL2, vascular endothelial growth factor, and CXCL12 (SDF1) by both the malignant and stromal tumor compartments.2,4,5 Early work by Mantovani et al showed that CCL2-induced recruitment of monocytes into fibrosarcomas enhanced tumor growth.6 Furthermore, reduced TAM infiltration into PyMT murine mammary tumors inhibited both tumor angiogenesis and metastasis.7,8 These findings are supported by many studies correlating increased numbers of macrophages with increased tumor angiogenesis and/or reduced patient survival in patients with various solid human tumors.3,4,9 This may also be the case for hematologic malignancies, such as follicular lymphoma as a gene expression signature associated with monocyte/macrophages was identified in follicular lymphoma patients with poor overall survival.10,11
Macrophages are highly versatile, multifunctional cells whose function depends on their anatomic location as well the physiologic or pathophysiologic context in which they are studied.12,13 Monocytes are recruited from the circulation into normal healthy tissues or at sites of injury, inflammation, infection, or malignancy where they then differentiate into tissue macrophages. They then acquire a distinct phenotype and activation status in response to factors present in the local tissue microenvironment. They are described as being “classically” activated by microbial products or interferon γ (IFN-γ) to express an M1 phenotype and express high levels of proinflammatory cytokines and major histocompatibility complex molecules, and are capable of killing of pathogens and tumor cells.14 On the other hand, stimulation with TH2 cytokines, such as interleukin (IL)–4, IL-10, and IL-13, drives macrophages toward an “alternatively” activated or M2 phenotype. In this state, they moderate the inflammatory response, promote angiogenesis and tissue remodeling, and clear cell debris.14,15 However, more recently, the plasticity of macrophage phenotypes has been acknowledged by the subdivision of the M2 classification into M2a, M2b, and M2c subgroups according to their inducing stimuli. M2a (induced by exposure to IL-4 and IL-13) and M2b (induced by combined exposure to immune complexes and Toll-like receptor [TLR] or IL-1R agonists) exert immunoregulatory functions and drive type II responses, whereas M2c macrophages (induced by IL-10) are more related to suppression of immune responses and tissue remodeling.16
Several studies have indicated tumors “educate” monocytes as they are recruited across the tumor vasculature to exhibit an alternatively activated, M2-like phenotype.15 For example, TAMs down-regulate both their expression of major histocompatibility complex class II and their ability to present antigen. They also show reduced antimicrobial and tumoricidal activity, while increasing production of mediators that promote angiogenesis, such as vascular endothelial growth factor and cyclo-oxygenase-2 (COX-2)–derived prostaglandin E2, as well as the anti-inflammatory cytokine IL-10. Another hallmark feature of alternative activation expressed by murine TAMs is the low expression of IL-12 and up-regulated levels of M2-specific genes, such as arginase-1 (Arg-1), macrophage galactose-type C-type lectin–2 (Mgl2), Fizz1, and Ym1.15,17
It should be noted, however, that recent studies have suggested that the activation status of TAMs may vary with both tumor type and stage of development, as well as being modulated by local signals within the tumor microenvironment, such as tumor necrosis factor α (TNF-α) and hypoxia.4,18,19 This diversity of function is evident from the variety of molecules expressed by TAMs, ranging from many proinflammatory (predominantly M1-like) functions, immunosuppressive (M2-like) characteristics, and even “mixed” phenotypes in some experimental and human tumors.4,17 The expression of markers of both classic and alternative activation has been observed for TAMs in murine tumors. For example, increased expression of inducible nitric oxide (iNOS or NOS2, an enzyme expressed by M1 macrophages) together with elevated levels of Arg-1 were observed in TAM (compared with splenic macrophages) in CT26 murine colon tumors and MethA− sarcoma.20 Similarly, coexpression of high levels of both NOS2 and Arg1 was noted in TAM in irradiated, early-stage murine prostate tumors.21
TAM, through the expression of suppressive factors such as IL-10, transforming growth factor-β (TGF-β), and Arg-1, prostaglandins were also suggested to contribute to suppression of T-cell activation and proliferation.22-24 IL-13–polarized TAMs have been shown to suppress T-cell activation in 4T1 mammary tumors.23 Similarly, in another study, TAMs from ovarian carcinoma have been shown to express the chemokine CCL22, which mediates trafficking of T-regulatory cells to the tumor.25 These observations indicate toward a role of TAM in subversion of adaptive immunity.
Myeloid-derived suppressor cells (MDSCs) are a population of immunosuppressive myeloid cells that are markedly increased both in both mouse models of cancer and in patients with head and neck, breast, non–small-cell lung, and renal cancers.26-29 Murine MDSCs are CD11b+, Gr-1+, IL-4α+, F4/80− cells with increased arginase activity and production of reactive oxygen species but are characterized mainly by their potent immunosuppressive functions.30-32 Characterization of human MDSCs is slightly more difficult to phenotype, as they appear to be a more variable population. MDSCs originate in the bone marrow from common myeloid progenitor and often differentiate into in CD11b+ Gr1medF4/80low/−IL-4Rα+ cells. Increased numbers appear in the blood during tumor growth in mice, with some being recruited to the tumor site where they often express the macrophage marker F4/80. Indeed, some studies have even suggested that they may differentiate into TAMs at such sites.20,31 The immunosuppressive repertoire of MDSCs is manifold. They inhibit CD8 T-cell activation by the expression of NOS2 and Arg1,30 induce the development of CD4+FOXP3+ T-regulatory cells,33 and induce an M2 polarization of TAMs through their release of high levels of IL-10.28 Knockout studies in IL-4Rα−/−, IL-13–deficient, STAT6−/−, and IFN-γ−/− mice have demonstrated the crucial role of IL-13/IL-4Rα and IFN-γ–mediated signaling pathways in promote MDSCs.23,34 In both the C26-GM and 4T1 tumor models, IL-13 (released by tumor cells and/or NKT cells) signals through the IL-4Rα/STAT6 pathway to drive Arg1 expression in MDSCs and TAMs, whereas IFN-γ (derived from NK cells or activated CD4+ T cells) triggers the expression of NOS2 in these cells.23,34
As can be seen, similarities between MDSCs and M2 macrophages include expression of Arg1, elevated levels of IL-10, inhibition of T-cell response, and promotion of angiogenesis.35 This argues against being too simplistic in the designation of myeloid cells such as TAMs or MDSCs into phenotypic categories or activation states (eg, M1 or M2).
Molecular pathways shaping macrophage polarization: the central role of NF-κB
The signaling pathways supporting the protumor functions of TAMs and the activation of these by distinct tumor-derived signals are now under considerable scrutiny.36 The transcription factor, nuclear factor–κB (NF-κB), has emerged as a central regulator of TAM function. This family of transcription factors, consists of 5 members: RelA (p65), c-Rel, RelB, p50, and p52 (which couple to form various dimers), although it should be noted that only RelA, RelB, and c-Rel contain transactivation domains and are therefore capable of activating transcription. Transgenic mice bearing deletions in each of these genes have been generated, but the exact physiologic role of different NF-κB dimers has proved difficult to elucidate because of functional compensation and redundancy within the Rel family.37
NF-κB activation is critical for macrophage responses to microbial/inflammatory stimuli, including TLR ligands, TNF-α, and IL-1β.16,38 Activation of NF-κB is regulated through the inhibitor of κB kinase (IKK) complex, which consists of 2 kinases, IKKα and IKKβ, and a regulatory protein IKKγ (or NEMO). During canonical NF-κB signaling, upstream signals converge at the IKK complex, phosphorylating IKKβ, which in turn phosphorylates the inhibitory molecule, inhibitor of κB (I-κB), resulting in its proteosomal degradation. This releases the p65/p50 NF-κB heterodimer, allowing its nuclear translocation and promoter binding for inflammatory gene transcription (Figure 1). However, many inflammatory cytokine genes require the activation of a number of transcription factors for maximal expression.39-43 For example, gene promoters for IL-12p40, NOS2, and CXCL10 contain one or more interferon-stimulated response elements, γ-activated sites, AP-1 response elements, and κB consensus sequences, which require the binding of signal transducer and activator of transcription 1 and 2 (STATs 1 and 2), interferon response factors (IRF1, 3, 5 7), AP-1, and NF-κB for their transcription.41,44-46 Production of type I IFNs downstream of TLR3, 4, 7, 8, and 9 forms an important autocrine loop for the expression of these genes.40,42 In macrophages through activation of STATs 1 and 2, the paracrine production of IFN-γ by NK or T cells also promotes increased expression of these genes through STAT1 activation.
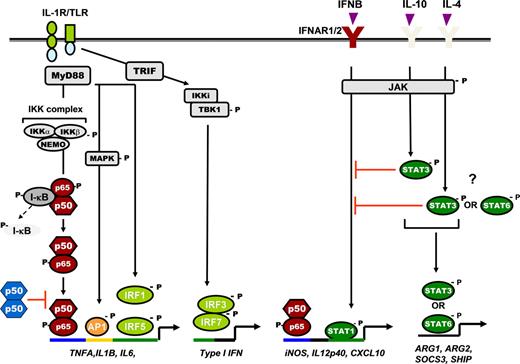
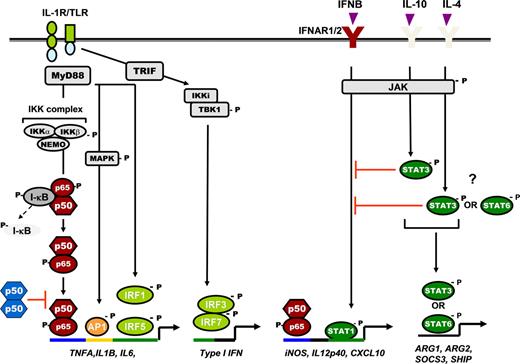
Schematic representation of NF-κB and STAT signaling pathway in regulation of cytokine gene expression in macrophages. TLR/IL-1R pathway, IL-1R, and all TLRs (except TLR3) signal through the adaptor protein MyD88, leading to NF-κB activation and transcription of proinflammatory cytokine genes. It also triggers activation of MAPKs (eg, JNK, p38) and its downstream transcription factor, AP-1, which coregulates proinflammatory cytokine expression. The TRIF or MyD88-independent signaling pathway triggered by TLR4 or TLR3 signals via the adaptor protein TRIF, which induces a phosphorylation cascade involving activation of IKKi, TBK1, and downstream transcription factor IRF3, leading to IFN-β transcription. IFN-β in turn binds to its cognate receptor (IFNAR1/2) and induces STAT1-mediated expression of IFN-inducible chemokine genes. IFNAR1/2 also serves as the receptor for IFN-α and IFN-γ. The requirement for IRFs is also shown. STAT3 and STAT6 activation induced by IL-10, IL-4, and IL-13 regulates the expression of several M2 genes and also inhibits STAT1 induction. p50 NF-κB homodimers also negatively regulated TLR/IL-1R signaling. Distinct transcription factor specific binding consensus sequences on gene promoters are indicated with different colors. “P” indicates phosphorylation; red T-bars indicate negative regulation.
Schematic representation of NF-κB and STAT signaling pathway in regulation of cytokine gene expression in macrophages. TLR/IL-1R pathway, IL-1R, and all TLRs (except TLR3) signal through the adaptor protein MyD88, leading to NF-κB activation and transcription of proinflammatory cytokine genes. It also triggers activation of MAPKs (eg, JNK, p38) and its downstream transcription factor, AP-1, which coregulates proinflammatory cytokine expression. The TRIF or MyD88-independent signaling pathway triggered by TLR4 or TLR3 signals via the adaptor protein TRIF, which induces a phosphorylation cascade involving activation of IKKi, TBK1, and downstream transcription factor IRF3, leading to IFN-β transcription. IFN-β in turn binds to its cognate receptor (IFNAR1/2) and induces STAT1-mediated expression of IFN-inducible chemokine genes. IFNAR1/2 also serves as the receptor for IFN-α and IFN-γ. The requirement for IRFs is also shown. STAT3 and STAT6 activation induced by IL-10, IL-4, and IL-13 regulates the expression of several M2 genes and also inhibits STAT1 induction. p50 NF-κB homodimers also negatively regulated TLR/IL-1R signaling. Distinct transcription factor specific binding consensus sequences on gene promoters are indicated with different colors. “P” indicates phosphorylation; red T-bars indicate negative regulation.
Many cytokines and growth factors signal through a related superfamily of cell-surface receptor tyrosine kinases that trigger the Janus kinases (JAK)/STAT pathway47 (Figure 1). Ligand-induced dimerization of the cytokine receptor induces the reciprocal tyrosine phosphorylation of the receptor-associated JAKs, which, in turn, phosphorylates tyrosine residues on the cytoplasmic tail of the receptor. These phosphorylated tyrosines serve as docking sites for the Src homology-2 domain of the STAT protein, and JAK catalyzes the tyrosine phosphorylation of the receptor-bound STAT. Phosphorylation of STAT induces their homodimerization or heterodimerization, followed by nuclear translocation and binding to the specific DNA response elements in the promoter region of target genes to activate gene expression. M2 stimuli, like IL-4, IL-10, and IL-13, stimulate distinct members of the STAT signaling proteins in macrophages. IL-4 and IL-13 activate STAT6,48 whereas IL-10 activates STAT3 in macrophages.49 Activation of these STAT members is linked to the transcription of several M2-specific genes, such as arginase-1, arginase-2, suppressor of cytokine signaling 3, Src homology-2-containing inositol phosphatase.36,48,50 In addition, STAT3 and STAT6 can inhibit NF-κB and STAT1 activation pathways leading to immunosuppression (Figure 1).36,50
Macrophage plasticity in tumor initiation and progression: the many faces of NF-κB
Activation of NF-κB in macrophages has been shown to be required for the onset of tumor development in several inflammation-induced cancer models.50-55 It is thought that, during chronic inflammation, NF-κB activation in inflammatory macrophages results in the release of cytokines, such as TNF-α and IL-6,54,56 which not only trigger prosurvival signals in tumor cells (through NF-κB or STAT3 activation in these cells) but also support their growth and progression.2,52,57,58 Experimental validation for this concept has come from elegant genetic studies of murine hepatocellular carcinoma and colitis-associated colon cancer.51,54,55 These are discussed in more detail below.
In the case of hepatocellular carcinoma, several studies have demonstrated that inhibition of NF-κB pathway in liver macrophages (Kupffer cells) by genetic deletion of IKKβ,54 or the inhibition of inflammatory cytokines, such as TNF-α produced by these cells,55 results in a marked reduction in tumor onset and tumor load. Alternatively, inhibition of NF-κB in hepatocytes by deletion of IKKβ,54 NEMO,53 or expression of an IκBα super-repressor,55 sensitized them to increased cell death, compensatory proliferation, predisposition to malignancy, and increased tumor load. NF-κB inhibition in hepatocytes results in inhibition of antiapoptotic genes, such as Bcl-XL and GADD45β, triggering increased cell death. Detection of these dying cells by Kupffer cell induces them to release more inflammatory cytokines, such as TNF-α and IL-6, which then support the growth and survival of malignant hepatocytes.52
Finally, in a dextran sodium sulfate–induced colitis-associated colorectal tumors, Greten and colleagues52 demonstrated that myeloid- or macrophage-specific depletion of IKKβ (through IKKβΔLysM mice) reduced both the size and number of tumors, correlating with decreased premalignant enterocyte proliferation. In this model, the oral administration of dextran sodium sulfate disrupted the intestinal endothelial lining, exposing the lamina propria macrophages to activation by enteric bacteria in the gut. Activation of these cells through NF-κB pathway led to release of inflammatory mediators that support tumorigenesis, including COX-2–derived PGE2 and IL-6. This finding was confirmed by the demonstration that neutralizing antibodies to the IL-6 receptor also reduced tumor growth.
These studies clearly emphasize the essential requirement of NF-κB activation in both TAMs and tumor cells for maintaining the inflammatory circuit(s) that promote tumor initiation and growth. In particular, NF-κB activation in TAMs shapes their protumoral repertoire, including the release of TNF-α, IL-6, and IL-1β, which sustain tumor cell growth and survival.4,18,36
However, an interesting picture of macrophage plasticity is now emerging, one in which these cells show essentially a proinflammatory phenotype in the early stages of inflammation-induced tumor initiation, then switch in established tumors to exhibit an alternate, immunosuppressive phenotype.36 Indeed, the immunosuppressive nature of TAMs is now well established. Furthermore, their defective expression of inflammatory cytokines, reactive nitrogen intermediates, and decreased tumoricidal activity in mouse mammary carcinoma has been demonstrated.59 Similarly, Sica et al have shown that TAMs from both established chemically induced murine fibrosarcoma and primary human ovarian carcinoma display an IL-10high/IL-12low phenotype.24,60 Hagemann et al also showed that ovarian cancer cells can polarize macrophages to express a tumor-promoting rather than tumor-inhibiting phenotype in vitro associated with a alternatively activated IL-10high/IL-12low phenotype.61,62 Finally, TAMs in Lewis lung carcinoma show significantly up-regulated expression of IL-10 and suppressed levels of the proinflammatory cytokines TNF-α, CCL3, CCL4, and IL-6, compared with peritoneal macrophages from normal mice.18 Further discussion of the plasticity of TAMs is beyond the scope of this review, so the reader is referred to several recent publications on this topic.4,18,36
The molecular characterization of TAMs in established murine fibrosarcomas has suggested that their tumor-promoting, M2-like phenotype develops because of defective NF-κB activation (Figure 2).60,63 Transcriptomal studies showed drastic down-regulation of several NF-κB–inducible genes, such as IL-12p40, TNF-α, CCL3, and IL-6 in TAMs on lipopolysaccharide (LPS) treatment in vitro. Defective NF–κB activation in the LPS-treated TAMs was evident from inhibition of p65 nuclear translocation and delayed phosphorylation of IκBα. Saccani et al also demonstrated that high levels of nuclear p50/p50 homodimers are responsible for the defective expression of NF-κB–inducible genes and the IL-10high/IL-12low M2-like phenotype.63 Although p50 is essential to the expression of many NF-κB target genes, as a binding partner for RelA (p65) and cRel, and as a homodimer or p52 heterodimer in complex with Bcl-3-p50 homodimers, it has also been described to inhibit NF-κB–regulated genes. It is thought that the p50/p50 homodimers (which lack the transactivation domain) compete with canonical p65/p50 heterodimers for the κB-binding sites on the inflammatory gene promoters, thereby blocking p65/p50 promoter binding and gene transcription. This mechanism has been described in the experimental phenomenon of LPS-induced tolerance and particularly inhibition of TNF-α expression.64 Saccani et al demonstrated that depletion of p50 in TAMs resulted in decreased tumor growth and prolonged host survival because of p50−/− TAMs showing an antitumoral, proinflammatory M1 phenotype of increased IL-12p40 and TNF-α, but decreased IL-10 production63 on IFN-γ/LPS treatment in vitro. Indeed, various studies have indicated that inhibition of NF-κB activation helps to drive the tumor-promoting phenotype of TAMs.63 Colombo et al, for example, reported that redirecting M2-like TAMs (and dendritic cells) toward an M1-like phenotype (by reactivating NF-κB using the TLR9 ligand CpG and an IL-10 receptor–specific antibody) induced significant antitumor immunity in a murine mammary carcinoma model.65,66 These studies are supported by the finding that 3′-methylcholanthrene–induced fibrosarcoma developed more rapidly in IL-12p40−/− mice and more slowly in IL-10−/− mice.67
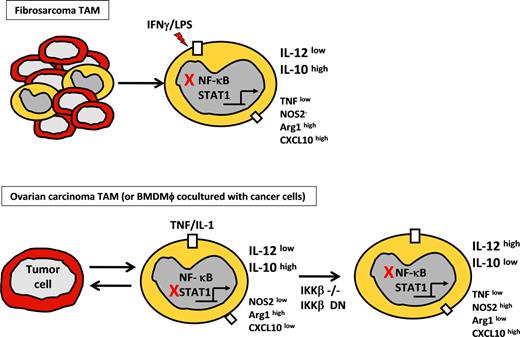
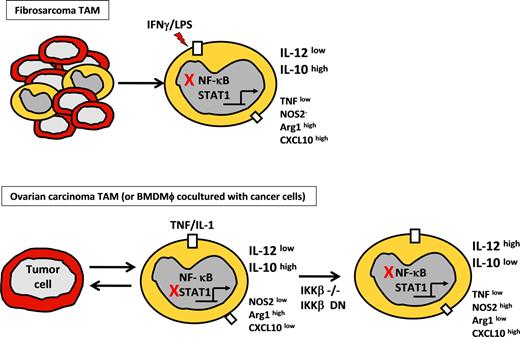
Phenotype of tumor-polarized macrophages from fibrosarcoma and ovarian carcinoma models. (Top panel) TAM phenotype from chemically induced fibrosarcoma model showing modulation of selected genes after LPS stimulation, compared with normal peritoneal macrophages. Red X indicates inhibition of NF-κB activation resulting from p50 homodimer overexpression. High or low expression is relative to that in peritoneal macrophages from non–tumor-bearing mice. (Bottom panel) Phenotype of bone marrow–derived macrophages or TAMs from ovarian carcinoma model on coculture with ID8 ovarian carcinoma cells. Red X indicates inhibition of STAT1 by IKKβ activation. (Right panel) Inhibition of NF-κB was done by means of targeted deletion of IKKβ (IKKβ−/−) or IKKβ-dn expression.
Phenotype of tumor-polarized macrophages from fibrosarcoma and ovarian carcinoma models. (Top panel) TAM phenotype from chemically induced fibrosarcoma model showing modulation of selected genes after LPS stimulation, compared with normal peritoneal macrophages. Red X indicates inhibition of NF-κB activation resulting from p50 homodimer overexpression. High or low expression is relative to that in peritoneal macrophages from non–tumor-bearing mice. (Bottom panel) Phenotype of bone marrow–derived macrophages or TAMs from ovarian carcinoma model on coculture with ID8 ovarian carcinoma cells. Red X indicates inhibition of STAT1 by IKKβ activation. (Right panel) Inhibition of NF-κB was done by means of targeted deletion of IKKβ (IKKβ−/−) or IKKβ-dn expression.
However, it should be noted that recent studies by Hagemann et al have demonstrated the requirement for IKKβ to maintain the IL-10high/IL-12low phenotype of TAMs in a mouse model of ovarian cancer62 (Figure 2). They showed that targeted deletion or inhibition of IKKβ in TAMs increased their tumoricidal activity through elevated NOS2 expression and IL-12–dependent recruitment and activation of NK cells. Because increased expression of IL-12, NOS2, and enhanced tumoricidal activity are associated with M1 characteristics, their data imply that inhibition of the IKKβ/NF-κB pathway promotes an M1-like phenotype in TAMs, whereas intact IKKβ/NF-κB activation maintains these cells in an alternative, tumor-promoting activation state. The apparent contradiction between the p50−/− TAM studies of Sica et al63 and these IKKβ studies by Hagemann et al62 is discussed in greater detail in the next section.
Understanding the apparent paradox
Many of the proinflammatory genes associated with the M1 phenotype, including IL-12p40 and NOS2, are well-known NF-κB–inducible genes. However, in addition to NF-κB, these genes possess STAT1/2-binding sites on their promoter.68 Hagemann et al suggested that IKKβ through inhibition of STAT1 causes the down-regulation of these genes, which is linked to decreased tumoricidal activity and the IL-12low/IL-10high, M2 phenotype.62 This accords well with the recent finding that IKKβ inhibits STAT1 activation, IL-12p40, and NOS2 expression in M1 macrophages.68 In line with these data, IKKβ−/− macrophages and IKKβ dominant-negative expressing TAMs cocultured with ID8 ovarian tumor cells exhibit higher levels of STAT1 activation than wild-type or mock-treated macrophages. Interestingly, Biswas et al showed that TAMs from murine fibrosarcomas also display higher basal levels of STAT1 activation and expression of STAT1-regulated chemokine genes, CCL5 and CXCL10.60 However, as opposed to the observation of an IL-12high/IL-10low phenotype of IKKβ-targeted TAMs in the ID8 tumor model, TAMs in murine fibrosarcoma (on LPS treatment in vitro) were IL-12low/IL-10high. There may be several reasons for this apparent discrepancy. First, the way in which macrophages were stimulated ex vivo was markedly different in the aforementioned studies. The IL-12p40high/IL-10low phenotype of IKKβ-targeted macrophages was observed after their coculture with ID8 tumor cells, or adoptive transfer of IKKβ-targeted macrophages into established tumors, whereas the IL-12low/IL-10high phenotype of both murine fibrosarcoma and human ovarian carcinoma TAMs was observed after IFN-γ/LPS stimulation in vitro.24
Second, the macrophage populations used in both of these studies were different. The ovarian tumor study used TAMs isolated directly from human/murine ovarian tumor ascites.62,69 In further mouse studies, Hagemann et al used both naive and tumor-educated macrophages.62 IKKβ was targeted in bone marrow–derived macrophages, by Cre/lox recombination or expression of an IKKβ-dn, before adoptive transfer to tumor-bearing mice. TAMs isolated from ascites of tumor-bearing mice were also used. The phenotype of these macrophages reflected alternative activation. These TAMs were adoptively transferred to tumor-bearing mice after infection with an IKKβ-dn adenovirus in vitro. Thus, IKKβ was targeted in both established TAMs and in naive macrophages before exposure to the tumor microenvironment. However, in previous studies, TAMs were isolated directly from either murine fibrosarcoma or human ovarian carcinoma, and stimulated in vitro with LPS/IFN-γ.24,63 Thus, the different source of cells used in the aforementioned studies (ie, blood monocytes, monocyte-derived macrophages, bone marrow–derived or peritoneal macrophages, ascites-derived macrophages or TAMs from solid tumors) must be taken into account comparing (or contrasting) their function. Despite these different experimental conditions, the results from studies in both ovarian carcinoma and fibrosarcomas are surprisingly complementary, although the mechanistic interpretation applied to the data may have differed. As mentioned before, the phenotype of TAMs can differ markedly between different tumor types4 and different stages of tumor development.18
It should also be remembered that macrophages isolated from transgenic mice lacking a single part of the NF-κB signaling cascade (ie, p50) may compensate in various ways for the long-term (ie, chronic) absence of an individual gene, so it is important to confirm the effects of such a genetic manipulation using short-term knockdown studies or transient inhibition NF-κB, for example, using the IKKβ-dn inhibitor.62 The IL-12low/IL-10high phenotype of TAMs from murine fibrosarcoma stimulated with LPS/IFN-γ in vitro was compared with that of peritoneal macrophages from non–tumor-bearing mice (as control cells). In the case of ovarian carcinoma, the IL-12low/IL-10high macrophage phenotype has been characterized after coculture with both human and mouse ovarian cancer cell lines in the absence of additional stimuli to reflect the tumor microenvironment.61,62 In further studies, IKKβ was targeted with recombinant adenoviruses in both TAM from ovarian tumors and macrophages cocultured with syngenic ovarian cancer cells, using mock-infected TAM/macrophages as controls. Resident peritoneal macrophages may have been a more appropriate control for TAM in epithelial ovarian cancer. However, because TAMs are derived from bone marrow progenitors recruited to the tumor by tumor-derived chemokines, the effects of malignant cells on the phenotype of bone marrow–derived macrophages may be equally appropriate.
Interestingly, LPS/TLR4 signaling through the MyD88 and TRIF pathways in macrophages leads to activation of NF-κB and through autocrine production of IFN-β also STAT1.70 LPS induced IL-12 production in macrophages is dependent on IFN-β, illustrating the critical role for STAT171 (T.L., unpublished observations, December 2006). Although the promoter of IL-12 gene requires both κB and interferon-stimulated response element (STAT1/STAT2) binding for maximal expression in reporter assays,41,43 it is possible that NF-κB binds the IL-12p40 promoter but only activates transcription minimally, acting more as a cofactor whereas full induction of IL-12 requires STAT1. IL-12 induction is increased in p50−/− p65+/− macrophages.72 Residual NF-κB activation after Cre/lox-mediated deletion of IKKβ, or expression of the IKKβ-dn, may be enough for the cofactor role of NF-κB on the p40 promoter.
Both TAM isolated from ovarian tumors and macrophages cultured with ovarian cancer cells showed up-regulated expression of the tumor-promoting cytokine, TNF-α.73 The effect of IKKβ inhibition on tumor cell–polarized macrophages was observed to be selective at the level of gene expression. Whereas NOS2 and IL-12 were up-regulated, expression of TNF-α was down-regulated in these cells.62 In TAMs from fibrosarcomas, defective NF-κB activation (on stimulation ex vivo) was seen to be concomitant with down-regulated TNF-α (Figure 2). Interestingly, TNF-α has been shown to have an antitumor role in models of fibrosarcoma.67,74 In these studies, TNF-α−/− mice showed higher tumor incidence, whereas transgenic expression of TNF-α by tumor cells alone arrested tumor growth.67,75 These observations illustrate the difficulty in drawing parallels between different tumor models. Wu et al75 used IFN-γ−/−, STAT1−/−, and IRF1−/− mice to also demonstrate that TNF-α-induced long-term suppression of tumor growth required a STAT1- and IRF1-dependent endogenous IFN-γ pathway. This observation is particularly relevant in light of the central role of STAT1 in regulating the tumor-promoting functions of TAMs.60,62
Defining tumor-polarized macrophages as M1- or M2-polarized cells based on the status of cytokine expression alone may not be an accurate assessment of their phenotype and function.14 It would be interesting to determine whether IKKβ or p50 is required for alternative activation of macrophages (as assessed by such markers as TGF-β, Fizz1, Ym1, and Mgl2).17,76,77 Neither Saccani et al63 nor Hagemann et al62 have shown that NF-κB regulates expression of M2 genes. However, it is clear that STAT1 regulates expression of M1 genes, IL-12, NOS2, CIITA, and probably other ISGs associated with the TAM phenotype, such as CXCL10 and CCL5.
Whereas the data reviewed here on the TAM repertoire in knockouts for NF-κB–related molecules (eg, p50, IKKβ) have clearly indicated the central role of this pathway in the function of these cells, identifying the tumor-derived factors that trigger this pathway in vivo has been difficult. Many studies have resorted to the ex vivo stimulation of macrophages or TAMs with specific cytokines known to be abundant in the tumor microenvironment (eg, IL-1β, TNF-α, and CD40L)24,60,63,78 or the in vitro coculture of macrophages with tumor cells.61,62,79 However, it remains to be seen whether these actually mimic the complex array of factors influencing macrophage function in tumors. Studying the effect of these factors in tumors grown in transgenic mice in which they have been “knocked out” has been argued to be a more physiologically relevant approach. For example, the contribution of IL-1β, an important NF-κB–inducing cytokine, in the tumor microenvironment to the regulation of TAM function has recently been investigated in several transgenic mouse models. In carcinogen-induced tumors grown in IL-1β–deficient mice, tumor incidence was lower and tumor growth retarded compared with wild-type mice. This correlated with reduced tumor macrophage infiltration in IL-1–deficient mice. Hagemann et al also showed that macrophages from IL-1R−/− mice failed to promote tumor growth in a syngeneic model of ovarian cancer compared with macrophages from wild-type mice.62 Interestingly, the tissue-specific (ie, gastric) expression of IL-1β results in local inflammation-induced cancer.79 Taken together, these studies suggest that IL-1β may stimulate the tumor-promoting functions of both TAMs and MDSCs in murine tumors.
The many faces of NF-κB in TAMs
The role of NF-κB in regulating TAM function may be complex and operate at many levels, as well as being context- and gene-dependent. For example, p50 homodimer overexpression in TAMs may result in repression of certain NF-κB target genes (IL-12p40, TNF-α)63 but may increase expression of other genes required to maintain the TAMs phenotype. Indeed, p50 homodimer has been shown to be necessary for the transcription of COX-2, which is also reported to be up-regulated by TAMs.80-83 Similarly, the crucial involvement of p50 NF-κB in the induction of IL-10 through the TLR4/TPL2/ERK pathway has also been demonstrated recently.84 Ongoing studies using macrophages isolated from p50 NF-κB−/− transgenic mice by Sica et al are attempting to clarify further the exact role of this molecule in TAM regulation.
Interestingly, the outcome of NF-κB activation can be quite different depending on the context in which it is being activated. For example, a study using the murine carrageenin pleurisy model demonstrated that, whereas NF-κB activation during the onset of inflammation is associated with the expression of proinflammatory genes such as NOS2, the same activation during the resolution phase is associated with the expression of anti-inflammatory genes such as TGF-β1.85 TAMs have many functions in common with resolution, such as release of anti-inflammatory cytokines IL-10, TGF-β, tissue remodeling, and angiogenesis.57 Under these settings, it is probable that NF-κB activation would support an anti-inflammatory, immunosuppressive M2 phenotype as suggested by Hagemann et al.62 Furthermore, it should be noted that macrophages when cocultured with tumor cells may crosstalk with a plethora of other signals found in tumors, such as hyaluronan, necrotic cell debris, HSP60, fibronectin, and HMGB1, many of which lead to an IL-10high, immunosuppressive M2 phenotype.86-88
The functional TRIF/IRF3/IFN-β/STAT1 pathways in fibrosarcoma TAMs and down-regulation of STAT1 in IKKβ-inhibited ovarian tumor cell–polarized macrophages are an interesting area of overlap between the 2 studies. However, the functional significance of this pathway in tumor progression awaits thorough investigation. In this context, STAT1−/− F4/80+ TAMs from EL4 tumor model failed to express NOS2 and Arg I and inhibit T-cell response.20 Here, STAT1 was implicated in mediating the TAM-induced suppression of T-cell activation.
Hypoxia fine-tunes the TAMs phenotype through crosstalk with NF-κB
Another tumor-derived signal that appears to impact on NF-κB signaling in TAMs is hypoxia (extremely low oxygen tension). The presence of many areas of hypoxia is a hallmark feature of most forms of solid tumor.89 Moreover, TAMs have been shown to accumulate in these areas where hypoxia promotes their pro-tumor TAM phenotype.3,90,91 Hypoxia-inducible factor 1 (HIF-1) has been shown to essentially control the cellular response to hypoxia. Hypoxia stabilizes the inducible α-subunit, preventing posttranslational hydroxylation and subsequent degradation via the proteasome. In recent years, clear evidence has emerged that HIF-1α is also responsive to many stimuli under normoxic conditions, including thrombin, growth factors, vasoactive peptides, insulin, LPS, and cytokines such as TNF-α. One putative mechanism underlying these responses is the transcriptional regulation of HIF-1α by NF-κB. More recently, short-term exposure (2-4 hours) of murine bone marrow–derived macrophages to hypoxia has been shown to up-regulate NF-κB activity, which in turn up-regulates HIF-1α levels.92 This study used macrophages from IKKβ−/− mice to show that NF-κB is a critical transcriptional activator of HIF-1α and that basal NF-κB activity is required for HIF-1α protein accumulation under hypoxia.
In conclusion, it is probable that NF-κB activation in such diverse innate effectors as macrophages contributes to tumor progression in a variety of ways. Evidence for considerable plasticity in NF-κB function in these cells is now emerging. Whereas NF-κB–induced expression of inflammatory cytokines, such as TNF-α and IL-6, by macrophages has been found to contribute crucially to tumor cell survival, and proliferation in inflammation-linked tumor onset, the modulation of NF-κB activation in TAMs in established tumors maintains their immunosuppressive, tumor-promoting phenotype. This plasticity in the TAM phenotype may reflect the presence of different combinations of local signals also acting on TAM at sites of chronic inflammation versus established tumors, but also the unique ability of NF-κB to induce or repress different gene subsets in the same cells under different conditions. Further studies of the spatiotemporal regulation and role of NF-κB in TAMs are now warranted. Further insights may be gained by investigating the relative contribution of individual NF-κB members (p65, c-Rel, p50, BCL3) and their combinatorial transcriptional partners, such as STATs and IRF3, to the function of NF-κB in TAMs.
Acknowledgments
This work was supported by the Medical Research Council, United Kingdom (T.H., T.L.); Biomedical Research Council, Singapore (S.K.B.); Associazione Italiana Ricerca sul Cancro, Italy; Fondazione Berlucchi, Italy; Ministero Università Ricerca, Italy; Ministero della Salute, Italy (A.S.); Cancer Research United Kingdom; the Breast Cancer Campaign, United Kingdom; and Yorkshire Cancer Research, United Kingdom (C.E.L.).
Authorship
Contribution: T.H., S.K.B., T.L., A.S., and C.E.L. helped write this review.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Claire E. Lewis, Academic Unit of Pathology, University of Sheffield Medical School, Beech Hill Rd, Sheffield S10 2RX, United Kingdom; e-mail: claire.lewis@sheffield.ac.uk.
References
Author notes
*All authors contributed equally to this work.