Abstract
Abstract 1236
Poster Board I-258
Glucocorticoids (GC) apoptotic effect in chronic lymphocytic leukemia (CLL) cells is known for many years. However, in CLL their use is often confined to their immunosuppressive activity in order to control autoimmune phenomena or as palliation. The fact that GC, particularly dexamethasone (DEX), can overcome p53 mediated resistance to therapy has renewed the interest in the use of GC as therapeutic agents in CLL. GC apoptotic induced cell death mechanism seems to depend on cell type and few studies were performed in CLL. GC increasing importance as apoptotic agent in CLL prompted us to analyze DEX apoptotic activity in CLL cells in order to clarify GC apoptotic mechanisms. For this, peripheral blood samples from 45 patients with CLL were selected for in vitro studies. Patients were analyzed for IGHV mutational status and ZAP-70 expression. Tumour cells were cultured over 24 hours with DEX at 13.25uM and viability was then determined by surface annexin V binding and propidium iodide (PI) staining flow cytometry analysis. To determine early apoptotic signal onset, BIM mRNA GC induced expression was quantified at different time points by quantitative RT-PCR. Genome-wide expression profile of CLL cells was done to discriminate genes involved in DEX apoptotic action.
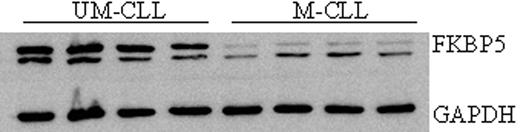
After 24 hours of exposure to therapeutic concentrations of DEX, cell viability was higher in mutated cases (M-CLL) than in unmutated IGHV cases (UM-CLL) (85.6% vs 69.5% in mean, respectively; p=0.000). mRNA BIM expression after 24 hours of treatment with DEX correlated with induced apoptotic cell death (R=0.496; p=0.000). As a consequence, UM-CLL had higher levels of induced mRNA levels of BIM than M-CLL cases (p=0.036). Time course experiments have shown that at 6h after DEX treatment BIM mRNA levels were induced 3 times without influence on cell viability. Genome-wide expression analysis of 12 CLL cases (7 UM-CLL, 5 M-CLL) was done after 6 hours of DEX treatment and was compared to the baseline gene expression. In both groups many genes were up and down regulated by DEX (UM-CLL 3359 genes and M-CLL 1008 genes, p adjusted < 0.01). Analysis of genes involved in the GC pathway revealed that basal mRNA levels of FKBP5, a protein essential to maintain the GC receptor (GR) complex suitable for GC binding, was more expressed in UM-CLL than in M-CLL cases (1.85 times, p= 0.027). Analysis by quantitative RT-PCR performed in 45 CLL patients to validate micro-array data confirmed that at baseline, FKPB5 expression was higher in UM-CLL than M-CLL (0.97 vs. 0.74 arbitrary units, respectively; p=0.042). The same was observed at protein level, WB analysis of FKBP5 basal levels showed that UM-CLL cases expressed more this protein than M-CLL (Fig. 1). In addition to that, genome-wide analysis revealed that GILZ, a glucocorticoid-induced leucine zipper, was differently induced by DEX in the studied groups. GILZ mRNA was less induced in M-CLL cases than in UM-CLL cases (difference fold change=0.52, p=0.0005). These results were also confirmed in 45 CLL cases by quantitative RT-PCR: in M-CLL, GILZ was induced 3.67 times whilst it was induced 5.62 times in UM-CLL cases (p=0.0001). In conclusion, treatment with GC induces more apoptotic cell death in UM-CLL than in M-CLL. As a downstream effect, BIM expression after GC exposure correlates with GC-induced apoptosis. Moreover, GC apoptotic effect in CLL is the result of several cell pathways imbalance as revealed by gene expression analysis. GILZ induction was proved to be necessary for DEX induced apoptosis in other cell types like multiple myeloma cell lines. In CLL, GILZ differential induction was observed at different degrees in GC-responders and non-responders. Finally, FKBP5 expression, upstream effecter of the GC pathway, correlated with cellular effect of GC and can be used to predict GC apoptotic activity in CLL cases.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.