Abstract
Abstract 1706
Poster Board I-732
in order to maintain efficacy and reduce toxicity of the treatment in elderly follicular lymphoma (FL) patients, we designed a study with a short chemo-immunotherapy R-FND with Rituximab consolidation followed by randomization between R maintenance or observation. Material and methods: From January 2004 to December 2007, 242 patients (age 60-75) with untreated advanced stage FL were enrolled by 33 IIL centres. Treatment plan was: 4 courses of R-FND (standard doses of Rituximab, Fludarabine, Mitoxantrone, Dexamethasone) every 28 days followed by 4 weekly Rituximab infusions as consolidation; responding (CR+CRu+PR) patients were randomized between Rituximab maintenance, one dose every 2 months for a total of 4 doses or observation. Qualitative and quantitative PCR monitoring for IgH/Bcl-2 rearrangement on bone marrow (BM) was performed at diagnosis, after R-FND and R consolidation and during maintenance/observation.
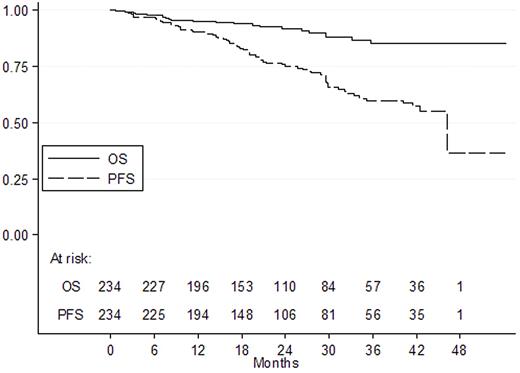
234 patients were eligible for the study: median age was 66 yrs; advanced stage II 14%, stage III 21% and stage IV 65%; BM involvement and B symptoms were documented in 55% and 18% respectively. FLIPI score was: Low 11%, Intermediate 34%, High 55%. One and 2 or more comorbidities were present in 36% and 23% of the patients respectively. Qualitative PCR analysis for IgH/Bcl-2 was performed in 223 patients at diagnosis and 51% were positive. Two hundred and two (86%) patients completed the induction treatment and were randomized between maintenance or observation; 32 were not because of: stable/progressive disease (16), adverse events (10) or other causes (6). Overall response at the end of treatment was 86% with 69% CR and 18% PR; PCR negativity at the end of treatment was 75%. Rituximab consolidation was able to induce CR in 37/90 (41%) partial responders and to increase PCR negativity from 61% to 75%. With a median follow-up of 22 months, two-years OS and PFS were 92% (95% CI 87%-95%) and 75% (95% CI 68%-81%), respectively (see Figure).
Two-years PFS rates according to FLIPI score were 85% for low/intermediate risk and 65% for high risk (p < 0.001); 2-years PFS rates were 57% and 79% respectively in patients with and without B symptoms (p < 0.001). The patients in more advanced decade (> 70 years) or those with 2 or more comorbidities did as well as younger ones or those with one or no comorbidities: 2-yr PFS rates for patients more or less seventy were 73% vs 76% (p = 0.39); for patients with ≥2 comorbidities or one or none were 84% vs 67% vs 76%, respectively (p = 0.82). A total of 1119 courses were delivered; the most frequent CTC grade 3-4 toxicity was neutropenia in 25% of the courses, with only 13 serious infections. One patients developed acute myelogenous leukaemia during treatment and died. Two toxic deaths during treatment (0.8%) occurred: 1 HBV reactivation and 1 Steven Johnson syndrome. So far 212 patients are alive; besides the above mentioned deaths, 15 patients died of lymphoma, 2 died of cardiac failure, 1 died of stroke and 1 died of drowning. So far too few events occurred to proper analyse the efficacy of the Rituximab maintenance. In the maintenance/observation phase the following severe (WHO grade 3-4) toxicities were recorded: 15 patients experienced neutropenia, seven cardiac events, four infections; no other relevant toxicities were recorded. The cumulative incidences of toxic events accounting for competing events at 18 months for maintenance arm (Arm A) versus observation arm (Arm B) were as follows: neutropenia 17% vs 1% (p<0.001); infections 2% vs 2% (p = 0.586); cardiac events 4% vs 4% (p= 0.627).
a short term chemo-immunotherapy R-FND + Rituximab consolidation is safe and effective with a good 2-yr PFS rate also in patients with high risk FLIPI score or in patients in more advanced decade or with comorbidities. Rituximab maintenance is safe with a more frequent neutropenia that was not associated with an increased risk of infections. Final results of the study will provide insights on the role of Rituximab maintenance after R-chemotherapy.
Vitolo:Roche: Lecture fees; Mundipharma: Lecture fees. Off Label Use: Study was supported by Roche: Rituximab maintenance in first line treatment is off-label. Boccomini:Roche: Lecture fees. Ladetto:Roche Italy: Research Funding; Amgen: Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.