Abstract
The transitional stage of B-cell development represents an important step where autoreactive cells are deleted, allowing the generation of a mature functional B-cell repertoire. In mice, 3 subsets of transitional B cells have been identified. In contrast, most studies of human transitional B cells have focused on a single subset defined as CD24hiCD38hi B cells. Here, we have identified 2 subsets of human transitional B cells based on the differential expression of CD21. CD21hi transitional cells displayed higher expression of CD23, CD44, and IgD, and exhibited greater proliferation and Ig secretion in vitro than CD21lo transitional B cells. In contrast, the CD21lo subset expressed elevated levels of LEF1, a transcription factor highly expressed by immature lymphocytes, and produced higher amounts of autoreactive Ab. These phenotypic, functional, and molecular features suggest that CD21lo transitional B cells are less mature than the CD21hi subset. This was confirmed by analyzing X-linked agammaglobulinemia patients and the kinetics of B-cell reconstitution after stem cell transplantation, which revealed that the development of CD21lo transitional B cells preceded that of CD21hi transitional cells. These findings provide important insights into the process of human B-cell development and have implications for understanding the processes underlying perturbed B-cell maturation in autoimmune and immunodeficient conditions.
Introduction
B-cell development begins in the bone marrow (BM) from pluripotent hematopoietic stem cells (HSCs), which progress through the pro– and pre–B-cell stages. Pre–B cells continue the developmental process by initiating Ig variable gene rearrangement and becoming an immature B-cell expressing a functional IgM B-cell receptor (BCR) capable of binding specific antigens. Before exiting the BM, immature B cells undergo negative selection to prevent self-reactive cells from entering in the periphery. After this first selection checkpoint, immature B cells enter the periphery as transitional B cells to continue the maturation process.1-3
The transitional stage of B-cell development represents a second stage where self-reactive B cells are purged from the mature B-cell repertoire.4 Indeed, approximately 40% of transitional cells in human peripheral blood (PB) are self-reactive; consequently, only approximately 50% develop into mature B cells.3,5,6 In mice, 2 classification systems have been described to distinguish subsets of transitional B cells. Loder et al defined 2 subsets: transitional type 1 (T1) and type 2 (T2), with them having CD24hiCD21loCD23loIgMhiIgDlo and CD24hiCD21hiCD23hiIgMhiIgDhi phenotypes, respectively.7 Alternatively, Allman et al identified 3 subsets of transitional cells as AA4+CD23−IgMhi (T1), AA4+CD23+IgMhi (T2), and AA4+CD23+IgMlo (T3).8 More recently, T3 cells have been proposed as being an anergic population that does not give rise to mature B cells.9,10 Each of these different stages of transitional B-cell development is differentially dependent on signals delivered through the BCR and cytokine receptors. Thus, mutations in syk, baff, or baff-r arrest B-cell development at the T1 to T2 stage,11-13 whereas mutations in btk, lyn, vav, BLNK, and cd45 prevent maturation of T2 B cells into mature B cells.7,8,14-19
Most of our knowledge of B-cell development has been derived from studies in mice, whereas several of the corresponding processes in humans remain incompletely defined. Because mutations in key genes (eg, BTK, BLNK, and CD45) result in profound differences in B-cell development in mice and humans,17,20-22 examination of human B-cell ontogeny is necessary to understand fully the cellular and molecular features and requirements of this process.
Human transitional B cells were first described as CD10+ or CD24++CD38++ B cells, which can be found at low frequencies in the blood and lymphoid tissues but are increased in cord blood (CB).5,23-25 Attempts have been made to separate further transitional B cells into T1-like and T2-like subsets based on the differential expression of IgD and CD3824 or CD24 and CD38.26 However, the delineation of subsets of transitional B cells according to these criteria is rather subjective, and the developmental and functional relationship between putative human transitional B-cell subsets remains unknown. Therefore, it is pertinent to identify markers that can better segregate transitional subpopulations to improve our understanding of the biology of human B-cell development.
We report the identification of human transitional B-cell subsets based on the differential expression of CD21, (ie, CD21lo and CD21hi). Detailed phenotypic, functional, molecular, and ontogenic assessment of these subsets demonstrated that the development of CD21lo transitional B cells preceded that of CD21hi transitional B cells, suggesting that the CD21lo subset represents the precursor of CD21hi transitional B cells. These findings provide important insights into human B-cell development and have implications for understanding the processes underlying perturbed B-cell maturation in autoimmune and immunodeficient conditions.
Methods
mAbs and reagents
The following monoclonal antibodies (mAbs) were used: fluorescein isothiocyanate (FITC)-conjugated anti-CD21 (Beckman Coulter); biotinylated anti-CD27 and anti-CD38, phycoerythrin-Cy7 (PE-Cy7) anti-CD20, PE-anti-CD23, PE-anti–B-cell activating factor belonging to the TNF family (BAFF) receptor (BAFF-R; eBioscience); biotinylated anti-CD44, PE-anti-CD27, allophycocyanin–anti-CD10, streptavidin-peridin chlorophyll protein; PE-, FITC-, allophycocyanin-, and PE-Cy7 IgG1 isotype control mAb (BD Biosciences PharMingen), Pacific Blue–anti-CD21 (Exbio), FITC–anti-Bcl2 (Dako North America), biotinylated anti-IgD (Southern Biotechnology), and FITC–anti-CD24 (Caltag). Recombinant human CD40L has been described.27 BAFF, interleukin-13 (IL-13), and IL-21 were from PeproTech; F(ab′)2 fragments of goat anti–human Ig were from Jackson Laboratories; IL-4 and IL-10 were provided by Dr Rene de Waal Malefyt (DNAX Research, Palo Alto, CA); CpG2006 was from Proligo.
Isolation of B-cell subsets
CB and PB were collected from healthy donors, patients with X-linked agammaglobulinemia (XLA; Table 1), or those recovering from HSC transplantation (HSCT). All studies were approved by institutional ethics review committees of all participating institutions, and informed consent was obtained in accordance with the Declaration of Helsinki. Normal spleens were obtained through the Australian Red Cross Blood Service. Mononuclear cells (MNCs) were prepared as previously described.27 Total human B cells were isolated by negative isolation, labeled with mAb specific for CD20, CD10, CD21, and CD27, and then sorted into subsets of CD21lo transitional (CD20+CD27−CD10+CD21lo), CD21hi transitional (CD20+CD27−CD10+CD21hi), naive (CD20+CD27−CD10−), and memory (CD20+CD27+CD10−) B cells (FACSAria, BD Biosciences).
Immunofluorescence staining
Five- and six-color flow-cytometric analysis was performed to characterize human transitional B-cell subsets. MNCs were labeled with anti-CD20, -CD10, -CD21, and -CD27 to resolve CD21lo and CD21hi transitional B cells, as well as an mAb to the protein of interest or control mAbs. To detect Bcl-2, cells were fixed in 2% formaldehyde and permeabilized and stained in phosphate-buffered saline-0.5% saponin with anti–Bcl-2 mAb. Single-color compensation controls were run for all experiments. Samples were acquired on a FACSCanto II (BD Immunohistochemistry Systems) and analyzed using FlowJo (TreeStar).
Rhodamine assay
PB B cells were labeled with anti-CD20, -CD10, -CD21, and -CD27 mAbs, washed in phosphate-buffered saline, and then stained with rhodamine 123 (R123, 12.5 μM; Sigma-Aldrich).29 Samples were collected at various time points, fixed with 1% formaldehyde, and analyzed for extrusion of R123.
KREC assay
RNA isolation and microarray
Total RNA was extracted from CB and PB B-cell subsets. cRNA was synthesized and amplified using biotin-labeled ribonucleotide and T7 polymerase (Affymetrix). Biotinylated cRNA was then hybridized to Human Genome U133 Plus 2.0 Affymetrix Arrays. The resulting data were analyzed by GeneSpring Software, Version 7.3.1. Detailed descriptions of each microarray experiment are provided at www.ncbi.nlm.nih.gov/geo (accession no. GSE17186).
Quantitative PCR
Polymerase chain reaction (PCR) primers (Integrated DNA Technologies) were designed using the Roche UPL primer design program. Primer sequences and Roche UPL probes are as follows: Glyceraldehyde 3-phosphate dehydrogenase, UPL probe 60, 5′ ctctgctcctcctgttcgac, 3′ acgaccaaatccgttgactc; Lymphoid enhancing binding factor-1 (LEF-1), UPL probe 17, 5′ cagatgtcaactccaaacaagg, 3′ ggagacaagggataaaaagtaggg; KIAA1199, UPL probe 43, 5′ ggaccagagtgctggaaaag, 3′ ctttgaagccaacgaatgc. Amplification reactions were performed using the Roche LightCycler 480 Probe Master Mix with the following conditions: denaturation at −95°C for 10 minutes; amplification, 45 cycles at 95°C for 10 seconds, 65°C for 30 seconds, and 72°C for 5 seconds, and cooling at 40°C for 30 seconds. All reactions were standardized to glyceraldehyde 3-phosphate dehydrogenase.
In vitro culture
Sorted B-cell subsets (5-15 × 103 cells) were cultured in media alone or with combinations of CD40L (1:200), CpG2006 (1 μg/mL), BAFF (500 ng/mL), anti-Ig (2.5 μg/mL), IL-4 (100 U/mL), IL-10 (100 U/mL), IL-13 (10 ng/mL), or IL-21 (50 ng/mL). To investigate survival, the cells were cultured for a total of 96 hours. At different times, a known number of CaliBRITE beads (BD Biosciences) were added to culture wells before harvesting, and the number of viable B cells was calculated as a function of the ratio of beads to live cells. For proliferation, cells were cultured for 5 days, and the incorporation of [3H]thymidine (0.5 μCi; MP Biomedicals) was determined during the final 18 hours.
Enzyme-linked immunosorbent assays
B-cell subsets (5-10 × 103/well per 200 μL) were cultured in medium alone or with CpG alone, CD40L alone, or in combination with IL-21. Supernatants were harvested on day 12, and secretion of IgM, IgA and IgG was determined by enzyme-linked immunosorbent assay.27,32 Autoreactivity was measured by antinuclear antibody (ANA) enzyme-linked immunosorbent assay as previously described5 except that bound IgM was detected with biotinylated goat F(ab′)2 antihuman IgM Ab (Southern Biotechnology) followed by horseradish peroxidase–streptavidin (Jackson ImmunoResearch).
Statistical analyses
All data were analyzed using GraphPad Prism software by repeated-measures analysis of variance and Tukey multiple comparison posttest when significance differences (P < .05) between groups were detected.
Results
Identification of 2 human transitional B-cell subsets
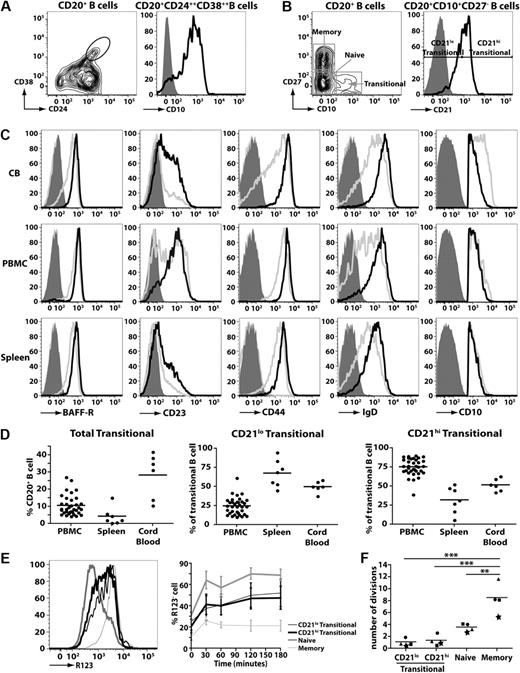
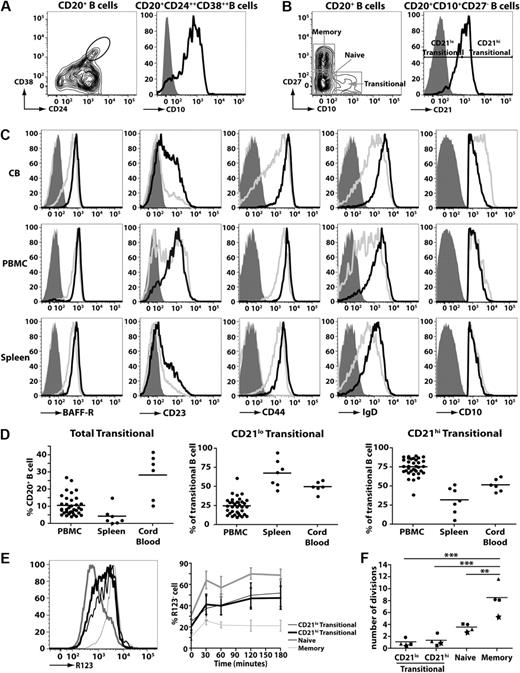
Human transitional B cells can be identified either by high expression of CD24 and CD3823-25 (Figure 1A left panel) or expression of CD10 (Figure 1A right panel).24,25 For the purposes of this study, we identified human transitional B cell as CD20+CD27−CD10+ cells, and naive and memory B cells as CD20+CD27− and CD20+CD27+ cells, respectively (Figure 1B left panel). A previous study of HIV-infected persons noted differential expression of CD21 on CD10+ transitional B cells.33 Based on this, we divided human transitional B cells into subsets corresponding to CD21lo and CD21hi populations (Figure 1B right panel). To ensure that the CD21lo and CD21hi transitional B-cell populations indeed represented distinct subsets, their phenotype was determined. IgD, CD44, and BAFF-R were consistently higher on CD21hi transitional B cells compared with CD21lo transitional cells (Figure 1C; supplemental Figure 1A-C, available on the Blood website; see the Supplemental Materials link at the top of the online article). When multiple donors were examined, there was a clear trend that the level of CD23 (expressed as mean fluorescence intensity [MFI]) and frequency of CD23+ cells were greater for CD21hi compared with CD21lo transitional B cells (ie, MFI: 532 ± 109 vs MFI: 171 ± 27 and 62.6% ± 7.0% vs 23.2% ± 2.7%, respectively; mean ± SEM [n = 8]; Figure 1C; supplemental Figure 1A-C). However, it is also clear that CD23 expression on CD21lo and CD21hi transitional B cells is heterogeneous. Thus, it was not feasible to isolate transitional B-cell subsets according to the differential expression of CD23 because the CD23+ subset of CD10+ transitional cells would include not only the CD21hi population but also some of the CD21lo transitional cells (Figure 1C; supplemental Figure 1A-C). Expression of CD10 on CD21lo and CD21hi transitional B cells was similar (Figure 1C). In contrast to CD10 and those markers presented in Figure 1C, expression of CD5, CD9, CD20, CD24, CD38, CXCR4, TACI, and BCMA on CD21lo and CD21hi transitional B cells was comparable (supplemental Figure 2). These phenotypic differences were noted irrespective of whether the transitional B cells were isolated from human CB, PB, or spleen (Figure 1C; supplemental Figure 2).
Identification of 2 subsets of human transitional B cells. (A-B) Peripheral blood mononuclear cells (PBMCs) were stained with mAbs against CD20, CD24, CD38, and CD10. (A) Left panel: Expression of CD24 and CD38 on CD20+ B cells; transitional B cells were initially defined as CD24hiCD38hi cells. Right panel: Expression of CD10 on CD24hiCD38hi B cells. (B) PBMCs were stained with mAbs specific for CD20, CD27, and CD10 to identify transitional (CD20+CD27−CD10+), naive (CD20+CD27−CD10−), and memory (CD20+CD27+CD10−) B cells (left panel). The transitional B-cell population was further divided into CD21lo and CD21hi subsets (right panel). (C) MNCs from CB, PB, and spleen were stained with mAbs against CD10, CD20, CD27, and CD21. Expression of BAFF-R, CD23 CD44, IgD, and CD10 on CD21lo (gray line) and CD21hi (black line) transitional B-cell subsets was determined by flow cytometry. The corresponding isotype control is represented by the light gray filled and dark gray filled histograms for CD21lo and CD21hi transitional B cells, respectively. Note that, for the depiction of the CD10 histograms, the solid gray overlay represents the fluorescence of all lymphocytes after labeling with an isotype control mAb. The plots are representative of more than 5 experiments using cells from different donors. (D) The frequencies of total transitional B cells as well as CD21lo and CD21hi subsets in PB (n = 35), spleen (n = 7), and CB (n = 6). The results are expressed (left to right) as percentage of CD20+ B cells, and percentage of CD20+CD27−CD10+ transitional B cells. (E) PB B cells were labeled with mAbs against CD10, CD21, and CD27, together with R123, and then incubated at 37°C. Samples were collected at various times and analyzed for extrusion of R123. Left panel: Representative data for R123 extrusion after 60 minutes. Right panel: Kinetics of extrusion of R123 (expressed as the percentage of R123− cells; mean ± SEM, n = 4). (F) The replication history of each subset was determined by KREC analysis.30,31 This was performed on B-cell subsets isolated from 4 unrelated donors; each symbol (●, ■, ★, ▲) represents an individual donor, whereas the horizontal line represents the mean. **P < .01. ***P < .001.
Identification of 2 subsets of human transitional B cells. (A-B) Peripheral blood mononuclear cells (PBMCs) were stained with mAbs against CD20, CD24, CD38, and CD10. (A) Left panel: Expression of CD24 and CD38 on CD20+ B cells; transitional B cells were initially defined as CD24hiCD38hi cells. Right panel: Expression of CD10 on CD24hiCD38hi B cells. (B) PBMCs were stained with mAbs specific for CD20, CD27, and CD10 to identify transitional (CD20+CD27−CD10+), naive (CD20+CD27−CD10−), and memory (CD20+CD27+CD10−) B cells (left panel). The transitional B-cell population was further divided into CD21lo and CD21hi subsets (right panel). (C) MNCs from CB, PB, and spleen were stained with mAbs against CD10, CD20, CD27, and CD21. Expression of BAFF-R, CD23 CD44, IgD, and CD10 on CD21lo (gray line) and CD21hi (black line) transitional B-cell subsets was determined by flow cytometry. The corresponding isotype control is represented by the light gray filled and dark gray filled histograms for CD21lo and CD21hi transitional B cells, respectively. Note that, for the depiction of the CD10 histograms, the solid gray overlay represents the fluorescence of all lymphocytes after labeling with an isotype control mAb. The plots are representative of more than 5 experiments using cells from different donors. (D) The frequencies of total transitional B cells as well as CD21lo and CD21hi subsets in PB (n = 35), spleen (n = 7), and CB (n = 6). The results are expressed (left to right) as percentage of CD20+ B cells, and percentage of CD20+CD27−CD10+ transitional B cells. (E) PB B cells were labeled with mAbs against CD10, CD21, and CD27, together with R123, and then incubated at 37°C. Samples were collected at various times and analyzed for extrusion of R123. Left panel: Representative data for R123 extrusion after 60 minutes. Right panel: Kinetics of extrusion of R123 (expressed as the percentage of R123− cells; mean ± SEM, n = 4). (F) The replication history of each subset was determined by KREC analysis.30,31 This was performed on B-cell subsets isolated from 4 unrelated donors; each symbol (●, ■, ★, ▲) represents an individual donor, whereas the horizontal line represents the mean. **P < .01. ***P < .001.
When the frequencies of B-cell subsets were enumerated, it was found that CB contained the greatest proportion of total transitional B cells (28.3% ± 5.4% of all B cells; n = 6) followed by PB (10.6% ± 1.0%; n = 35), whereas the spleen contained the lowest frequency (4.3% ± 1.9%; n = 7). The highest frequency of CD21lo transitional B cells was detected in the spleen (67.6% ± 6.7% of all transitional B cells), whereas they were least represented in PB (24.7% ± 1.9%) (Figure 1D). Accordingly, CD21hi transitional B cells were most abundant in PB (Figure 1D). In contrast, transitional B cells in CB were composed equally of both the CD21lo and CD21hi subsets (Figure 1D).
Human transitional, naive, and memory B cells can be resolved from one another by their differential expression of the ABCB1 transporter and their relative abilities to extrude the dye R123.29 We confirmed this finding by demonstrating that naive B cells extruded R123, whereas memory B cells could not (Figure 1E). Both subsets of transitional B cells extruded R123 to a comparable level, however not as efficiently as naive B cells (Figure 1E). The comparable extrusion of R123 by CD21lo and CD21hi transitional B cells was mirrored by the similar expression of ABCB1 mRNA in these cells as determined by microarray analysis (not shown).
We determined the replication history of the 2 transitional subsets using a genomic DNA PCR assay that measures KRECs.30 This analysis revealed that the in vivo proliferative activity of memory B cells exceeded that of naive cells by more than 2-fold (mean number of cell divisions ± SEM: 8.5 ± 1.2 and 3.60 ± 0.39, respectively, n = 4, P < .01; Figure 1F).30,31 The in vivo proliferation of memory B cells also significantly exceeded that of both subsets of transitional B cells (P < .001; Figure 1F). The proliferation of both subsets of transitional B cells (<1.5 divisions in vivo) was less than naive B cells, and there was no significant difference between the in vivo replicative history of the CD21lo and CD21hi transitional B cells (Figure 1F; P > .05). Thus, both the CD21lo and CD21hi subsets of transitional cells correspond to recently generated quiescent B cells.
CD21lo and CD21hi transitional B-cell subsets exhibit a unique gene expression profile
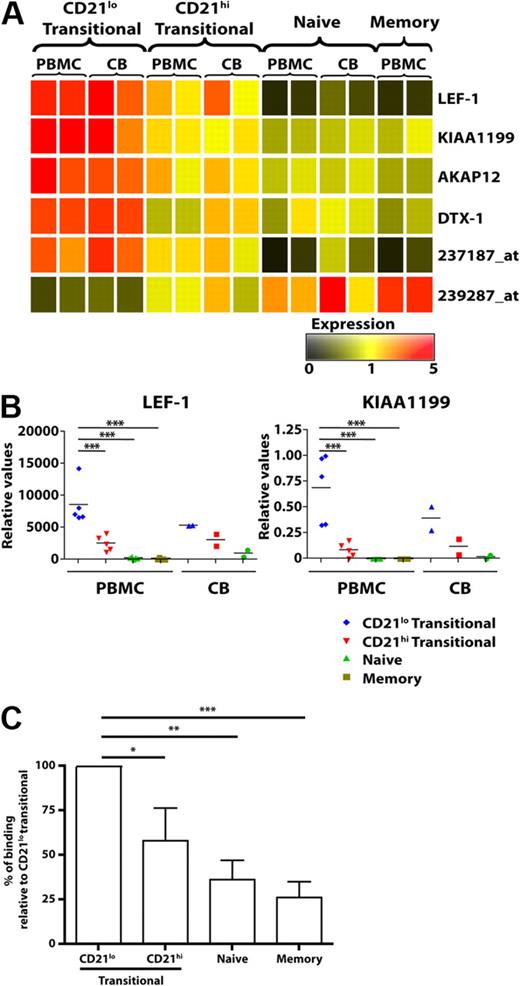
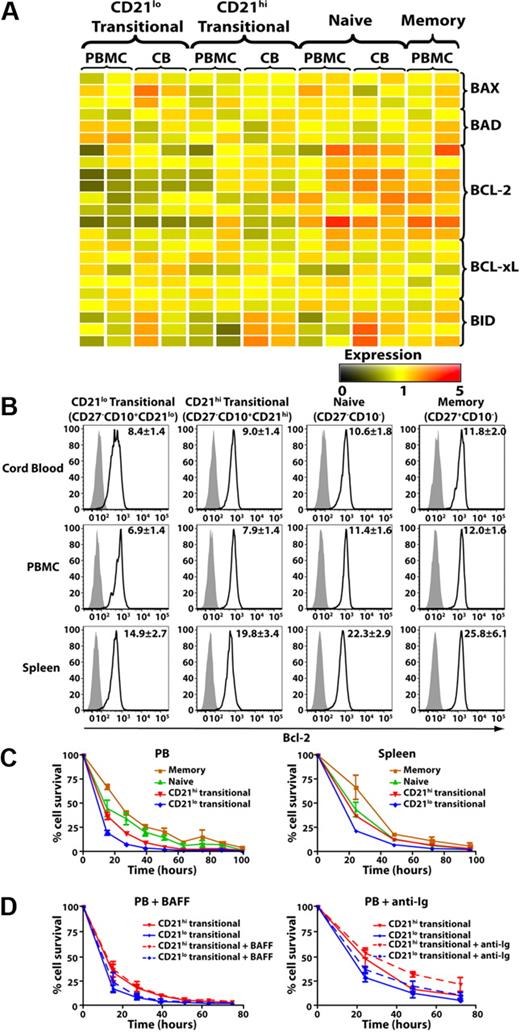
To demonstrate further that the CD21lo and CD21hi transitional B cells were distinct subsets, we performed microarray analysis on PB and CB B-cell subsets. We identified 6 genes that differed in their level of expression between CD21lo and CD21hi transitional B-cell subsets by more than 2-fold. LEF-1, Deltex-1 (DTX1), KIAA1199, A Kinase Anchor Protein 12 (AKAP12), and 1 unknown gene (Affymetrix probe 237187_at) were expressed at the highest levels in CD21lo transitional B cells, followed by the CD21hi transitional population, whereas expression in naive and memory B cells was even lower or extinguished (Figure 2A). In contrast, expression of another unknown gene (239287_at) was lowest in CD21lo transitional B cells, increased in CD21hi transitional B cells, and further up-regulated in naive B cells (Figure 2A). Quantitative PCR analysis confirmed that LEF1 and KIA1199 were expressed at significantly higher levels in CD21lo transitional B cells compared with the CD21hi transitional, naive, and memory B-cell subsets (P < .001; Figure 2B). Collectively, this phenotypic and molecular analysis of human B cells suggests that CD10+CD21lo and CD10+CD21hi B cells corresponds to 2 separable subsets of transitional cells that differ not only from each other but also other mature B-cell subsets, ie, naive and memory cells.
Differential gene expression in, and production of autoreactive IgM by, human B-cell subsets. (A) Microarray gene expression profiles of sort-purified CD21lo transitional, CD21hi transitional, naive, and memory B cells from PB and CB. The range of expression is represented by color shading from black (low expression) to red (high expression). Each column represents 2 pooled donors for the PBMCs and different groups of pooled donors for the CB (range of 2 or 3 different donors for each pooled data). (B) Differential expression of LEF1 and KIA1199 was confirmed by quantitative PCR. Each symbol represents an individual donor (PB, n = 5; CB, n = 2); horizontal lines represent the mean. No values are presented for memory B cells in CB as such cells are infrequent. *P < .05. ***P < .001. (C) B-cell subsets were purified from different healthy donors and cultured with CD40L/IL-21. After 12 days, the amounts of antinuclear IgM antibody were determined. The results are expressed as percentage binding relative to that of the CD21lo transitional B-cell subset (normalized to equal 100%), and are the mean ± SEM of 5 independent experiments. *P < .05; P < .01; ***P < .001.
Differential gene expression in, and production of autoreactive IgM by, human B-cell subsets. (A) Microarray gene expression profiles of sort-purified CD21lo transitional, CD21hi transitional, naive, and memory B cells from PB and CB. The range of expression is represented by color shading from black (low expression) to red (high expression). Each column represents 2 pooled donors for the PBMCs and different groups of pooled donors for the CB (range of 2 or 3 different donors for each pooled data). (B) Differential expression of LEF1 and KIA1199 was confirmed by quantitative PCR. Each symbol represents an individual donor (PB, n = 5; CB, n = 2); horizontal lines represent the mean. No values are presented for memory B cells in CB as such cells are infrequent. *P < .05. ***P < .001. (C) B-cell subsets were purified from different healthy donors and cultured with CD40L/IL-21. After 12 days, the amounts of antinuclear IgM antibody were determined. The results are expressed as percentage binding relative to that of the CD21lo transitional B-cell subset (normalized to equal 100%), and are the mean ± SEM of 5 independent experiments. *P < .05; P < .01; ***P < .001.
In vitro analysis of survival of human B-cell subsets
The microarray analysis also revealed that expression of Bcl-2 was lower in CD21lo transitional B cells compared with the CD21hi subset and was increased further in naive and memory B cells (Figure 3A). These differences were confirmed by intracellular staining and flow cytometry (Figure 3B). This analysis revealed significantly higher expression of Bcl-2 protein in both naive and memory B cells relative to CD21lo and CD21hi transitional B cells (P < .001). The differential expression of Bcl-2 among B-cell subsets appeared to be unique for proapoptotic and antiapoptotic molecules because BAD, BAX, BID, and BCLXL were detected at similar levels in the transitional, naive, and memory cells in both CB and PB (Figure 3A). We next determined whether differential expression of Bcl-2 correlated with in vitro survival of human B-cell subsets. There was a trend that CD21hi transitional B cells survived better than CD21lo transitional cells (Figure 3C left), but this difference was not statistically significant. However, consistent with their increased expression of Bcl-2, survival of naive and memory B cells was significantly greater than that of both transitional B-cell populations (ie, ∼ 24, 40, and 50 hours; naive vs transitional: P < .05; memory vs transitional: P < .001). This pattern of survival was also seen for splenic B-cell subsets (Figure 3C right). We then questioned whether survival of transitional B cells could be improved by exogenous factors. Unexpectedly, stimulation with BAFF, a crucial survival factor for murine B cells,34 had no effect on either subset of human transitional B cells (Figure 3D left). In contrast, anti-Ig improved survival of both transitional subsets, with the greatest effect being on CD21hi transitional B cells (Figure 3D right).
Expression of Bcl-2 and survival of human B-cell subsets. (A) Microarray of survival genes expressed by PB and CB CD21lo transitional, CD21hi transitional, naive, and memory B cells. The range of expression is represented by shading from black (low expression) to red (high expression). Each row corresponds to a different probe for the indicated gene of interest. (B) MNCs from CB, PB, and spleen were stained with anti-CD20, -CD27, -CD10, and -CD21 mAbs to identify transitional, naive, and memory B-cell subsets. The cells were then fixed, permeabilized, and incubated with anti–Bcl-2 (black line) or isotype control (gray filled) mAb. The values in each histogram represent the ratio of MFI of cells incubated with anti-Bcl-2 mAb over that of cells incubated with an isotype control ± SEM (CB, n = 4; PBMCs, n = 11; spleen, n = 9). (C-D) CD21lo transitional, CD21hi transitional, naive, and memory B cells purified from PB (C top left panel) or spleen (C top right panel) were cultured in media alone (C), or with (D) BAFF (0.5 μg/mL; left panel) or F(ab′)2 fragments of anti-Ig (2.5 μg/mL; right panel) for 100 hours. The number of viable cells was determined at the indicated times. Results shown are the mean ± SEM of 3 independent experiments for PBMCs and 2 independent experiments for spleen.
Expression of Bcl-2 and survival of human B-cell subsets. (A) Microarray of survival genes expressed by PB and CB CD21lo transitional, CD21hi transitional, naive, and memory B cells. The range of expression is represented by shading from black (low expression) to red (high expression). Each row corresponds to a different probe for the indicated gene of interest. (B) MNCs from CB, PB, and spleen were stained with anti-CD20, -CD27, -CD10, and -CD21 mAbs to identify transitional, naive, and memory B-cell subsets. The cells were then fixed, permeabilized, and incubated with anti–Bcl-2 (black line) or isotype control (gray filled) mAb. The values in each histogram represent the ratio of MFI of cells incubated with anti-Bcl-2 mAb over that of cells incubated with an isotype control ± SEM (CB, n = 4; PBMCs, n = 11; spleen, n = 9). (C-D) CD21lo transitional, CD21hi transitional, naive, and memory B cells purified from PB (C top left panel) or spleen (C top right panel) were cultured in media alone (C), or with (D) BAFF (0.5 μg/mL; left panel) or F(ab′)2 fragments of anti-Ig (2.5 μg/mL; right panel) for 100 hours. The number of viable cells was determined at the indicated times. Results shown are the mean ± SEM of 3 independent experiments for PBMCs and 2 independent experiments for spleen.
CD21hi transitional B cells are functionally more mature than CD21lo transitional B cells
We next investigated the functionality of the CD21lo and CD21hi transitional B-cell subsets and compared their responses with those of naive and memory cells. Stimulation with CD40L resulted in greater [3H]thymidine incorporation by all B-cell subsets compared with that occurring in unstimulated B cells (Table 2). Although stimulation through the BCR failed to induce proliferation of any B-cell subset, it synergized with CD40L resulting in a proliferative response exceeding that induced by CD40L alone by 2- to 4-fold (Table 2). Notably, proliferation of CD40L or CD40L/anti-Ig–stimulated CD21lo transitional B cells was consistently and substantially less than that of CD21hi transitional B cells, whereas proliferation of CD21hi transitional B cells resembled the naive subset (Table 2). We determined the effect of cytokines on proliferation of the transitional B-cell subsets. Although addition of IL-4, IL-10, IL-13, or IL-21 improved proliferation of all CD40L-stimulated B-cell subsets, the magnitude of the response by CD21lo transitional B cells was always less than that of CD21hi transitional and naive B cells (Table 2).
To assess Ig secretion, B-cell subsets were stimulated with CpG, CD40L, or CpG/CD40L in the absence or presence of IL-21, a potent inducer of human B-cell differentiation.32,35,36 Both transitional subsets secreted low amounts of IgM but neither IgG nor IgA in response to CD40L alone or CpG with or without IL-21 (Table 3). CD40L/CpG increased IgM secretion and induced secretion of low amounts of IgG and IgA by CD21lo and CD21hi transitional B-cell subsets (Table 3). Under these conditions, the levels of Ig secreted by CD21hi transitional B cells were 2- to 5-fold greater than CD21lo transitional B cells. These results demonstrate that CD21lo transitional B cells are functionally less mature than the CD21hi subset. In contrast to proliferation, differentiation of CD21hi transitional B cells into Ig-secreting cells was inferior to that of naive B cells. However, both subsets of transitional B cells secreted comparable amounts of IgM, IgG, and IgA in response to stimulation with CD40L/IL-21, with or without CpG (Table 3).
Increased production of autoreactive IgM antibodies by CD21lo transitional B cells
A previous study discovered that more than 40% of human transitional B cells expressed an autoreactive BCR.5 It was therefore of interest to determine whether the 2 transitional subsets described here produce different levels of autoreactive Ab. B-cell subsets were stimulated with CD40L/IL-21, and the level of IgM ANA was determined. CD21lo transitional B cells produced IgM that contained the greatest amount of ANA, and this amount was significantly higher than the self-reactive IgM produced by CD21hi transitional cells (P < .05; Figure 2C). The levels of self-reactive Ab produced by naive (P < .01) and memory (P < .001) B cells were also significantly less than CD21lo transitional B cells (Figure 2C). Thus, whereas previous studies elegantly established that transitional B cells are enriched for self-reactive B cells,5 our findings demonstrate greater autoreactivity within the CD21lo subset of transitional B cells.
CD21loCD10+ B cells represent the predominant population in cases of B-cell deficiency
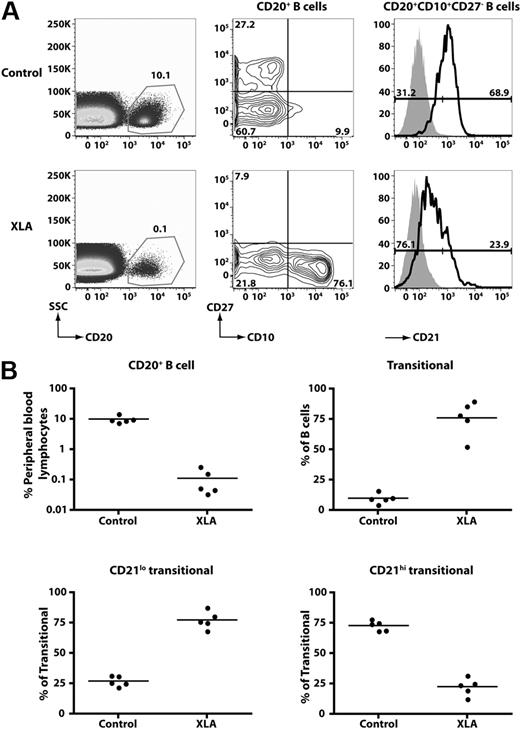
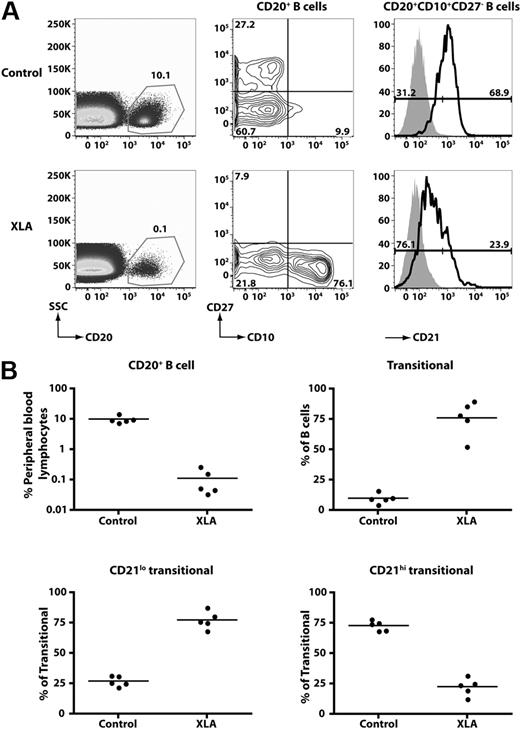
To gain further insight into the relationship between CD21lo and CD21hi transitional B cells, we analyzed the B-cell compartment of patients with XLA resulting from mutations in BTK. XLA patients have a block in B-cell development in the BM resulting from impaired BCR signaling21 such that the frequency of PB B cells was 0.1% plus or minus 0.1% (n = 5; range, 0.03%-0.27%), compared with 10.1% plus or minus 2.8% in normal healthy persons (Figure 4). Although the absolute number of B cells was drastically diminished in these patients, most of these had a transitional phenotype (XLA, 76.1% ± 14.6% of all B cells; healthy donors, 9.9% ± 1.9%; Figure 4). Furthermore, most transitional B cells in XLA belonged to the CD21lo subset (∼ 75%). This contrasts with normal donors, in whom only approximately 25% of transitional B cells belong to this subset. These findings provide strong evidence that CD21loCD10+ and CD21hiCD10+ B cells are developmentally distinct populations of transitional B cells.
CD21lo transitional B cells are the predominant B-cell population in the blood of B-cell–deficient XLA patients. (A) PBMCs from a healthy donor and an XLA patient were stained with anti-CD20, -CD27, -CD10, and -CD21 mAbs. (Left panel) The frequency of CD20+ B cells; these cells were divided into transitional, naive, and memory B-cell subsets based on differential expression of CD10 and CD27 (middle panel). The transitional B cells were further divided into CD21lo and CD21hi subsets. The histograms presented for CD21 expression (right-hand panel) were obtained by gating on the CD20+CD10+CD27− B cells (which appear in the lower right-hand quadrant of the contour plot depicted as the middle panel). The overlay solid gray histogram represents the fluorescence of lymphocytes incubated with an isotype control mAb. Flow-cytometric analysis of transitional B-cell subsets detected in donors and XLA patients was performed on the same day with identical instrument settings. The values in each plot are the mean percentages of each of the indicated B-cell subsets from all studied healthy donors and patients. (B) Summary of the frequency of total B cells, transitional B cells, and transitional B-cell subsets in healthy donors (HD) and XLA (n = 5).
CD21lo transitional B cells are the predominant B-cell population in the blood of B-cell–deficient XLA patients. (A) PBMCs from a healthy donor and an XLA patient were stained with anti-CD20, -CD27, -CD10, and -CD21 mAbs. (Left panel) The frequency of CD20+ B cells; these cells were divided into transitional, naive, and memory B-cell subsets based on differential expression of CD10 and CD27 (middle panel). The transitional B cells were further divided into CD21lo and CD21hi subsets. The histograms presented for CD21 expression (right-hand panel) were obtained by gating on the CD20+CD10+CD27− B cells (which appear in the lower right-hand quadrant of the contour plot depicted as the middle panel). The overlay solid gray histogram represents the fluorescence of lymphocytes incubated with an isotype control mAb. Flow-cytometric analysis of transitional B-cell subsets detected in donors and XLA patients was performed on the same day with identical instrument settings. The values in each plot are the mean percentages of each of the indicated B-cell subsets from all studied healthy donors and patients. (B) Summary of the frequency of total B cells, transitional B cells, and transitional B-cell subsets in healthy donors (HD) and XLA (n = 5).
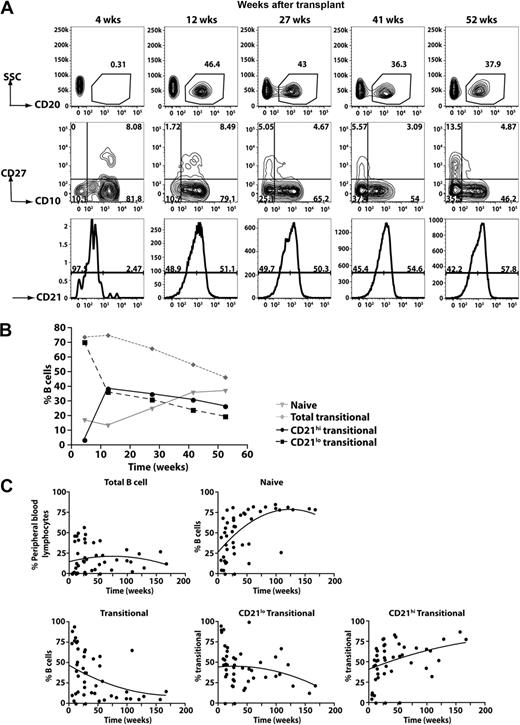
CD21lo transitional B cells are the first B cells to appear in the periphery after HSCT
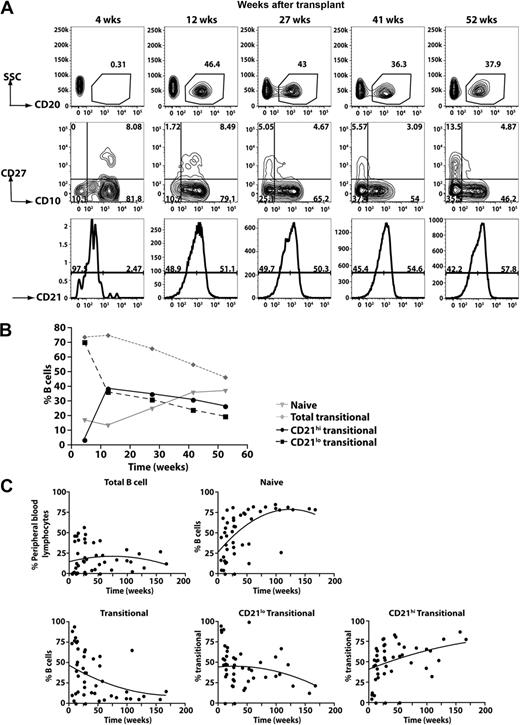
The data derived from the analysis of XLA patients suggested that the generation of CD21lo transitional B cells precedes that of CD21hi transitional B cells. An extension of this is that CD21lo transitional B cells are precursors of CD21hi transitional B cells. To examine this in more detail, we analyzed the kinetics of B-cell reconstitution in a cohort of patients undergoing HSCT. Figure 5A-B depicts the data from one patient, whereas the data for all patients are compiled in Figure 5C. In the initial stages after HSCT (0-10 weeks), only a low frequency of PB CD20+ cells was detected (<1% of all lymphocytes). However, the majority of them (>75%) were CD10+ transitional B cells. At subsequent times, the frequency of CD20+ lymphocytes increased. This was accompanied by a decrease and increase, respectively, in the frequency of transitional and naive B cells (Figure 5). Interestingly, the composition of the transitional B-cell population also changed after HSCT such that, at the earliest times examined, more than 95% of them were CD21lo whereas over time the CD21hi subset emerged and increased in frequency (Figure 5B-C). Overall, the B-cell reconstitution data demonstrate that the first B-cell population to be exported from the BM to the periphery is the CD21lo transitional subset, which subsequently gives rise to CD21hi transitional B cells, the precursor to the naive B-cell pool.
Kinetics of B-cell reconstitution after HSCT reveals the early appearance of CD21lo transitional B cells. (A) PBMCs were collected from patients at different times after HSCT and then stained with anti-CD20, -CD27, -CD10, and CD21 to determine the B-cell reconstitution profile. CD20+ B cells were gated (top panel) and divided into transitional, naive, and memory B-cell subsets based on differential expression of CD10 and CD27 (middle panel). The transitional B cells were further divided into CD21lo and CD21hi populations (bottom panel). The numbers indicate the percentage of total B cells (top panel) and the corresponding subsets (middle and bottom panels). (B) Summary of the reconstitution of naive and transitional B-cell subsets after HSCT in the patient presented (A) as a percentage of all B cells. (C) Combined B-cell reconstitution profile in 12 post-HSCT patients.
Kinetics of B-cell reconstitution after HSCT reveals the early appearance of CD21lo transitional B cells. (A) PBMCs were collected from patients at different times after HSCT and then stained with anti-CD20, -CD27, -CD10, and CD21 to determine the B-cell reconstitution profile. CD20+ B cells were gated (top panel) and divided into transitional, naive, and memory B-cell subsets based on differential expression of CD10 and CD27 (middle panel). The transitional B cells were further divided into CD21lo and CD21hi populations (bottom panel). The numbers indicate the percentage of total B cells (top panel) and the corresponding subsets (middle and bottom panels). (B) Summary of the reconstitution of naive and transitional B-cell subsets after HSCT in the patient presented (A) as a percentage of all B cells. (C) Combined B-cell reconstitution profile in 12 post-HSCT patients.
Discussion
Much is known of the biology of murine transitional B cells, yet our understanding of human transitional B cells is only just beginning to be unraveled. Here, we identified and characterized 2 populations of human transitional B cells: CD21lo and CD21hi. Our data revealed that these 2 populations have distinct phenotypes, with higher expression of CD23, CD44, IgD, and BAFF-R by the CD21hi subset (Figure 1). Notably, the greater expression of CD21, CD23, and IgD on the CD21hi versus CD21lo transitional subsets correlates with the phenotypes of murine transitional B cells, with expression of these molecules also increasing along a T1 → T2 → T3 gradient7,8
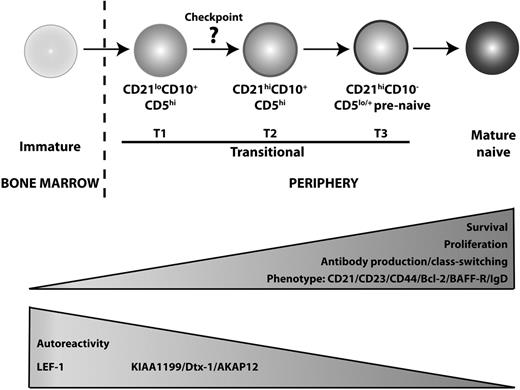
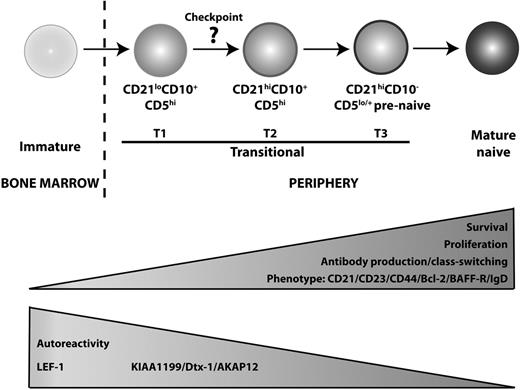
The phenotypic data, together with greater proliferation and Ig secretion by the CD21hi transitional B cells (Tables 2–3), suggest that CD21lo and CD21hiCD10+ B cells correspond to distinct stages of B-cell development, with the CD21lo subset representing a less mature population. More compelling support for this conclusion came from assessment of the composition of the B-cell compartment in patients with XLA or those recovering from HSCT, as well as microarray analyses. First, although BTK mutations cause a severe block in B-cell development21 (Figure 4), the few circulating B cells detectable in XLA patients were predominantly CD21lo transitional cells, suggesting that loss of Btk prevents CD21lo transitional B cells from maturing into CD21hi transitional, and subsequently naive, B cells (Figure 4). Second, and consistent with the observations in XLA patients, CD21lo transitional B cells were the first B-cell subset to populate the periphery after HSCT. Over time, these cells declined in number, and this coincided with the appearance of CD21hi transitional cells, thereby suggesting that CD21lo transitional B cells represent the initial population of BM emigrant B cells that subsequently develops into the CD21hi transitional subset (Figure 5). Third, gene profiling revealed that LEF-1 was expressed at the highest level in CD21lo transitional B cells, with expression being reduced in CD21hi transitional B cells and extinguished in mature naive and memory cells (Figure 2). Hystad et al recently reported that LEF-1 was absent in human early B cells (CD34+CD10+CD19−), induced in pro–B cells (CD34+CD10+CD19+), further up-regulated in pre–B cells (CD34+CD10+CD19−), and reduced, but still expressed, in immature B cells (CD34−CD10+CD19+).37 The declining level of LEF-1 expression between human BM pre–B cells and immature B cells,37 and CD21lo and CD21hi subsets of transitional B cells, and further down-regulation between the CD21hi transitional and naive B cells suggest that B-cell development progresses in the following linear manner: immature → CD21lo transitional → CD21hi transitional → naive (Figure 6). The finding that CD21lo transitional cells were enriched for autoreactive Ab is also consistent with the notion that CD21lo transitional B cells are the earlier BM emigrants and that they are still undergoing negative selection.
Proposed model of human B-cell development. We propose that B cells emigrating from the BM to the periphery are CD21lo transitional/T1 B cells, which subsequently differentiate into CD21hi transitional/T2 B cells. This maturation step requires Btk, indicating a role for BCR engagement, and is accompanied with improved survival and the potential to proliferate and differentiate into Ig-secreting cells. There is also a reduction in autoreactivity, suggesting that a checkpoint for self-tolerance exists at the step between these 2 stages of transitional B-cell development. Phenotypically, the CD21hi transitional B cells increase expression of CD21, CD23 CD44, Bcl-2, BAFF-R, and IgD. Meanwhile, expression of LEF-1, KIAA1199, Dtx-1, and AKAP12 is reduced as the cells enter the mature B-cell repertoire. CD21hi transitional/T2 B cells then give rise to T3/prenaive CD5+ B cells,38 which are characterized by loss of expression of CD10, high expression of CD21, but impaired extrusion of R123. The auto-reactive specificity of this population remains to be determined.
Proposed model of human B-cell development. We propose that B cells emigrating from the BM to the periphery are CD21lo transitional/T1 B cells, which subsequently differentiate into CD21hi transitional/T2 B cells. This maturation step requires Btk, indicating a role for BCR engagement, and is accompanied with improved survival and the potential to proliferate and differentiate into Ig-secreting cells. There is also a reduction in autoreactivity, suggesting that a checkpoint for self-tolerance exists at the step between these 2 stages of transitional B-cell development. Phenotypically, the CD21hi transitional B cells increase expression of CD21, CD23 CD44, Bcl-2, BAFF-R, and IgD. Meanwhile, expression of LEF-1, KIAA1199, Dtx-1, and AKAP12 is reduced as the cells enter the mature B-cell repertoire. CD21hi transitional/T2 B cells then give rise to T3/prenaive CD5+ B cells,38 which are characterized by loss of expression of CD10, high expression of CD21, but impaired extrusion of R123. The auto-reactive specificity of this population remains to be determined.
The results from our study provide novel and significant insights into human B-cell development in the periphery and clarify our understanding of the requirements for this process. First, we identified several genes (eg, LEF1, KIA1199) that are highly expressed in CD21lo transitional B cells and down-regulated at subsequent stages of B-cell development (Figure 2). The exact roles of these genes in human B-cell development are unknown. However, LEF-1–deficient mice have a reduction in the total number of B cells in the fetal liver and perinatal BM,39 thus highlighting the importance of this gene in controlling B-cell development. In contrast, overexpression of KIAA119940 in cancer cells was associated with increased cell death and/or growth suppression. Based on these observations, it is possible that the heightened expression of KIAA1199 in transitional B cells contributes to their reduced survival and proliferation in vitro relative to naive B cells. Further examination of these genes in immune cells should reveal more about their role during human B-cell development.
Our finding that BTK mutations severely block B-cell development at the CD21lo transitional stage (Figure 4) questions a previous finding that BTK is required to establish B-cell tolerance in humans.41 Ng et al, noting the predominance of transitional (ie, CD10+) B cells in XLA PB,41 demonstrated that these cells had a greater frequency of autoreactivity (∼ 55%-60%) than CD10+ B cells from normal donors (37%).41 They concluded that altered signaling through the BCR resulting from mutations in BTK allowed the escape of an increased proportion of autoreactive B cells; thus, intact BTK function was necessary for the maintenance of B-cell tolerance.41 However, an alternative interpretation is that the increase in autoreactivity noted in XLA B cells41 simply reflects the fact that approximately 80% of transitional B cells in these patients belong to the CD21lo subset (Figure 4), and a greater frequency of these cells are autoreactive Abs compared with the CD21hi subset (Figure 2C), which composes most of the CD10+ population in healthy donors (Figure 1D). Thus, it is possible that BTK does not play a role in establishing tolerance; rather, it is necessary for the maturation of CD21lo transitional B cells into CD21hi transitional B cells.
Although the CD21lo and CD21hi transitional B-cell subsets exhibited numerous differences, they did share several phenotypic and functional attributes. For instance, based on replication history, these 2 B-cell subsets were both nonproliferating cells in vivo (Figure 1F). Thus, the development of CD21lo transitional cells into CD21hi transitional cells is unlikely to involve proliferation. Interestingly, naive B cells had undergone several divisions in vivo (Figure 1F),30,31 implying that differentiation of transitional B cells into naive cells may involve a proliferative phase. Notably, BAFF had very little effect on the survival of either transitional B-cell subset despite detectable expression of BAFF-R (Figures 1,3). We also found that BAFF did not modulate expression of CD21 or CD23 on human transitional B cells in vitro (supplemental Figure 3). Because BAFF is critical for the in vivo survival13,42 of and up-regulation of expression of both CD21 and CD2343 on murine B cells, it is possible that B-cell development in humans is less dependent on BAFF than that in mice. This is supported by the recent description of 2 related persons with mutations in TNFRSF13C (encoding BAFF-R) who, in contrast to Baffr-deficient mice,13,42 still developed naive and memory B cells, albeit at reduced frequencies compared with normal healthy controls.44
Several studies have partitioned human transitional B cells into subsets according to phenotype and/or function. Anolik et al defined human “T1” and “T2” transitional cells based on expression levels of CD24 and CD38, with T1 B cells being CD38+++CD24+++ and T2 B cells CD38++CD24++.26 These transitional subsets were detected in the PB of patients recovering from B-cell depletion therapy.26 More recently, this group identified a third population of human transitional B cells (termed T3) that exhibit a phenotype of naive B cells (ie, CD10−CD24intCD38intIgD+CD27) but can be resolved from naive cells by their inefficiency in extruding R123.45 Consistent with our data from HSCT patients, the appearance of the T1 subset preceded that of the T2 and T3 populations in the setting of B-cell reconstitution after B-cell depletion.26,45 Interestingly, another study described a population of human CD5+ prenaive B cells that can be distinguished from transitional and naive B cells by their intermediate levels of expression of CD9, CD10, and CD38, and efflux of R123.38 Based on surface phenotype, their predominance in CB, and ability to mature into naive B cells in vitro,38,45 it is probable that T3 cells and CD5+ prenaive B cells represent the same population of developing B cells. Because our study relied on CD10 expression to identify transitional B cells, neither the CD21lo nor CD21hi subset corresponds to T3/CD5+ (CD10−/lo) prenaive B cells. Our analysis demonstrated that CD21lo and CD21hi transitional B cells in PB of normal donors express comparable levels of CD24 and CD38, raising the possibility that they do not correspond to CD38+++CD24+++ T1 and CD38++CD24++ T2 transitional cells, respectively. However, when we assessed CD24 and CD38 on transitional B-cell subsets in post-HSCT patients, we, like Anolik et al,26 observed that, at the earliest times of analysis, CD21lo transitional B cells expressed higher levels of both markers compared with CD21hi transitional B cells (supplemental Figure 4). Despite this initial difference, expression of CD24 and CD38 on the transitional B-cell subsets eventually approximated one another (supplemental Figure 4), thus suggesting that expression levels of CD24 and CD38 on transitional B-cell subsets reflect the time since their generation and export from the BM, rather than a delineation between transitional subsets. Based on this, it is probable that the CD21lo and CD21hi transitional B-cell subsets are the CD38+++CD24+++ T1 and CD38++CD24++ T2 cells.26 This is further supported by the findings that expression of CD21 and CD23 was increased on T2/CD21hi compared with T1/CD21lo transitional B cells and that neither population was responsive to BAFF in in vitro survival or differentiation assays.26,45
Combining the data from our current study and those recently published, it is possible to propose a map of human peripheral B-cell development whereby immature B cells exit the BM as CD21lo transitional/T1 cells that develop into CD21hi transitional/T2 cells in a BTK-dependent manner (Figure 6). Interestingly, the reduction in autoreactivity between the CD21lo and CD21hi transitional stages suggests that a checkpoint for self-tolerance exists at this step. This is analogous to the checkpoints between early immature and immature B cells, and transitional and naive B cells where autoreactive B cells are deleted from the repertoire,5 and is consistent with the increased frequency of T1/CD21lo transitional B cells in some patients with systemic lupus erythematosus.24,46 CD21hi transitional B cells then develop into T3/prenaive B cells, which undergo a final maturation process to become naive B cells (Figure 6). It will be important to investigate the autoreactivity and proliferative history of the T3/prenaive B-cell population to determine whether replication is required for the development of these cells into the naive compartment and if deletion of autoreactive cells occurs beyond the CD10+ stage of B-cell development. Overall, this framework will allow refined molecular investigation of human B-cell development in cases of immunologic dyscrasias characterized by perturbed B-cell behavior, such as immunodeficiency, autoimmunity, and malignancy.
Presented in oral form in the Keystone Symposia, B cells in context, Taos, NM, February 28, 2009.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Jerome Darakdjian and Chris Brownlee for cell sorting as well as Anne Maree Johnston, Kay Montgomery, and Katherine Stephen for the collection and delivery of the HSCT blood samples.
This work was supported by research grants and fellowships awarded by the National Health and Medical Research Council of Australia and Cancer Institute New South Wales.
Authorship
Contribution: S.S. designed the research, performed experiments, analyzed and interpreted results, and wrote the manuscript; B.S.-N. and R.N. provided CB samples; D.A.F., J.G., M.W., A.W., and P.J.S. provided patient samples; S.G.T conceptualized and designed the research and wrote the manuscript; and all authors commented on the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Stuart G. Tangye, Garvan Institute of Medical Research, 384 Victoria St, Darlinghurst 2010, NSW, Australia; e-mail: s.tangye@garvan.org.au.