To the editor:
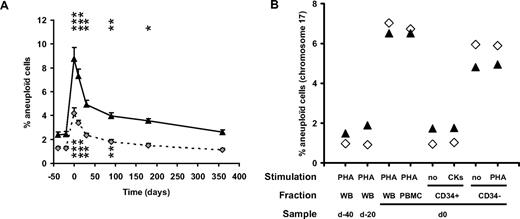
Lymphocytes from granulocyte colony-stimulating factor (G-CSF)–mobilized donors display epigenetic and genetic alterations similar to those observed in leukemia patients.1 To further evaluate the scope and duration of G-CSF–induced genetic alterations, 24 healthy donors were followed for 1 year after peripheral blood stem cell mobilization by fluorescence in situ hybridization (FISH) analysis (Figure 1A). The chromosomes 8 and 17 aneuploidy increased an average of 4.5- and 4.3-fold, respectively, at the time of G-CSF mobilization and subsequently decreased, but remained significantly higher than baseline at 3 and 6 months after mobilization (Figure 1A). The frequency of cells with monosomy was higher, and returned to baseline more gradually than that of cells with trisomy or multisomy (data not shown).
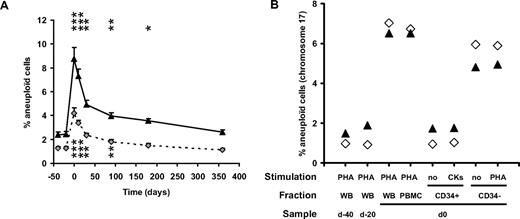
FISH analysis of PBMC from healthy donors after G-CSF mobilization. (A) Twenty-four healthy consenting donors (9 males, 15 females; mean age 44.2 ± 2.5 years; range, 23.3-57.9 years) were recruited in 3 transplantation centers (Besançon, Nancy, and Lyon, France), after approval by the Besançon University Hospital ethical committee. Blood samples harvested at baseline (2 control samples harvested before G-CSF administration, at day −30 ± 6 and −5 ± 1, respectively), just after G-CSF (Granocyte, Lenograstim, Chugai Pharma, Paris, France, n = 6 or Neupogen, Filgrastim, Amgen, Neuilly sur Seine, France, n = 18) mobilization (day 0, ie, before the first cytapheresis), after the last cytapheresis (day 0.7 ± 0.2) and at 1 (day 32. ± 1), 3 (day 92 ± 1), 6 (day 184 ± 1), and 12 (day 366 ± 2) months after mobilization were analyzed for aneuploidy, quantified after FISH of chromosomes 8 (gray diamonds, dashed line) and 17 (▲, full line) specific centromeric probes on PHA-stimulated whole-blood samples. For clarity, data are arbitrarily set at −40, −20, 0, 10, 30, 90, 180, and 360 days, respectively. Each time point is compared with the mean of baseline values using a paired t test; n = 24 except at days 180 (n = 20) and 360 (n = 17). *P < .05; **P < .005; ***P < .001. (B) CD34+ cells were purified to 96% and 89%, respectively, from 2 donors' peripheral blood mononuclear cells (PBMCs) harvested at day 0 (D21, ▲; D22, ◊) by positive immunomagnetic sorting (Miltenyi Biotec) according to the manufacturer's recommendations. The CD34+ fraction was either analyzed immediately after sorting, without stimulation (no), or was incubated in 200 μL of Iscove modified Dulbecco media (IMDM) in the presence of 20% FCS, 1% essential amino acid and stimulated with the hemopoietic growth factors Fms-like tyrosine kinase 3 ligand (Flt-3L; 300 ng/mL), stem cell factor (SCF; 300 ng/mL), IL-3 (10 ng/mL), IL-6 (10 ng/mL), and G-CSF (50 ng/mL) for 7 days at 37°C in a 5% CO2 environment (CK) before hybridization with a chromosome 17–specific centromeric probe. The CD34− fraction was either analyzed immediately after sorting, without stimulation (no) or stimulated by PHA (10 μg/mL), before hybridization. The CD34+ and CD34− fractions were compared with their unsorted PHA-stimulated PBMC counterpart or to PHA-stimulated whole blood (WB) harvested from the same donors before G-CSF mobilization (control samples) and at day 0. Similar results were obtained, although at lower frequencies, when cells were hybridized with chromosome 8–specific centromeric probes.
FISH analysis of PBMC from healthy donors after G-CSF mobilization. (A) Twenty-four healthy consenting donors (9 males, 15 females; mean age 44.2 ± 2.5 years; range, 23.3-57.9 years) were recruited in 3 transplantation centers (Besançon, Nancy, and Lyon, France), after approval by the Besançon University Hospital ethical committee. Blood samples harvested at baseline (2 control samples harvested before G-CSF administration, at day −30 ± 6 and −5 ± 1, respectively), just after G-CSF (Granocyte, Lenograstim, Chugai Pharma, Paris, France, n = 6 or Neupogen, Filgrastim, Amgen, Neuilly sur Seine, France, n = 18) mobilization (day 0, ie, before the first cytapheresis), after the last cytapheresis (day 0.7 ± 0.2) and at 1 (day 32. ± 1), 3 (day 92 ± 1), 6 (day 184 ± 1), and 12 (day 366 ± 2) months after mobilization were analyzed for aneuploidy, quantified after FISH of chromosomes 8 (gray diamonds, dashed line) and 17 (▲, full line) specific centromeric probes on PHA-stimulated whole-blood samples. For clarity, data are arbitrarily set at −40, −20, 0, 10, 30, 90, 180, and 360 days, respectively. Each time point is compared with the mean of baseline values using a paired t test; n = 24 except at days 180 (n = 20) and 360 (n = 17). *P < .05; **P < .005; ***P < .001. (B) CD34+ cells were purified to 96% and 89%, respectively, from 2 donors' peripheral blood mononuclear cells (PBMCs) harvested at day 0 (D21, ▲; D22, ◊) by positive immunomagnetic sorting (Miltenyi Biotec) according to the manufacturer's recommendations. The CD34+ fraction was either analyzed immediately after sorting, without stimulation (no), or was incubated in 200 μL of Iscove modified Dulbecco media (IMDM) in the presence of 20% FCS, 1% essential amino acid and stimulated with the hemopoietic growth factors Fms-like tyrosine kinase 3 ligand (Flt-3L; 300 ng/mL), stem cell factor (SCF; 300 ng/mL), IL-3 (10 ng/mL), IL-6 (10 ng/mL), and G-CSF (50 ng/mL) for 7 days at 37°C in a 5% CO2 environment (CK) before hybridization with a chromosome 17–specific centromeric probe. The CD34− fraction was either analyzed immediately after sorting, without stimulation (no) or stimulated by PHA (10 μg/mL), before hybridization. The CD34+ and CD34− fractions were compared with their unsorted PHA-stimulated PBMC counterpart or to PHA-stimulated whole blood (WB) harvested from the same donors before G-CSF mobilization (control samples) and at day 0. Similar results were obtained, although at lower frequencies, when cells were hybridized with chromosome 8–specific centromeric probes.
To investigate whether such cytogenetic abnormalities affected CD34+ cells and required the induction of cell division, CD34+ and CD34− fractions, purified from 2 donors at time of mobilization, were analyzed either immediately or after ex vivo culture. The frequencies of chromosome 8 (data not shown) and chromosome 17 (Figure 1B) aneuploidy were increased in the CD34− fractions of samples harvested immediately after G-CSF mobilization, similar to the levels observed in whole blood or peripheral blood mononuclear cells from the same blood harvests. However, the frequencies of aneuploidy in the CD34+ fractions did not increase compared with control samples harvested before G-CSF administration, suggesting that G-CSF–induced cytogetenic abnormalities (1) affect only mature CD34− cells and not CD34+ cells and (2) are present before the induction of proliferation and therefore are not in vitro artifacts resulting from cell division. Several possibly complementary mechanisms may account for these observations: (1) the increased hematopoiesis, not G-CSF per se, may lead to an increased production in the marrow and release in the blood of aneuploid cells; (2) aneuploid cells that are normally sequestered in the marrow are specifically released in the circulation upon G-CSF mobilization; (3) pre-existing clones with monosomy or multisomy may expand upon G-CSF treatment, as reported with chromosome 7 monosomy in patients with aplastic anemia2 ; or (4) G-CSF directly contributes to aneuploidy by modulating the expression of genes involved, for example, in chromatin condensation, mitotic spindle formation, chromosome segregation, or kinetochore activity, such as genes governing the anaphase-promoting complex/cyclosome,3 with, as a consequence, improper chromatid separation during the anaphase.
Because up to 10% of cells were aneuploid for either chromosome 8 or 17 at time of mobilization, we hypothesized that cytogenetic abnormalities would be present on at least one chromosome of any pair in a majority of cells. Among a total of 365 karyotypes analyzed on 15 (n = 1 donor) or 50 (n = 7 donors) metaphases of G-CSF–mobilized, phytohemagglutinin (PHA)–activated blood samples, only 1 abnormality (chromosome 15 translocation) was observed. When further analyzed by FISH, all the metaphases (4.2%-8.5% of total cells) were euploid for both chromosome 8 and 17 whereas aneuploid nuclei were found exclusively in the interphasic fraction (9.0%-44.5% within 200 interphases analyzed/donor), suggesting that the absence of abnormalities upon caryotyping might be in relation with an inability of aneuploid cells to complete metaphases and to undergo cell division, which may be counterintuitively associated with a protective effect on transformation.4 A better understanding of the mechanisms leading to cytogenetic abnormalities upon G-CSF–induced mobilization is therefore mandatory to better assess the risks of transformation in healthy donors.5
Authorship
Acknowledgments: We are grateful to the donors who volunteered to be enrolled in this study. This work was supported by the Agence de la Biomédecine and the Ligue Contre le Cancer, Comité du Doubs. C.M. is a recipient of a fellowship of Région Franche-Comté and of the Fondation Transplantation (FDTSFV).
Contribution: C.M.-S. performed research, recorded and analyzed data, and contributed to paper writing; F. Larosa, F. Legrand, B.W., M.M., and D.R. informed and recruited donors; P.L. performed donor management, data management, and data recording; M.P. performed statistical analyses; N.R. performed donor management; M.T., S.M.-N., and O.H. informed donors and performed HSC harvests; J.-R.P. and F.V. performed research experiments; M.-A.C.-R. and J.-L.B. contributed to data analysis; F.P. contributed to study design; E.D. initiated and designed the study, recruited donors and contributed to paper writing; P.T. initiated and designed the study and wrote the paper; and E.R. designed and managed the study, performed data analysis, and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
The current address for E. R. is Inserm, U748, 67000 Strasbourg, France.
Correspondence: Eric Robinet, PhD, Inserm U748, 3 rue Koeberlé, 67000 Strasbourg, France; e-mail: e.robinet@unistra.fr.