Abstract
Abstract 1435
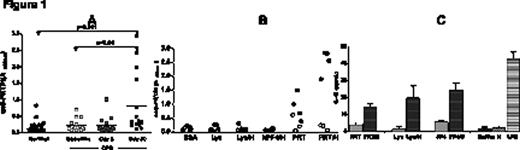
Platelet Factor 4 (PF4)/heparin (H) multimolecular complexes initiate an immune response that can ultimately lead to complications of Heparin-Induced Thrombocytopenia (HIT), a life-threatening prothrombotic disorder. We have previously shown that PF4:H multimolecular complexes assemble through non-specific electrostatic interactions and that other unrelated positively-charged proteins such as protamine (PRT) and lysozyme (Lys) exhibit similar biophysical interactions with heparin (ASH 2009; abstract # 1316). In these earlier studies, we showed that PRT/H and Lys/H, like PF4/H, show heparin-dependent binding over a range of heparin concentrations and that formation of multimolecular complexes occurs at distinct stoichiometric ratios (PRT/H at 3:1 and Lys/H at 5:1 molar ratios). We now extend these observations in vivo to show relevance to human disease. Using a murine immunization model, we show that mice injected with PRT/H and Lys/H multimolecular complexes, but not PRT alone, Lys alone or buffer, develop antigen-specific immune responses. In additional studies, we show that the immune response to PRT/H or Lys/H shares important biologic similarities with the humoral response to murine (m) PF4/H multimolecular complexes. Specifically, we demonstrate that antibody formation to PRT/H and Lys/H is heparin-dependent (occurs optimally at certain stoichiometric ratios) dose-dependent (requires threshold amounts of multimolecular complexes) and shows serologic transience. To demonstrate the clinical relevance of our findings, we examined patients undergoing cardiopulmonary bypass (CPB) for development of PRT/H antibodies. For these studies, we assayed the plasma from healthy subjects (n=45) and patients undergoing CPB (n=15) at three time points {baseline (BL), 5 days (5D) and 30 days (30D) after CPB} for the presence of PRT/H antibodies. As shown Figure 1A, plasma from normal subjects and patients undergoing CPB patients at BL and D5 displayed minimal reactivity in the PRT/H ELISA. However, by 30D, we observed that 4/15 patients (27%) developed significantly elevated levels of antibodies to PRT/H as compared to normals, or their respective samples obtained at baseline or 5D after surgery. Seropositive patients (filled symbols, n=4) as compared to seronegative patients (open symbols, n=3) recognized PRT/H and to some extent, PRT alone, but did not cross-react with other antigens including PRT/H, BSA, Lys, Lys/H or human PF4/H, Figure 1B; p<0.001). To identify the mechanism by which protein/heparin multimolecular complexes triggered immune activation, we incubated murine dendritic cells from non-immunized C57Bl/6 mice with heparin or buffer, protein (mPF4, PRT or Lys), or protein/H complexes and measured IL-12, a marker of dendritic cell activation. As shown in Figure 1C, we demonstrated that IL-12 levels were significantly increased in wells containing protein/H complexes as compared to wells containing uncomplexed protein, buffer or heparin. Taken together, these studies indicate that heparin significantly alters the biophysical and biological properties of positively-charged compounds through formation of macromolecular complexes that lead to dendritic cell activation and trigger immune responses in vivo.
Arepally:Glaxo Smith Kline: Speakers Bureau; Paringenix: Research Funding; University Of New Mexico: Patents & Royalties; Amgen: Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.