Abstract
Abstract 1880
AL is a clonal plasma cell disorder with a reported median survival (OS) of 13 months from diagnosis. Therapy involves suppression of amyloidogenic light chain production from clonal plasma cells. Hematological response (HR) is critical to improve organ function and survival. Autologous transplantation (AHCT) despite its risks, if performed in expert centers leads to rapid HR and organ function improvement. Those with poor Karnofsky performance score (KPS), multisystem disease or cardiac AL are poor candidates for AHCT.
We assessed the efficacy of bortezomib (V) based combination therapy in newly diagnosed AL in a population with poor KPS or ineligible for AHCT.
Among 35 consecutive newly diagnosed, biopsy proven AL patients (pts) since 2006, 29 pts received at least 2 cycles of V based therapy and were evaluable. Non evaluable pts (n=6) did not complete 2 cycles of therapy: liver function abnormalities (1), neuropathy (1); or early death (4 within 6 weeks of diagnosis). Two treatment protocols were used sequentially over this period. From 2006 – 4/2009, V twice weekly with dexamethasone weekly (VD) for 2 out of 3 weeks (n= 14) and thereafter weekly VD with cyclophosphamide (CVD) x3 on a q 4 week schedule (n=15). For complete or partial responses (CR/PR) after 6 cycles of therapy, V maintenance was administered every 2 weeks. Response evaluation was according to the 10th international AL symposium criteria.
Median age was 68 years (range 35–88 years; 55% female). Subtype of AL was lambda in 55%. KPS was less than 80 in more than 50%. Majority had 3 or more organs involved: 62% cardiac, 62% renal, 41% GI, 20% neurologic and 17% liver involvement. Cardiac stage was Mayo III in 50%. Hematological response (HR) was achieved in 86% including 34% CR and 52% PR. Among non-responders (14%), 2 have died after 2 cycles and 1 discontinued therapy. Median number of cycles to achieve a HR was 3 cycles (range, 1 to 9 cycles). CR was achieved after a median of 4 cycles for VD and 3 cycles for CVD. At median follow up of 22 mo (range 2–57 mo), median overall and progression free (PFS) survival have not been reached. Median OS for patients with cardiac involvement (n= 18) was 11 mo (range 2 – 29 mo). Among those with HR, 60% had at least one organ response including 44% renal and 55% cardiac responses. Among those with HR, 2 died following hematological progression; while 6 died of AL related organ failure. Half the deaths due to AL organ dysfunction were within the first 4 cycles. Median duration of response has not been reached. Hematopoietic cell harvest was successful in 41% (n=12) of which 4 pts completed consolidative AHCT.
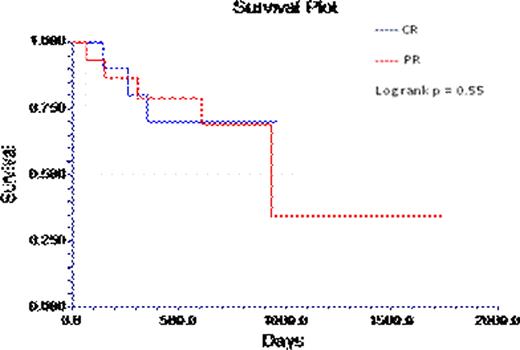
There was no difference in survival between those achieving CR vs. PR (log rank p = 0.5, Fig 1) or between recipients of CVD vs. VD (log rank p= 0.47 and Table 1).
| . | VD (n = 14) . | CVD (n = 15) . |
|---|---|---|
| ORR | 93% (13) | 80% (12) |
| CR | 36% (5 pts) | 40% (6 pts) |
| After first 4 cycles | ||
| ORR | 64% (9) | 66% (10) |
| CR | 14% (2) | 29% (4) |
| . | VD (n = 14) . | CVD (n = 15) . |
|---|---|---|
| ORR | 93% (13) | 80% (12) |
| CR | 36% (5 pts) | 40% (6 pts) |
| After first 4 cycles | ||
| ORR | 64% (9) | 66% (10) |
| CR | 14% (2) | 29% (4) |
Therapy related non-hematologic adverse effects (all grades) included peripheral neuropathy (27%), gastrointestinal events with nausea, vomiting and diarrhea (17%), infections (3%) and blood clots (3%). Grade ≥ 3 toxicities were 3% and discontinuations/dose reductions resulting from toxicities were 10% and 3% respectively. Worsening of preexisting neuropathy was rare (2 pts out of 6).
In this cohort of advanced multisystem AL with poor KPS; VD or CVD established rapid responses in 86%. In comparison with upfront AHCT, this approach has the advantages of lower toxicity, wider applicability and similar response time although durability of response is unknown at this time. Stem cell harvest was feasible when attempted. Addition of cyclophosphamide was well tolerated with the benefit of rapid onset of CR and allowed once weekly V without compromising efficacy. In good risk pts, AHCT should be compared against CVD in prospective randomized fashion.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.