Abstract
Abstract  2504
2504
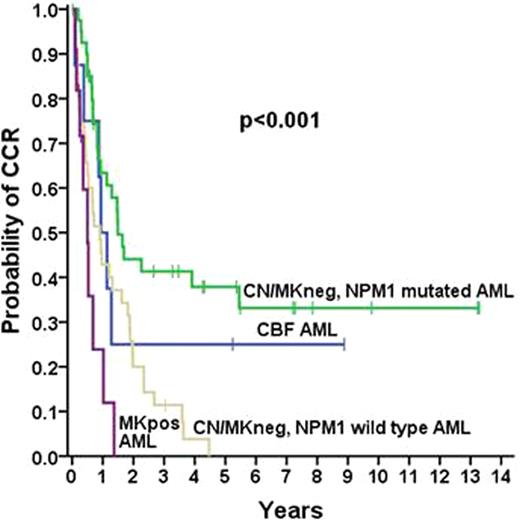
Whereas treatment strategies for AML patients (pts) aged <60 years are well established, therapy in the elderly deserves special consideration. In the present study, we analysed survival and risk factors predicting outcome in 192 consecutive patients (pts) with de novo AML aged ≥60 years (median age: 70.5 years, range 60–89 years). Pts were treated at a single center (Medical University of Vienna) between November 1994 and January 2011, and received conventional induction chemotherapy with DAV (daunorubicin, 45 mg/m2/day, days 1–3; etoposide, 100 mg/m2/day, days 1–5; ARA-C, 2 × 100 mg/m2/day, days 1–7; between 1994 and 2008). Pts achieving a complete hematologic remission (CR) were scheduled to receive a full consolidation program with up to 4 cycles of IDAC (ARA-C, 2 × 1 g/m2/day i.v. on days 1, 3, and 5). Of the 192 pts, 115 (60%) achieved a CR following induction therapy, 57 pts (30%) did not achieve a CR, and 20 pts (10%) died within 40 days after start of induction therapy. Among several parameter including the leukocyte count, hemoglobin, platelet count, LDH, and fibrinogen, only the karyotype according the the Breems-Score was an independent predictor for achieving CR in our multivariate analyses. CR rates in pts with core binding factor (CBF) AML, cytogenetically normal (CN) AML, non-monosomal karyotype (Mkneg) AML, and monosomal karyotype (Mkpos) AML, were 100% (8/8), 66% (57/87), 56% (33/59), and 44% (11/25), respectively (p<0.05). Of the 115 pts in CR, 106 (92%) received up to 4 cycles of IDAC (4 cycles, n=53 pts [50%]; 3 cycles, n=20 pts [19%]; 2 cycles, n=20 [19%]; and 1 cycle, n=13 [12%]). Treatment was well tolerated without grade 3 or 4 neurotoxicity. The most frequent non hematologic toxicity was febrile neutropenia requiring antibiotics (1st consolidation: 50.5%; 2nd consolidation: 58.4%, 3rd consolidation: 55.6%, 4th consolidation: 51.0%). A remarkable finding was that the median time of absolute neutropenia in all IDAC cylces (8–9 days/consolidation) was relatively short when compared to induction chemotherapy or other consolidation regimens. Only two patients died during consolidation with IDAC (cardiac failure, n=1, severe sepsis, n=1). The median overall survival (OS) for all pts was 9.7 months, and for pts achieving a CR, the OS amounted to 24.2 months. The most important predictive variables for continuous complete remission (CCR) were the karyotype and the presence of an NPM1 mutation determined by PCR and melting point analysis. As assessed by uni- and multivariate analysis, both parameters were found to be independent predictive variables (p<0.05), whereas the other markers examined, i.e. FLT3 ITD, overexpression of ERG, MN1, or BAALC, as well as the number of induction cycles required to achieve CR, showed no predictive value. The median CCR and CCR after 5 years were 17.9 and 31.3%, respectively in pts with CN/Mkneg-NPM1 mutated AML, and 11.3 months and 25% for pts with CBF AML (p<0.001; see figure). In pts with CN/Mkneg and wild type NPM1 AML and Mkpos AML, the median CCR rates were 10.8 and 6.0 months, respectively. Together, our data show that elderly patients with CN/Mkneg-NPM1 mutated AML or CBF AML can achieve long term CCR when treated with intensive chemotherapy and up to 4 consolidation cycles of IDAC, whereas pts with CN/Mkneg-NPM1 wild type AML and Mkpos AML did not achieve long term CCR despite intensive consolidation. These results are in favour of individualized chemotherapy in elderly pts with AML and suggest that patient-selection in advanced age should not only be based on patient-related factors and the karyotype but also on the NPM1 mutation status.
Disclosures:
No relevant conflicts of interest to declare.
Author notes
*
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract
© 2011 by The American Society of Hematology
2011