Abstract
Abstract 55
Recurrent chromosomal translocations involving the mixed lineage leukaemia (MLL) gene initiate aggressive forms of leukaemia, which confer a poor prognosis and are often refractory to conventional therapies. Recent efforts have begun to unravel the molecular pathogenesis of these malignancies. Several groups have demonstrated that MLL-fusions associate with two macromolecular chromatin complexes; the polymerase associated factor (PAFc) complex, which interacts with the N-terminal domain of MLL, a portion of the protein that is retained in all the described fusions, or the super elongation complex (SEC), via interaction with the C-terminal fusion partner. These complexes play an integral role in regulating transcriptional elongation and this function appears to be aberrantly co-opted by the MLL-fusions to initiate and perpetuate transcriptional programmes that culminate in leukaemia.
In this study we used a systematic global proteomic survey incorporating quantitative mass spectrometry to demonstrate that MLL-fusions, as part of SEC and PAFc complexes, are associated with the BET family of acetyl lysine recognition chromatin “adaptor” proteins. These data provided the basis for therapeutic intervention in MLL-fusion leukaemia, via the displacement of the BET family of proteins from chromatin. Targeting the BET proteins to alter aberrant transcriptional elongation has recently been demonstrated to be possible using small molecule inhibitors that selectively bind the tandem bromodomain at the amino-terminus of the ubiquitously expressed BET proteins (BRD2/BRD3/BRD4).
We developed a novel class of potent small molecule inhibitors to the BET family, which is chemically distinct to previously published BET-inhibitors. We then used this new compound (I-BET151) to demonstrate its profound and selective efficacy against human MLL-fusion leukaemic cell lines in liquid culture as well as clonogenic assays in methylcellulose. We also establish that primary murine progenitors retrovirally transformed with MLL-ENL and MLL-AF9 are equally susceptible to treatment with I-BET151. We show that the main phenotypic consequence of BET inhibition in MLL fusion leukaemia is a dramatic early induction of cell cycle arrest and apoptosis. Global gene-expression profiling, following I-BET151 treatment in two different human MLL-fusion leukaemia cell lines (expressing MLL-AF4 and MLL-AF9), highlights a common differentially expressed gene signature that accounts for this phenotype. Importantly, chromatin immunoprecipitation analyses at direct MLL target genes including BCL2, C-MYC and CDK6, indicate that I-BET151 selectively inhibits the recruitment of BET family members BRD3/BRD4, and SEC and PAFc components. These events result in the inefficient phosphorylation and release of paused POL-II from the TSS of these genes providing mechanistic insight into the mode of action of I-BET151 in MLL-fusion leukaemia.
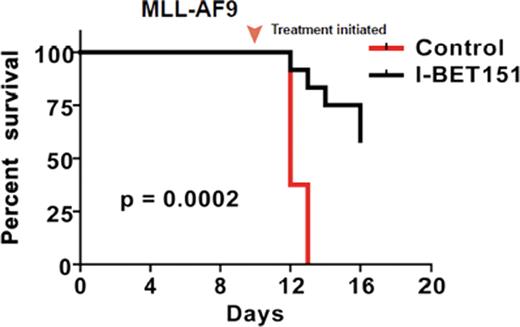
We subsequently established the therapeutic efficacy of I-BET151 in vivo by demonstrating dramatic disease control in murine models of MLL-AF4 and MLL-AF9 leukaemia. Finally, we also demonstrate that I-BET151 accelerates apoptosis in primary leukaemic cells from a large number of patients with various MLL-fusion leukaemias, by affecting a similar transcription programme to that identified in the human leukaemic cell lines. Importantly, we also demonstrate that I-BET151 significantly reduces the clonogenic potential of isolated primary leukaemic stem cells, suggesting that disease eradication may be possible.
Prinjha:GSK: Employment. Chung:GSK: Employment. Lugo:GSK: Employment. Beinke:GSK: Employment. Soden:GSK: Employment. Mirguet:GSK: Employment. Jeffrey:GSK: Employment. Lee:GSK: Employment. Kouzarides:GSK: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.