Abstract
Abstract 1084
We have previously demonstrated that monoclonal antibodies to the CD44 antigen can ameliorate murine ITP as successfully as IVIg. We have also shown that, similar to IVIg, these antibodies do not require expression of the neonatal Fcγ receptor FcRn. Unlike IVIg, however, CD44 antibodies do not require the Fc portion to function in the murine ITP model. A lack of requirement for the Fc region of CD44 antibodies would indicate that Fcγ receptors should not be necessary for antibody function, but CD44 antibodies do not ameliorate ITP in FcγRIIB deficient mice (B6;129S-Fcgr2btm1Ttk/J). Recent data from some groups have suggested that IVIg can in fact ameliorate murine ITP in the absence of FcγRIIB, depending on the background strain of the mouse. These data cast doubt on the simplistic view that IVIg ameliorates murine ITP by upregulating macrophage FcγRIIB expression.
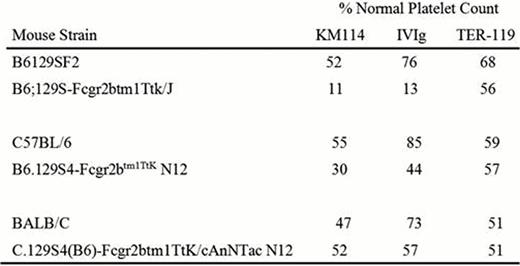
The requirement for the expression of the inhibitory IgG receptor FcγRIIB has been a prominent theory as to how IVIg ameliorates ITP. Similar to IVIg, the CD44 antibody KM114 does not function in B6;129S-Fcgr2btm1Ttk/J FcγRIIB deficient mice. Another IVIg product which has ameliorative effects similar to IVIg but appears to function via a different mechanism is anti-D. We have previously shown that IVIg and a monoclonal antibody with “anti-D like” activity, TER-119, can successfully ameliorate thrombocytopenia in a murine model of ITP. In contrast to KM114 and IVIg, TER-119 is fully functional in B6;129S-Fcgr2btm1Ttk/J mice. To further characterize the therapeutic CD44 antibody KM114, we have analysed KM114 function in FcγRIIB deficient mice on 3 different backgrounds. Specifically, mice were pretreated with nothing, 50 ug KM114, 50 mg IVIg, or 50 ug TER-119 thirty min prior to administration of the anti-platelet antibody MWReg30.
Here, we have confirmed that TER-119, but not KM114 or IVIg, successfully ameliorates murine ITP in B6;129S-Fcgr2btm1Ttk/J mice. These mice are not fully congenic, and have been reported to be approximately a 50:50 mix of B6 and 129S4 (Leontyev, et. al Blood. 31;119(22):5261-4). To investigate whether or not KM114 required the presence of FcγRIIB in mice from different backgrounds, we next employed B6.129S4-Fcgr2btm1TtK N12 FcγRIIB deficient mice, which, unlike B6;129S-Fcgr2btm1Ttk/J mice, are congenic on the B6 background. KM114 and IVIg were both effective in treating thrombocytopenia in these mice. TER-119 was also effective, as expected. To further explore these findings, we next employed FcγRIIB deficient mice which are congenic on the BALB/C background, C.129S4(B6)-Fcgr2btm1TtK/cAnNTac N12 mice. Again, we found that KM114, IVIg and TER-119 were all effective in treating thrombocytopenia in these mice.
As KM114 functions in congenic B6.129S4-Fcgr2btm1TtK N12 and C.129S4(B6)-Fcgr2btm1TtK/cAnNTac N12 mice, but not in mixed background B6;129S-Fcgr2btm1Ttk/J mice, these data suggest that rather than FcγRIIB expression being necessary, it may be the mouse background genes that affect KM114's ability to function. Further experiments using mice of different genetic backgrounds may assist in understanding these findings.
No relevant conflicts of interest to declare.