Abstract
Abstract 2403
Pediatric Burkitt Lymphoma (BL) is the most common histological subtype of Non Hodgkin Lymphoma (NHL) in children and adolescents (Cairo et al., Blood, 2007; Miles/Cairo, BJHaem., 2012). We previously identified in a subset analysis that children with BL and a 13q deletion, particularly 13q14.3, had significantly poorer outcome and inferior overall survival (OS) despite aggressive short, intensive multiagent chemotherapy (Poirel/Cairo et al., Leukemia, 2009; Nelson/Cairo/Sanger et al., BJHaem., 2009). Deleted in Lymphocytic Leukemia 1 (DLEU1) is a long non codign RNA gene with the BL classifier genes as reported by Dave et al (NEJM. 2006) on chromosome 13q14.3 region and the expression levels of RASSF1, TUBB2C and UBR1 were significantly higher in BL than in diffuse large B-cell lymphoma (DLBCL) (Day/Cairo et al., AACR, 2008). Sequence-specific Zinc Finger Nucleases (ZFN) and Transcription Activator-Like Effector Nucleases (TALENs) technologies have been developed for precision targeted genome editing in in vitro experiments with high efficiency and TALEN technology has been applied in a variety of organisms and stem cells for new experimental and therapeutic tools (Carlson et al., Mol Ther., 2012).
We hypothesize that 1) TALEN technology is suitable for the modification of and silencing of DLEU1 locus and 2) DLEU1 may have function as a tumor suppressor gene and therefore examined whether the loss of DLEU1 by TALEN-mediated DLEU1 knockdown Raji BL cells result in change in proliferation and programmed cell death.
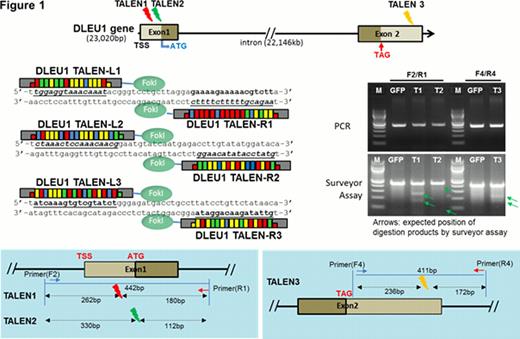
TALENs were constructed based on REAL (restriction enzyme and ligation) assembly methods (Sander et al., Nat. Biotech, 2011) for DLEU1 gene modification. DLEU1 TALEN pairs or combinations were transfected into 293T and Raji cells using lipofectamin reagent (Invitrogen) and Amaxa nucleofection kit, respectively. Genomic DNA and total RNA were extracted using genomic DNA extraction kit (Promega) and Trizol reagent (Invitrogen). Surveyor mutation detection assay (Transgenomic) was applied for TALENs validities and confirmed by sequencing analysis. Quantitative RT-PCR was performed by CFX96 Real-time system (Bio-rad) using qScript™ cDNA Synthesis Kit (Quantas) and SsoFast™ EvaGreen® Supermix (Bio-rad). Transfected cells were re-plated (1×104) into 48 well plates at 24hours post transfection and then counted every 24hrs for cell proliferation assay. For spontaneous apoptosis assay, DLEU1 TALENs transfected Raji cells were re-plated into 96 well plates at 24hours post-transfection and measured caspase 3/7 activities (Promega) using Clarity Luminescence microplate reader (Biotek) and statistical significance was determined by a one or two-tailed Student t-test.
Three pairs of functional DLEU1 TALENs (T1, T2, and T3) targeting endogenous DLEU1 gene generated and confirmed their validities with gene modification caused by non-homologous end joining (NHEJ) in TALENs mediated 293T [Figure 1]. Forty eight hours after transiently transfection in Raji cells, combined DLEU1 TALENs, T1/T2 (T12), T1/T3 (T13) and T2/T3 (T23) mediated Raji cells were dramatically reduced expression of DLEU1 mRNA (about 82–93% reduction) compared to untransfected Raji cells [92% reduction (p<0.002), 93% (p<0.0006), and 92% (p<0.0003)], respectively, and compared to empty vector as a control [82% reduction (p<0.05), 87% (p<0.0001), and 82% (p<0.001, respectively)]. T12 and T13-mediated DLEU1 knockdown Raji cells showed significantly an increase of cell proliferation [1.2-fold (p<0.02) and 1.5-fold (p<0.04) at day 5, respectively]. In spontaneous apoptotic assays, T13 mediated Raji showed significantly 20% (p<0.05) of reduction of caspase 3/7 activities. In comparison of mRNA expression of DLEU1 network genes in T13 mediated Raji cells, there were no significant differences in mRNA expression of c-myc compared to control, however, there was significantly reduced expression of RASSF1 and UBR1 mRNA (<7.7-fold, p<0.005 and <11.1-fold, p<0.0005), respectively, and significantly increased expression of TUBB2C mRNA (>7.5-fold, p<0.005).
We demonstrate that 1) DLEU1-TALENs are useful tools for the modification of endogenous DLEU1 gene expression, 2) DLEU1 may have as a tumor suppressor gene in BL, and 3) DLEU1 TALEN mediated gene knockdown resulted in inhibition of BL apoptosis and increase in cell proliferation.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.