Abstract
Abstract  746
746
The clinical benefit of high dose chemotherapy programs in patients with diffuse large B cell lymphomas (DLBCL) with unfavorable presentation (IPI>2) is still matter of debate and several prospective randomized clinical trials have been conducted to address this point.
In year 2005 the cooperative study group GITIL launched a multicenter phase III trial (R-HDS 0305, Clinical Trials.gov.number NCT00355199) in DLBCL patients without CNS involvement and the following inclusion criteria: age between 18–60 years, stage > II B-bulk with ECOG-PS=0-3 and age adjusted IPI (aaIPI) 2–3 or age 61–65 years with ECOG-PS = 0–2 and IPI >3. The control group received 8 courses of the conventional R-CHOP-14 chemotherapy program, followed by IFRT, in patients who achieved at least a partial response (PR) after 4 cycles. Cases refractory to R-CHOP-14 could be rescued by R-HDS. The experimental arm (R-HDS) included: 3 courses of doxorubicin-containing chemotherapy (APO), followed by high-dose cyclophosphamide (CTX) 7 g/sqm, Ara-C (2g/sqm every 12 hours for 6 days), etoposide 2 g/sqm plus cisplatin 100 mg/sqm. After R-HDS chemotherapy, a mitoxantrone-melphalan (60 and 180 mg/sqm, respectively) or BEAM conditioning regimen with autologous stem cell transplantation (ASCT) + IFRT, as above. Rituximab (375 mg/sqm) was given for a total of 6 doses, twice after CTX and Ara-C based cycles, as in vivo purging before CD 34+ cells harvest, and twice after ASCT (Tarella C. et al Leukemia 21:1802–1811, 2007). The primary end-point of the study was to test an increase of 2-year Event Free Survival (EFS) from 50% in the R-CHOP arm to 65% in the intensified R-HDS arm. Secondary end-points were Disease Free Survival (DFS), Overall Survival (OS) and toxicity.
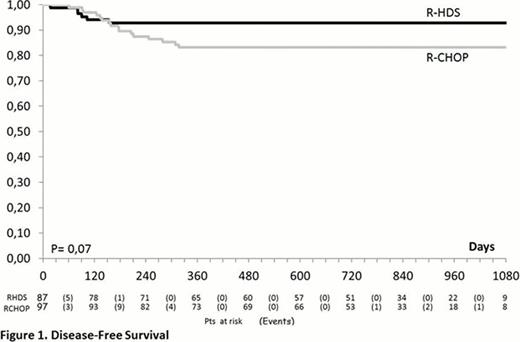
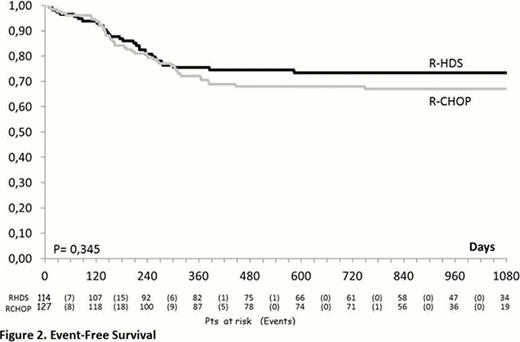
From June 2005 to June 2011, 248 patients were enrolled in the study (R-CHOP-14=127; R-HDS=121) and 241 were evaluable as to completeness of information. The median age was 51 years (range, 18–65), 32 subjects (13.3%) had> 60 years and the M/F ratio was 1.39. Clinical features were as follows: advanced Ann Arbor clinical stage (93%), BM infiltration (22%), bulky disease (69%), elevated LDH (87%), elevated beta 2-microglobulin (60%), poor ECOG-PS (61%), B-symptoms (57%), ≥2 extranodal sites (71%), IPI 2 (51%) and IPI >3 (49%). The two arms were well balanced for all these features. No significant difference could be detected in the response rates of patients treated with R-CHOP and R-HDS that in detail, were the following: complete response (CR, 76% vs. 76%), partial response (PR, 4.7% vs. 7.9%), progressive disease (PD, 15% vs. 8.8%), stable disease (SD, 1.6% vs. 0.9%), treatment discontinuation for any reason (0 vs. 1.8%), early death (ED, 2.4% vs. 4.4%). On the contrary, the relapse rate was higher in the R-CHOP vs. the R-HDS arm (14% vs. 4.6%, p= 0.025). After a median follow-up of 27.7 months (range 0.3 – 52.5) the 2-year DFS of patients treated with R-CHOP or R-HDS is 83% vs. 93% (p=0.07) while the EFS is 68% vs. 73% (p= 0.345) (Figure 1 and 2). Similarly, with the current follow up, no significant difference could be detected in OS (77% vs. 80%, p= 0.398). The OS was better for patients with IPI 2 in comparison to those with IPI >3 (84% vs. 73%; p= 0.04), but without any significant difference according to treatment arm. Although the rate of death in remission (2% in both arms) was similar, the overall toxicity of the R-HDS arm was higher in terms of hematologic toxicity (CTC G>2) (p<0.001) and infectious complications (p<0.001). So far, no secondary malignancies have been observed.
In DLBCL patients with unfavorable presentation a conventional R-CHOP and a more intensive R-HDS chemotherapy achieved the same excellent rate of response. Despite a trend for a better DFS observed in patients treated with R-HDS, the EFS (primary endpoint of the study) and the OS are not different. A longer follow-up is needed in order to definitively rule out the role of intensified R-HDS program as first line treatment for poor-prognosis DLBCL patients.
Tarella:Hoffmann-La Roche: Consultancy, Honoraria. Gianni:Hoffmann-La Roche: Consultancy, Honoraria. Pizzolo:Hoffmann-La Roche: Consultancy, Honoraria. Rambaldi:Hoffmann-La Roche: Consultancy, Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract