Abstract
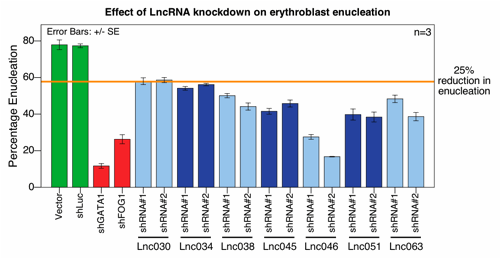
Mammalian genomes encode thousands of long noncoding (lnc) RNAs, although the extent to which lncRNAs regulate biological functions is not known. Indeed, most lncRNAs show low levels of conservation among mammals, which could suggest limited or species-restricted functions. We used RNA-seq to identify 683 and 1189 lncRNAs expressed during mouse and human erythropoiesis respectively, of which ∼25% are erythroid-specific. Primary cell chromatin immunoprecipitation (ChIP) studies indicate that most mouse erythroid lncRNAs arise from classical promoters, and their genomic loci are bound by the hematopoietic transcription factors GATA1 and SCL/TAL1. Using the G1E-ER4 cell line, which has an inducible form of GATA1, we found that lncRNAs whose genomic loci are bound by GATA1 in primary cells are upregulated on GATA1 induction in the cell line, similar to coding genes.
We compared RNA-Seq transcriptomes of erythroblasts from 8 different mice strains and showed that lncRNA expression is highly conserved within strains of the same species. A comparison of the human and mouse lncRNA transcriptomes showed that only 15% of mouse erythroblast lncRNAs have detectable syntenic transcripts in human erythroblasts, and that most lncRNAs are not detectable in humans.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.