Abstract
Hematopoietic stem and progenitor cells (HSPCs) reside in a particular and specialized microenvironmental niche. Interaction of HSPCs with their niche is fundamental for cell proliferation, differentiation and quiescence. While alterations of HSPC-niche interactions have been shown to play an important role in animal models, methods to study the human HSPC niche are lacking. In order to characterize the adhesion of various HSPC subsets to various extracellular matrix proteins found in the bone marrow, we used high-throughput fluorescent microscopy to study cell adhesion by quantifying the ratio of adherent versus non-adherent cells being detached by gravity. This allowed the analysis of low cell numbers making the study of clinical samples feasible. Multiparameter flow cytometry was used to analyze both the adherent and non-adherent fractions to study the major subsets of CD34+ cells, evaluating CD34, CD38, CD45RA, CD123 and CD90 surface levels.
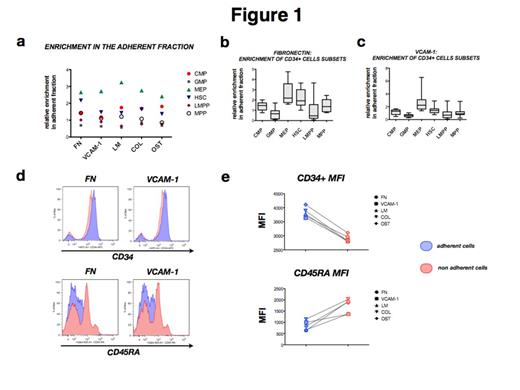
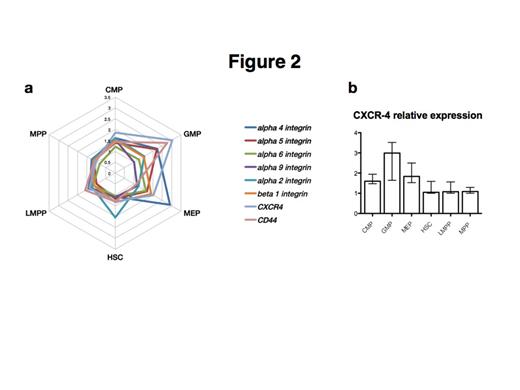
More than 80% of plated CD34+ cells adhered to fibronectin, 70% to VCAM-1, while only a minor proportion adhered to laminin, osteopontin and collagen-1 (11%-19%). Inhibitory antibodies against α4 and α5 integrin were used to inhibit adhesion of CD34+ cells to fibronectin and VCAM-1. α4 integrin antibody significantly decrease the number of adherent CD34+ cells to fibronectin from 65% to 38% (p< 0.0001) and on VCAM-1 from 51% to 12% (p< 0.0001). No effect was obtained by α5 integrin antibody. When comparing the major subsets of CD34+ cells, megakaryocyte-erythroid progenitors (MEPs) were the subpopulation that adhered strongest to each substrate followed by common myeloid progenitors (CMPs) and hematopoietic stem cells (HSCs). By contrast, granulocyte-macrophage progenitors (GMPs) adhered less to each of the studied matrix proteins (Fig. 1a-1b). Expression levels of CD34 were higher in adherent than in non-adherent cells, while CD45RA+ cells showed the opposite pattern (Fig. 1c-1d). These data suggest that the level of differentiation influences the adhesion properties and that more immature cells have a higher affinity to niche elements. We then analyzed the expression of adhesion markers in the different CD34+ cells subsets. Unexpectedly, GMPs, the least adhesive population, showed higher expression of CXCR4, a key chemokine receptor involved in HSPCs’ homing and differentiation. GMPs have been described to constitute the leukemic stem cell population in AML. GMPs also showed a high expression of CD44, a glycoprotein required for migration, cell interactions and adhesion. HSCs showed higher levels of α2 integrin while MEPs expressed more α4 integrin. The other subpopulations, common myeloid progenitors (CMPs), multipotent progenitors (MPPs) and lymphoid-primed multipotent progenitors (LMPPs) showed lower levels of all studied adhesion markers.
In conclusion, we present a method to quantify the adhesion of different human HPSC subpopulations. The assay is suitable for analysis of human myeloid neoplasms and may elucidate aberrant patterns as well as reveal potential targets for anti-adhesive therapy inhibiting the niche«s ability to sustain the leukemic stem cell population.
Analysis of adhesion preferences of CD34+ stem and progenitor cells.
MEPs show more adhesiveness to all the substrates studied, followed by CMPs and HSCs. HSCs exhibits a preference for fibronectin compared to the other proteins tested. The less adhesive seems to be GMPs (a,b,c). Difference between GMPs and MEPs adhesiveness is statistically validated (p< 0.01). CD34 expression is higher on adherent cells; on the contrary, CD45RA is more expressed on non adherent cells (d,e).
GMPs express more CXCR-4 and CD44 than the other subsets, MEPs exhibit high levels of α4 integrin, explaining why this population seems more adhesive to all the substrates. α4 and α5 integrins are important also for GMPs (a).
Expression of CXCR-4 in all the subpopulations (b).
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.