Osteoporosis is a multifactorial major health problem affecting over 200 million people worldwide. It is long known as a complication of marked iron overload, both primary (i.e. genetic hemochromatosis) and secondary (i.e. transfusional iron overload), but only recently cellular and animal models have shed some light on the pathogenetic link between iron and bone metabolism. Iron has been shown to activate osteoclasts (Ishii KA, Nat Med 2009) and to inhibit osteoblasts (Yamasaki K, Toxicol Lett 2009), which express ferroportin regulated by hepcidin (Xu Y, Inflammation 2012). A murine model has shown that iron overload causes bone loss through induction of Reactive Oxygen Species (ROS) (Tsay J, Blood 2010). Of note, a recent longitudinal study in a Korean population has demonstrated that serum ferritin, even at concentrations generally not considered as “iron overload”, is an independent predictor of bone mass deterioration and incident vertebral fractures (Kim BJ, J Bone Miner Res 2012), an effect most prominent in women ≥ 45 years of age (Kim BJ, Osteoporos Int 2013). Taking advantage from the recently completed iron section of the Val Borbera Study (Traglia M, J Med Genet 2011), this study aimed to evaluate for the first time the association between iron status (including serum hepcidin levels) and bone mass in a Caucasian population.
This survey included 921 subjects (564 females, 357 males) aged 53.8 ± 16.3 years for whom complete data regarding bone mass (measured by transportable Quantitative Ultrasonography, QUS-based approach) and iron status (including serum hepcidin-25 levels measured by Mass Spectrometry) were available. Subjects with known inflammatory and renal disorders, as well as hereditary hemochromatosis had been previously excluded. Analyses were performed separately in males and females, due to known gender-related differences in either iron or bone metabolism.
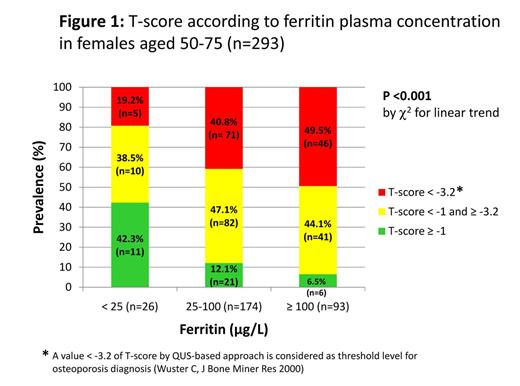
No significant association was found in males, while in females both ferritin (r= -0.42, P<0.001) and hepcidin (r= -0.30, P<0.001) were inversely correlated with T-score at univariate analyses. However, after including both ferritin and hepcidin in an age-adjusted linear regression model, only ferritin remained a significant predictor of T-score variability (beta coefficient= -0.115, P=0.042). Subsequent regression models adjusted for age, BMI, and C-Reactive Protein highlighted ferritin levels as independent predictors of T-score in females. After stratification for age and ferritin categories, T-score decreased linearly with increasing ferritin levels especially in females aged 50-75 years (n=293), i.e. the age known to be at major risk of accelerated bone loss (P<0.001 – Figure 1).
This study confirms that iron status significantly associates with bone loss at population level even in Caucasians, particularly in post-menopausal women. Increasing iron stores, even not clearly “pathologic”, may influence bone metabolism through increased ROS and/or hepcidin-mediated altered iron handling of osteoblasts and osteoclasts.
No relevant conflicts of interest to declare.