Abstract
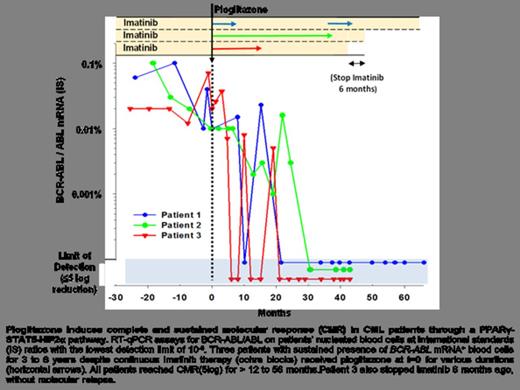
Whether cancer is maintained by a small number of stem cells or is composed of proliferating cells with approximate phenotypic equivalency is a central question in cancer biology. In the stem cell hypothesis, relapse after treatment may occur by failure to eradicate cancer stem cells. The chronic myeloid leukemia (CML) is quintessential to this hypothesis. CML is a myeloproliferative disorder that results from dysregulated tyrosine kinase activity of the fusion oncoprotein BCR-ABL. During the chronic phase, this sole genetic abnormality (chromosomal translocation Ph1+: t(9;22)(q34;q11)) at the stem cell level causes increased proliferation of myeloid cells without loss of their capacity to differentiate. Without treatment, most patients progress to the blast phase when additional oncogenic mutations result in a fatal acute leukemia made of proliferating immature cells. Imatinib mesylate and other tyrosine kinase inhibitors (TKIs) that target the kinase activity of BCR-ABL have improved patient survival dramatically. However, fewer than 10% of patients reach the stage of complete molecular response (CMR), defined when BCR-ABL transcripts become undetectable in blood cells3. Failure to reach CMR results from the inability of TKIs to eradicate quiescent CML leukemia stem cells (LCSs). Here we show that the residual CML LSC pool can be gradually purged by the glitazones, antidiabetic drugs that are agonists of peroxisome proliferator-activated receptor gamma (PPARg). We found that activation of PPARg by the glitazones decreases expression of STAT5 and its downstream targets HIF2α and CITED2, which are key guardians of CML LSC's quiescence and stemness. When pioglitazone was temporarily given to three CML patients in chronic residual disease in spite of continuous treatment with imatinib, all of them achieved sustained CMR, up to 4.7 years after withdrawal of pioglitazone. This suggests that clinically relevant cancer eradication may become a generally attainable goal by combination therapy that erodes the cancer stem cell pool.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.