Key Points
LIN28B regulates HbF expression in erythroblasts that are cultured from umbilical cord and adult human blood.
LIN28B expression manifested a more fetal-like phenotype among adult human erythroblasts.
Abstract
Reactivation of fetal hemoglobin (HbF) holds therapeutic potential for sickle cell disease and β-thalassemias. In human erythroid cells and hematopoietic organs, LIN28B and its targeted let-7 microRNA family, demonstrate regulated expression during the fetal-to-adult developmental transition. To explore the effects of LIN28B in human erythroid cell development, lentiviral transduction was used to knockdown LIN28B expression in erythroblasts cultured from human umbilical cord CD34+ cells. The subsequent reduction in LIN28B expression caused increased expression of let-7 and significantly reduced HbF expression. Conversely, LIN28B overexpression in cultured adult erythroblasts reduced the expression of let-7 and significantly increased HbF expression. Cellular maturation was maintained including enucleation. LIN28B expression in adult erythroblasts increased the expression of γ-globin, and the HbF content of the cells rose to levels >30% of their hemoglobin. Expression of carbonic anhydrase I, glucosaminyl (N-acetyl) transferase 2, and miR-96 (three additional genes marking the transition from fetal-to-adult erythropoiesis) were reduced by LIN28B expression. The transcription factor BCL11A, a well-characterized repressor of γ-globin expression, was significantly down-regulated. Independent of LIN28B, experimental suppression of let-7 also reduced BCL11A expression and significantly increased HbF expression. LIN28B expression regulates HbF levels and causes adult human erythroblasts to differentiate with a more fetal-like phenotype.
Introduction
In humans and some other mammals, the composition of hemoglobin tetramers in erythrocytes switch from fetal hemoglobin (HbF) (α2γ2) to adult hemoglobin (HbA) (α2β2) during the last stages of fetal development until early infancy.1 HbF is the most important known modifier of the clinical symptoms for patients with sickle cell disease (SCD) and β-thalassemias, which are among the most common genetic disorders worldwide.2,3 In patients with SCD, the polymerization of sickle hemoglobin results in erythrocyte deformation and hemolysis.4 SCD patient’s clinical outcomes are largely improved by inhibition of the polymerization by HbF.5 In β-thalassemias, decreased production of β-globin causes imbalanced globin polypeptide chain synthesis, and leads to severe effects on the erythroid cells’ maturation and survival. The loss of β-globin expression may be compensated by an increase in HbF production that leads to improvement of the clinical phenotype.6
The molecular mechanisms underlying the switch from HbF to HbA are still largely unknown. Genome-wide association studies (GWAS) in both normal individuals and patients with β-hemoglobinopathies have identified BCL11A, HSB1L-MYB, and HBB clusters as having an association with the persistence of HbF in adults.7-10 Suppression of the BCL11A transcription factor causes an increase in HbF levels.11
Lin28 is a highly conserved gene that is expressed in the early stages of development of multicellular organisms. In the nematode, Caenorhabditis elegans, lin28 regulates developmental timing at the larvae L2 stage.12 Mammals express 2 homologs of the C. elegans lin28 gene known as LIN28A and LIN28B.13,14 Human LIN28 genetic variation correlates with developmental timing characteristics, such as variation in height,15 timing of puberty,16-18 and age at natural menopause.16 The effects of LIN28 protein depend, in part, on the cell type in which it is expressed, as evidenced by its role in promoting the pluripotency of stem cells,19 as well as the differentiation of skeletal muscle cells.20
In part, LIN28 acts through the 3′ end uridylation of the precursor molecule pre-let-7, ultimately leading to the degradation of microRNA (miRNA) molecules.21-23 Interestingly, expression of the let-7 family of miRNAs was correlated with the fetal-to-adult developmental transition in circulating human reticulocytes.24 Those data suggest a potential link between LIN28B expression, the regulation of the let-7 miRNAs, and vertebrate hematopoiesis. In addition to binding to the terminal loop of let-7 miRNAs, LIN28 binds to GGAGA motifs in messenger RNAs (mRNAs).25 Moreover, a predicted fetal stage-specific enhancer for LIN28B was identified using human primary erythroid cells,26 and in murine lymphocytes, Lin28b expression regulates the fetal-like phenotype.27 Here, we investigated LIN28B as a candidate regulator of the fetal-to-adult transition of hemoglobin expression and the erythroblast phenotype using an experimental approach developed for ex vivo engineering of human erythrocytes.
Methods
Cell culture
All related studies were performed after human subjects review and National Institutes of Health Institutional Review Board approval. These studies were conducted in accordance with the Declaration of Helsinki. Human cord blood CD34+ cells were obtained from All Cells (Emeryville, CA) and ReachBio LLC (Seattle, WA). For the ex vivo culture, a 21 day serum-free system consisting of 3 phases was used. During phase I of the culture (day 0 to day 7): adult or cord blood CD34+ cells were placed in medium containing StemPro-34 complete medium (l-glutamine, pen-strep, and StemPro-34 nutrient supplement) (Invitrogen, Carlsbad, CA) with 50 ng/mL SCF (HumanZyme, Chicago, IL), 50 ng/mL FLT3 Ligand (HumanZyme) and 10 ng/mL IL-3 (HumanZyme). After phase I, the cells were transferred to phase II (day 7 to day 14). During this expansion phase, the cell count was monitored on days 9, 11, and 14 to maintain cell counts less than 2 × 106 cells/mL. Phase II medium was comprised of the following: StemPro-34 complete medium, 4 U/mL erythropoietin (EPO) (Amgen, Thousand Oaks, CA), 10 ng/mL SCF, 10 µg/mL insulin (Sigma-Aldrich, St. Louis, MO), 3 U/mL heparin (Hospira, Lake Forest, IL), and 0.8 mg/mL holo Transferrin (Sigma-Aldrich). On culture day 14, cells were counted and transferred to phase III medium at 8 × 105 cells/mL for the remaining 7 days of culture. Phase III medium was comprised of the following: StemPro-34 complete medium, 4 U/mL EPO, 3 µM RU486 (Sigma-Aldrich), 10 µg/mL insulin, 3 U/mL heparin, and 0.8 mg/mL holo Transferrin.
RNA IP
RNA immunoprecipitation (IP) was performed according to the manufacturer’s instructions (Magna RIP RNA-Binding Protein Immunoprecipitation Kit, Millipore, Billerica, MA) with equal amounts of lysates from human cord blood cells using antibodies against LIN28B (catalog #4196; Cell Signaling, Danvers, MA) or IgG (catalog #PP64; Millipore). The immunoprecipitated RNA was purified before reverse transcription-polymerase chain reaction with primers to primary let-7d miRNA (Assays-on-Demand Gene Expression Product #Hs03302562_pri; Applied Biosystems, Grand Island, NY).
Recombinant viral transduction
Human LIN28B was subcloned into an MSCV-IRES-Puro-based vector, as previously described.27 Human LIN28A overexpression lentiviral particles were purchased from Qiagen (Valencia, CA) and used for comparison studies. Human LIN28B knockdown clones were purchased from Sigma Aldrich. BCL11A knockdown clones were selected as previously described.11 A miRNA sponge experimental approach was used for suppression of let-7.28 The let-7 sponge plasmid (MSCV puro let-7 sponge) was purchased from Addgene (Cambridge, MA; plasmid, catalog #29766). Tissue culture dishes were coated with 20 μg/mL poly-l-lysine (Sigma-Aldrich) for 1 hour at 37°C. One day before transfection, the 293T cells were seeded at 4 × 106 cells per dish. Retroviral supernatants were produced by transient Lipofectamine 2000 (Invitrogen) cotransfection of 293T cells with the RetroMax packaging vector pCL-Eco (Imgenex, San Diego, CA) and the let-7 sponge plasmid vector (20 μg each) following the manufacturer's instructions. Seventy-two hours post-transfection, cell supernatant was harvested and concentrated with Retro-X concentrator solution (Clontech, Mountain View, CA) according to the manufacturer’s instructions. Empty vectors were used as controls. On culture day 3 of phase I, the cells were transduced with either LIN28, BCL11A, or let-7 sponge viral particles (estimated multiplicity of infection: 12). After 24 hours, Puromycin (Sigma-Aldrich) was added to the culture. On culture day 7, cells were transferred to phase II medium containing EPO and Puromycin until culture day 9. After culture day 9, cells were cultivated at the conditions previously described without Puromycin.
Flow cytometry analyses
Immunostaining with antibodies directed against CD71 and glycophorin A (Invitrogen) were performed to assay erythroid differentiation on culture day 21, as previously described,29 using the BD FACSAria I flow cytometer (BD Biosciences, San Jose, CA). Enucleation was quantitated using thiazole orange staining. Enucleated erythroid cell populations were isolated by filtering day 21 cells through Purecell Neo Leukocyte Reduction filters (Pall, Covina, CA).
Confocal microscopy
For confocal microscopy, slides containing culture day 14 cells were probed against human LIN28B (Cell Signaling). Laser scanned confocal images were obtained from LSM 5 Live Duoscan (Carl Zeiss, Oberkochen, Germany) and analyzed with Zen2007 software.
HPLCs for HbA and HbF
Samples for hemoglobin profiles (HPLCs) were prepared and analyzed as previously described.29
Quantitative polymerase chain reaction analysis
Quantitative reverse-transcription polymerase chain reaction (qRT-PCR) assays and polymerase chain reaction conditions were performed as previously described.30 Additional primers and probes used in this study are described in supplemental Table 1. To evaluate miRNA expression, reactions, and data normalization were performed as previously described.24
Western blot analysis
Nuclear and cytoplasmic extracts were prepared from culture day 14 erythroblasts using NE-PER Nuclear Protein Extraction Kit (Pierce Biotechnology, Rockford, IL) according to the manufacturer’s protocol. Equal amounts of protein (20 to 30 µg) were separated by NuPAGE Novex 4% to 12% Bis-Tris gel electrophoresis in 4-morpholinepropanesulfonic acid buffer and transferred using the iBlot Blotting System onto nitrocellulose membranes (Invitrogen). Blots were probed with antibodies against human CA1 (Abcam, Cambridge, MA), glucosaminyl (N-acetyl) transferase 2 (GCNT2) (Santa Cruz Biotechnology, Dallas, TX), BCL11A (Abcam), and LIN28B (Cell Signaling), and the appropriate horseradish peroxidase-conjugated secondary antibody (Santa Cruz Biotechnology). Immunoreactive proteins were detected and visualized using ECL Plus Western blotting detection reagents (GE Healthcare, Waukesha, WI). All blots were probed with β-actin (Abcam) antibody as a loading control.
Statistical analysis
Replicate data are expressed as mean value ± standard deviation with significance calculated by two-tailed Student t test.
Results
LIN28B binds primary let-7 transcripts and regulates HbF in human cord blood CD34+ cells
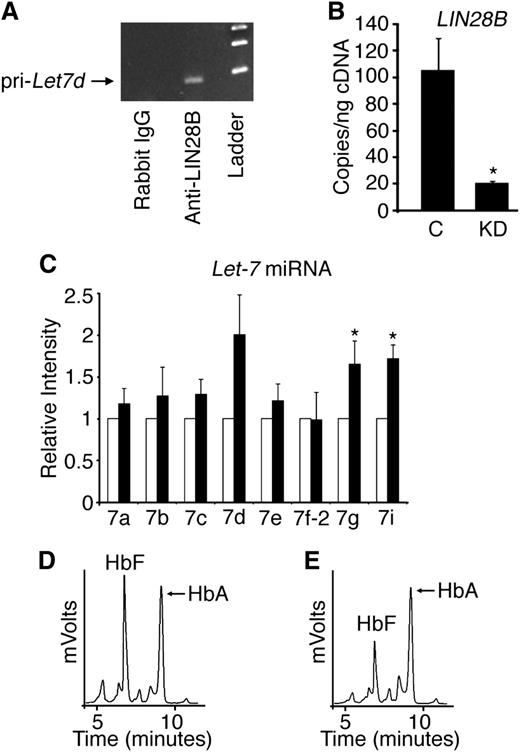
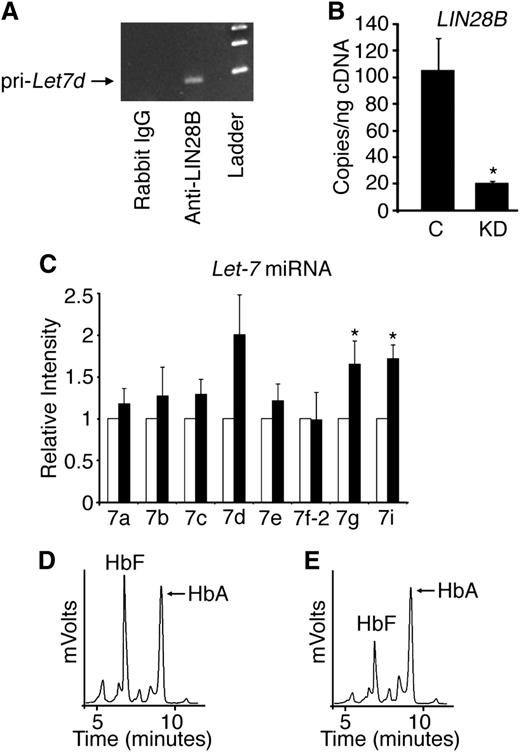
We previously reported that decreased HbF expression in adult human reticulocytes compared with those present in cord blood is associated with increased let-7 levels in adult reticulocytes.24 Because let-7 miRNA expression is downregulated by the binding of LIN28 proteins, the absence of LIN28B expression in adult human bone marrow compared with its detection in fetal liver27 suggests that both LIN28B and let-7 are developmentally regulated in erythroid tissues. Initial studies were performed to determine if LIN28B binds endogenous let-7 primary transcripts in cord blood-derived erythroblasts. RNA IP in CD34+ cord blood lysates was performed and quantitated by reverse transcription-polymerase chain reaction for primary let-7d. The let-7d was chosen based on its expression level in cord blood cells.24 Primary let-7d was enriched by LIN28B IP compared with the IgG control (Figure 1A).
LIN28B regulates HbF levels in cultured human cord blood erythroblasts. (A) Detection of primary let-7d miRNA in erythroblasts cultured from human CD34+ cord blood cells after RNA IP with antibodies against LIN28B or control (rabbit IgG) with DNA ladder in the right lane shown for comparison. (B) LIN28B knockdown (LIN28B-KD) confirmation by qRT-PCR quantitation of copy number per nanogram complementary DNA (cDNA) (copies/ng cDNA). LIN28B-KD and control samples were evaluated. (C) The relative expression levels of the let-7 family of miRNAs were determined by qRT-PCR. Open bars represent control samples and black bars represent LIN28B-KD. Standard deviation bars are shown. *P < .05 in triplicate experiments. Hemoglobin profiles demonstrated by HPLC analysis are shown for (D) control and (E) LIN28B-KD cultures. HbF and HbA peaks are labeled on each graph (y-axis: mVolts; x-axis: elution time in minutes). C, empty vector control; KD, LIN28B knockdown; Pri-Let7d, primary let-7d miRNA.
LIN28B regulates HbF levels in cultured human cord blood erythroblasts. (A) Detection of primary let-7d miRNA in erythroblasts cultured from human CD34+ cord blood cells after RNA IP with antibodies against LIN28B or control (rabbit IgG) with DNA ladder in the right lane shown for comparison. (B) LIN28B knockdown (LIN28B-KD) confirmation by qRT-PCR quantitation of copy number per nanogram complementary DNA (cDNA) (copies/ng cDNA). LIN28B-KD and control samples were evaluated. (C) The relative expression levels of the let-7 family of miRNAs were determined by qRT-PCR. Open bars represent control samples and black bars represent LIN28B-KD. Standard deviation bars are shown. *P < .05 in triplicate experiments. Hemoglobin profiles demonstrated by HPLC analysis are shown for (D) control and (E) LIN28B-KD cultures. HbF and HbA peaks are labeled on each graph (y-axis: mVolts; x-axis: elution time in minutes). C, empty vector control; KD, LIN28B knockdown; Pri-Let7d, primary let-7d miRNA.
To investigate a potential role for cord blood LIN28B expression in HbF regulation, CD34+ cord blood cells were transduced with lentivirus encoding a short hairpin RNA (shRNA) against LIN28B and cultured ex vivo in erythropoietin-supplemented serum-free media for 21 days. Two shRNA clones (TRCN0000122191 and TRCN0000122599) demonstrated equivalent results; the results using clone TRCN0000122191 are shown in Figure 1. LIN28B knockdown (LIN28B-KD) was confirmed by qRT-PCR (control: 105 ± 24 copies/ng; LIN28B-KD: 20.7 ± 0.9 copies/ng; P = .02), as demonstrated in Figure 1B. Let-7 miRNAs were measured by qRT-PCR in LIN28B-KD cells. As shown in Figure 1C, LIN28B-KD caused increased expression of several let-7 species (let-7a, let-7b, let-7c, let-7d, and let-7e), and the increased levels of let-7g and let-7i reached statistical significance compared with the controls (P = .05 and P = .01, respectively). Hemoglobin expression profiles for control and LIN28B-KD were determined by standard HPLCs (Figures 1D-E). Triplicate experiments demonstrated a significant reduction in HbF expression in LIN28B-KD cells compared with the controls (LIN28B-KD: 31.0 ± 5.3%; control: 54.2 ± 7.7%; P = .03).
LIN28B expression activates HbF during adult human erythropoiesis
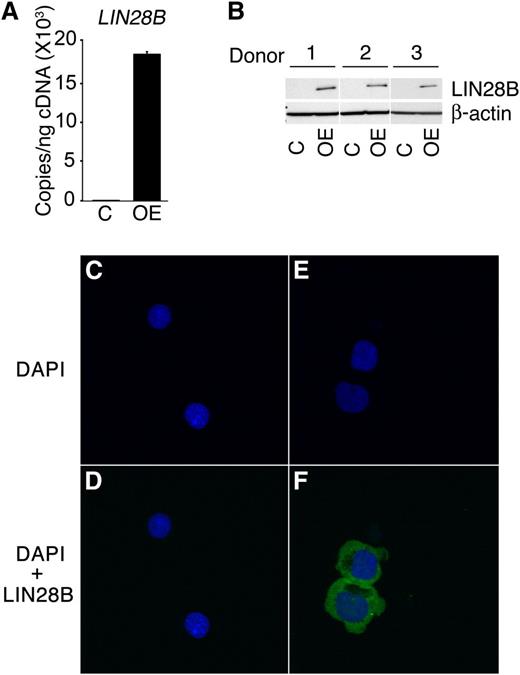
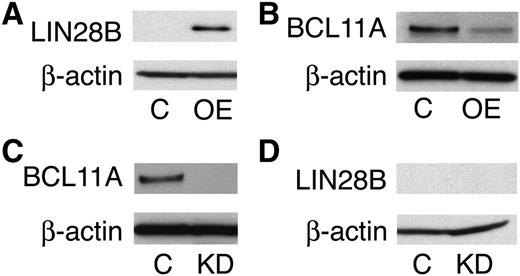
In addition to studies of cord blood-derived erythroblasts, LIN28B expression in adult erythroblasts was examined. When compared with the cord blood erythroblasts, adult erythroblasts demonstrated a major reduction in LIN28B expression (0.1 ± 0.4 copies/ng). To increase LIN28B expression, the cells were transduced with a retrovirus encoding LIN28B. LIN28B overexpression (LIN28B-OE) was confirmed by qRT-PCR (LIN28B-OE: 1.8E+04 ± 3.5E+02 copies/ng; P = .01) and Western blot analyses (Figure 2A-B). In transduced erythroblasts, LIN28B was expressed predominantly in the cytoplasm without distinct nuclear localization as shown by confocal microscopy (Figure 2C-F).
LIN28B overexpression in adult human erythroblasts.LIN28B-OE was confirmed by (A) qRT-PCR quantitation of copy number per nanogram complementary DNA (cDNA) (copies/ng cDNA); (B) Western blot analysis and (C-F) confocal images of control and LIN28B-OE cells were stained with DAPI (4′,6-diamidino-2-phenylindole) (blue) and LIN28B (green). Cells were transfected with empty vector (C-D), and LIN28B (E-F). Analyses were performed at culture day 14. Mean value ± standard deviation of 3 independent donors for each condition. C, empty vector control; OE, LIN28B overexpression.
LIN28B overexpression in adult human erythroblasts.LIN28B-OE was confirmed by (A) qRT-PCR quantitation of copy number per nanogram complementary DNA (cDNA) (copies/ng cDNA); (B) Western blot analysis and (C-F) confocal images of control and LIN28B-OE cells were stained with DAPI (4′,6-diamidino-2-phenylindole) (blue) and LIN28B (green). Cells were transfected with empty vector (C-D), and LIN28B (E-F). Analyses were performed at culture day 14. Mean value ± standard deviation of 3 independent donors for each condition. C, empty vector control; OE, LIN28B overexpression.
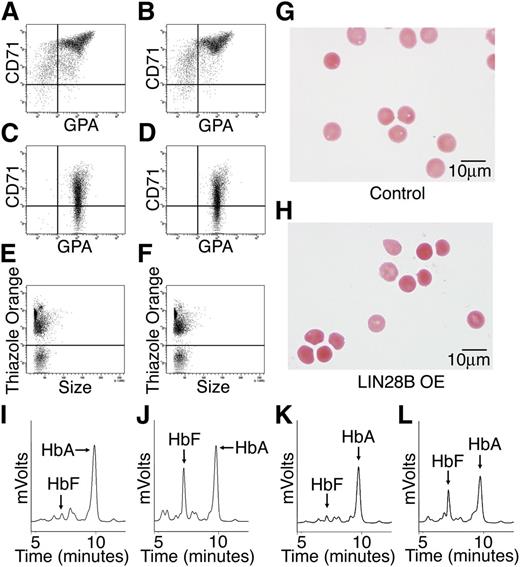
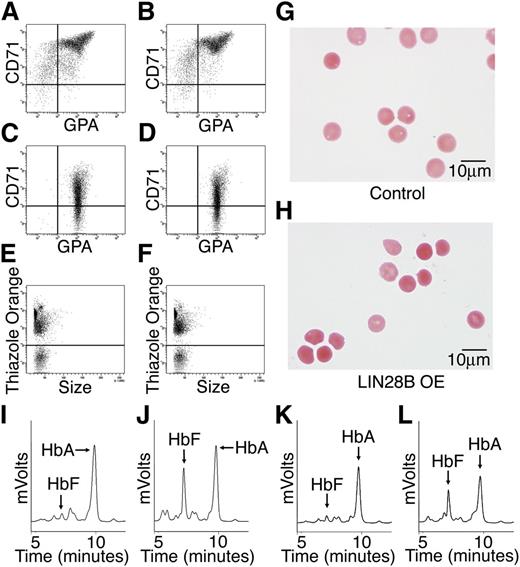
Erythroblast differentiation and the patterns of hemoglobin expression were compared between control and LIN28B-OE cells. Flow cytometric analysis of erythroblast plasma membrane markers, transferrin receptor (CD71), and glycophorin A were performed at culture days 14 and 21 for erythroblast differentiation (Figure 3A-D). Surprisingly, LIN28B-OE did not overtly affect erythroid cell maturation when compared with empty vector controls. Furthermore, thiazole orange staining of nuclei on culture day 21 in LIN28B-OE and control cells showed comparable results (Figure 3E-F) (LIN28B-OE enucleation: 38 ± 2.1% compared with control: 25 ± 9.3%), thus indicating the ability of LIN28B-OE erythroblasts to undergo terminal maturation. The culture-generated erythrocytes were purified and further studied. The LIN28B-OE erythrocytes were morphologically equivalent to controls for all analyzed samples (Figure 3G-H). HPLCs showed markedly increased HbF levels in the LIN28B-OE cells (Figure 3I-J) (LIN28B-OE: 33.6 ± 9.4%; control: 5.8 ± 4.5%; P = .01). For comparison, a different lentiviral vector encoding the other human LIN28 gene (LIN28A) was used. A robust increase in HbF expression was also detected in LIN28A transduced cells (Figure 3K-L). Taken together, these data demonstrate that overexpression of LIN28A or LIN28B is sufficient for the manifestation of high-level HbF expression in cultured adult human erythroblasts.
LIN28B overexpression enhances HbF levels in adult human erythroblasts. Flow cytometry analyses of (A) control and (B) LIN28B-OE at culture day 14, and (C) control, and (D) LIN28B-OE at culture day 21 stained with anti-transferrin receptor (CD71) and anti-glycophorin A (GPA) antibodies. Enucleation is represented by staining of culture day 21 cells with thiazole orange for (E) control and (F) LIN28B-OE. The enucleated cells were imaged in panels (G) control and (H) LIN28B-OE. HPLC analysis of hemoglobin from (I) control and (J) LIN28B-OE, and (K) control and (L) LIN28A-OE samples performed at culture day 21 shown for comparison with LIN28B-OE. HbF and HbA peaks are labeled on each graph (y-axis: mVolts; x-axis: elution time in minutes). Data are representative of more than 3 independent experiments. LIN28B-OE, LIN28B overexpression; LIN28A-OE, LIN28A overexpression.
LIN28B overexpression enhances HbF levels in adult human erythroblasts. Flow cytometry analyses of (A) control and (B) LIN28B-OE at culture day 14, and (C) control, and (D) LIN28B-OE at culture day 21 stained with anti-transferrin receptor (CD71) and anti-glycophorin A (GPA) antibodies. Enucleation is represented by staining of culture day 21 cells with thiazole orange for (E) control and (F) LIN28B-OE. The enucleated cells were imaged in panels (G) control and (H) LIN28B-OE. HPLC analysis of hemoglobin from (I) control and (J) LIN28B-OE, and (K) control and (L) LIN28A-OE samples performed at culture day 21 shown for comparison with LIN28B-OE. HbF and HbA peaks are labeled on each graph (y-axis: mVolts; x-axis: elution time in minutes). Data are representative of more than 3 independent experiments. LIN28B-OE, LIN28B overexpression; LIN28A-OE, LIN28A overexpression.
Adult erythroid cells expressing LIN28B manifest a more fetal-like phenotype
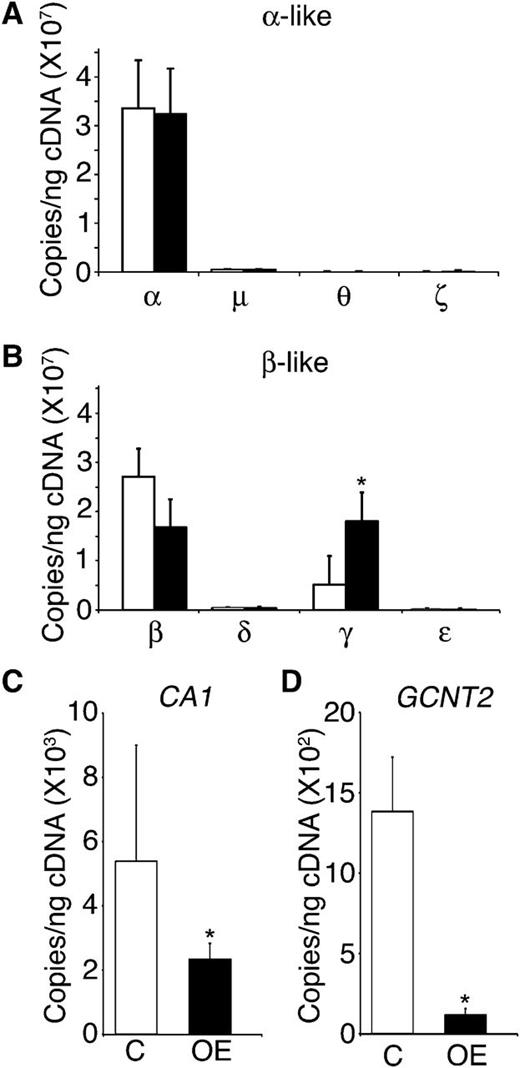
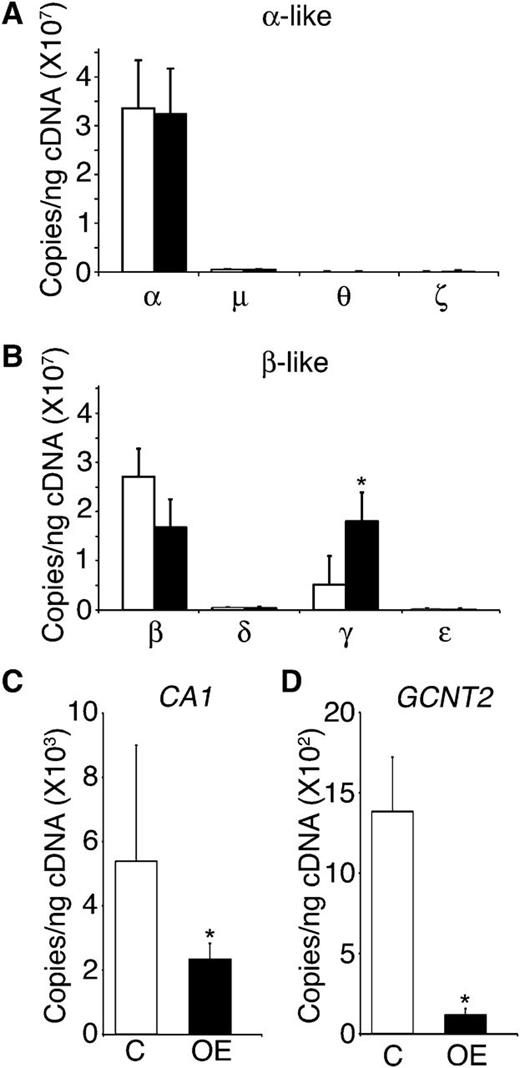
The fetal-to-adult hemoglobin transition in erythrocytes encompasses a reduction in the expression of the fetal (Aγ/Gγ-globin) genes among erythroblasts, coinciding with an increased expression of the adult (β- and δ-globin) genes. This process begins during the later stages of fetal development and continues through early infancy. Based on the high level of HbF expression in LIN28B-OE cells, globin gene expression patterns were quantitated by qRT-PCR. The expression levels of α, μ, θ, ζ, β, δ, γ, and ε globin genes were evaluated for LIN28B-OE and compared with controls. As shown in Figure 4A-B, only the fetal γ-globin mRNA expression is significantly heightened with LIN28B-OE (control: 5.1E+06 ± 2.6E+06 copies/ng; LIN28B-OE: 1.8E+07 ± 5.8E+06 copies/ng; P = .04). The increased expression of γ-globin was balanced by reductions in β-globin and δ-globin. No changes were observed in α, μ, and θ globins, and the low level expression patterns of ζ and ε globins indicated only minor increases.
LIN28B expression regulates γ-globin and erythroid-related fetal genes in adult erythroblasts. (A) α, μ, θ, and ζ globins, (B) β, δ, γ, and ε globins, (C) CA1, and (D) GCNT2 mRNA expression analysis of LIN28B-OE compared with control samples. Open bars represent control and black bars represent LIN28B-OE. qRT-PCR analyses were performed at culture day 14. Mean value ± standard deviation of 3 independent donors for each condition. P values were calculated using two-tailed Student t test. C, empty vector control; OE, LIN28B overexpression. *P < .05.
LIN28B expression regulates γ-globin and erythroid-related fetal genes in adult erythroblasts. (A) α, μ, θ, and ζ globins, (B) β, δ, γ, and ε globins, (C) CA1, and (D) GCNT2 mRNA expression analysis of LIN28B-OE compared with control samples. Open bars represent control and black bars represent LIN28B-OE. qRT-PCR analyses were performed at culture day 14. Mean value ± standard deviation of 3 independent donors for each condition. P values were calculated using two-tailed Student t test. C, empty vector control; OE, LIN28B overexpression. *P < .05.
The fetal-to-adult transition in humans is further characterized by an increase in the carbonic anhydrase I (CA1) gene expressed in erythrocytes,31 as well as the carbohydrate modification due to the augmented expression of GCNT2.32-34 Therefore, expression of CA1 and GCNT2 were investigated by qRT-PCR with LIN28B-OE. As shown in Figure 4C-D, both CA1 and GCNT2 were significantly reduced with LIN28B-OE (CA1, control: 5E+03 ± 4E+03 copies/ng; LIN28B-OE: 2E+03 ± 5E+02 copies/ng; P = .01; GCNT2, control: 1E+03 ± 3E+02 copies/ng; LIN28B-OE: 1E+02 ± 4E+01 copies/ng; P = .01). Western blot analysis using anti-human CA1 antibody in LIN28B-OE was compared with control samples at culture day 21 confirming a reduction in CA1 levels; however, a major change in GCNT2 protein expression was not detected (supplementary Figure 1A-B).
LIN28B suppresses let-7 and miR-96 miRNAs
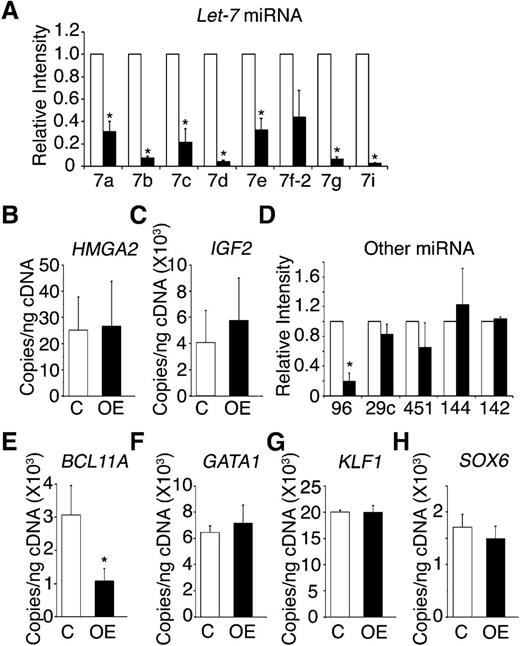
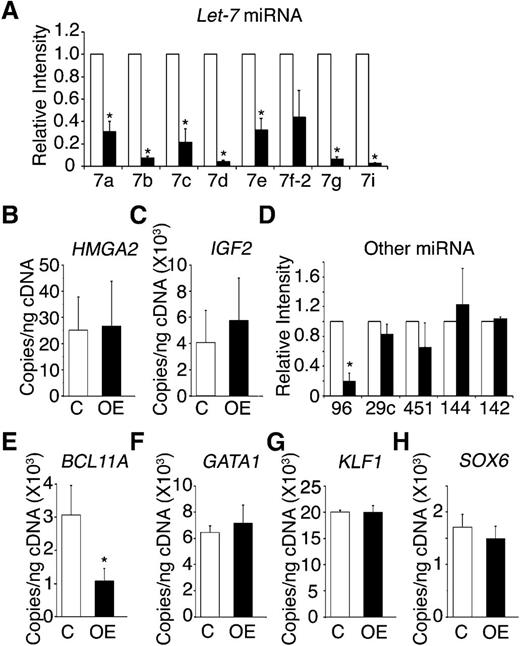
One characteristic effect of LIN28 proteins is the suppressed maturation of the let-7 family of miRNAs.21,35 Therefore, the expression patterns for several let-7 family members (let-7a, let-7b, let-7c, let-7d, let-7e, let-7f-2, let-7g, and let-7i) were investigated in LIN28B-OE erythroblasts. The let-7 family members demonstrated robust and consistent suppression with a greater than 70% reduction in LIN28B-OE compared with the control (Figure 5A). The chromatin modifier HMGA2 is a validated target of let-7, which also modulates growth and differentiation.36-38 Retroviral integration of globin encoding vectors into the HMGA2 locus resulted in hematopoietic clonal dominance at the integration site, as well as increased HbF expression.39 Lin28 protein also increases the efficiency of insulin-like growth factor 2 (IGF2) protein translation in muscle cells.20 IGF2 is expressed at high levels in fetal hepatocytes and supports expansion of hematopoietic stem cells in the fetal liver.40 Despite the strong suppression of the let-7 family, HMGA2 was not significantly up-regulated in LIN28B-OE samples (Figure 5B). IGF2 expression was slightly increased in the LIN28B-OE samples (Figure 5C).
LIN28B modulation of adult CD34+ cells toward a fetal-like phenotype involves the let-7 family of miRNAs, miR-96, and BCL11A.LIN28B-OE compared with control samples in (A) the relative expression levels of the let-7 family of miRNAs (open bars represent control and black bars represent LIN28B-OE), the mRNA expression levels of (B) HMGA2 and (C) IGF2, (D) the relative expression levels of miR-96, miR-29c, miR-451, miR-144, and miR-142, and the mRNA expression of the transcription factors (E) BCL11A, (F) GATA1, (G) KLF1, and (H) SOX6. The qRT-PCR analyses were performed at culture day 14. The miRNAs relative expression levels (y-axis) in the control cells were defined as a level of one for comparison. Error bars denote ± standard deviation of 3 independent donors for each condition. P values were calculated using two-tailed Student t test. C, empty vector control; OE, LIN28B overexpression. *P < .05.
LIN28B modulation of adult CD34+ cells toward a fetal-like phenotype involves the let-7 family of miRNAs, miR-96, and BCL11A.LIN28B-OE compared with control samples in (A) the relative expression levels of the let-7 family of miRNAs (open bars represent control and black bars represent LIN28B-OE), the mRNA expression levels of (B) HMGA2 and (C) IGF2, (D) the relative expression levels of miR-96, miR-29c, miR-451, miR-144, and miR-142, and the mRNA expression of the transcription factors (E) BCL11A, (F) GATA1, (G) KLF1, and (H) SOX6. The qRT-PCR analyses were performed at culture day 14. The miRNAs relative expression levels (y-axis) in the control cells were defined as a level of one for comparison. Error bars denote ± standard deviation of 3 independent donors for each condition. P values were calculated using two-tailed Student t test. C, empty vector control; OE, LIN28B overexpression. *P < .05.
In addition to the let-7 family of miRNAs, miR-96 and miR-29c are preferentially expressed in adult human peripheral blood reticulocytes when compared with cord blood samples.24 Recently, miR-96 expression in reticulocytes was demonstrated to increase during human ontogeny,24 and miR-96 knockdown resulted in increased γ-globin expression in human erythroblasts.41 However, the miR-96 effects on HbF expression in mature erythroid cells were not defined. Three erythroid miRNAs that are not developmentally regulated (miR-451, miR-144, and miR-142) were tested as controls. Among the group, a significant reduction was observed only in miR-96 (Figure 5D), suggesting possible targeting of this miRNA by LIN28B.
LIN28B regulates BCL11A expression
Additional genetic mechanisms underlying γ-globin expression and HbF production were sought, including the potential for LIN28B-OE to regulate erythroid transcription factors. BCL11A is an important transcriptional silencer of HbF in model systems, as well as in humans.11,26,42 KLF1 knockdown cells were reported to downregulate BCL11A and activate γ-globin.43 In addition, SOX6 expression has been detected during erythroid development and was implicated in playing a cooperative role with BCL11A in the regulation of γ-globin.26 The expression patterns of the transcription factors BCL11A, KLF1, and SOX6 were investigated, along with the erythroid transcription regulator, GATA1 (Figure 5E-H). Among the group, only BCL11A was affected by LIN28B-OE, and LIN28B-OE caused a 65% reduction in BCL11A expression (control: 3.07E+03 ± 1.5 E+02 copies/ng; LIN28B-OE: 1.07E+03 ± 18 copies/ng; P = .02) (Figure 5E).
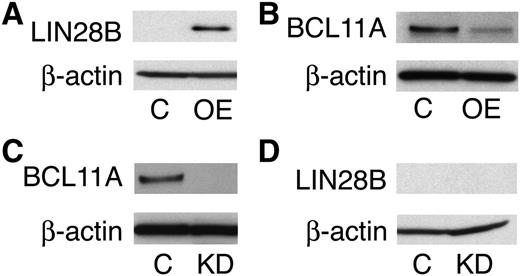
Next, we investigated whether LIN28B and BCL11A regulate each other. In accordance with our mRNA expression data, LIN28B-OE downregulated BCL11A at the protein level, as shown in Figure 6A-B. To investigate whether BCL11A modulates LIN28B expression, primary CD34+ cells were transduced with the shRNA lentivirus knockdown vector of BCL11A and lentiviral controls. BCL11A knockdown (Figure 6C) was evaluated and the results showed no effect on LIN28B expression (Figure 6D).
LIN28B is an upstream regulator of BCL11A. Western blot analyses with protein extracts from empty vector control (C) versus LIN28B-OE (OE) cells (A-B), or BCL11A knockdown (KD) cells (C-D). The blots were probed with anti-LIN28B, and anti-BCL11A as labeled at the left of each band pair. An anti-β-actin probe was used as a loading control.
LIN28B is an upstream regulator of BCL11A. Western blot analyses with protein extracts from empty vector control (C) versus LIN28B-OE (OE) cells (A-B), or BCL11A knockdown (KD) cells (C-D). The blots were probed with anti-LIN28B, and anti-BCL11A as labeled at the left of each band pair. An anti-β-actin probe was used as a loading control.
Let-7 miRNAs regulate BCL11A expression and HbF
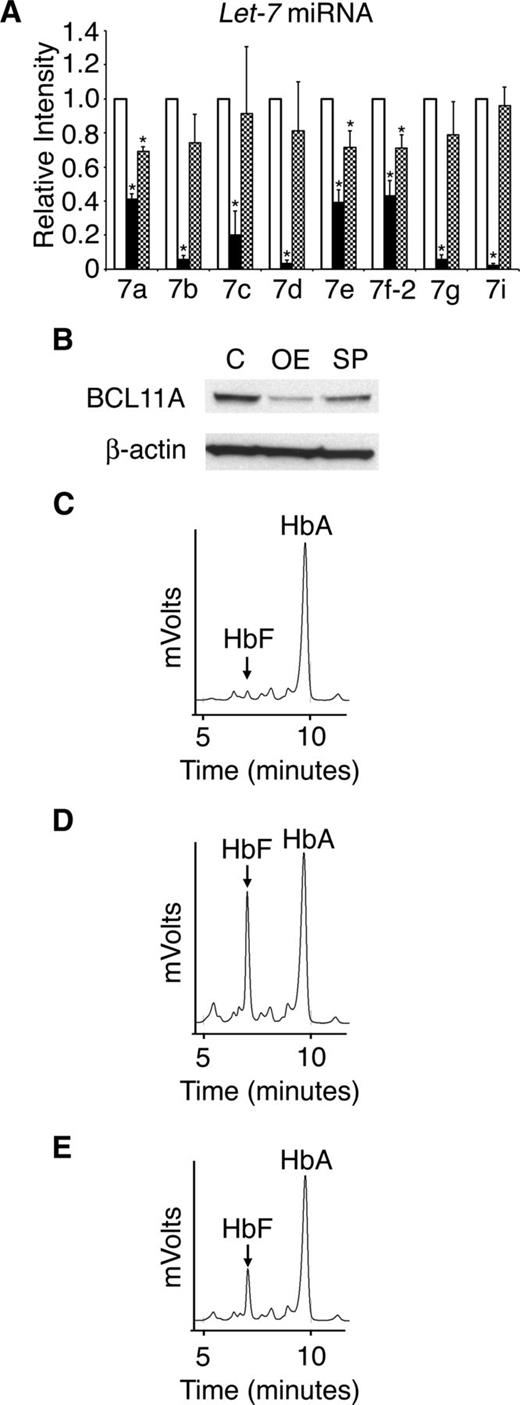
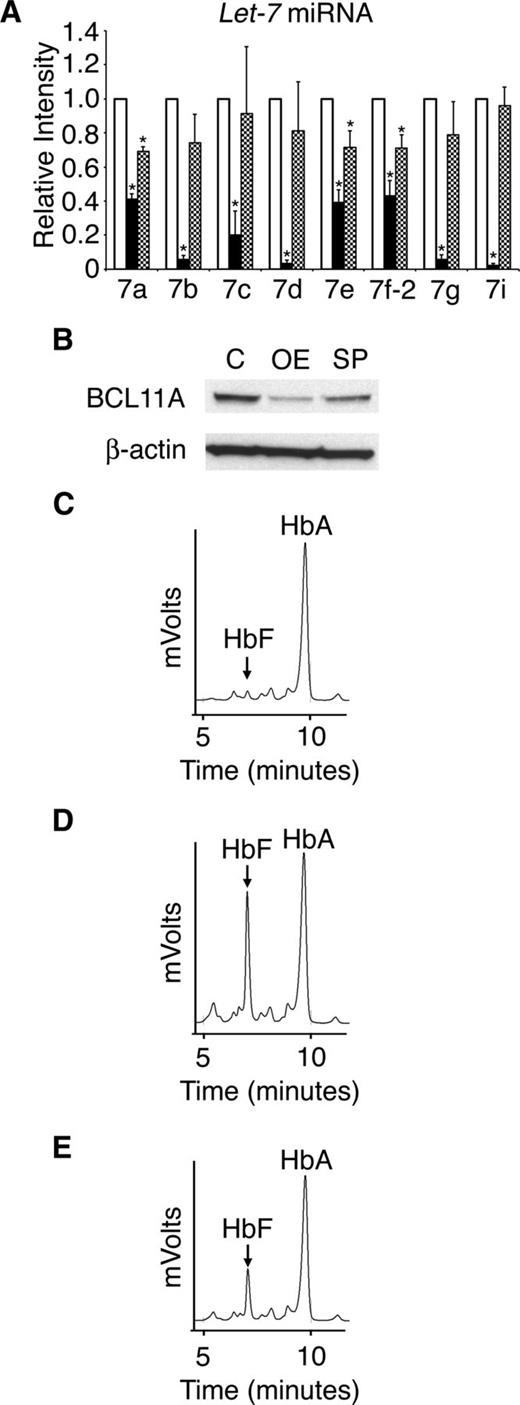
To investigate if the main targets of LIN28B, the let-7 family of miRNAs, are responsible for the regulation of HbF, adult CD34+ erythroid cells were transduced with a let-7 sponge retrovirus. LIN28A and LIN28B expression remained less than the qRT-PCR detection threshold after transduction (data not shown). Transduction with the sponge retrovirus resulted in significant suppression of let-7 (Figure 7A). However, the level of let-7 suppression was more robust with LIN28B-OE than the let-7 sponge. Comparison of LIN28B-OE and let-7 sponge experiments also demonstrated greater effects of LIN28B-OE on BCL11A (Figure 7B) and HbF than achieved after let-7 suppression (Figure 7C-E). HPLCs demonstrated significantly increased HbF levels in adult erythroid cells with either LIN28B-OE or let-7 sponge compared with controls (control: 3.5 ± 0.3%; LIN28B-OE: 31.1 ± 2.90%; P = .0029; let-7 sponge: 19.1 ± 0.2%; P = .0003). These results demonstrate that suppression of let-7 miRNAs is sufficient to reduce BCL11A and increase HbF in adult human erythroblasts, even in the absence of LIN28A or LIN28B expression.
Retroviral suppression of the let-7 family of miRNAs regulates BCL11A and HbF. (A) Relative expression levels of the let-7 family of miRNAs (open bars represent control, black bars represent LIN28B-OE, and hatch-marked bars represent let-7 sponge) after transduction of the let-7 sponge or LIN28B-OE encoding retrovirus. The qRT-PCR analyses were performed at culture day 14, and compared with control transductions. The miRNAs relative expression levels (y-axis) in the control cells were defined as a level of one for comparison. Error bars denote ± standard deviation of 3 independent donors for each condition. (B) Western blot analyses with protein extracts from the empty vector control (C) versus LIN28B-OE (OE) and let-7 sponge (SP) cells. The membranes were probed with anti-BCL11A. Anti-β-actin probe was used as a loading control. HPLC analyses of hemoglobin from (C) control, (D) LIN28B-OE, and (E) let-7 sponge samples were performed at culture day 21. HbF and HbA peaks are labeled on each graph (y-axis: mVolts; x-axis: elution time in minutes). Data are representative of 3 independent experiments. P values were calculated using two-tailed Student t test. C, empty vector control; OE, LIN28B overexpression; SP, let-7 sponge. *P < .05.
Retroviral suppression of the let-7 family of miRNAs regulates BCL11A and HbF. (A) Relative expression levels of the let-7 family of miRNAs (open bars represent control, black bars represent LIN28B-OE, and hatch-marked bars represent let-7 sponge) after transduction of the let-7 sponge or LIN28B-OE encoding retrovirus. The qRT-PCR analyses were performed at culture day 14, and compared with control transductions. The miRNAs relative expression levels (y-axis) in the control cells were defined as a level of one for comparison. Error bars denote ± standard deviation of 3 independent donors for each condition. (B) Western blot analyses with protein extracts from the empty vector control (C) versus LIN28B-OE (OE) and let-7 sponge (SP) cells. The membranes were probed with anti-BCL11A. Anti-β-actin probe was used as a loading control. HPLC analyses of hemoglobin from (C) control, (D) LIN28B-OE, and (E) let-7 sponge samples were performed at culture day 21. HbF and HbA peaks are labeled on each graph (y-axis: mVolts; x-axis: elution time in minutes). Data are representative of 3 independent experiments. P values were calculated using two-tailed Student t test. C, empty vector control; OE, LIN28B overexpression; SP, let-7 sponge. *P < .05.
Discussion
Increasing levels of HbF in adult erythroblasts represent an important therapeutic target for patients with SCD and β-thalassemias. In this study, we demonstrated that LIN28B regulates HbF levels during human erythroid cell development. LIN28B knockdown in CD34+ cord blood cells significantly reduced HbF expression, and overexpression of LIN28B in adult erythroblasts significantly increased HbF levels. Interestingly, HbF levels were also increased in adult cells transduced with a separate vector encoding LIN28A. LIN28A and LIN28B are thus functionally similar in this regard. LIN28B is a more likely candidate as a physiological regulator of HbF, because LIN28B is expressed in the fetal liver27 and the gene possesses fetal-specific chromatin marks26 in erythroid cells.
Because each cellular transcriptome helps determine its repertoire of RNA species that may be targeted by LIN28 proteins, the LIN28 effects on cellular fate and phenotype are predicted to be variable. GWAS identified LIN28 genes as regulators of temporally defined events in humans.15,16 LIN28 proteins are expressed during normal human development, as well as disease states including cancer. In embryonic stem cells, differentiation is associated with decreased LIN28 protein expression.44 LIN28 acts with other factors to induce the pluripotency of somatic cells.19 During mammalian development, expression of LIN28 proteins is reduced after embryogenesis. With the exception of cardiac and skeletal muscles, LIN28 proteins are nearly absent in adult tissues. In contrast, increased expression of LIN28 causes differentiation of myoblasts.20 Unlike stem cells and myoblasts, erythroblast differentiation was largely unaffected by the overexpression of a LIN28 protein. However, significant changes were noted for a subset of genes that are coordinated with developmental timing in humans. The increased expression of the fetal globin in adult CD34+ cells coincided with reduced expression of the adult globin (HBB). In addition to HBB, decreased expression of CA1 and GCNT2 genes were detected. These genes mark the developmental transition from fetal-to-adult erythropoiesis.32-34 The reductions of let-7, HBB, CA1, and GCNT2 genes on LIN28B-OE suggest a more fetal-like erythroid phenotype beyond the expression of HbF. These data support the notion that LIN28B and its targeted RNAs may be involved and should be explored further in the context of developmental changes in human erythroid biology.45,46
Examination of LIN28B mRNA in cord blood erythroblasts suggests that low level expression is sufficient to regulate let-7 and HbF in those cells. Significant reduction in HbF expression in cord blood erythroblasts was mediated by reductions in the erythroblast LIN28B mRNA levels (105 copies/ng reduced to 21 copies/ng). The effects of reducing LIN28B expression are thus amplified to regulate γ-globin RNA levels that are normally detected at 5-log higher levels (107 copies/ng) (Figure 4B). In the adult erythroblasts, LIN28A or LIN28B expression was less than the detection limit in the control cultures. LIN28B overexpression in the adult cells reached levels that were approximately 100-fold higher than the cord blood erythroblasts (104 copies/ng) (Figure 2A). The viral vector designs did not permit studies of adult-derived erythroblasts that express LIN28B in the comparable range of cord blood cells. However, higher level expression of the transduced LIN28B in the adult cells caused increases in HbF that approached, but did not exceed, that of cord blood-derived erythroblasts. The lack of increases in HbF to levels greater than detected in cord blood, despite higher LIN28B expression, suggests saturation of the globin gene regulating effects in adult cells. These results may be relevant in the context of gene therapy applications,47 because the requirements for high level γ-globin gene expression may be achieved via lower level expression of a transduced γ-globin gene regulator.
Our studies of LIN28B overexpression, as well as let-7 suppression in the absence of LIN28A or LIN28B, caused decreased expression of BCL11A as a predominant mechanism for increasing γ-globin mRNA in the adult erythroblasts. Increased BCL11A was also detected after LIN28B-KD in the cord blood erythroblasts (data not shown). An initial GWAS suggested that the BCL11A locus accounts for approximately 15% of the HbF trait variance in European populations.7 The role of BCL11A in globin gene regulation was confirmed experimentally using human erythroblasts11 and genetically-modified mice.48 Because neither BCL11A knockdown nor direct let-7 suppression had effects on LIN28A or LIN28B expression, our data suggest that LIN28B affects the expression of HbF primarily by targeting let-7 to act as an upstream regulator of BCL11A. We did not observe changes in KLF1, GATA1, or SOX6 gene expression in response to LIN28B. In addition to binding and inhibiting let-7, LIN28 proteins may affect non-let-7 RNA species25,49,50 ; however, our let-7 sponge studies suggest that the let-7 miRNAs are critical targets of LIN28 proteins. Nevertheless, systems biology approaches may be useful to more fully understand how LIN28 proteins and let-7 regulate the subset of genes described here, as well as the overall erythroid phenotype.
Ultimately, these studies support the original hypothesis that regulated expression of the let-7 miRNA family is involved in hemoglobin switching during the fetal-to-adult transition of human ontogeny.24 Because let-7 and other miRNAs function by reducing the expression of the mRNA to which they bind, it is proposed that suppression of let-7 causes decreased expression of BCL11A by targeting other RNAs that are expressed in human erythroblasts. Although predicted erythroid mRNA targets of let-7 were previously reported,24 the potential role for those genes in regulating BCL11A expression remains vague.
In summary, we demonstrated that LIN28B regulates HbF expression in erythroblasts cultured from cord blood and adult CD34+ cells without detected interference with terminal maturation or enucleation. In the adult cells, LIN28B acts as an upstream regulator of several genes with an associated fetal erythroid phenotype, including CA1, GCNT2, and miR-96. Overexpression of LIN28B, as well as suppression of its major RNA target (the let-7 family of miRNAs) suppresses BCL11A, a well-characterized transcription factor that represses γ-globin gene expression. Our findings have important clinical implications introducing LIN28 proteins and their target RNAs as potential tools to generate therapeutic erythrocytes or aid in the identification of novel candidate molecules for treating SCD and β-thalassemias.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors wish to thank Alan N. Schechter for his mentoring and helpful comments in preparation of this manuscript. S.A.M thanks William E. Paul for his mentoring.
The Intramural Research Programs of the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Institute of Allergy and Infectious Diseases supported this work. A provisional patent application was filed by the US government (National Institutes of Health) in conjunction with this work.
Authorship
Contribution: Y.T.L., J.F. de V., J.Y., C.B., S.-J.N., E.R.M., K.S.K., A.R., and M.K. performed experiments; Y.T.L., J.F. de V., J.Y., C.B., S.-J.N., E.R.M., K.S.K., A.R., and M.K. analyzed data; Y.T.L., J.F. de V., J.L.M., and S.A.M. wrote the paper; and J.L.M. and S.A.M. conceived, assisted, and directed research.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
The current affiliation for J.Y. is Molecular Medicine and Gene Therapy, Lund University, Lund, Sweden.
Correspondence: Jeffery L. Miller, Molecular Medicine Branch, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, 10 Center Dr, Building 10, Room 9N311, Bethesda, MD 20892; e-mail: jm7f@nih.gov.
References
Author notes
Y.T.L. and J.F. de V. contributed equally to this work.
S.A.M. and J.L.M. co-directed this work.